Abstract
Findings from polymerase chain reaction–based methods have suggested a role of Felis catus papillomavirus 2 (FcaPV-2) in the development of feline cutaneous squamous cell carcinoma (SCC). However, because polymerase chain reaction cannot localize deoxyribonucleic acid or ribonucleic acid within the lesion, it is difficult to differentiate a coincidental FcaPV-2 infection and a causative association. Given that a key event in the pathogenesis of human papillomavirus–induced cancer is the expression of viral E6 and E7 oncogenes, localization of FcaPV-2 E6 and E7 transcription within neoplastic cells in feline SCCs would support a causative role for this papillomavirus. Therefore, RNAscope in situ hybridization was used to localize FcaPV-2 E6 and E7 transcripts in 18 formalin-fixed paraffin-embedded samples of cutaneous SCC. Positive signals were present within 5 of 9 samples (56%) from ultraviolet-protected sites and 0 of 9 samples from ultraviolet-exposed sites. In the 4 in situ hybridization–positive samples that contained adjacent hyperplastic skin, hybridization patterns in these regions were characterized by intense nuclear signals within the superficial epidermis and punctate signals within the basal epithelial layers. However, within the 5 SCCs, punctate signals were present within all layers of the epidermis, with progressive loss of intense nuclear signals within the superficial epidermis. This hybridization pattern is consistent with unregulated E6 and E7 transcription and decreased viral replication and is similar to the pattern observed in human papillomavirus–induced cancers as they progress from hyperplastic lesions containing productive infections to nonproductive neoplasms. These findings support a causative role for FcaPV-2 in the pathogenesis of feline SCC.
Cutaneous squamous cell carcinoma (SCC) is the most common malignant neoplasm of the skin of cats, accounting for 15% to 48% of feline skin tumors. 7 The majority of SCCs are thought to be caused by chronic ultraviolet (UV) light exposure and develop because of progression from actinic keratosis. These cancers are most common in lightly haired areas such as the pinnae, nasal planum, and eyelids. 7,19 A smaller proportion of feline cutaneous SCCs develop at UV-protected sites. These have been hypothesized to develop as a progression of Bowenoid in situ carcinomas. 7,9 Because Bowenoid in situ carcinomas in cats are thought to be caused by papillomavirus (PV) infection, this suggests that a proportion of feline SCCs could develop as a result of PV infection. PVs are a well-recognized cause of cancer in humans, especially cancers of the cervix and oral cavity. PVs are also recognized to cause some human skin SCCs, usually in association with chronic exposure to UV light or immunosuppression, such as subsequent to organ transplantation. 4,22
PVs are double-stranded deoxyribonucleic acid (DNA) viruses that infect keratinocytes at both mucosal and cutaneous sites. 5 These are highly host- and tissue-specific viruses that have been identified from a wide variety of species. 20 Most often PV infections are asymptomatic, although a small proportion of PV types can result in the development of hyperplastic papillomas (warts) or cutaneous plaques. Rarely, these lesions can progress to cancer. 5 In humans, progression from a preneoplastic lesion to cancer is attributed to unregulated expression of the viral oncogenes E6 and E7. 5 These viral genes are responsible for driving terminally differentiated keratinocytes back into the cell cycle and preventing apoptosis. 5 Thus, unregulated expression of viral E6 and E7 oncogenes is a key event in the pathogenesis of human PV-driven cancer. 5
There is growing evidence that PV infection may promote the development of feline cutaneous SCCs. 10 –13,15,16 A papillomaviral etiology appears more common in SCCs from UV-protected skin, in which PV DNA was detected in 9 of 11 SCCs developing at UV-protected sites compared with just 6 of 19 from UV-exposed skin. 14 Felis catus PV type 2 (FcaPV-2) is thought to cause feline viral plaques and Bowenoid in situ carcinomas and is also the PV type most often identified within SCC. 9,15,17 This virus was identified in 17 of 20 cutaneous SCCs using specific primers for FcaPV-2 16 , and recently, abundant FcaPV-2 E6/E7 transcripts and high PV DNA copy numbers were identified within 20 of 60 feline cutaneous SCCs. 21 These studies, however, all relied on polymerase chain reaction (PCR)–based methods to detect PVs, which does not allow localization of the viral DNA and/or ribonucleic acid (RNA) within the sample.
