Abstract
Pigmented tumors have been reported infrequently in captive deer. We document herein the clinical progression and gross and histopathologic features of pigmented tumors diagnosed as melanoma and pigmented schwannoma in 11 white fallow deer (Dama dama). Affected animals were part of a captive herd maintained at a drive-through park in southern Oregon and were 5–17 y of age during the study period (2004–2013). Primary lesion locations included periocular, perineal, and neck tissues, with cutaneous and internal metastases later identified at autopsy in some cases of malignant melanoma. Diagnoses included 7 malignant melanomas, 2 benign melanomas, and 2 pigmented schwannomas. Diagnosis of melanoma was based on typical histomorphologic features, and final diagnosis of pigmented schwannomas was based on histomorphologic features with negative staining for melan A and positive staining for laminin. Metastasis was found in 3 of 7 cases diagnosed as malignant melanoma; 2 had extensive pulmonary involvement and resulted in euthanasia of the animal; 1 animal developed eyelid and ear lesions that also resulted in euthanasia.
Introduction
Neoplasia is an uncommon finding in surveys of cervid disease.1,18 The most common neoplasms reported in deer are virally induced tumors that include various types of fibromas and papillomas. 21 Papillomavirus infection has been documented in several cervid species including white-tailed deer, mule deer, European elk, reindeer, red deer, roe deer, Sika deer, fallow deer, black-tailed deer, and caribou.2,21 In North America, the most widely distributed papillomavirus in cervids, particularly white-tailed deer, is the deer papillomavirus, which causes cutaneous and pulmonary fibromatosis. 2 Literature citations of other types of neoplasia in cervids are sporadic, and include lymphosarcoma, melanoma, schwannoma, and oligodendroglioma.4,9,10,15,16 We describe the histopathologic characteristics and clinical history of an unusual series of 11 fallow deer with pigmented tumors.
The majority of the tumors described in our report could be easily identified as melanomas (benign or malignant) given their pigmentation and typical histomorphologic features. In 2 tumors, poorly melanized melanoma and pigmented schwannoma were the major differential diagnoses, and tumor identity could not be determined by hematoxylin and eosin (H&E) staining alone.
Pigmented schwannomas and melanomas can share diagnostic features, but differentiation is important because of prognostic implications and treatment strategies in humans and dogs.13,25 Schwann cells are surrounded by a complete external lamina given their inherent property of synthesizing external lamina components. There is variable expression of S100 in canine and feline schwannomas 7 and, to our knowledge, this antigen has only rarely been pursued in cervids. 13 Given the variable expression in some species and poorly documented utility in cervids, we did not pursue S100 as a diagnostic marker. Immunohistochemical staining of laminin and collagen IV has been used to differentiate pigmented schwannomas from malignant melanomas in humans, 25 and laminin expression has been reported in schwannomas of dogs, cats, and horses.3,7,11,17 Histochemical staining for reticulin fibrils with Gordon and Sweet stain has also been described as being useful in the identification of schwannomas in dogs and cats. 5
Diagnosis of amelanotic melanomas often requires ancillary histochemical and immunohistochemical methods. Fontana–Masson is a silver stain that reacts with melanin, although a disadvantage is lack of specificity because of cross-reaction. 20 Melan A has been described as a less sensitive but more specific positive marker for the diagnosis of melanoma, although its utility in cervids is uncertain. 20
Ultraviolet (UV) damage is commonly implicated in the development of melanomas in humans and Angora goats.8,20 Solar elastosis is believed to result from UV alteration of dermal matrix proteins in damaged areas of the skin of humans and multiple domestic animal species. 6 Degeneration of collagen and elastic fibers in the superficial dermis can be highlighted by Verhoeff elastin silver (VES) staining. 5 To investigate possible UV damage contributing to melanoma development in this case series, VES was performed when sufficient cutaneous tissue was present.
Materials and methods
Animals
The deer in our study were part of a captive herd in a drive-through park in southern Oregon. The annual herd size was ~40 during the study period (2004–2013). Animals were predominantly females with contraceptive programs in place to prevent overpopulation. All of these deer were leucistic (white color phase).
