Abstract
A 2-year-old male free-ranging white-tailed deer (Odocoileus virginianus) was diagnosed with bilateral expansile tumors of antler origin. The deer was found dead by a landowner in High Springs, Florida. Two roughly spherical, multilobular, broad-based, bony, velvet-covered masses originated from each antler pedicle. These masses replaced or displaced many of the bones and soft tissues of the skull and extended through the left cribriform plate and the right petrous temporal bone, compressing portions of the brain. Microscopically, the masses closely resembled normal-growing antler, containing all the elements thereof but with areas of necrosis and hemorrhage suggestive of ischemi or trauma. Tumorlike outgrowths termed antleromas have been described in free-ranging and captive cervids and typically are associated with disruptions in the seasonal rise and fall of circulating testosterone necessary for normal antler growth, casting, and regeneration.
The term antleroma was first used by Richard Goss to describe tumorlike fibrocartilaginous outgrowths of skin from retained velveted antlers of castrated fallow deer (Dama dama). 4 Other terms have been used to describe similar lesions albeit in different cervid species, including perukes, cactus antlers, and wig antlers. 4,6 Goss included these other manifestations when defining an antleroma. 4 In moose, the term velericorn was used to describe nodular bony transformations of velveted antlers. 1 More recent histologic studies of antlers from castrated fallow deer described numerous bony protuberances in addition to the fibrocartilaginous outgrowths of skin. 6 These studies suggest that tumors of antler origin may manifest differently among species, even within a species. To avoid confusion, we use the term antleroma in a general sense to describe any tumor or neoplasm of growing antler origin. Since normal-growing antler is composed of bone, cartilage, fibrous connective tissue, and velveted skin, antleromas may consist of one or multiples of these tissues.
In cervids, lesions consistent with antleromas have been described in captive and free-ranging Columbian black-tailed deer (Odocoileus hemionus columbianus), fallow deer, moose (Alces alces), mule deer (Odocoileus hemionus), roe deer (Capreolus capreolus), red deer (Cervus elaphus), Sitka black-tailed deer (Odocoileus hemionus sitkensis), and white-tailed deer (Odocoileus virginianus). These lesions were associated with alterations in circulating testosterone levels due to castration, cryptorchidism, hypogonadism, or endocrine disrupting agents. 1 –4,6,11 –13 This association strongly suggests that alterations to the normal circulating levels of testosterone may be necessary for antleroma development; however, the mechanisms by which these alterations directly affect antleroma development and produce the variable antler tumors reported in the literature are poorly understood. It is important to note that these are rare occurrences in free-ranging populations with little population significance. However, local populations may have higher incidences due to poor genetic diversity or increased exposure to toxins. 13
Antler formation and regeneration are tightly controlled, well-coordinated processes closely associated with seasonal fluctuations in the concentration of circulating androgens. These processes have been studied in red deer, sika deer (Cervus nippon), and fallow deer. 5,7 –9 It is likely that species-specific differences exist, for example, in reindeer and caribou where both sexes develop antlers; however, for the purposes of this report, we assume that the processes elucidated in red deer, sika deer, and fallow deer hold true for white-tailed deer.
Antlers grow from permanent bony outgrowths called pedicles. During the first year of life, pedicles develop from the antlerogenic periosteum, a specialized thickened periosteum overlying the lateral crest of the frontal bone. 8 Pedicles grow by intramembranous ossification, and growth is associated with rising blood testosterone concentrations. The first antlers form as direct continuations of the antler pedicles. Transition from intramembranous ossification to a modified process of endochondral ossification appears to be necessary for continued antler elongation and antlerogenesis; however, antler thickening continues via intramembranous ossification. 5,9 The first antlers elongate via this modified form of endochondral ossification in the presence of low testosterone levels until a sharp increase in circulating testosterone concentrations occurs before the first rut. The antlers harden, and the velvet is shed following this increase in testosterone. Deletion and transplantation studies confirm that the antlerogenic periosteum is necessary for pedicle and first antler growth and that it contains specific morphogenetic information for autonomous differentiation and axial orientation of the antler. 5
Antler regeneration begins in late winter or early spring of a male’s second year of life, immediately after the first antlers are cast. A sharp decrease in circulating testosterone concentrations following the rut is associated with osteoclasia at the distal pedicle and subsequent casting of the antlers. Appropriate wound healing at the casting site is necessary for normal antler regeneration. The exposed bone of the pedicle is covered by granulation tissue, followed by lateral growth and simultaneous thickening of the distal pedicle periosteum and, finally, second intention healing of the pedicle skin. 5,7 Regenerating antlers develop from the pedicle periosteum by endochondral ossification. Vascularization and innervation of the developing bone originate from the overlying antler velvet. The process of antler regrowth occurs in association with continued low concentrations of circulating testosterone and increasing day lengths throughout the spring and summer months. Antler growth continues until blood testosterone concentrations rise before the rut, at which time the antlers harden and the velvet is shed. 5 This entire process is repeated once the antlers are cast. The specialized cells of the antlerogenic periosteum and pedicle periosteum are pluripotent stem cells, and deer antlers are the only known mammalian appendages capable of full renewal. 7
History and Gross Findings
A free-ranging male white-tailed deer was found dead in a field in High Springs, Florida (Alachua County) on June 17, 2011. Based on tooth wear, its age was estimated to be 2 years 10 ; it weighed 37 kg; and it was emaciated. Each antler was grossly misshaped, consisting of multilobular, broad-based, bony, velvet-covered masses (Figs. 1, 2). Numerous lacerations and abrasions were scattered across the velvet of both antlers. Approximately 15 to 20 engorged ticks were attached to the caudal aspects of the antler velvet. Both ears were displaced ventrally and caudally by the antler masses. The scrotal testicles appeared grossly normal.
