Abstract
Amyloidosis is classified according to the amyloid precursor protein, and accurate diagnosis of the amyloidosis type may guide appropriate treatment. Immunohistochemistry and Congo red staining are the most frequently used methods used to distinguish types of amyloidosis, but problems with specificity and sensitivity indicate the need for an alternative diagnostic method. In this study, we evaluated laser microdissection-liquid chromatography-tandem mass spectrometry (LMD-LC-MS/MS) for the diagnosis of amyloid light-chain (AL) amyloidosis in animals. Plasmacytomas with amyloid deposits from 15 dogs and 2 cats were subjected to Congo red staining with or without potassium permanganate pretreatment, immunohistochemistry for kappa and lambda light chains, and LMD-LC-MS/MS. Congo red staining was diagnostic in 12 of 17 cases based on resistance to potassium permanganate pretreatment, but in 5 of 17 cases the pretreatment unexpectedly reduced Congo red staining or abrogated the birefringence and a definitive diagnosis could not be reached. Immunohistochemistry detected kappa or lambda light chains in 6 of 17 cases. With LMD-LC-MS/MS, immunoglobulin lambda light chain was detected in all 17 cases. The amyloid signature proteins ApoA-I, ApoA-IV, and ApoE were detected in 9, 1, and 3 of the 15 canine cases by LMD-LC-MS/MS, but not in the feline cases. In conclusion, LMD-LC-MS/MS consistently determined the amyloid type in all examined specimens, while Congo red staining after potassium permanganate treatment and immunohistochemistry were less sensitive tests.
Keywords
Amyloidosis is a group of diseases in which fibrillar insoluble proteins called amyloid are deposited systemically or locally. Amyloidosis is classified according to the amyloid precursor protein. In light-chain (AL) amyloidosis, monoclonal immunoglobulin light chains are produced in excess due to plasma cell proliferative disorders such as multiple myeloma or plasmacytoma, leading to amyloid formation and deposition in a single organ (localized) or multiple organs (systemic). 4,11,20,29,30 Systemic AL amyloidosis is one of the major amyloidoses in humans, 30 and it is commonly associated with multiple myeloma. 29 In animals, AL amyloidosis is usually localized within an extramedullary plasmacytoma. 5,30
Accurate diagnosis of the type of amyloidosis is important for appropriate treatment. 14 AL amyloidosis may be diagnosed by Congo red staining based on its resistant to pretreatment with potassium permanganate, whereas the staining of some other types of amyloid is abolished by this pretreatment. 30 Immunohistochemistry (IHC) for immunoglobulin kappa and lambda light chains is another method of diagnosis, 9,14 but it is neither specific nor sensitive for the diagnosis of AL amyloidosis in human and animals because the immunoglobulin light chains have variable regions leading to changes in antigenicity. 13,14,23,24 Hence, the development of an alternative diagnostic method would be useful for the diagnosis of AL amyloidosis.
In recent years, laser microdissection-liquid chromatography-tandem mass spectrometry (LMD-LC-MS/MS) has been used for diagnosis of human amyloidosis. 12,13,28 The sensitivity and specificity of LMD-LC-MS/MS has been proven in many studies, 12,13 and amyloid precursor protein can be detected from even a small quantity of tissue. 9 Moreover, it is useful to detect not only the precursor protein but also amyloid signature proteins (or amyloid-associated proteins) such as apolipoprotein A-I (ApoA-I), apolipoprotein A-IV (ApoA-IV), and apolipoprotein E (ApoE). 12 In this study, we analyzed AL amyloidosis in dogs and cats by using LMD-LC-MS/MS, IHC, and Congo red staining after potassium permanganate treatment and examined the benefits of LMD-LC-MS/MS over the other methods.
Materials and Methods
Samples and Histopathology
This study used 17 formalin-fixed paraffin-embedded (FFPE) specimens from 15 dogs and 2 cats that had been diagnosed as plasmacytoma with amyloid deposition (Table 1). Specimens were processed routinely and 3-μm-thick histologic sections were stained with hematoxylin and eosin or Congo red with or without potassium permanganate pretreatment as described previously. 27 Amyloid deposition was confirmed by Congo red staining under polarized light. Amyloid deposits were considered to be sensitive to potassium permanganate when the deposits lost their affinity for Congo red after potassium permanganate treatment, or resistant if there was staining following pretreatment. For the histological analyses, the following were examined: the pattern of amyloid deposition, the degree of plasma cell proliferation, and the presence or absence of inflammation, calcification, and multinucleated giant cells.
Clinical and Histological Findings in 17 Cases of Plasmacytoma With Amyloidosis.