In all cases, biopsy samples were collected under general anesthesia following identification of cutaneous masses during routine observation or annual health check and in accordance with routine health care protocols at the facility. In 3 cases, the lesions were so severe that the animal was euthanized at the time of initial diagnosis or biopsy collection. Samples were collected into 10% neutral-buffered formalin and routinely processed for embedding in paraffin. Sections cut at 5 μm thickness were stained with H&E for evaluation. Samples with sufficient non-neoplastic cutaneous tissue were also stained with VES to check for solar elastosis. Some of the samples were additionally stained with Gordon and Sweet reticulin stain and Fontana–Masson stain.
Immunohistochemistry
Immunohistochemistry for laminin and melan A was also performed on a subset of samples using the following procedure. Tissue blocks were sectioned at 4–5 µm, collected on charged slides, and baked at 60°C for 1 h. Slides were rehydrated through 2 changes of xylene, 2 changes of 100% ethanol, 1 change of 80% ethanol, and water. For melan A, high-temperature antigen retrieval was performed in a microwave pressure cooker using a target retrieval solution (Dako North America, Carpinteria, CA) for 10 min after pressure was reached, and then cooled to room temperature. Slides were placed on a autostainer (Dako) and washed in Tris-buffered saline with Tween (TBST; TWB945M, Biocare Medical, Concord, CA) followed by 3% H2O2 (Sigma-Aldrich, St. Louis, MO) in TBST for 10 min. For the laminin-staining procedure, slides were treated with proteinase K for 10 min, followed by a serum-free protein block (Dako). The primary antibodies were diluted in an antibody diluent (Dako) and applied for 30 min at room temperature. A horseradish peroxidase (HRP) rabbit polymer (MaxVision Biosciences, Mukilteo, WA) was applied for 7 min at room temperature for laminin and HRP mouse polymer (ImmunoBioScience [IH-8064-custom-OrSU or IH-8062-custom-OrSU], Mukilteo, WA) for 10 min at room temperature for melan A. Slides were again washed in TBST before the chromogen (SK-4800, Vector Laboratories, Burlingame, CA) was applied for 5 min. Slides were then washed in dH20 followed by hematoxylin (Dako) diluted 1:3 in dH20 for 5 min, rinsed in dH20, rinsed in TBST, run down to xylene, and coverslipped. A kit provided the rabbit antibody (Dako) used as the negative control. Internal positive controls were available (basement membranes for laminin; epidermal melanocytes for melan A), but we also stained known canine melanoma and schwannoma specimens.
Diagnostic criteria
Melanomas can have variable histologic appearance; however, the melanomas in our case series were mainly epithelioid variants, with round-to-polygonal cells, discrete cell borders, abundant cytoplasm often containing brown-black (melanin) granules, and nuclei with prominent nucleoli.7,20 Malignancy was based on histologic criteria used for distinguishing benign and malignant melanomas in dogs.19,20 Greater than 3 mitotic figures in ten 400× high-power fields (HPF), nuclear atypia (>20% of nuclei showing atypia), presence of ulceration, and extension beyond the dermis were considered features of malignancy.19,20 Dermal melanocytomas have been described as less cellular with small neoplastic spindle cells a variable amount of collagenous stroma, and the cells may have neuroidal morphology. 7 There were 2 tumors in our case series with the aforementioned histologic features and poor pigmentation in which the differential diagnosis included poorly melanized melanocytoma and pigmented schwannoma. Intermixing streams of elongated, spindle cells with closely associated reticulin fibers were seen with reticulin stain; additional immunohistochemical staining was performed in these cases. Membranous and cytoplasmic positive laminin staining of neoplastic cells and negative staining for melan A led to a diagnosis of pigmented schwannoma.
Results
Details of the signalment, lesion location, and other features are summarized in Table 2. In this series of 11 fallow deer, 7 cases were diagnosed as malignant melanoma, 2 cases were diagnosed as benign melanoma, and 2 cases were diagnosed as pigmented schwannoma after additional immunohistochemical staining. Cases of melanoma were definitive by H&E staining; melan A staining was not pursued in these cases because we did not feel it was necessary for diagnosis.
