Abstract
The association of feline morbillivirus (FeMV) with kidney disease in cats is controversial. Two cats with a history of severe hematuria had eosinophilic inclusion-like bodies in the renal tubular epithelial cells, without any inflammatory cellular reaction. Ultrastructurally, aggregations of electron-dense viral-like particles were found where the inclusion-like bodies were located. Immunohistochemistry (IHC) using antibodies against FeMV matrix protein labeled these inclusion-like bodies, and also labeled the cytoplasm of tracheal and bronchiolar epithelial cells, and lymphocytes and macrophages in spleen and mesenteric lymph node. Using double IHC, FeMV antigen was detected in astroglia and oligodendroglia but not in microglia. Phylogenetic characterization of the fusion and hemagglutinin gene sequences revealed FeMV-1A genotypes in both cats. These findings indicated an active viral infection with FeMV. We propose that FeMV is a renal epitheliotropic virus and also localizes in various other tissues.
Feline morbillivirus (FeMV) was first discovered in stray cats in Hong Kong in 2012. 22 Early reports suggested that FeMV was involved in chronic kidney disease (CKD) in domestic cats 21,22 and black leopards 10 and that it may be associated with feline urinary tract disease. 19 However, the causal role of FeMV in kidney disease in cats was challenged by several studies that also detected FeMV antigens in healthy cats. 2,9,17 So far, recent studies have reported that FeMV-infected cats can shed the virus via the urine for several months without abnormal clinical signs. 3,17 Thus, the relationship between kidney disease and the presence of the FeMV antigen is still questioned. 3,7,8,16,20,23
Two intact male domestic short-haired cats, both 2 years old, showed clinical signs of severe hematuria before death and had no previous history of other clinical illnesses. They were raised in different households and died in August and September 2019, respectively.
Postmortem examination of both cats revealed similarly prominent macroscopic findings in the urinary bladder, with severe acute hemorrhagic cystitis and marked accumulation of red urine (Fig. 1). Multifocal-to-coalescing areas of hemorrhage were present on the mucosal and serosal surfaces of the bladder. In addition, case 1 had moderate frothy exudate in the trachea with pulmonary edema, multifocal petechiae in the adrenal cortex, and mild peritoneal effusion, while case 2 had mild hemorrhage of the mesenteric lymph node (Suppl. Fig. S1). Other organs including brain, liver, and kidneys were congested (Fig. 2; Suppl. Fig. S2; Suppl. Table S1).
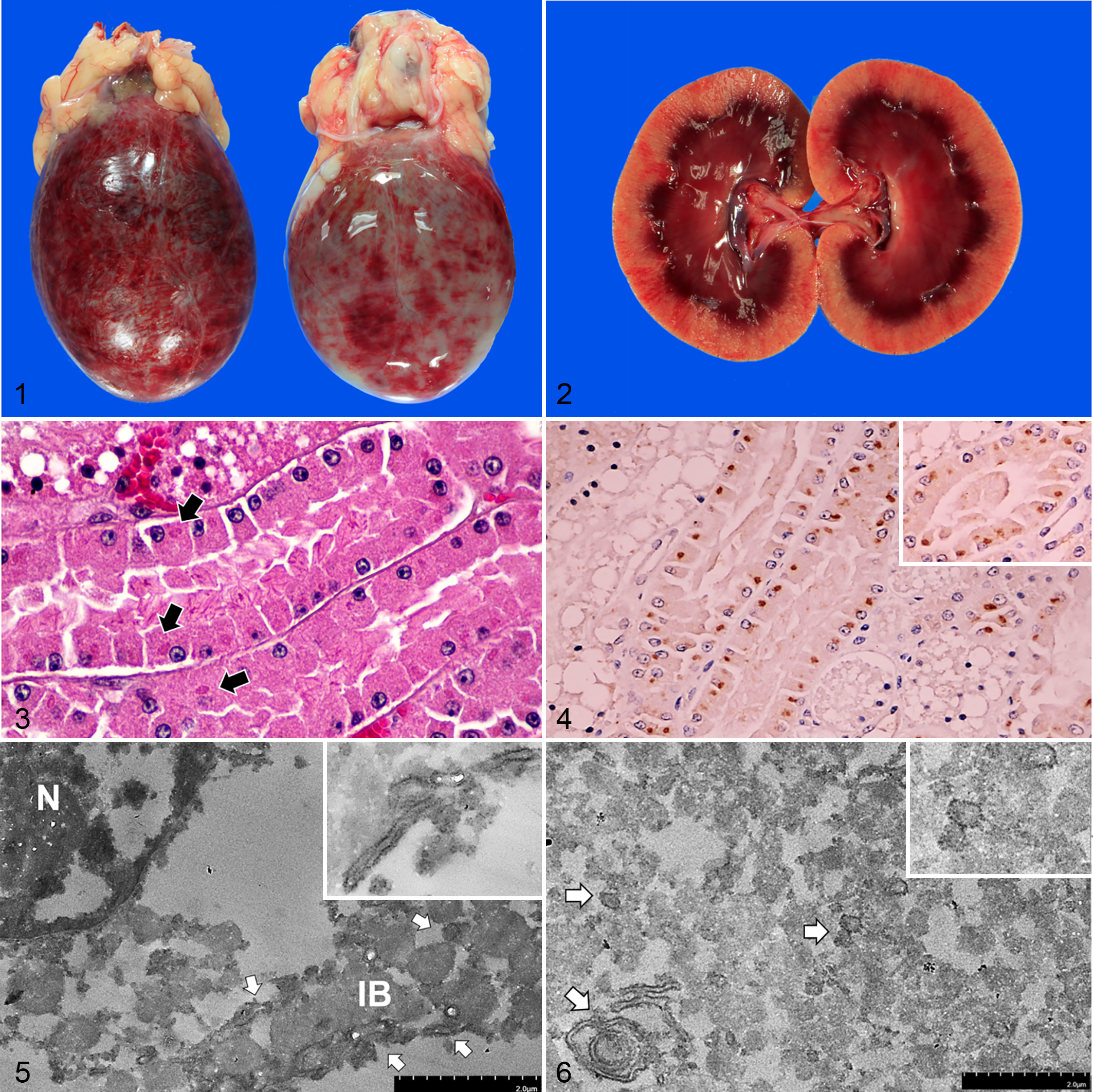
Feline morbillivirus (FeMV) infection, cat.