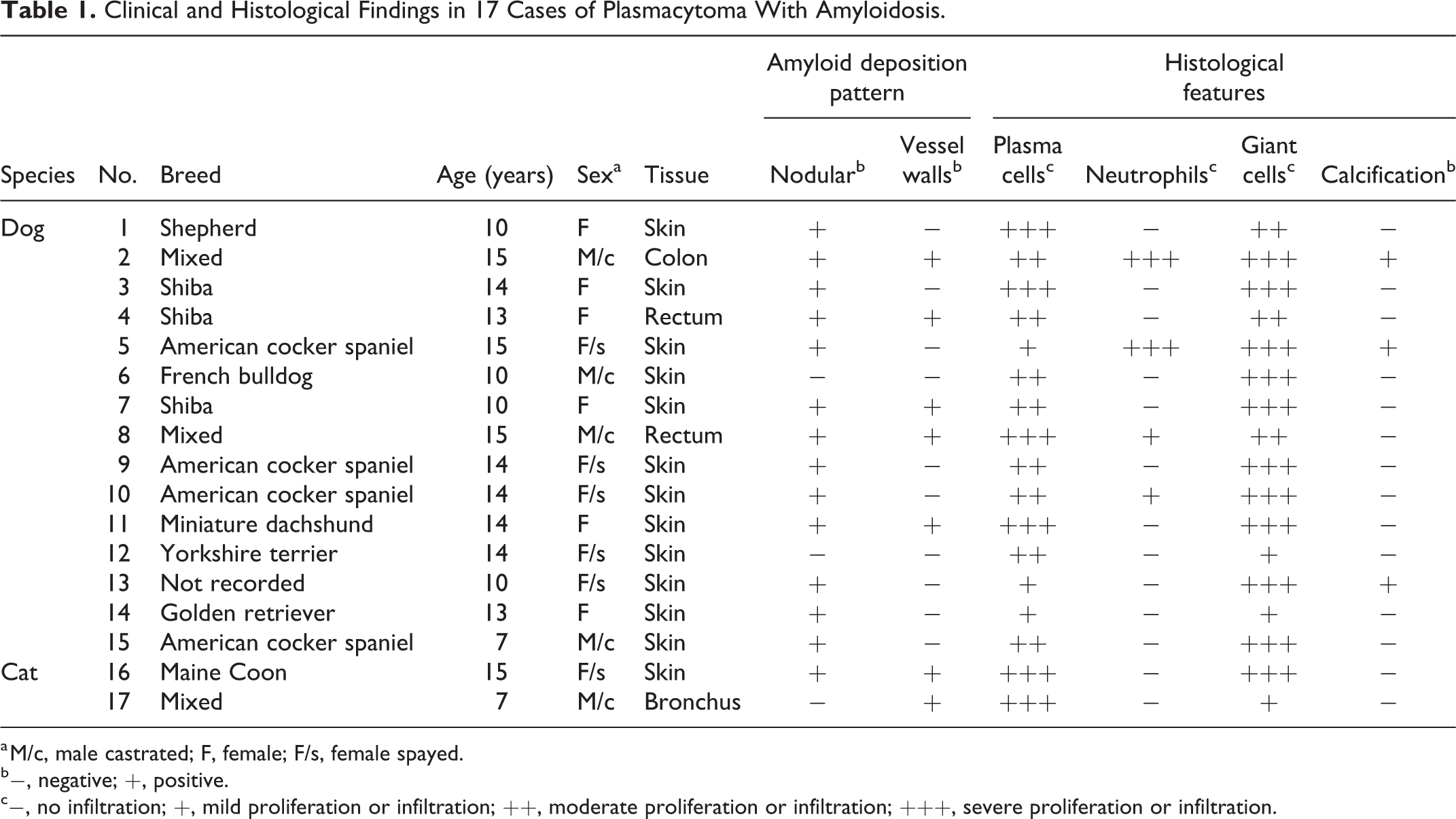
a M/c, male castrated; F, female; F/s, female spayed.
b−, negative; +, positive.
c−, no infiltration; +, mild proliferation or infiltration; ++, moderate proliferation or infiltration; +++, severe proliferation or infiltration.