Therefore, although these data are quite compelling, the question of causality remains. Because cats are often subclinically infected with PVs, 18 and PVs can be amplified from non-SCC skin, 16 PCR alone cannot distinguish among a bystander contaminant, a transient infection, or a biologically relevant infection that drives carcinogenesis. In humans, for a human PV to be causally associated with SCC, there needs to be evidence of (1) viral DNA within tumor samples, (2) active viral oncogene transcription in tumor cells, and (3) interaction of viral oncoproteins with tumor suppressor genes. 3 So far, we have evidence for some of these requirements in feline cutaneous SCC, including demonstration of viral DNA within tumor samples, viral oncogene transcription in SCC samples (but not yet localized to tumor cells), and interaction of FcPV2 E6 and E7 with tumor suppressor genes. 2
This study was initiated to investigate whether active viral oncogene transcription was present in the tumor cells. Therefore, the aim of this study was to detect and localize FcaPV-2 E6 and E7 messenger RNA (mRNA) within feline SCCs. This was done using colorimetric RNAscope in situ hybridization (ISH). Demonstration of active E6 and E7 gene transcription within tumor cells would add to the growing support for a causal role of FcaPV-2 in the development of feline cutaneous SCCs. Furthermore, the hybridization patterns of FcaPV-2 E6 and E7 within the tumors were compared with the patterns observed in human PV-induced cervical lesions.
Methods
Case Selection and Histopathology
Formalin-fixed paraffin-embedded (FFPE) tissue samples of feline cutaneous SCCs and non-SCC skin were selected from archived biopsies from North Carolina State College of Veterinary Medicine Anatomic Pathology Service. Cases were selected on the basis of location on the body, quality of specimen (lack of autolysis and adequate sample remaining), and presence of intact bordering skin. Histologic confirmation of SCCs was determined on hematoxylin and eosin–stained sections from each case by a board-certified veterinary pathologist (J.L.). SCCs were characterized by disorderly keratinocyte differentiation containing markedly dysplastic keratinocytes with invasion through the basement membrane. In addition to the SCC, the tissue sections were also evaluated for the presence of adjacent hyperplastic skin, characterized by epidermal hyperplasia with orderly differentiation and no to minimal epithelial dysplasia.
PCR for FcaPV-2
Because FcaPV-2 is the PV type that is detected most frequently within feline cutaneous SCC, 16 primers specific for FcaPV-2 were used to maximize the sensitivity of the PCR. Conventional PCR was performed using extracted genomic DNA (gDNA) from FFPE tissues. Two 25-μm scrolls were cut from the FFPE tissues, ensuring that the microtome blade was cleaned or replaced between samples to avoid cross-contamination. The gDNA was extracted using a commercially available kit following their recommended protocol (DNeasy Blood and Tissue Kit; Qiagen) and as previously described. 8
The primers JMPF (5′-GTG TCC TGT AGT TCC TAT AC-3′) and JMPR (5′-GTG CCG AAG GTC TCC TCT TC-3′) were used to amplify FcaPV-2 DNA, as previously described. 14,16 The positive control was gDNA extracted from a feline viral plaque. A sample without template DNA served as the negative control. PCRs were performed as previously described. 14,16 PCR for GAPDH was performed to verify adequate quality of extracted gDNA. The primers were felGAPDH-For (5′-GAT TGT CAG CAA TGC CTC CT-3′) and felGAPDH-Rev (5′-AAG CAG GGA TGA TGT TCT GG-3′). Samples of gDNA were diluted with distilled water to yield a concentration of 10 ng/μL. The DNA sample (100 ng) was amplified by PCR using a commercially available kit (HotStar Taq DNA polymerase; Qiagen). The reaction mixture contained 1× buffer, 1.5 mmol/L MgCl2, 200 μmol/L deoxynucleoside triphosphate, 1 U (0.2 μL) taq polymerase, and 1 μmol/L of each primer, for a total reaction volume of 50 μL. All PCRs were performed on a GeneAmp PCR system 2700 thermocycler (Applied Biosystems). An initial activation step of 95°C for 10 minutes was followed by 40 cycles of (1) 30 seconds of denaturation at 95°C, (2) 1 minute of annealing at 58°C, and (3) 1 minute of elongation at 72°C. There was a hold for 7 minutes at 72°C and a final hold at 4°C. Omission of the template DNA served as the negative control. All PCR products were separated by electrophoresis through 1% agarose at 80 V for 1 hour and then stained with the DNA stain GelRed (Fisher Scientific).
Colorimetric ISH
Colorimetric ISH was performed using the proprietary RNAscope ISH method (Advanced Cellular Diagnostics). A 13ZZ probe named V-FPV-E6-E7 was designed and synthesized. This probe targets the sequence between nucleotides 461 and 1154 (E6 and E7 genes) of FcaPV-2 (Genbank EU796884.1). A probe to the bacterial gene dihydrodipicolinate reductase (DapB) served as the negative control. ISH was performed on 4-μm-thick FFPE tissues using the RNAscope 2.5 detection kit according to the manufacturer’s protocol. Successful hybridization requires binding of adjacent probe pairs on the targeted nucleic acid, initiating a cascade that leads to deposition of diaminobenzidine. The final deposit is visualized as brown, punctate precipitate. In one sample, the amount of pigment dispersal precluded definitive localization of hybridization signals. In this case, ISH was performed using the RNAscope 2.5 Red detection kit, which results in deposition of red precipitate.