Histologically, both cats had diffuse renal tubular vacuolation with mild (case 1) to moderate (case 2) segmental multifocal membranous glomerulonephropathy. In both cats, the renal epithelial cells occasionally contained 2 to 4 µm, round to oval, intracytoplasmic, eosinophilic, hyalinized globules, especially in the proximal convoluted tubules at the corticomedullary junction and in the renal pelvic epithelium (Fig. 3). The urinary bladder had severe locally extensive to multifocal necrotizing hemorrhagic cystitis with marked submucosal hemorrhage. Numerous neutrophils infiltrated throughout the bladder wall and adjacent tissues, and there was multifocal fibrinoid vasculitis (case 2). The lungs of both cats had edema, diffuse congestion, and alveolar histiocytosis. The brain sections had mild to moderate diffuse perineuronal satellitosis in case 1 and focal cerebral rarefaction and vacuolation with multifocal cerebral hemorrhage in case 2. The liver of case 1 had multifocal lobular collapse, central vein fibrosis, moderate sinusoidal leukocytosis, and neutrophilia, while the spleen of case 2 had lymphoid follicular depletion with mild extramedullary hematopoiesis. The small intestine in case 2 had severe necrotizing enteritis with mild lymphocytic epitheliotropism. Details of the histopathological findings are included in Supplemental Table S2. Escherichia coli and Pseudomonas aeruginosa were cultured from urinary bladder samples from cases 1 and 2, respectively.
The histological findings of intracytoplasmic, eosinophilic, hyalinized globules in the renal tubules corresponded to the ultrastructural examination using transmission electron microscopy (TEM; HT7800) with the pop-off technique. 6,11 There were pleomorphic viral ribonucleocapsid-like particles, about 100 to 250 nm in diameter, that were aggregated and localized adjacent to the endoplasmic reticulum of the degenerated renal tubular epithelium. The viral particles were also rarely found in the perinuclear area (Figs. 5, 6).
The findings of viral ribonucleocapsid-like particles in the renal tubular epithelium prompted us to identify further the potential causative agent using pan-morbillivirus polymerase chain reaction (PCR) 22 screening of urine samples from both cases and fresh tissue from case 2. Positive pan-morbillivirus PCR fragments were identified in the urine samples of both cats, and in the fresh tissues of case 2 including kidney, lung, liver, small intestine, urinary bladder, spleen, mesenteric lymph node, and brain. The cycle threshold (Ct) value from the RT-qPCR reaction (Rotor-Gene Q real-time PCR cycler, Qiagen) targeting the polymerase (L) gene was lowest in the urine sample (Ct = 24.8), followed by the spleen (Ct = 32), kidney and mesenteric lymph node (Ct = 34.2), urinary bladder and small intestine (Ct = 34.8), and lung (Ct = 37.4), with the melting peak at 79.5 to 80 °C. Ct values could not be determined for the liver and brain samples (data not shown).
