Abstract
Equus caballus papillomavirus type-2 (EcPV-2) has been proposed as a causal factor in equine genital squamous cell carcinoma (SCC). This study had 2 objectives: first, calculate the frequency of papillomavirus (PV) and EcPV-2 infection in papillomas, carcinomas in situ (CIS), and SCCs in Western Canadian horses; and second, determine if EcPV-2 status of equine SCCs is associated with overall survival (OS). EcPV-2 status of 115 archived tissue samples, spanning 6 years, was determined using broad spectrum (MY09/11) and EcPV-2-specific polymerase chain reaction (PCR) assays, EcPV-2-E6/E7 chromogenic RNA in situ hybridization (R-ISH), and amplicon sequencing. A retrospective survey gathered data on history, outcome, breeding, treatment, and rationales of referring veterinarians when managing PV-associated diseases. Histologic grade and completeness of surgical margins of SCCs were also considered. EcPV-2 DNA was identified in 10/58 (17%) SCC, 8/27 (30%) papillomas, 0/5 CIS, and 0/11 lesions identified as “other.” Overall, 18/101 (18%) of these lesions were positive for EcPV-2. EcPV-2 was identified in 10/35 (29%) SCCs arising from genital tissues but in 0/22 SCCs from other locations. There was no association between breeding history and EcPV-2 status of genital SCCs. EcPV-2 status of genital SCCs was not associated with OS (P = .76). The strongest negative predictors of OS were a lack of treatment (P < .01) and recurrence post-treatment (P < .01). Weaker predictors of OS included older age at time of diagnosis (P = .02). Completeness of margins at surgical excision, concurrent disease, treatment type, anatomic location of the SCC (anogenital vs other), and histologic grade of the SCC did not influence OS (P > .1).
Papillomaviruses are a group of ancient, highly host adapted, nonenveloped, double-stranded DNA viruses. 3,4,20 Horses are susceptible to infection by Equus caballus papillomavirus (EcPV), for which 9 types (EcPV-1–9) have been identified. 17 –19,21 EcPV-1, commonly seen in younger horses, is associated with cutaneous papillomatosis and is highly contagious and typically self-limiting. 15 EcPV-2 has been associated with anogenital lesions and neoplastic transformation to SCCs. 2,12,14,16 EcPV-3 is responsible for aural plaques that typically afflict mature horses and are persistent but not considered pre-neoplastic. 15 EcPV types 4, 5, and 6 have been identified in aural plaques, often as part of mixed infections. 7 Additionally, EcPV-4 has been identified in inguinal and vulvar plaques. 17 EcPV-7 has been associated with penile masses. 17 EcPV-8 has been identified in 3 horses with generalized papillomatosis 19 as well as a fourth horse with inguinal plaques, papillomas, and SCC. 24 EcPV-9 was identified in the semen of a horse with a penile lesion. 18
To date, the possible oncogenic mechanisms of EcPV-2 have not been definitively determined but whole genome sequencing of EcPV-2 has shown the presence of both E6 and E7 genes. 25 In contrast to high-risk human papillomaviruses (hr-HPVs) known to cause cervical cancer in women, the EcPV-2 E7 lacks the pRb-binding motif, making E6 the oncogene of major interest in EcPV-2. In hr-HPVs, the E6 protein interacts with various host cell pathways including those of the cell cycle; inactivation of p53 by E6 by signaling for proteasomal degradation is one such example. 8 –11,23,27 The finding of EcpV-2 DNA within SCCs is not, on its own, sufficient to infer that it causes neoplastic transformation. EcPV-2 DNA has been identified in up to 20/70 (29%) of asymptomatic horses (Greenwood et al, 2020, in press). An alternative hypothesis is that EcPV-2 replication is in response to the increased proliferation of neoplastic cells. Thus, confirming that EcPV-2 causes SCC will require further study of the virus’s oncogenic mechanisms.
The focus of our study was evaluation of EcPV-2 infection of Western Canadian horses with mucocutaneous lesions, including SCCs, from a variety of anatomic locations. We analyzed the impact of EcPV-2 status on overall survival (OS) while also addressing confounding factors such as treatment variations, comorbidities, and election of euthanasia for non-SCC-related reasons. In addition, a survey gathered data regarding age of onset, duration of disease prior to treatment, and breeding history.
In contrast to the relatively small body of research on EcPV-2 and SCCs, extensive studies on human papillomavirus (HPV) have shown a clear prognostic impact of HPV status on a subset of cancers including head and neck squamous cell carcinoma (HNSCC). 1,26 HPV-positive HNSCCs have a favorable prognosis when compared with their PV-negative counterparts, affecting both prognostication and therapeutic recommendations. To the authors’ knowledge, only one study has evaluated the prognostic impact of EcPV-2 status on penile and preputial SCCs, and it concluded that there was no impact on overall or disease free-survival times, although this study did not account for variation in treatments. 32
Materials and Methods
Case Selection
A search of the Prairie Diagnostic Services Inc (PDS; Saskatoon, Saskatchewan) database was performed of equine submissions between 2009 and 2014 using the criteria for histologic diagnosis of any of the following terms: “squamous cell carcinoma,” “carcinoma in situ,” or “papilloma.” The search parameters also allowed for the inclusion of the above terms as differential diagnoses listed by the histopathologist; such lesions were reviewed and designated as “other.” The formalin-fixed, paraffin-embedded (FFPE) tissue samples and hematoxylin- and eosin-stained (HE) slides were retrieved. Where more than one block existed for a single submission, a review of the glass slides was used to select the block with the most affected tissue and, where possible, some adjacent, normal epithelium. Information collected from the requisition forms included age, breed, and sex of the horse, anatomic location of the lesion, date that the lesion was sampled, as well as any pertinent historical information.