Characterization of the ISH hybridization patterns was based on an evaluation of human PV E6 and E7 hybridization patterns in human cervical PV-induced preneoplastic lesions. 6 Hybridization patterns were categorized on the basis of the location of positive ISH signals within the neoplasm and within adjacent hyperplastic epidermis. The hybridization patterns were assessed as one of the following categories: dot-like hybridization signals from the basal layer to midepithelium, dot-like hybridization signals throughout the full thickness of the epithelium, and intense diffuse nuclear hybridization signals in the most superficial layer of the epidermis.
Colorimetric ISH Control Assay
Two samples with positive FcaPV-2 hybridization signals were used to investigate the contribution of viral DNA and viral mRNA to the hybridization signal. ISH was performed as above, with the addition of a pretreatment with either RNase A (Qiagen; 10 mg/mL in phosphate-buffered saline, 30 minutes, 40°C) or DNase I (Sigma-Aldrich; diluted 1:50 in 10 mmol/L TrisCl, 2.5 mmol/L MgCl2, 0.5 mmol/L CaCl2 [pH 7.5], 30 minutes, 40°C). The samples were then washed in water 5 times before addition of the targeting probes. Targeting probes included FcaPV-2 E6 and E7 probes and a predesigned control probe to the reference gene human peptidylprolyl isomerase B (PPIB) (Advanced Cellular Diagnostics). ISH was performed using the RNAscope 2.5 Red detection kit, as it was easier to compare signals between RNase- and DNase-treated samples using this method.
Results
Samples
This study comprised 18 SCCs, including 9 from areas of the skin classified as UV-protected and 9 from areas of the skin classified as UV-exposed (Table 1). The mean age of cats with lesions from UV-protected regions was 14 years (range, 11–18 years). The ages of 2 cats were not known. The mean age of cats with lesions from UV-exposed regions was 12 years (range, 3–18 years). Ten non-SCC cases, including inflamed or hyperplastic skin from various locations, were included (cases 19–28; Suppl. Table S1).