Subsequently, we analyzed the FeMV lineages by characterizing the full-length fusion (F) and hemagglutinin (H) genes in both cats using conventional RT-PCR assays with a set of specific primers targeting the F and H genes and total nucleic acids extracted from the cat’s urine as the template. 2 The sequencing results revealed homogeneity among both strains and that they were clustered in FeMV genotype 1A (FeMV-1A; Suppl. Fig. S11). The nucleotide similarity among them was 98.2% and 98.6% for the F and H genes, respectively, while that for the deduced amino acid sequences was 97.2% and 97.6%, respectively. The FeMV-1A sequences obtained from this study were submitted to GenBank database with accession numbers MT338572 (case 1) and MT338573 (case 2). More details on the similarities of the nucleotide and deduced amino acid sequences of the F and H genes obtained from this study, compared to the former FeMV strains (FeMV-1A, -1B, -1C, -1D, and FeMV-2), are included in Supplemental Figures S12 and S13.
To confirm the concordant results of viral-like particles by TEM and FeMV nucleic acid detected by RT-qPCR, the kidney sections of both cats were subjected to immunohistochemistry (IHC) to determine the presence of FeMV antigen using Pab-His-rFeMV-M. 10,12 Prominent FeMV immunoreactivity was localized in renal tubular epithelial cells, where the eosinophilic, intracytoplasmic, hyalinized globular materials were found (Fig. 4, inset). In other organs, FeMV immunoreactivity was diffusely present in the cytoplasm of various epithelial cells, including transitional, tracheal, bronchial, and bronchiolar epithelial cells (Suppl. Figs. S3, S4), and macrophages and lymphoid cells of the spleen and mesenteric lymph node were occasionally positive (Suppl. Fig. S5). In the brain, immunolabeling was present in the cytoplasm of neuroglial cells in the cerebrum and cerebellum (Suppl. Fig. S6). To differentiate FeMV-infected neuroglia, double-label IHC was performed targeting FeMV and either glial fibrillary acidic protein (GFAP; astrocytes), oligodendrocyte transcription factor 2 (Olig-2; oligodendroglial cells), or ionized calcium-binding adapter molecule 1 (Iba-1; microglial cells), respectively, as previously described with some modifications. 14 With these methods, FeMV antigen was localized in astroglia and oligodendroglia but not in microglia (Figs.7, 8; Suppl. Fig. S8). There was no immunoreactivity in negative control slides (Suppl. Figs. S7–S10). The IHC scores were evaluated and are summarized in Supplemental Table S3.
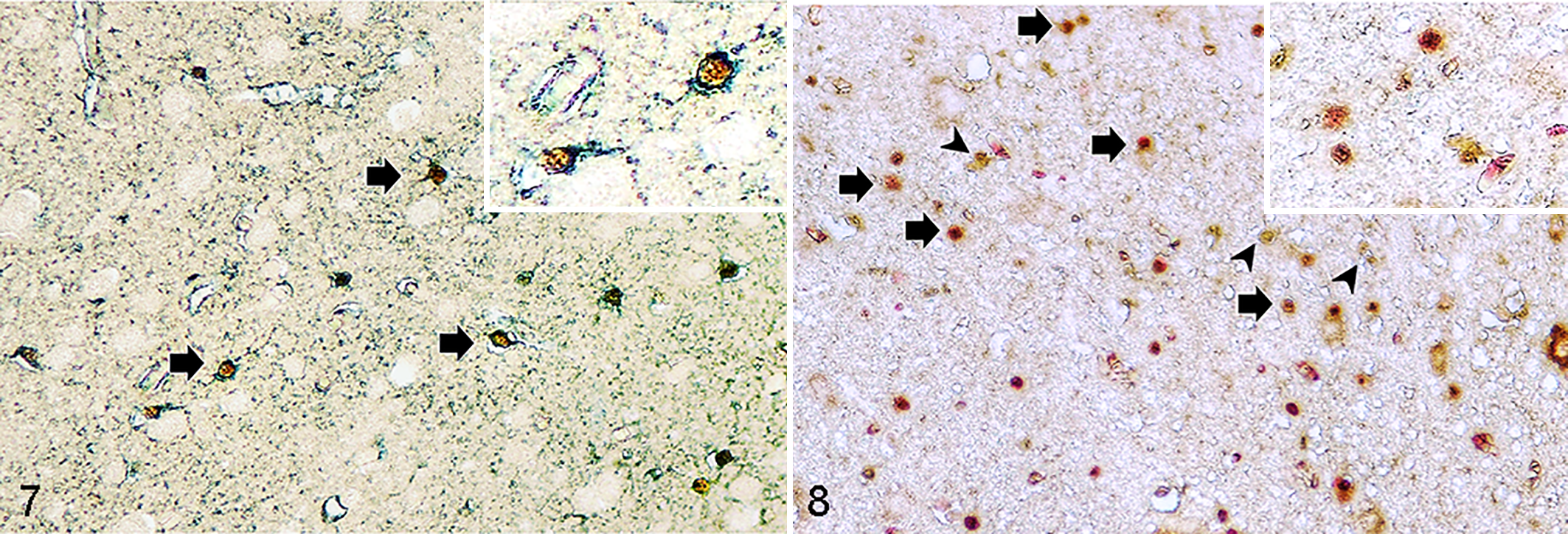
Feline morbillivirus (FeMV) infection, cat. Double-label immunohistochemistry.