Summary of cases of pigmented tumors in 11 white fallow deer (Dama dama).*

BM = benign melanoma; F = female; LTF = lost to follow-up; M = male; MM = malignant melanoma; Met = metastasis; N = no; NA = not applicable; NE = not examined; PS = pigmented schwannoma; R = recurrence; TFI = tumor-free interval; Y = yes. Age is defined as at diagnosis of pigmented mass; clinically healthy is defined as being related to the mass.
Euthanized because of trauma unrelated to melanoma.
Malignant melanomas
Melanomas diagnosed as malignant had consistent features of variable melanin pigment, cellular pleomorphism (generally epithelioid), and mitotic activities ranging from 2 to 3 figures per HPF up to 10 mitotic figures per HPF (Fig. 1). Sometimes bizarre nuclear shapes, high nuclear-to-cytoplasmic ratio, and abnormal mitotic figures were seen in these specimens, as were binucleate and multinucleate cells. Necrosis, ulceration, and melanophages were inconsistently seen in these growths. Although some of the samples lacked sufficient non-neoplastic tissue for proper assessment, several of these cutaneous locations exhibited some degree of solar elastosis with VES.
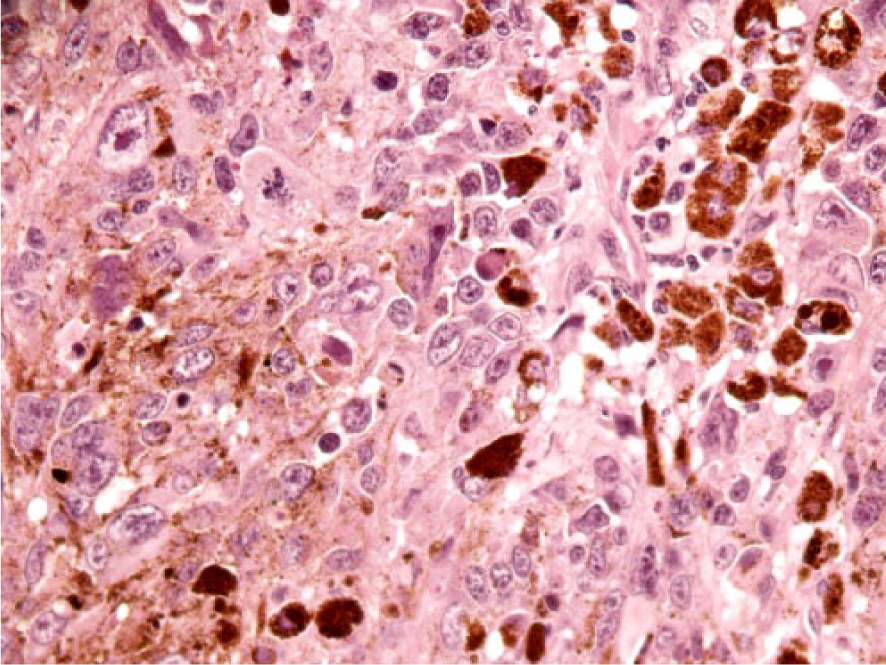
Periocular, malignant melanoma in fallow deer 1. There is marked anisocytosis and anisokaryosis with bizarre mitotic figures. Melanomacrophages are scattered throughout. H&E. 400×.
Four of the 7 deer with malignant melanoma in our report were euthanized, and in 3 of these 4 cases the original mass occurred on the eyelid with evidence of metastasis at the time of autopsy (Fig. 2). The fourth deer that was euthanized had multiple concurrent pulmonary lesions (bronchiolar adenocarcinoma) and this may have resulted in insufficient tissue sampling to detect melanocytic metastases. In total, 4 of 4 eyelid melanomas in this series metastasized or recurred. Sites of metastasis included mandibular skin, aural skin, and lung.