Signalment, Lesion Location, and Detection of FcaPV-2 Nucleic Acid for Selected Cases of Feline Cutaneous Squamous Cell Carcinoma.
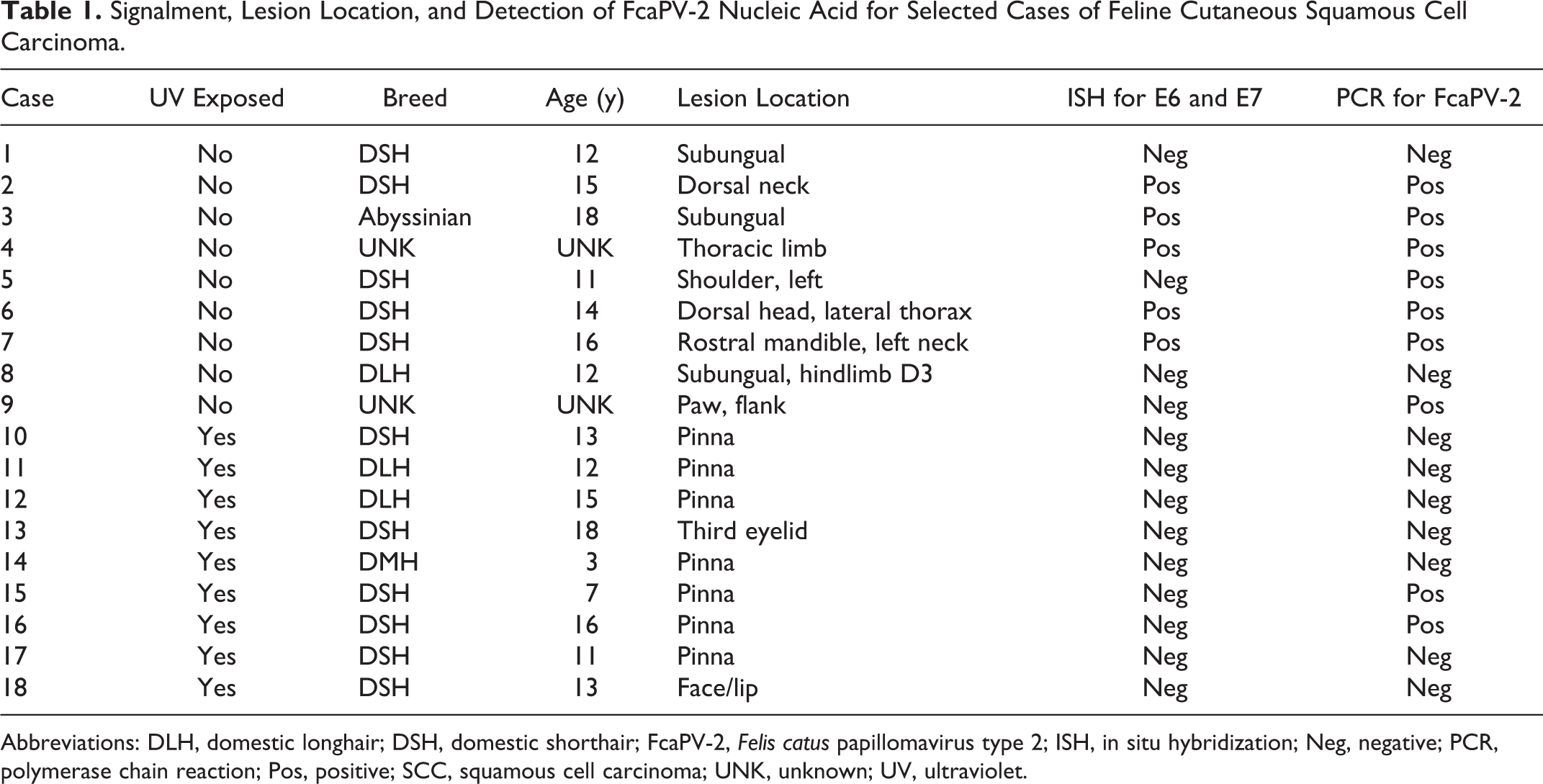
Abbreviations: DLH, domestic longhair; DSH, domestic shorthair; FcaPV-2, Felis catus papillomavirus type 2; ISH, in situ hybridization; Neg, negative; PCR, polymerase chain reaction; Pos, positive; SCC, squamous cell carcinoma; UNK, unknown; UV, ultraviolet.
PCR for FcaPV-2
GAPDH DNA was amplified from all 18 SCCs, indicating that all samples contained amplifiable DNA. FcaPV-2 DNA was amplified from 9 of 18 SCCs (50%) (Table 1). This included 7 of 9 SCCs (78%) from UV-protected sites and 2 of 9 (22%) from UV-exposed sites.
Colorimetric ISH
Colorimetric ISH was performed to identify the cellular localization of FcaPV-2 E6 and E7 nucleic acid. Positive hybridization signals were present within 5 of 9 SCCs (56%) from UV-protected sites, while none of the SCCs from UV-exposed sites were positive. All 5 SCCs with positive ISH hybridization signals also contained FcaPV-2 DNA amplified by PCR. Four SCCs were PCR positive for FcaPV-2 DNA but were negative using ISH. These included 2 SCCs from UV-protected sites and 2 SCCs from UV-exposed sites. Nine SCCs did not have either ISH or PCR evidence of FcaPV-2 infection. No FcaPV-2 hybridization signals were detected in the 10 non-SCC skin samples tested (cases 19–28; Suppl. Table S1).
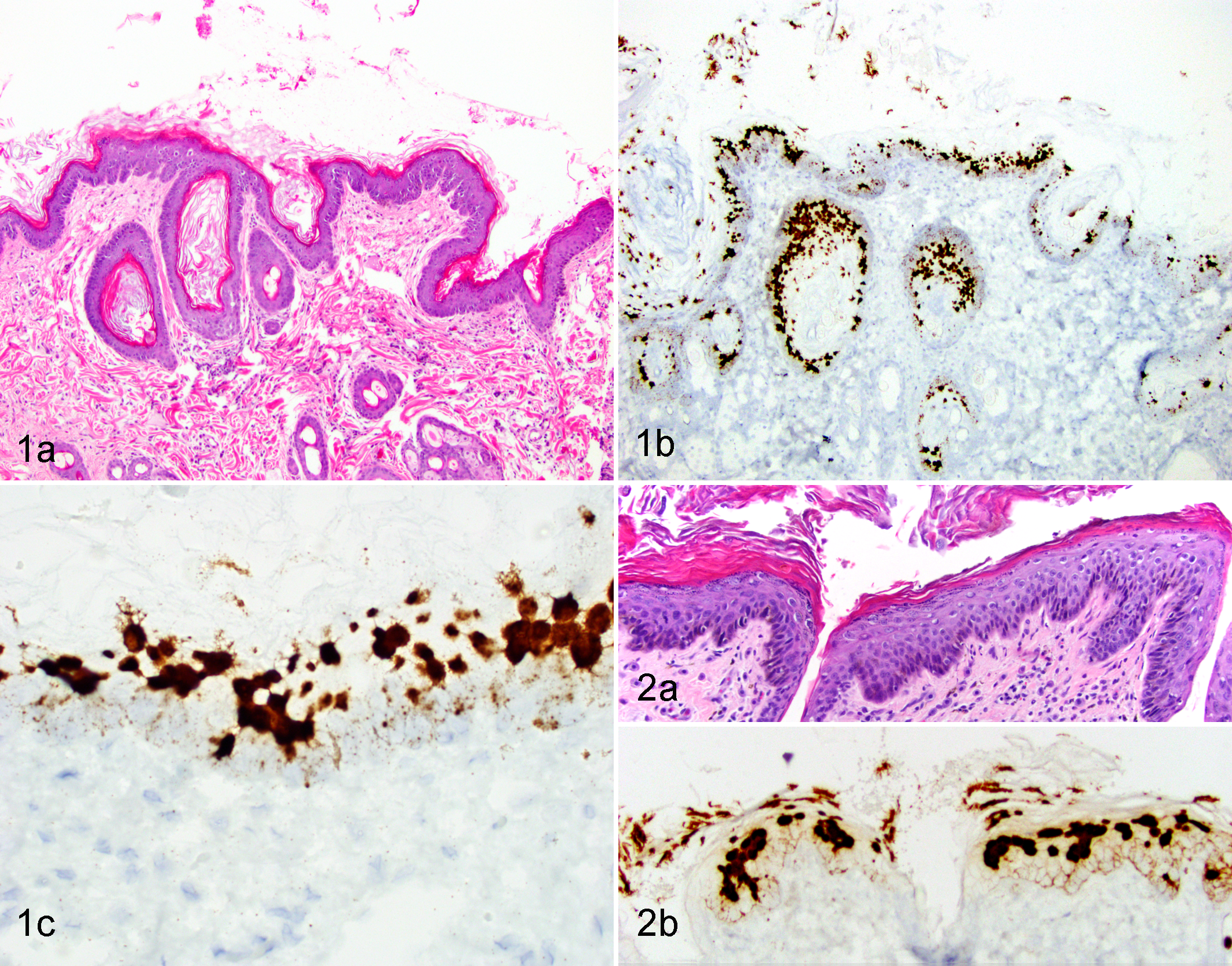
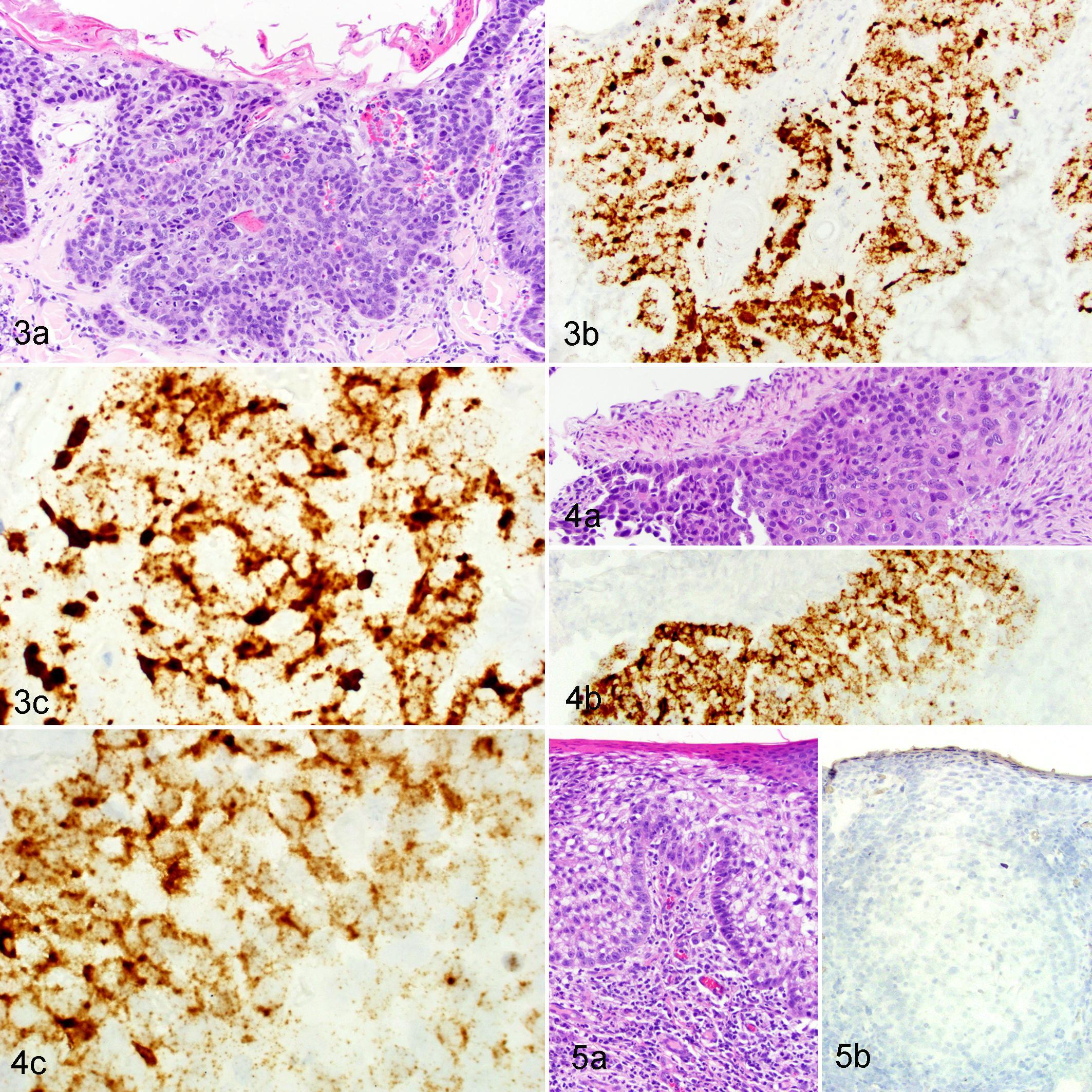