Age was reported in years and then further categorized as “young” for horses being 10 years or younger and “old” for those 11 years or older. Sex was recorded as either mare, gelding or stallion and categorized as female or male.
All samples with available FFPE material were first subjected to MY09/11 polymerase chain reaction (PCR; described below) and those that were positive were categorized as “PV-positive.” Samples that were negative on MY09/11 PCR were considered “PV-negative.” “PV-positive” lesions were then subjected to EcPV-2-specific RNA in situ hybridization (R-ISH); samples that were positive were classified as “EcPV-2-positive.” R-ISH-negative lesions were further analyzed by EcPV-2-specific PCR to identify potential false-negative R-ISH reactions; samples that were positive were classified as “EcPV-2 positive.” Samples that were MY09/11 PCR-positive, EcPV-2 R-ISH-negative, and EcPV-2 PCR-negative were categorized as “EcPV-2-negative/PV-positive”; these samples were sequenced to identify the PV. As such, samples were considered “EcPV-2-positive” only if they were positive for MY09/11 PCR and any of the following: EcPV-2-specific PCR, R-ISH, or identification of EcPV-2 by sequencing.
PCR and R-ISH
Chemical DNA extraction was performed as described previously. 31 In brief, deparaffinization was performed using xylene and ethanol washes. Overnight incubation in a 56 °C water bath with lysis buffer and proteinase K was followed with sequential extractions using phenol-chloroform, followed by precipitation with sodium acetate and ethanol overnight at −20 °C. Nucleic acid pellets were washed in ethanol, dried, and then resuspended in Tris-EDTA buffer. Extracted DNA was stored at −20 °C until amplification could be performed. Separate, conventional PCR reactions were performed using 2 different primer pairs, both of which target the L1 gene. First, PCR using a broad spectrum papillomavirus degenerate primer, MY09/11, was performed on all samples as previously described. 13 The second PCR reaction used EcPV-2-specific primers, which are described elsewhere by Greenwood et al (2019, in press). In brief, consensus primers targeted EcPV-2 to amplify a 306 bp segment of the L1 gene; sequences were as follows: EcPV-2For 5′-TCCTCCACCAATTTTAAAACCTAT-3′ and EcPV-2Rev 5′-ATCCAAGTCAAGGGAAAG-3′. All amplicons were visualized with UV light applied to an agarose gel with 1.2% ethidium bromide. A sample was considered positive if a band of the appropriate size was visible. Internal negative controls included blank blocks, extraction blanks, and nontemplate controls. The presence of amplifiable DNA was confirmed by a positive PCR reaction using primers for housekeeper gene equine β-actin. Positive controls for both MY09/11 and EcPV-2 PCR reactions included a previously sequenced EcPV-2 sample.
EcPV-2 E6/E7 chromogenic R-ISH was performed on all EcPV-2 PCR-positive tumors for the purpose of identifying the cells infected with EcPV-2 and to rule out possible false-positive PCR reactions. The methods used were similar to those previously described by Zhu et al (2015) 33 and Greenwood et al (2020, in press), which use the RNAScope assay (RNAscope, Advanced Cell Diagnostics, Hayward, CA). The designed probe targeted a 972 nucleotide portion of the E6/E7 region of EcPV-2 (EU503122.1). 33 R-ISH probe hybridization (referred to herein as “signal”) was visualized at 400× under routine light microscopy. The EcPV-2 signal presented as a red, predominantly nuclear stain, and a sample was considered EcPV-2-positive if a signal could be clearly visualized within at least one epithelial cell. Whenever present, adnexa and adjacent normal epithelium were also evaluated for the presence of R-ISH signal.
For tissues that were positive on MY09/11 PCR (PV-positive) but negative on both EcPV-2-specific R-ISH and -PCR (ie, PV-positive/EcPV-2 negative), DNA was amplified in triplicate using MY09/11 PCR and sent for sequencing (Macrogen, Seoul, South Korea). Samples were assembled using Staden Package software program and BLAST analysis performed for identification against Genbank entries.
Histologic Grading
The histologic grading system was initially published for human penile SCCs 5 and adopted for equine mucocutaneous SCCss. 30,32 Under this system, grade 1 represents the most well-differentiated tumors, grade 3 the most anaplastic, and grade 2 all those that fail to classify as grades 1 or 3. Under this system, a tumor was assigned a grade based on the most abnormal cells identified, irrespective of their overall percentage of the tumor cell population. The 77 HE-stained slides of available SCCs and CISs were reviewed by a single ACVP board-certified veterinary anatomic pathologist (BW) who was blinded to the original histologic diagnoses and patient information.