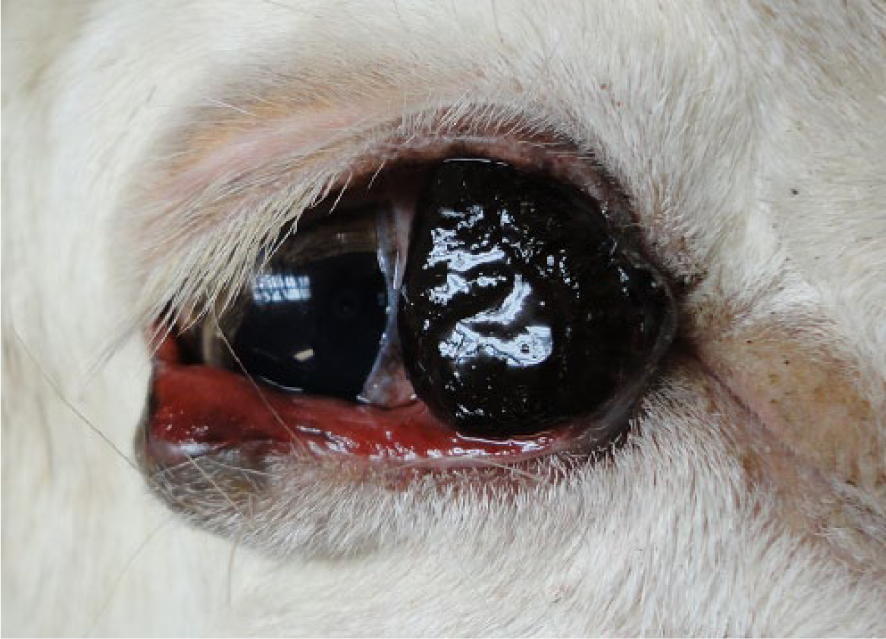
Focal, black, soft, conjunctival malignant melanoma in fallow deer 1.
Benign melanomas
Two cases were diagnosed as benign melanoma based on identification of pigmented polygonal-to-spindle–shaped cells with no mitotic figures and restriction to the dermis. One of the benign melanomas was a perineal mass identified on routine physical examination of a 9-y-old female. This was an exophytic, pigmented, dermal lesion. The neoplastic cells were loosely arranged in interweaving streams and were polygonal-to-spindle–shaped with abundant fine granular melanin pigment. Although the cells exhibited moderate anisokaryosis and occasional prominent nucleoli, no mitotic figures were identified, and the mass remained within the dermis. VES staining identified a moderate degree of solar elastosis. Reticulin staining was performed, and reticulin fibers were neither increased nor associated with neoplastic cells. The mass was completely removed with large surgical margins. Three years later, a poorly differentiated sarcoma was removed from the vulva, and a fibroma from the abdominal skin. The animal was alive at completion of the study period, with no recurrence of the pigmented perineal mass in the intervening 6 y. The other animal diagnosed with a benign melanoma was a 12-y-old female with a periocular mass. When anesthetized, a fibroma was also removed from the neck and a basal cell epithelioma from the perineum. The periocular benign melanoma was exophytic, discrete, and restricted to the dermis. Neoplastic cells were polygonal-to-spindle–shaped with pigmentation and numerous melanomacrophages; no mitotic figures were identified (Fig. 3). There was mild solar elastosis based on VES staining. One month after the removal of the melanoma, the animal was found trapped under a log and dyspneic. The animal was euthanized given its poor condition, and autopsy revealed gastric perforation, uterine adenocarcinoma with pulmonary metastasis, and no evidence of melanoma recurrence or metastasis.