In 4 of the 5 ISH-positive SCCs, the invasive cancer was surrounded by hyperplastic epidermis characterized by focally extensive areas of epithelial hyperplasia with variable hyperkeratosis (Figs. 1A and 2A). The hybridization signal pattern in all 4 regions of hyperplasia was characterized by intense nuclear hybridization signals within the superficial epidermis and punctate nuclear and cytoplasmic hybridization signals limited to the basal epithelial layers (Table 2, Figs. 1B, 1C, and 2B). In contrast to these areas of hyperplasia, the hybridization pattern within the 5 SCCs was consistently cytoplasmic and nuclear punctate signals within all layers of the epidermis, with progressive loss of intense diffuse nuclear signals within the superficial epidermal layers (Table 2; Figures 3 and 4). Adjacent histologically normal skin and the negative control probe were negative in all cases. An SCC that did not show FcaPV-2 hybridization signals is shown in Figure 5.
Scoring System Categorization for ISH-Positive Samples.

Abbreviations: BME+, hybridization signals occurring in all epithelial layers; ≤BME, hybridization signals occurring in the basal and suprabasal layers; ISH, in situ hybridization; NA, not present; Pos, positive; SCC, squamous cell carcinoma; SupD, intense nuclear hybridization signals.
Colorimetric ISH Control Assay
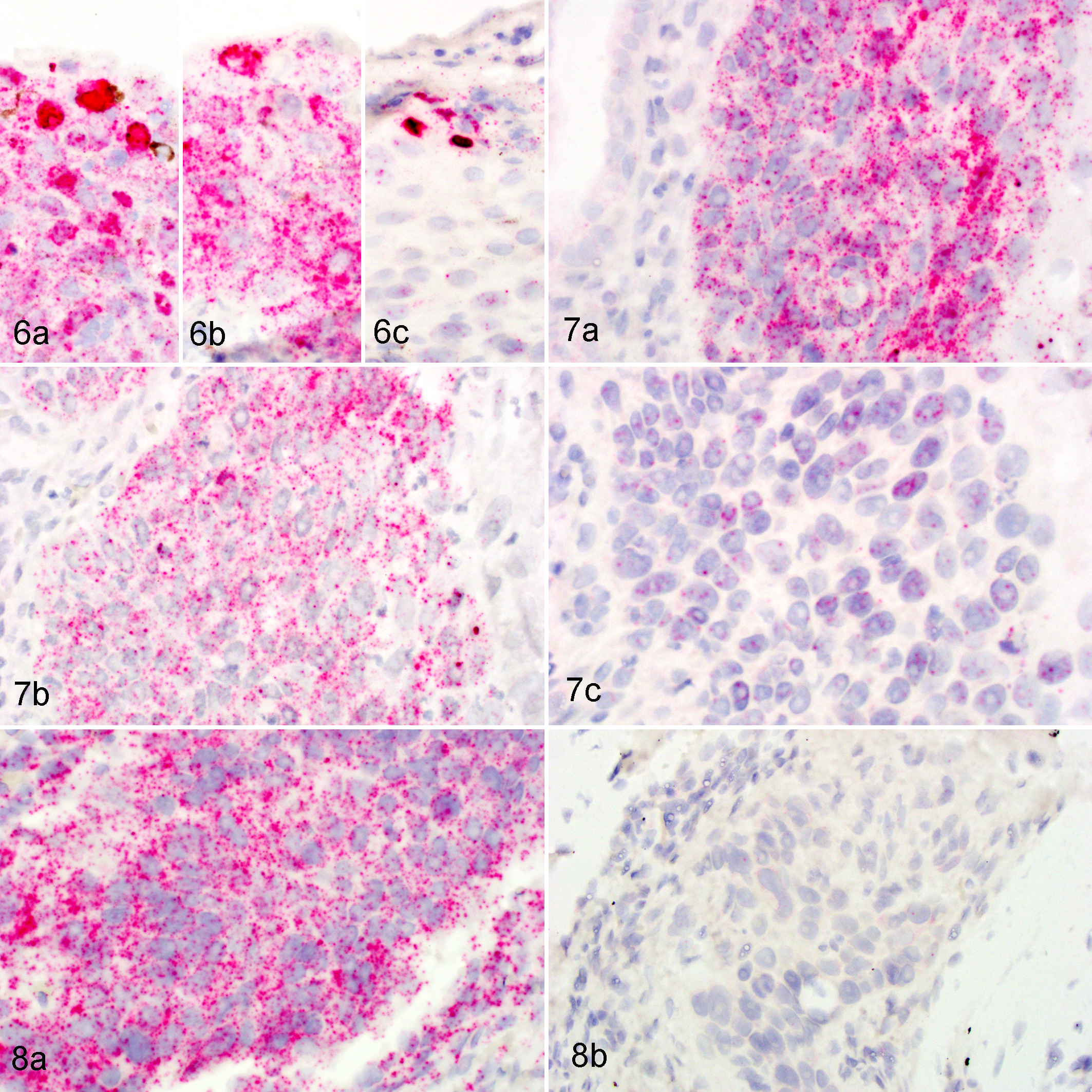
To determine if the hybridization signals were due to detection of mRNA or DNA, 2 tissues were pretreated with RNase A or DNase I prior to hybridization with targeting probes. In a region of epithelial hyperplasia merging into dysplasia, strong diffuse nuclear signals were present within the upper epidermal layers and punctate cytoplasmic and nuclear signals throughout all layers of the epidermis (Fig. 6A). Pretreatment with DNase I resulted in loss of a portion of the diffuse nuclear hybridization signals, but the punctate signals remained (Fig. 6B). Pretreatment with RNase A resulted in