Retrospective Survey and Statistical Analysis
A 10-question survey (see Supplemental Table S1) was sent to each referring veterinarian responsible for the submitted biopsy samples identified by the database search. The survey addressed historical factors such as duration of the lesion, number of lesions, prior treatment, considerations pertaining to the decision to treat, type of treatment selected, and outcome following histopathologic diagnosis (eg, post-treatment recurrence, survival time, and cause of death or euthanasia). In addition, veterinarians were asked to provide information regarding the presence of in-contact animals with similar lesions and historical information regarding the breeding status of the affected animal. Hard copy surveys were mailed to each clinic, and two follow-up attempts were made, four weeks apart, by email and telephone. For those submissions that listed the primary referring veterinary clinic as the Western College of Veterinary Medicine Veterinary Medical Clinic (WCVM VMC), information was gathered directly from the hospital’s archived records. Similarly, where horses had been referred to the WCVM VMC by general practitioners, data were collected from the WCVM VMC database in addition to sending a survey to the primary care veterinarian.
Descriptive statistics were performed using Microsoft Excel (2017) to analyze the proportion of PV and EcPV-2 positivity by histologic diagnosis, anatomic site, age, breed, and sex categories. OpenEpi software (www.openepi.com) was used for χ2 calculations to compare the proportion of EcPV-2 positivity of tissue samples by age categories and anatomic sites and to compare results from the current study to previous related publications. Significance was set as P < .05.
Survival analysis was used to analyze the time to event data, given that there were a large number of horses lost to follow-up during the time frame covered by the retrospective study time period. Outcomes for the analysis include (1) “study-time,” which was the time from the submission of the diagnostic sample to the end of the study, or date of death or loss to follow-up, and (2) “owner-time,” which was the time from when the owner or referring veterinarian first noted the lesions to the end of the study, or date of death or loss to follow-up. The data were analyzed using the test for equality of survivor functions (log rank test) in STATA 15 (P = .20 for consideration for model building). A Cox proportional hazard model with a single predictor using a χ2 test was used for analysis of continuous predictors (age and duration). A visual assessment of the assumption of proportionality of the survivor functions for categorical predictors involved graphing the Kaplan-Meier survival estimates and global test for proportional hazards for continuous predictors. Since a large number of cases were lost to follow-up shortly after the diagnostic sample submission, a few different datasets were assessed: (1) all deaths where cause of death could not be confirmed (unknown deaths) or censored cases for which little information existed to confirm the time to event were excluded (N = 36); (2) all censored cases for which little information existed to confirm the time to event were excluded but the unknown deaths were treated as either (a) all censored or (b) all failures (N = 40); and (3) all censored cases for which little information to confirm time to event were given the value of 7 days (lost shortly after the diagnostic submission) and the unknown deaths were treated as either (a) all censored or (b) all failures (N = 54). To account for possible confounders, which included horse’s age at the time of histologic diagnosis of SCC (categorized dichotomously by greater than 10 years of age), sex (gelding, stallion, or mare), anatomic site (anogenital vs other) and margins (complete or incomplete), and duration (only for study-time outcome), a stratified log-rank test was used.
Logistic regression (with extension to exact logistic regression for some variables) was used to assess what factors were associated with EcPV-2 status (positive or negative). Variables of anatomic site and histologic diagnosis did not have enough cases for each of the categories and were condensed to dichotomous variables of genital site and SCC. Each variable was assessed independently with the outcome, using a conservative P value (.20) for further model building. Potential confounders included age category and sex using a 20% change in the risk factor of interest’s coefficient. Model building used a forward stepwise selection process (using exact logistic regression) using a P value of .05 for inclusion.
Results
Clinical and Pathologic Findings
The initial database search identified 123 submissions that met inclusion criteria, representing 115 distinct lesions once repeated biopsies were excluded. Of the 115 identified lesions, 14 were excluded because FFPE blocks were not available or DNA could not be amplified (β-actin PCR negative). The remaining 101 submissions were included in the study. Ages of horses were available for 89/101 submissions and ranged from 2 to 30 years old, with a mean of 14.5 years and a median of 14 years. Age category distribution was young in 30% (27/89) and old in 70% (62/89). Information regarding breed (including crosses) was available for 82/101 submissions and included the following: Quarter horse (29), American paint (17), Appaloosa (13), Warmblood (5), Arabian (4), Draft (4), Tennessee walker (4), Thoroughbred (2), and one each of Morgan, Donkey, Miniature horse, and Pony. There were 81 males (72 geldings, 9 stallions), 19 mares, and 1 of unknown sex.
Table 1 summarizes the included submissions by anatomic location, histologic diagnosis, and PV and EcPV-2 status. Additional data are presented in Supplemental Table S2. Of the 101 submissions, 100 had known anatomic locations and were as follows: penis or sheath (51), vulva (8), perianal (2), periocular (22), “other” (17). “Other” locations included those described as arising from the face, nose, shoulder, coronary band, frog, lower lip, tail head, muzzle, ear, mouth, gingiva, pastern, and rectum.
Prevalence of Equus caballus Papillomavirus Type 2 (EcPV-2) in Squamous Cell Carcinomas (SCC), Carcinomas In Situ (CIS), Papillomas (P), and From Various Anatomic Locations in 101 Horses.