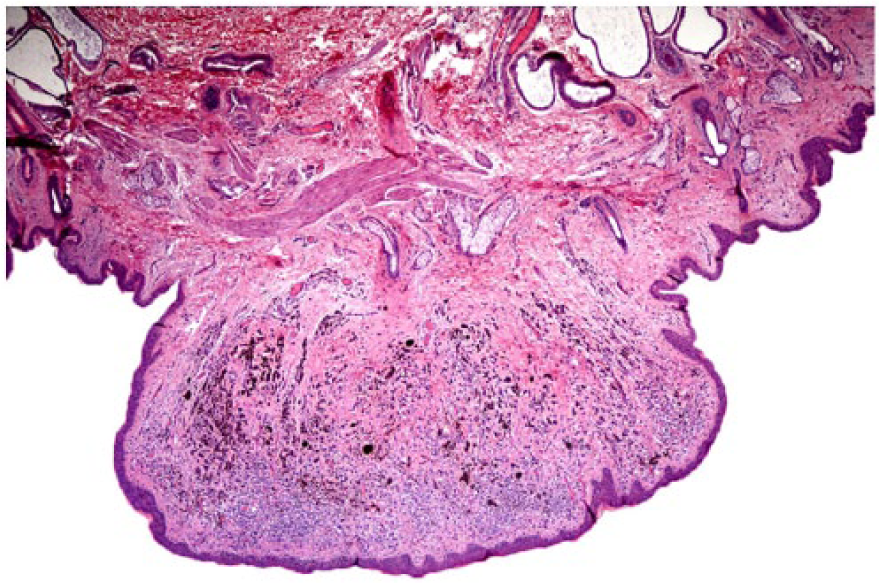
Perineum, dermal periocular, exophytic, discrete, benign melanoma in fallow deer 6. Neoplastic cells are moderately pleomorphic with variable pigmentation and are intermixed with numerous melanomacrophages. No mitotic figures are identified, and neoplastic cells do not extend beyond the dermis. H&E. 20×.
Pigmented schwannomas
Two pigmented tumors were slightly atypical of melanoma, and with additional staining and expression of intra- and extracellular laminin, these were considered most consistent with pigmented schwannomas. One mass was from a 12-y-old male with a history of a ~0.25 cm black pigmented lesion on the face that grew to ~1 cm within 1 mo. This unencapsulated mass consisted of mildly pleomorphic cells with oval-to-tapered nucleoli and ~1 mitotic figure per HPF. The interlacing pattern of cells in some quadrants was reminiscent of neural tissue. Solar elastosis was not identified with VES. There were multifocal superficially located scattered cells positive for melan A that clearly contained melanin and were interpreted to be melanomacrophages. Melanocytes were also identified within the basal layer of the epithelium, confirming reactivity of melan A in this species. Reticulin fibers were associated with neoplastic cells. Laminin staining was positive, making the neoplasm consistent with a pigmented schwannoma.
The other schwannoma was from a 14-y-old female with a smooth-surfaced perineal mass found on routine physical exam. A second mass removed from the neck with clear surgical margins was diagnosed as a fibroma. Microscopically, the perineal mass was poorly pigmented, poorly demarcated, multilobulated, and restricted to the dermis. The neoplastic cells showed mild anisocytosis and anisokaryosis. Melanin pigment was identified in cells bordering the epidermis and in a few isolated cells in the deeper dermis. Melan A was negative in the vast majority of the tumor cell population. There were increased cell-associated reticulin fibers (Fig. 4), and laminin IHC (Fig. 5) was positive, consistent with a diagnosis of pigmented schwannoma. One of the surgical margins of this mass did not appear clear of neoplastic cells, but there was no recurrence of either growth during a 6-y follow-up period. Other neoplasms identified in these fallow deer included fibroma, fibrosarcoma, basal cell epithelioma, bronchioloalveolar adenocarcinoma, and uterine adenocarcinoma with pulmonary metastases.
Perianal, pigmented schwannoma in fallow deer 5. Spindle-shaped neoplastic cells are arranged in interweaving streams within the dermis. The neoplastic cells within the dermis are closely associated with reticulin fibers. Gordon and Sweet reticulin stain. 500×.