the loss of most punctate cytoplasmic and nuclear signals, but the diffuse nuclear staining remained (Fig. 6C). Islands of neoplastic SCC revealed strong punctate cytoplasmic and nuclear signals, which were largely removed with pretreatment with RNase A but not with DNase I (Fig. 7). RNase A pretreatment of samples hybridized for mRNA detection of the reference gene PPIB resulted in loss of most punctate cytoplasmic and nuclear signals (Fig. 8).
Discussion
FcaPV-2 E6 and E7 mRNA was detected within the neoplastic cells of 5 of 18 feline cutaneous SCCs (28%). These results are in accord with a previous study that identified FcaPV-2 E6 and E7 mRNA in 33% of 60 feline cutaneous SCCs using reverse transcription quantitative PCR. 21 Another study, however, detected FcaPV-2 E6 and E7 mRNA within 4 of 6 feline cutaneous SCCs (67%). 2 This higher detection rate may reflect differences in sample location (UV-exposed vs UV-protected regions) or the comparatively small sample size. 2 In the present study, 5 of 9 SCCs (56%) from UV-protected skin contained FcaPV-2 mRNA, whereas FcaPV-2 RNA was not detected in any SCCs from UV-exposed sites. Similarly, in an earlier study, 4 of 6 SCCs (67%) from UV-protected sites were found to contain FcaPV-2 E6 and E7 gene transcription by reverse transcription quantitative PCR. 21 The detection of E6 and E7 mRNA within the SCCs suggests that FcaPV-2 was influencing the growth and regulation of these cells. Therefore, these results provide additional evidence that FcaPV-2 may influence the development and growth of feline cutaneous SCCs, especially those that develop in haired, pigmented skin.