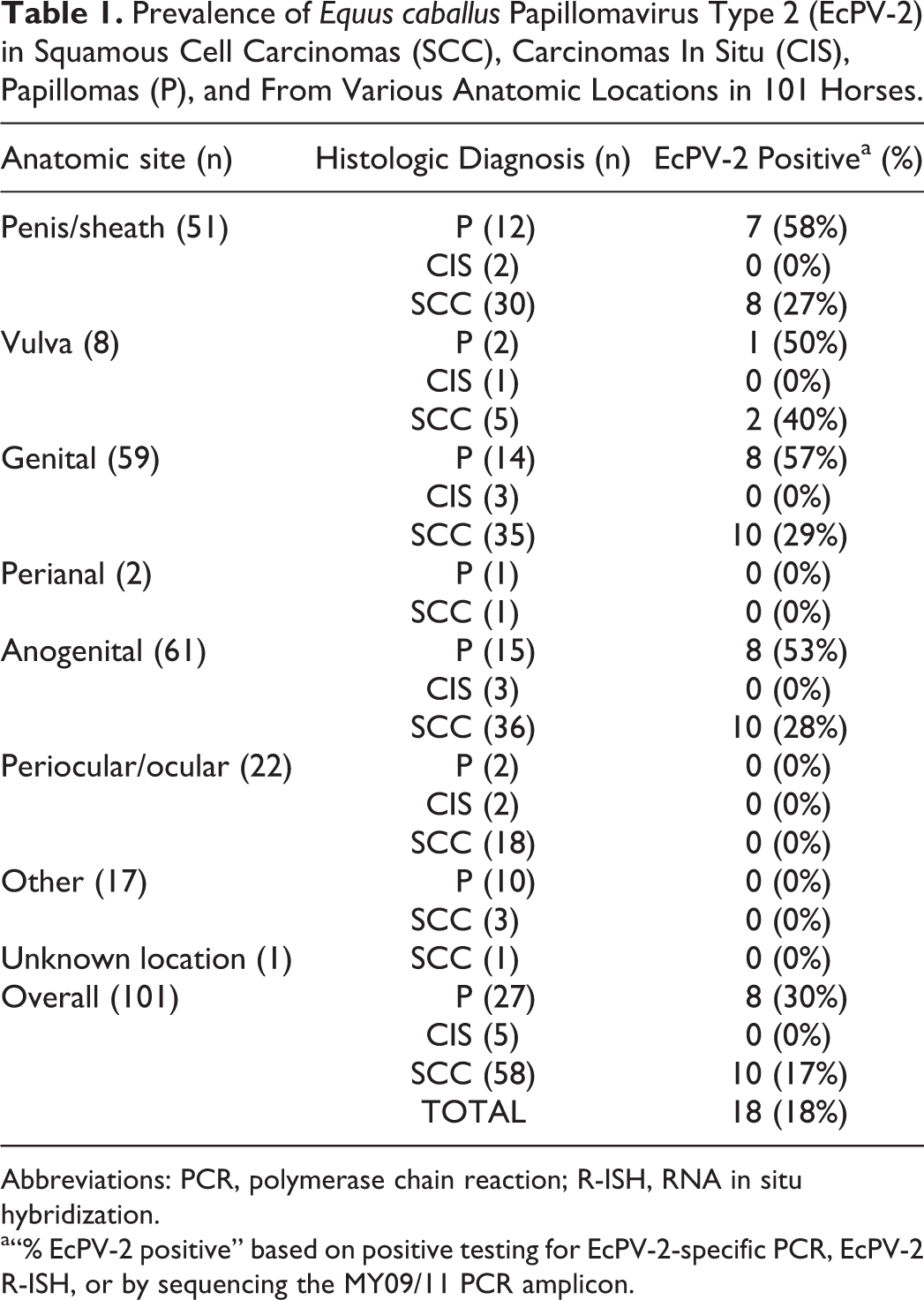
Abbreviations: PCR, polymerase chain reaction; R-ISH, RNA in situ hybridization.
a“% EcPV-2 positive” based on positive testing for EcPV-2-specific PCR, EcPV-2 R-ISH, or by sequencing the MY09/11 PCR amplicon.
Histologic diagnoses included 27 papillomas, 5 CISs, 58 SCCs, and 11 “other.” “Other” lesions were included whenever a differential diagnosis of squamous cell carcinoma had been included in the pathologist’s histology report. Lesions classified as “other” included basosquamous carcinomas (1), hyperplastic (2), inflammatory polyp (1), normal (1), inflammation (2), sarcoid (1), soft tissue sarcoma (1), fibroma (1), dyskeratosis (1).
Detection of Papillomaviruses by PCR
PV DNA was identified in 13/27 (48%) papillomas, 0/5 CISs, 13/58 (22%) SCCs, and 1/11 “other lesions.” When lesions were categorized by anatomic site, 16/51 (31%) of penile lesions contained PV DNA. Of these, 15/16 were identified as EcPV-2 by PCR, while the other case (a SCC) was EcPV-2-negative and the sequence of the MY09/11 PCR amplicon had no significant match in Genbank. Three of the 8 vulvar lesions were PV-positive and all were identified as EcPV-2 by PCR. Neither of the 2 perianal lesions contained PV DNA. Two of the 22 periocular/ocular lesions were PV-positive, and the sequence of the MY09/11 PCR amplicons were identified as Deer PV (1) and Feline PV-3 (1). The remaining 17 lesions from “other locations” included 6 PV-positive lesions, which were identified based on sequencing the MY09/11 PCR amplicon as Deer-PV (1 SCC from the nose) or EcPV-1 (papillomas from the face [2], shoulder [1], and ear [1]), and HPV-107 from an oral ossifying fibroma.