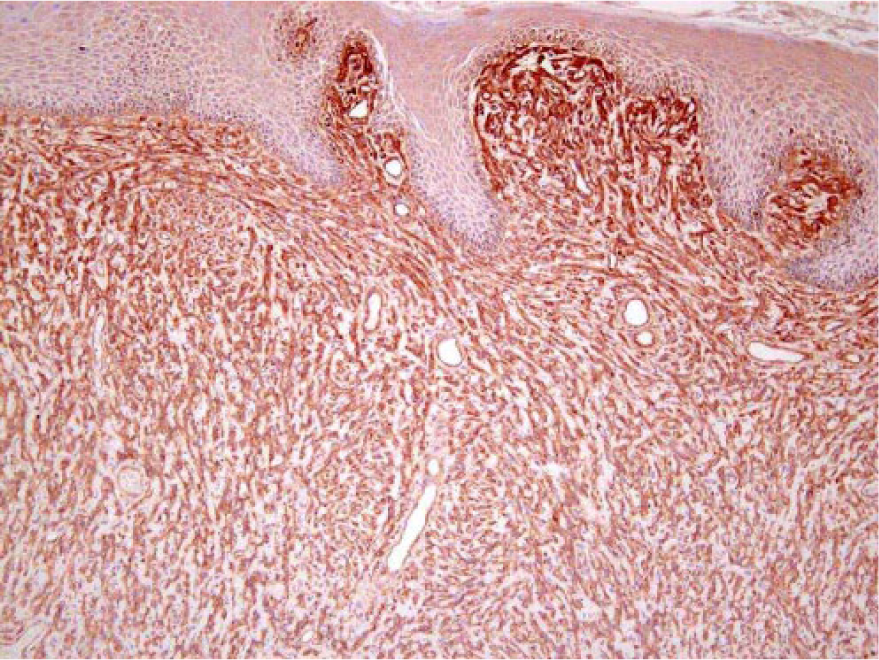
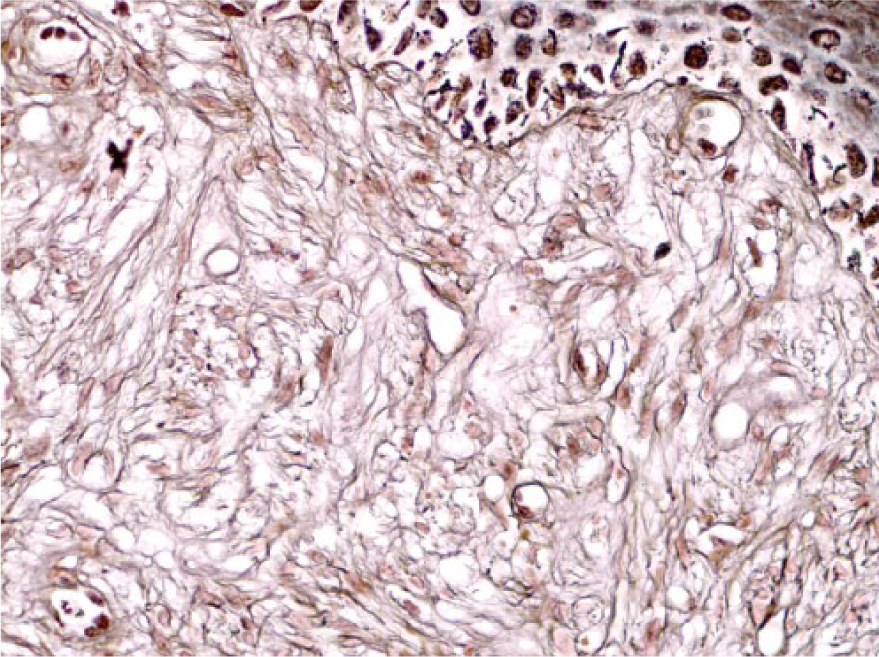
Perianal, pigmented schwannoma in fallow deer 5. The spindle-shaped neoplastic cells within the dermis are surrounded by laminin with scattered granular cytoplasmic immunolabeling. Laminin immunohistochemical stain. 100×.