All of the ISH-positive cases contained amplifiable FcaPV-2 DNA, consistent with an active infection. However, FcaPV-2 DNA was also amplified by PCR from 4 cases that did not show any hybridization. These cases may have represented latent infections, in which DNA could be detected using PCR, but no viral transcription was present. Alternatively, low levels of viral transcription could have been present, but this low-level transcription was not detected by ISH.
Two of the SCCs from UV-protected sites did not contain PV DNA or FcaPV-2 hybridization signals. These SCCs appear unlikely to have been caused by FcaPV-2 infection. However, as the techniques used only detect FcaPV-2, it remains possible that other PV types could have influenced the development of these SCCs. Alternatively, nonviral factors may promote the development of a minority of non-UV-induced SCCs in cats.
Only 2 of 9 SCCs (22%) from UV-exposed skin contained FcaPV-2 DNA. The rates of detection were lower than previously observed in a study of 45 UV-exposed SCCs in which PV DNA was detected in 42% of the samples. 14 In the previous study, the highest rates of detection of PV DNA were in SCCs from the nasal planum. It is therefore possible that the inclusion in the present study of mainly pinnal and eyelid SCCs, which were previously identified to have the lowest rate of PV infection, is responsible for the lower rates of detection. None of the SCCs from UV-exposed sites contained detectable FcaPV-2 E6 and E7 RNA. In contrast, E6 and E7 RNA was previously detected by PCR in 5 of 28 SCCs (18%) from UV-exposed sites. 21 Again, this may reflect differences in precise sites of the UV-exposed SCCs used in the 2 studies. Alternatively, it is possible that PCR has higher sensitivity and therefore can detect smaller quantities of RNA than the ISH techniques used in the present study. The failure to detect FcaPV-2 RNA in any of the UV-exposed SCCs in the present study suggests that FcaPV-2 is unlikely to be a common cause of SCCs from the pinnae or eyelids of cats.