Ten of the 58 SCCs (17%) were positive for EcPV-2, including 8/30 (27%) from the penis and prepuce, 2/5 from the vulva and none from perianal (n = 1) or periocular-ocular (n = 18) tissues, “other” sites (n = 3), nor the single lesion from an unknown location. When EcPV-2% positivity of SCCs from the current study was compared to previous reports, it was reduced, but a statistical difference existed only for a subset of studies (Supplemental Table S3). The current study observed an EcPV-2% positivity of SCCs that was similar (>0.05) to that previously reported in normal tissues (Greenwood et al, 2020, in press; Table 2).
Comparison of EcPV-2 Positivity of Squamous Cell Carcinomas (SCC) in Western Canadian Horses’ Normal Tissues From Canadian Horses and SCCs From Non-Canadian Horse Populations.

When we compared EcPV-2 positivity by age category (all lesions, any location), there was no significant difference between young horses (3/27; 11%) and old horses (14/62; 33%), P = .22). When considering SCC submissions from horses of known ages, SCC was more likely in older horses (48/58; 83%) than younger horses (10/58; 17%), P < .001. However, there was no significant difference in EcPV-2% positivity of SCCs from older horses (8/40; 20%) and younger horses (1/9), P = .6.
EcPV-2 E6/E7 R-ISH
Of the 27 samples identified as PV positive by MY09/11 PCR, 17 were positive by EcPV-2 E6/E7 R-ISH and 10 were negative. The 10 cases that were MY09/11 PCR-positive but EcPV-2 E6/E7 R-ISH-negative were tested by EcPV-2-specific PCR; one case was EcPV-2 positive and 9 were negative. Of those 9 samples (which were MY09/11 PCR-positive, EcPV-2 E6/E7 R-ISH-negative, EcPV-2 PCR-negative), the sequences of the MY09/11 PCR amplicons matched that of Deer PV (2), EcPV-1 (3), Feline PV-3 (1), and HPV-107 (1), and one case had no significant match. To evaluate the specificity of EcPV-2 E6/E7 R-ISH, 5 known PV (MY09/11) PCR-negative SCCs from a separate study were tested by EcPV-2 R-ISH and all were negative.
All samples that were R-ISH positive contained at least one intralesional epithelial cell with a diffuse nuclear signal. However, the distribution of the R-ISH-positive epithelial cells within the lesions was usually random and multifocal
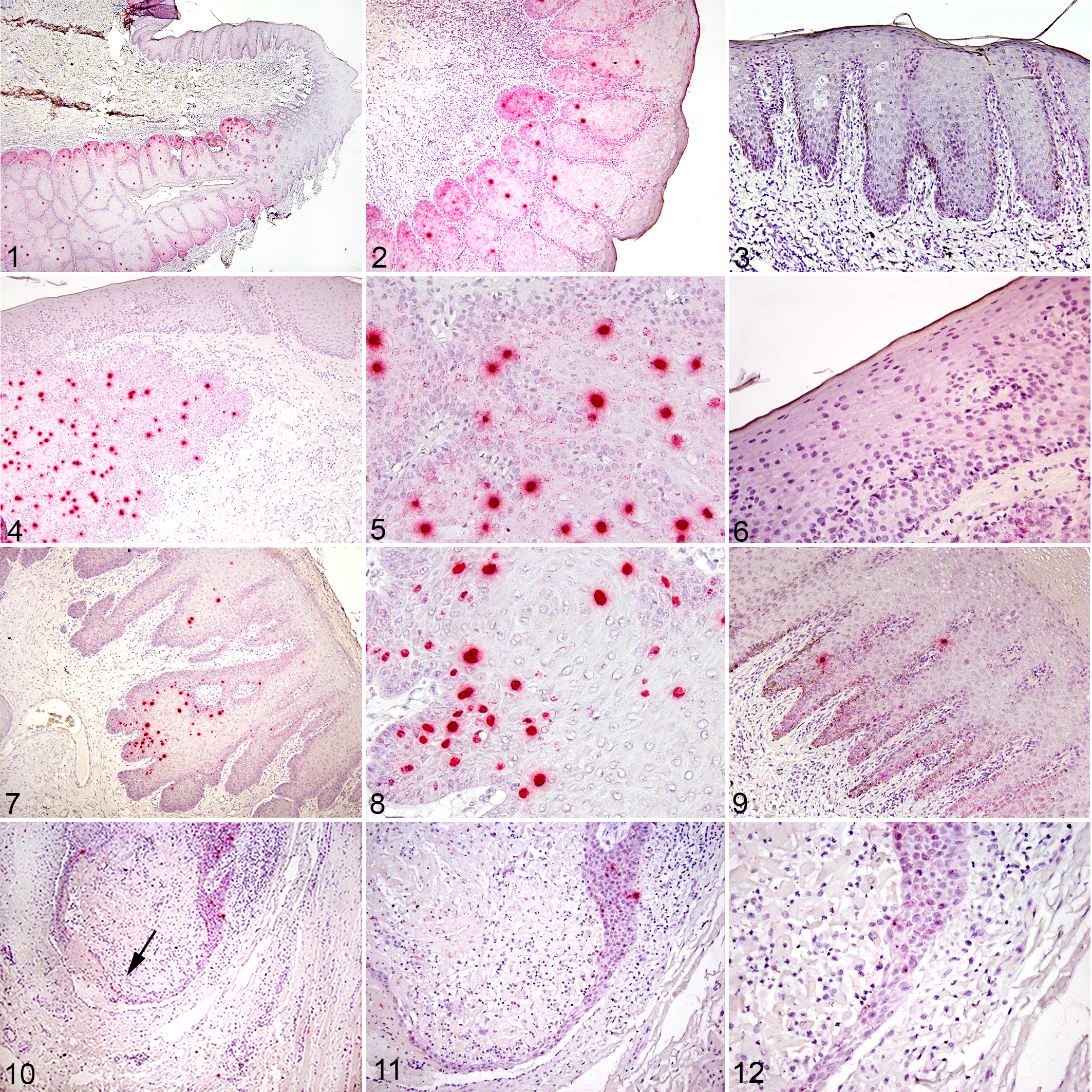
EcPV-2 positivity is multifocally distributed throughout the papilloma.