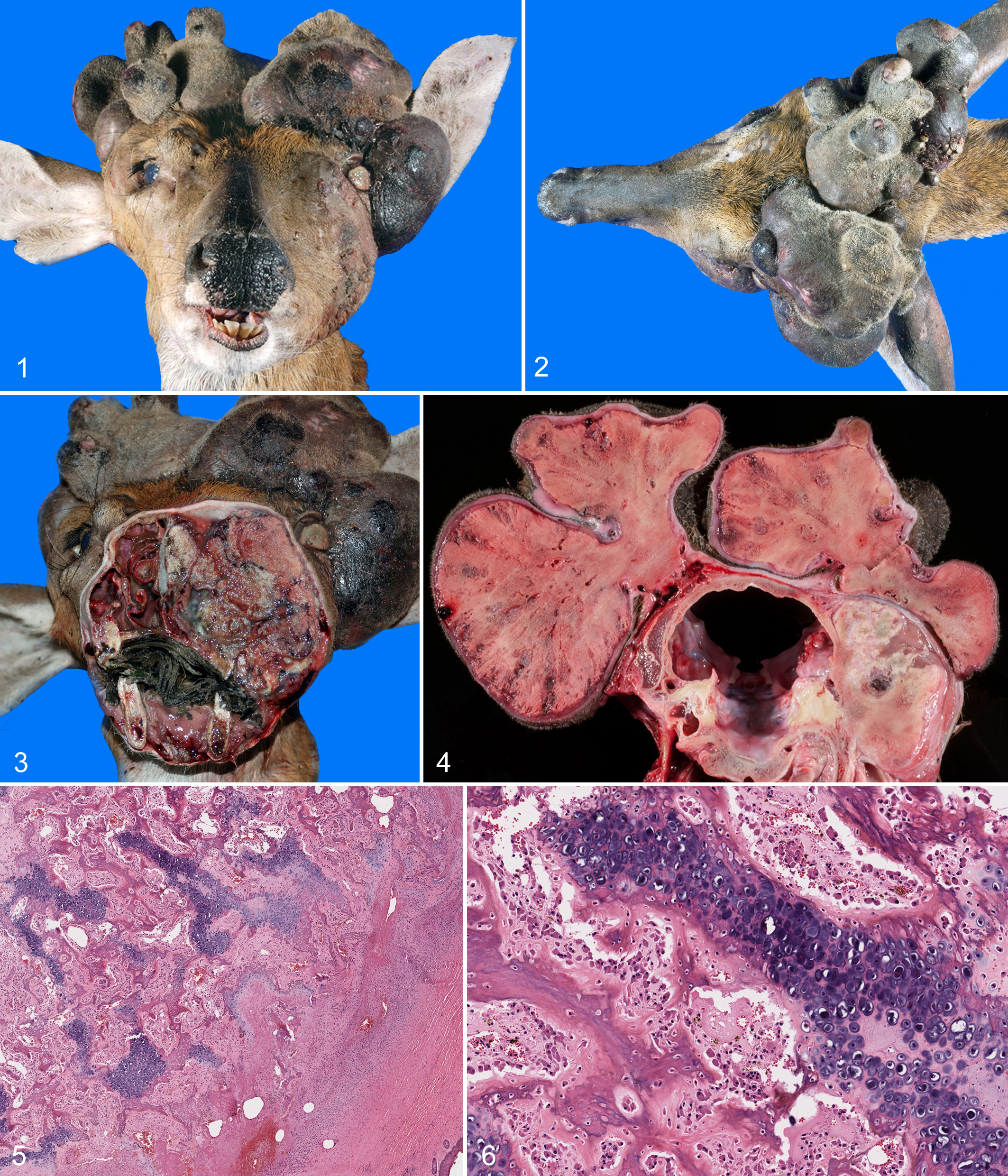
The left antler was approximately 10 × 15 × 7 cm, with similarly sized medial and lateral lobes. On cut section, the lobes consisted of hard white to tan material most consistent with bone. The medial lobe was multilobular and the lateral lobe spherical (Figs. 1, 2). Hard expansile material, similar to the antler masses in color, character, and consistency, displaced and likely replaced portions of the parietal, frontal, maxillary, and zygomatic bones. This hard expansile tissue displaced the left eye rostrally and laterally, constricting the left orbit (Figs. 1, 3). While the left globe was intact, the cornea was thickened, tan, and opaque. The left ethmoid turbinates were obliterated, and the nasal septum deviated 3 cm to the right (Fig. 3). An abscess in the left maxillary bone was associated with the upper molars and resorbed roots. The skin between the left eye and rostrum was variably ulcerated with variable necrosis and edema in deeper tissues. A hard, nodular subcutaneous mass (2-cm diameter) extended from the cartilage at the base of the left pinna. Adjacent to this was a hard pedunculated subcutaneous mass (1-cm diameter). A multilobular mass (2 × 1.8 cm) extended through the left side of the cribriform plate 1.5 cm, compressing the left olfactory bulb and cranial portions of the left frontal lobe.