In addition to using ISH to determine whether FcaPV-2 nucleic acid was present within neoplastic cells, the localization of E6 and E7 hybridization signals was also determined, because evidence from studies of human cervical cancer has shown that PV hybridization signals change as lesions progress from hyperplasia to neoplasia. 6 In the cats, areas of hyperplastic epithelium surrounding the SCCs consistently contained intense ISH nuclear hybridization signals in the upper epithelial layers and dot-like cytoplasmic and nuclear hybridization signals restricted to the basal and suprabasal keratinocytes. A similar hybridization pattern was reported for the earliest precursor lesion of cervical cancer and is thought to indicate the presence of a productive PV infection. 6 The presence of a similar hybridization pattern in the feline samples therefore suggests that the hyperplastic epidermis surrounding SCCs contains a productive PV infection. This is supported by the high copy numbers of FcaPV-2 that have been previously reported in some feline SCCs. 21
Within the feline SCCs that contained hybridization signals, the hybridization pattern appeared as intense dot-like signals throughout all layers of the epidermis or with progressive loss of nuclear hybridization signals in the upper layers of the epidermis. In humans, this hybridization pattern is seen as preneoplastic lesions progress to true neoplasms. 6 This is thought to be due to the deregulation of the viral genome leading to increased E6 and E7 transcripts throughout all layers of the epithelium. 5 Normally, in a productive infection, E6 and E7 transcription is restricted to basal and suprabasal keratinocytes. Because of the deregulation of normal viral gene expression, such neoplastic lesions contain no or reduced viral replication. Because of the loss of normal regulation of E6 and E7 gene transcription, these lesions would be expected to contain abundant E6 and E7 transcripts using reverse transcription quantitative PCR, which was reported in a proportion of feline SCCs. 21
One potential disadvantage of RNAscope ISH is the possibility that both RNA and DNA could be detected by the hybridization. Therefore, we assessed hybridization patterns after a pretreatment with either DNase or RNase. The intense diffuse nuclear signals represent codetection of RNA and DNA, likely because of the presence of single-stranded forms of viral DNA that occur during upregulated episome synthesis when the DNA load is high, as in cells in the productive phase of infection. 6 The majority of punctate signals within the hyperplastic regions and SCC are consistent with RNA, as these were largely removed with pretreatment with RNase and not with DNase.
In humans, the distinction between an early preneoplastic lesion and a lesion that has undergone malignant transformation is vital in determining the appropriate treatment and prognosis. 1 To help with this differentiation when histology alone cannot provide a clear distinction, evaluation of the RNA expression patterns using RNAscope ISH can be used as a biomarker for transformation. 6 In the present study using cats, there was a clear distinction in the hybridization pattern between hyperplastic lesions and SCC, with the latter having a hybridization pattern consistent with a transformative infection. Identification of feline transformative phase lesions could help determine treatment strategies; however, in veterinary medicine, the high cost of RNAscope ISH precludes its use, at least at this time, as a diagnostic aid. However, it is interesting that the pattern of E6 and E7 hybridization signals within the feline SCCs and the human cervical lesions were so similar. This suggests that, as in cervical cancers, the papillomaviral gene transcription changes as lesions progress from preneoplastic to neoplastic. The altered gene transcription within the SCCs provides further evidence that PVs are not present simply as an incidental infection within the neoplasms.
Conclusions
FcaPV-2 E6 and E7 mRNA was detected within 5 of 18 feline cutaneous SCCs, including 5 of 9 SCCs from UV-protected skin. The presence of FcaPV-2 E6 and E7 transcription within the neoplasms provides additional evidence that PVs can influence the development of these common cancers in cats. The hybridization patterns of E6 and E7 within the feline lesions were similar to that observed in human PV-induced cervical cancers.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985817750456 - Localization of Felis catus Papillomavirus Type 2 E6 and E7 RNA in Feline Cutaneous Squamous Cell Carcinoma
Supplemental Material, DS1_VET_10.1177_0300985817750456 for Localization of Felis catus Papillomavirus Type 2 E6 and E7 RNA in Feline Cutaneous Squamous Cell Carcinoma by Nathan Hoggard, John S. Munday, and Jennifer Luff in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by research funds provided by North Carolina State University, College of Veterinary Medicine.
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