Retrospective Survey
The 123 submissions represented 115 different lesions from 44 veterinary clinics in British Columbia (3), Alberta (20), Saskatchewan (17), Manitoba (3), and Yukon Territory (1). Survey responses were obtained for 91/115 (79%) horses. For 24 submissions, surveys were not returned (21), returned with little/no information (2), or the clinic was no longer in business (1).
Survey responses that included known mortality status, and results of EcPV-2 testing were available for 27/57 SCCs and these data are shown in Table 3. This subset of SCCs had an average age of 15.3 years (range, 4–27 years) and included 11 Quarter horses, 10 American paints, 8 Appaloosas, 2 draft horses, 2 Tennessee walkers, 2 Warmbloods, and one each of Morgan, Pony, and Arabian.
Median Overall Survival Time (OS) of Horses With Squamous Cell Carcinoma by EcPV-2 Status and Anatomic Location.

Abbreviations: PCR, polymerase chain reaction; R-ISH, RNA in situ hybridization; SCC, squamous cell carcinoma.
aEcPV-2 positivity is based on positive testing using EcPV-2-specific PCR, EcPV-2 R-ISH, or by sequencing the MY09/11 PCR amplicon.
b“Median OS EcPV-2 positive” refers to the median number of days, as measured from the time of initial perception of the SCC, by either the owner or veterinarian, to death of the horse, and for which the SCC was positive for EcPV-2 DNA.
c“Median OS EcPV-2 negative” refers to the median number of days, as measured from the time of initial perception of the SCC, by either the owner or veterinarian, to death of the horse, and for which EcPV-2 DNA was not identified.
d Other denotes a single SCC from each the nose and gingiva.
Of the cases with SCC, none of surveys identified other in-contact horses as being also afflicted. Multifocal distribution of SCCs was not uncommon (15/43). Comorbidities were identified at the time of histologic diagnosis of SCC in 10/28 horses and included laminitis, pituitary pars intermedia dysfunction, navicular disease, recurrent airway obstruction, dental disease, sinus cysts, lameness, heart disease, and sarcoids. The most common treatment of SCCs was surgical excision alone (21/46). Surgery with additional treatment was reported for 14/46 cases and included modalities such as cryotherapy, cisplatin, 5-fluorouracil, and systemic piroxicam. A single SCC was treated with cisplatin alone. Ten of 46 cases were not treated following histologic diagnosis, and the most common reasons were the financial limitations of the owner, perceived low value of the horse., advanced age of the horse, and the veterinarian’s opinion that the invasiveness of the disease made surgical treatment difficult or that recurrence was likely and the prognosis poor. Of the horses that were treated for SCCs and for which follow-up information was available, 14/17 reported no recurrence by the end of the animal’s life or at the time of survey closure.
Of the 30 horses with SCC and a known mortality status, 15 had been euthanized or died by the time of the survey closure. Of the 11 with a known cause of death, 10 were related to SCC. Results for the owner-time, herein referred to as overall survival (OS), for horses with SCCs and for which EcPV-2 status was known are summarized in Table 3. The median OS for horses with SCC was 1245 days. Horses with SCC arising from the periocular and ocular tissues had the longest median OS (1474 days) followed by penis/prepuce (1468 days), vulva (1199 days), and “other tissues” 183 days.
Survival Analysis and Logistic Regression
Only survival analysis for the second scenario with OS is reported here, in which all censored cases for which little information existed to confirm the time to event were excluded, but the unknown deaths were all treated as failures (N = 40). In the Kaplan-Meier analysis, the difference in OS was significant for failure to receive treatment (P = .003) and recurrence following treatment (P = .004) but not anogenital location (P = .42). Weaker predictors of OS, further only considered as cofounders of OS, included the horse’s age at the time of histologic diagnosis of SCC (P = .02), completeness of surgical margins at time of excision (P = .17), and sex (P = .14). After accounting for age and sex, differences in OS by failure to receive treatment was no longer significant (P = .13); and recurrence following treatment was also no longer significant (P = .18). Factors that had no difference in OS for horses with SCCs included EcPV-2 status (P = .76), histologic grade (P = .21), and comorbidities (P = .98). Additionally, when only those horses that were treated were considered (n = 32), there was no difference in OS when treatment type was considered (surgical alone or with adjunctive therapy; P = .57). Duration prior to histological diagnosis of SCCs was considered as a variable in the “study-time” model and was only a weak predictor of OS (P = .07). In addition, there was little difference in the overall conclusions of important variables based on the owner-time model (that includes duration) and study tie models (results not presented). The completeness of surgical margins at excision was also a weak predictor of OS (P = .17).