Feline CKD has been associated with both noninfectious and infectious factors. A number of viruses causing CKD have been documented, such as feline immunodeficiency virus, feline leukemia virus, feline infectious peritonitis virus, 1 feline paramyxovirus, 19 and FeMV. However, the association of FeMV with kidney injury remains controversial.
In the current cases, these moribund cats suffering from acute hemorrhagic cystitis were found to be infected with the FeMV-1A genotype, which was confirmed by microscopic and ultrastructural examination along with molecular testing and IHC. The significance of the observed intracytoplasmic inclusion bodies, viral nucleic acid, and viral antigen in these cats cannot be determined as there was no associated inflammatory infiltrates within the renal tissue. Other studies also reported that cats infected with FeMV do not have renal inflammation. 7,8,20,23
Since the discovery of FeMV in 2012, the ultrastructural morphology of this virus has been seen only in a cell culture system. 22 Multiple attempts have been conducted to identify FeMV antigen and nucleic acid in the urinary tract, 4,5,17,18,22,23 but there has been no in situ confirmation of FeMV. Here, we report the presence of the viral ribonucleocapsid of FeMV associated with noninflammatory kidney disease in cats. These findings suggest that FeMV infection could be dormant without stimulating a host immune response.
Morbilliviruses possess the properties of lymphotropism and epitheliotropism by invading host cells via specific cellular receptors, including signaling lymphocytic activation molecule and nectin-4, respectively. 13 We demonstrated here the cellular localization of FeMV in infected cats using IHC-based analysis against the FeMV matrix protein. The results here are in accordance with previous research. 4,22 The identified FeMV-1A genotype also had in vivo tropism for various epithelial cells in the trachea, bronchi, bronchioles, kidneys, and urinary bladder, suggesting the epitheliotropism of FeMV. 12 FeMV immunoreactivity was also evident in the cytoplasm of lymphoid cells in the spleen and lymph nodes, supporting the lymphotropic nature of FeMV that was recently described. 10 Moreover, viral antigen was also detected in astroglia and oligodendroglia cells in the brain, supporting FeMV neurotropism as for other morbillivirus infection. However, the inconsistency between the positive RT-qPCR and the negative IHC results in the intestine might suggest hematogenous spreading of FeMV as indicated by previous reports. 18 The IHC results in urinary bladder are in agreement with the Ct value of the qRT-PCR assays. Regarding to these cats, we speculated that infection with the FeMV may lead to immunosuppression as found as in other morbillivirus infections, 15 and opportunistic bacterial infections might have led to septicemia in these cases. However, the role of FeMV in the cystitis or systemic disease in these cats is not known, and determining causality would require further studies.
In conclusion, we provided supportive evidence of FeMV infection in noninflammatory kidney disease in cats and indicated the dormant existence of FeMV in the kidney in the absence of a host immune response, but also demonstrated active FeMV infection in the kidney. FeMV nucleic acid and antigen were identified in various tissues. We propose that FeMV is a renal epitheliotropic virus of cats and may cause viral inclusions.
Supplemental Material
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211045441 - Renal epitheliotropism of feline morbillivirus in two cats
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211045441 for Renal epitheliotropism of feline morbillivirus in two cats by Surangkanang Chaiyasak, Chutchai Piewbang, Jakarwan Yostawonkul, Suwimon Boonrungsiman, Tanit Kasantikul, Anudep Rungsipipat and Somporn Techangamsuwan in Veterinary Pathology
Footnotes
Acknowledgements
We are grateful to Dr Jadsada Ratthanophart, Ms Prukswan Chetanachan, and Mr Poowadon Chai-in for excellent technical assistance. Animal Virome and Diagnostic Development Research Group and Chulalongkorn Academic Advancement Into Its 2nd Century Project, Faculty of Veterinary Science, Chulalongkorn University, are also acknowledged.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Thailand Research Fund (RSA6180034) to ST; a Grant for Joint Funding of External Research Project, the Ratchadaphisek Somphot Endowment Fund, and Veterinary Science Research Fund (RES_61_364_31_037), Chulalongkorn University, to ST. Surangkanang Chaiyasak was supported by the 100th Anniversary Chulalongkorn University Fund for Doctoral Scholarship. Chutchai Piewbang was funded by a grant from the Ratchadaphisek Somphot Fund for Postdoctoral Fellowship, Chulalongkorn University.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