The deformed right antler was approximately 8 × 12 × 6 cm, similar in appearance to the left antler on external and cut surfaces but more elongate and lobulated (Figs. 1, 2). A poorly delineated, hard, bony mass extended from the right ear to the right eye, obliterating portions of the temporal and parietal bones (Fig. 4). The pale and edematous soft tissues caudal to the right eye were elevated and compressed by the mass. A 2.2-cm-diameter nodular mass extended from the parietal and petrous temporal bones 1.7 cm into the cranial vault, compressing the caudal right cerebral hemisphere (Fig. 4).
Histopathology
The surfaces of the antler masses were covered by sparsely haired skin. The dermis contained few sebaceous glands and a thick layer of dense fibrous connective tissue, consistent with normal antler velvet. Mesenchymal cells subjacent to the dermis were transitioning from fibroblasts to a deeper layer of chondrocytes (Fig. 5). This cartilaginous cap interdigitated with spicules of immature bone, trabeculae of woven bone, and trabeculae of variably mineralized cartilage, consistent with endochondral ossification. Bony trabeculae were variably covered by wide osteoid seams or numerous osteoclasts within resorption lacunae (Fig. 6). Multiple foci within the developing bone were necrotic and hemorrhagic. Variably sized foci of fibrous connective tissue were scattered throughout sections of the bony masses. Portions of the expansile masses that obliterated cranial bones and expanded into the cranial vault had a similar microscopic appearance.
The small nodular and pedunculated masses noted at the base of the left pinna consisted of islands of mature and immature bone adjacent to mature cartilage, all circumscribed by a subdermal layer of loose connective tissue. These masses elevated a variably thickened epidermis and a dermis expanded by dense connective tissue, areas of necrosis, and hemorrhage. They were likely produced by the same process that produced the bony masses.
Seminiferous tubules contained a single layer of primary spermatocytes, and there was no evidence of developing spermatids within these tubules. Samples of testes from normal male deer, collected at the same time of year, had a similar appearance. Further interpretation of the testes was precluded by autolysis. No other significant lesions were identified.
Discussion
Previously described tumors of antler origin consisted of slow-growing, benign, bony, fibrocartilaginous, and/or dermal outgrowths extending from antlers with little or no involvement of adjacent tissues. 1,4,6,11,13 Experimentally, antleromas can be produced by castration performed anytime after antler pedicle formation has been completed. Retained velveted antlers can subsequently be cast with administration of testosterone. 4 –6,8 In free-ranging cervid populations, development of antleromas has been linked to alterations in androgen production associated with testicular lesions (eg, cryptorchidism, hypogonadism) or toxins. 2,3,11 –13 The antler masses described in this report were markedly thickened, misshapen, and expanded well beyond the pedicles to displace or obliterate adjacent bones of the skull, and they extended into the cranium compressing portions of the brain (Figs. 1–4). Likely differentials included osteoma, osteosarcoma, multilobulated osteochondrosarcoma, and ossifying fibromas. Despite the aggressive presentation, the masses maintained the basic histologic structure of growing antler. The occasional areas of hemorrhage or necrosis were likely due to loss of blood supply or trauma. There was no indication of an infectious process.
Antleromas can be considered neoplasms in the most basic sense, in that they are abnormal growths derived from normal tissues that expand beyond normal anatomical borders and lack normal growth controls. When normal androgen production is disrupted and circulating testosterone does not rise above a certain threshold prior to rut, the normal growth control over antlerogenic tissues is lost, and antleromas may form. The pathogenesis of bilateral antleroma development in this case is unknown. This deer died at a time when androgen levels are expected to be low, spermatogenesis absent, and antlers growing and covered in velvet. While it was impossible to directly assess androgen production in this animal, we were able to assess the testes. Despite significant autolysis, possibly masking subtle lesions, both testicles appeared normal when compared to healthy age-matched deer collected during the summer months. A more revealing assessment of the tissues might have been possible if the deer had died during periods of expected peak testosterone activity. There have been no other reports of bucks with persistently velveted or malformed antlers from this area of Florida.
Footnotes
Acknowledgements
We thank Kelly Straka for her help with the initial necropsy, and we thank the personnel at the Florida Fish and Wildlife Conservation Commission for collecting and submitting this diagnostic case.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded through the continued support of the Southeastern Cooperative Wildlife Disease Study through cooperative agreements with member states and federal agencies.