Logistic regression analysis identified anatomic location of SCCs as the only predictor of EcPV-2 status (P < .001), as only genital SCCs were EcPV-2-positive in the current study. Breeding history was not predictive of EcPV-2 status (P = .13).
Discussion
In the current study, EcPV-2 was only identified in genital lesions, and in these sites, EcPV-2% positivity was greater in papillomas (57%) as compared with SCCs (28%). We found that 7/12 (58%) penile papillomas and 8/30 (26%) penile SCCs were EcPV-2 positive. This is less than what has been previously described (Lange et al 2012), which reported 100% of penile papillomas and SCCs contained EcPV-2 DNA. 16 Possible explanations for this disagreement include geographic variability and differences in methodology. The Lange et al 16 study included Swiss horses and used 2 broad-spectrum primers targeting E1 and L1 genes (followed by sequencing for identification), thus increasing the opportunity for an EcPV-2-positive result. The absence of EcPV-2 in nongenital SCCs is consistent with prior studies. 22,25 EcPV-2-positive SCCs have been reported from non-anogenital sites including the oropharynx 12 or metastasis to draining lymph nodes or other distant sites, 2,12,28 none of which were included in the current study. The low number of CIS sampled may account for not identifying EcPV-2 in any of these lesions.
The large number of papillomas that were negative for PV nucleic acids on PCR is likely due to 2 factors: the nature of FFPE tissues and the affinity of the MY09/11 primer. Treatment with formalin and embedding with paraffin have a negative impact on nucleic acid integrity as does extraction using phenol-chloroform, 6 and, as such, the current study is likely underestimating the prevalence of PV and EcPV-2 infection. The use of FFPE tissues was important in our study, however, as it allowed for comparison of results to other publications on SCCs as well as our own work on normal equine tissues. The MY09/11 primer is a degenerate primer with an affinity for detecting mucosal-adapted PVs, such as EcPV-2. Cutaneous-adapted PVs, such as EcPV-1 and -3 and BPV-1 and -2, may have been missed using only the MY09/11 degenerate broad-spectrum primer; these would have been better detected using a consensus pair degenerate primer such as FAP59/64. For the purpose of this study, however, our interest was in detecting EcPV-2.
We found a lower prevalence of EcPV-2 infection in genital SCCs from Canadian horses as compared to some previous reports of non-Canadian horses. 28 This may reflect a difference in methodology, horse populations, host or environmental factors (UV exposure), or geographic distribution of the virus. It has been proposed that EcPV-2 infection plays a causal role in the development of genital SCCs because the prevalence of EcPV-2 is higher in tissues from diseased horses as compared to those sampled from healthy horses. 2,12,25,29 However, when EcPV-2 prevalence in SCCs in the current study was compared to the prevalence in comparable normal tissues taken at necropsy from asymptomatic Canadian horses (Greenwood et al, 2020, in press), they were statistically similar. In that study, we did, however, report an increased proportion of EcPV-2 positivity as compared to prior studies on asymptomatic horses and reasons cited included differences in sampling methods. Prior studies sampled “normal” horses using cytobrush, swabs or bodily fluids as sources of nucleic acids, which do not capture infected basal cells of stratified epithelium where a quiescent infection is most likely to be present. Additionally, noncomparable populations were used in some previous studies. For example in Knight et al, normal horses in New Zealand were compared to diseased tissues from horses in New York. 12 In contrast, in the current study, we compared surgical submissions of SCCs to full-thickness tissue samples (not superficial swabs) of necropsied asymptomatic horses, both from Western Canadian horses. The finding of similar EcPV-2 prevalence in normal tissues as compared to SCCs does not preclude the possibility that the virus plays a necessary but insufficient role in the development of neoplasia, similar to that of high-risk HPVs in human neoplasia. It is possible that the etiopathogenesis of EcPV-2-positive SCCs is like that of HPV-associated neoplasia in humans: asymptomatic infection is more frequent than lesion development, and neoplastic transformation is not inevitable as it requires a combination of host and environmental circumstances. Conversely, the current results do not rule out the alternative theory that EcPV-2 infection of SCCs represents only viral replication within a proliferating cell population and that the virus is not responsible for the transformation of host cells.
SCCs were more frequently submitted from older horses than younger horses, and age was a predictor of OS. However, there was no significant difference in EcPV-2 positivity when age was considered for SCCs alone or in combination with papillomas and carcinomas in situ. If EcPV-2 infection is no more likely to occur in older horses than younger ones, one must consider that either many infections are cleared or that horses are infected very early in life. The concept that EcPV-2 infection is common is supported by our previous study (Greenwood et al, 2019, in press) as is the possibility that horses can develop infection early in life, even in utero. The idea that many PV infections are likely cleared is not novel as this is well-described for HPV-associated diseases. 4
The most commonly identified PV infection in the current study was EcPV-2; however, a small number of non-equine-adapted PVs were identified including Feline PV-3, Deer-PV, and HPV 107. The significance of these findings is unclear.