Discussion
The fallow deer is a cervid native to Eurasia but has been introduced to at least 38 countries including those in North America. These deer weigh 30–80 kg, and their pelage coloration (white, menil, common, and black) is the most variable of any species of deer (https://goo.gl/vRDDKd). A solitary case of malignant melanoma arising from the conjunctiva in a captive fallow deer has been reported, but the color phase was not mentioned. 15 Lymphatic invasion and 1 or 2 mitotic figures per HPF were observed. A case report of malignant melanoma on the foot of a red deer (Cervus elaphus) included metastasis to a regional lymph node and many of the histologic features seen in malignant melanomas in our study. 16 Melanomas in cattle can occur as a congenital lesion or develop at any age and are usually benign. 23 Melanocytic tumors in goats and sheep are uncommon, occurring in the skin or subcutaneous tissue and are often malignant, locally invasive, and prone to metastasis.12,23 In other domestic animals, melanomas arising from the conjunctiva are rare but often found to be behaviorally malignant, whereas melanomas arising from the haired skin of the eyelid are often benign.23,24 Limbal melanomas (epibulbar) are considered to be benign with slow, localized behavior, and are responsive to local excision.23,24 None of the periocular melanomas in our group of fallow deer had direct connection to the globe.
There was metastasis or recurrence in 4 of 4 eyelid melanomas and 1 of 2 periocular melanomas reported in our series. It is not clear whether eyelid masses in the cases presented in this series arose from the skin or the conjunctiva, but periocular melanomas in white fallow deer appear to have an aggressive biologic behavior.
Melanomas commonly arise from sun-exposed surfaces, although other factors undoubtedly contribute to oncogenic transformation. 8 In Angora goats, it was estimated that 65% of cutaneous melanomas occur as a result of DNA damage from UVA (ultraviolet A)- and UVB-induced solar radiation. 20 In our fallow deer, 7 of 9 melanomas arose from thinly haired areas: periocular, face, and perineum, and all of the deer were leucistic. Six of the 7 malignant melanoma cases that could be assessed featured solar elastosis, suggesting that UV exposure may also be a risk factor for melanoma in leucistic fallow deer. The drive-through park also has a variety of other cervid species, including sika deer (Cervus nippon), which has extensive white spotting on the pelage, but melanomas were only found in the white fallow deer during the study period. Melanoma development in these fallow deer likely has a genetic component in addition to UV exposure.
Two tumors in our study were not diagnosed definitively with H&E staining. The differential diagnoses for these pigmented tumors included amelanotic melanoma and pigmented schwannoma. A lack of positivity with melan A as well as increased reticulin fibers surrounding neoplastic cells and expression of laminin surrounding cells and intracytoplasmically resulted in diagnosis of pigmented schwannoma in both cases. A malignant schwannoma located on the left hindlimb has been reported in a red deer. 14 Histopathologic features of a spindle cell tumor with frequent palisade arrangements (Antoni type A pattern) were noted. 14 In this red deer, the neoplasm was positive with vimentin and S100 and negative for neurofilament, glial fibrillary acidic protein, and keratin, but laminin staining was not performed. 14 Diagnosis of schwannoma has been described to be confirmed by immunoreactivity to laminin or collagen IV, which define the basal lamina external to the neoplastic Schwann cell cytoplasmic membrane.22,25 In our case series, both diagnosed schwannomas showed immunoreactivity to laminin outlining the basal lamina with granular cytoplasmic staining, which has also been described in dogs. 3
Melanomas were common in white fallow deer in our case series, with thinly haired areas often being the site of origin. Periocular melanomas, in particular, appear to have an aggressive biologic behavior in white fallow deer, and UV solar radiation may play a part in the pathogenesis. Pigmented schwannomas were differentiated from melanomas by decreased melan A reactivity, increased cellular associated reticulin fibers, and positive laminin staining of neoplastic cells.
Footnotes
Acknowledgements
We thank Dr. Bermudez and the Department of Biomedical Sciences at Oregon State University for supporting this research. We also thank Kay Fischer (Histology Laboratory, OSU-VDL) for slide preparation and outstanding support with immunohistochemistry.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We received no financial support for the research, authorship, and/or publication of this article: Funding for some slide preparation and for publication was provided by Oregon State University Veterinary Diagnostic Laboratory.