EcPV-2 status of SCCs was not associated with OS in horses of this study, even when treatment was accounted for. In this respect, the current study agrees with the only other publication on the topic of prognostic impact of EcPV-2 status on SCCs. 32 This is in contrast to what is known about certain human SCCs (eg, HNSCC) where HPV status has a significant impact on both therapeutic recommendations and prognostic outcome. 1 However, the literature on HPV-associated SCCs is vast and larger studies on equine SCCs are warranted to identify subcategories of SCCs that might be influenced by PV status. One of the limitations of the current study was that despite a strong survey response rate, many horses were lost to follow-up at the level of the submitting practitioner. Larger, prospective studies may be able to identify smaller differences pertaining to EcPV-2 status on OS than we could detect.
Unlike the study by van den Top, 32 we did not demonstrate an impact of histologic grade of SCC on OS. The authors opted to select the same grading system for equine mucocutaneous squamous cell carcinomas that was previously used in both the van den Top study 32 and a review paper. 30 However, the relatively subjective nature of the grading system used may have contributed to the disagreement between the current study and van den Top 32 as it pertains to the impact of histologic grade of SCCs on OS.
Factors identified in the current study as affecting OS included whether treatment was administered, recurrence of the SCC following treatment, age of the horse at time of diagnosis of SCC, and completeness of surgical margins at excision. Interestingly, in the current study, OS was similar irrespective of whether adjunctive modalities such as cryotherapy, chemotherapy, or systemic NSAIDs (nonsteroidal anti-inflammatory drugs) were administered in addition to surgical excision (P = .57). The completeness of surgical margins at excision was also a very weak predictor of OS (P = .17). Duration of a lesion prior to treatment was not a predictor of OS. However, given that owner, or vet, perception of when a lesion began was considered t = 0 in the study-time model, and we recognize that perceptions vary among observers, it is possible that we have failed to recognize the impact of duration in our analysis.
The finding that only SCCs arising from the penis, prepuce, or vulva contained EcPV-2 nucleic acids might suggest that the virus is sexually transmitted. However, breeding status did not predict EcPV-2 positivity of SCCs. Only 5/36 horses with a SCC and a positive EcPV-2 status had a history of ever having been bred. When EcPV-2% positivity of SCCs was compared by breeding history, there was no significant difference between nonbreeders (2/22; 9%) and breeders (2/5; 40%), P = .14, suggesting that if sexual contact is a means of transmitting EcPV-2, it is likely not the only route. This is perhaps not surprising considering that in the authors’ other study, EcPV-2 nucleic acids were identified in a variety of normal tissues from asymptomatic horses, including those from fetuses (Greenwood et al, 2020, in press). It is possible that some of the horses in the current study were infected vertically or by nonsexual routes. Transmission of EcPV-2 in horses should be further investigated.
Conclusion
EcPV-2 prevalence in SCCs in Western Canadian horses was lower than that reported in some previous studies of American and European populations. The EcPV-2 status of genital SCCs in Western Canadian horses was not associated with OS, even when treatment type was accounted for; this finding agreed with the only other published study of the prognostic impact of EcPV-2 infection on penile and preputial SCCs. 32 The most important prognosticating factors in equine genital SCC appeared to be whether treatment was provided, and the presence of recurrence posttreatment. However, these factors were strongly influenced by both sex and age of the horse at the time of diagnosis. OS was not influenced by whether treatment consisted of surgical excision alone or included adjunctive therapies. The duration of the lesion prior to histologic diagnosis did not influence OS; this was consistent with the general biological behavior of SCCs being locally invasive but slow to metastasize. Given our finding that the prevalence of EcPV-2 nucleic acids in healthy horse tissues from a comparable population (Greenwood et al, 2020, in press) was similar to that reported in SCCs in the current study, it remains unclear what role the virus plays in the development of equine neoplasia. We propose that, similar to hr-HPVs in human neoplasia, EcPV-2 might be a causal but insufficient factor in the etiopathogenesis of equine genital SCCs and that additional host and environmental factors are likely also important. In contrast to human HNSCC, there did not seem to be any value in determining the PV status of equine SCCs for the purpose of treatment planning or prognostication. However, knowing the EcPV-2 status of a horse with a SCC may be of value if other horses are in contact, as it is currently unknown if infected tumors shed virus and it appears likely that nonsexual transmission routes exist.
Supplemental Material
Supplemental Material, Combined_supplemental_materials-Greenwood_et_al - Prevalence and Prognostic Impact of Equus caballus Papillomavirus Type 2 Infection in Equine Squamous Cell Carcinomas in Western Canadian Horses
Supplemental Material, Combined_supplemental_materials-Greenwood_et_al for Prevalence and Prognostic Impact of Equus caballus Papillomavirus Type 2 Infection in Equine Squamous Cell Carcinomas in Western Canadian Horses by Sarah Greenwood, Betty Chow-Lockerbie, Tasha Epp, Cameron Knight, Garrett Wachoski-Dark, Valerie MacDonald-Dickinson and Bruce Wobeser in Veterinary Pathology
Footnotes
Acknowledgements
The authors would like to acknowledge Prairie Diagnostic Services Inc for their provision of archived material and histologic preparations.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for the current study was provided by the Townsend Equine Research Health Fund.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