Immunohistochemistry and Western Blot
IHC was conducted to identify the amyloid precursor proteins and amyloid signature proteins. Two antibodies were used for each of the amyloid proteins, kappa and lambda light chains: Kappa1 (anti-human kappa light chain rabbit polyclonal antibody, A80-111A, Bethyl Laboratories, Montgomery, TX), Kappa2 (anti-human kappa light chain rabbit polyclonal antibody, GTX73364, GeneTex, Irvine, CA), Lambda1 (anti-human lambda light chain rabbit polyclonal antibody, RB-334-A0, Thermo Scientific, Waltham, MA), and Lambda2 (anti-human lambda light chain rabbit polyclonal antibody, IS507, Dako, Kyoto, Japan). To confirm the specificity of the antibody, sections of canine and feline spleen containing normal plasma cells was used as positive control. IHC for amyloid signature proteins was performed by using anti-human ApoA-I rabbit polyclonal antibody (PAA519Ca01, Cloud-Clone, Katy, TX), anti-human ApoA-IV rabbit polyclonal antibody (PAB967Hu02, Cloud-Clone), and anti-canine ApoE rabbit polyclonal antibody (PAA704Ca01, Cloud-Clone). For the semiquantitative evaluation of IHC staining, the percentage of positive reactions in amyloid deposits were graded (−, <1%; +, 1% to 33%; ++, 34% to 66%; +++, 67% to 100%).
Western blot was performed to confirm the specificity of the antibodies. Normal sera of dog and cat were provided from Prof Ryuji Fukushima in Tokyo University of Agriculture and Technology Animal Medical Center. The normal sera were dissolved in sodium dodecyl sulfate (SDS)-sample buffer (50 mM Tris-HCl, pH 6.8, 2% SDS, 6% β-mercaptoethanol, 10% glycerol, and bromophenol blue) and boiled for 10 minutes before electrophoresis. These samples were loaded onto a 10% SDS-polyacrylamide gel and electrophoresed. Proteins were transferred onto a polyvinylidene difluoride membrane (Immobilon-P, Millipore), blocked with 5% nonfat milk in 10 mM Tris-HCl, pH 8.0, 150 mM NaCl, and 0.1% Tween 20 for 1 hour at room temperature. Subsequently, the membrane was incubated with the primary antibodies (Kappa1, Kappa2, Lambda1, Labda2 antibodies) for 1 hour at room temperature. After 3 washes, the membrane was incubated with horseradish peroxidase-conjugated anti-mouse IgG antibodies (GE Healthcare, Chicago, IL) for 45 minutes at room temperature. After 3 washes, peroxidase activity was detected using an ECL Prime detection kit (GE Healthcare) and visualized by STAGE-5100 analyzer (AMZ System Science, Tokyo, Japan). With all 4 antibodies to kappa and lambda light chains, positive bands around 25 to 27 kDa were confirmed in dog and cat sera, indicating that these antibodies reacted with the target antigens of dogs and cats (data not shown).
LMD-LC-MS/MS Analysis
For LMD, 5-µm FFPE sections were placed on polyethylene napthalate membrane glass slides and stained with Congo red. Amyloid deposits were collected using the LMD System LMD7000 (Leica Microsystems, Wetzlar, Germany) or Palm Microbeam (Zeiss Microscopy, Jena, Germany), and tryptic digestion was carried out as described previously. 19 The tryptic peptides were then subjected to the following proteomic analysis with LC-MS/MS (micrOTOF-QII, Bruker Daltonics Co, Billerica, MA). The digested peptides were separated using a PEGASIL ODS SP300-3 column (ϕ1 mm × 100 mm, 3 μm; Senshu Scientific Co, Suginami-ku, Tokyo, Japan) and eluted with a linear gradient of 0% to 100% buffer B (100% acetonitrile and 0.1% trifluoroacetic acid) in buffer A (0.1% trifluoroacetic acid in water) at a flow rate of 0.04 ml/min. MS and MS/MS data were acquired using the data-dependent top 10 method. The resulting MS/MS data were correlated to theoretical fragmentation patterns of tryptic peptide sequences from the NCBIprot database using Mascot Server (Matrix Science Inc, Boston, MA). Statistically significant proteins/peptides were extracted by Mascot’s probability-based scoring algorithm.
Results
Histopathology
Histologically, round cells resembling plasma cell-like proliferated in cords or nests. These cells were mononuclear, binucleated, or multinucleated and had moderate eosinophilic to amphophilic cytoplasm, a peripheral nucleus, and a clear zone in the perinuclear area. With Congo red staining, amyloid deposits were identified in all 17 cases and the histologic patterns are shown in Table 1. In 14 of 17 cases, nodular amyloid deposits replacing the tissue were observed (Fig. 1). In 7 cases, vascular amyloid deposits were observed, and the deposits became less severe with increasing distance from the lesion center (Fig. 4). In cases 5, 13, and 14, neoplastic plasma cells were rarely observed, and the tissue was almost completely replaced by nodular amyloid deposits. In cases 2 and 5, marked neutrophil infiltration was observed around the amyloid deposits. In all 17 cases, multinucleated giant cells were observed on the edge of amyloid deposits (Fig. 2), and amyloid was often observed in their cytoplasm (Fig. 3). In cases 2, 5, and 13, mineralization was observed in the areas of amyloid deposition (Fig. 2).
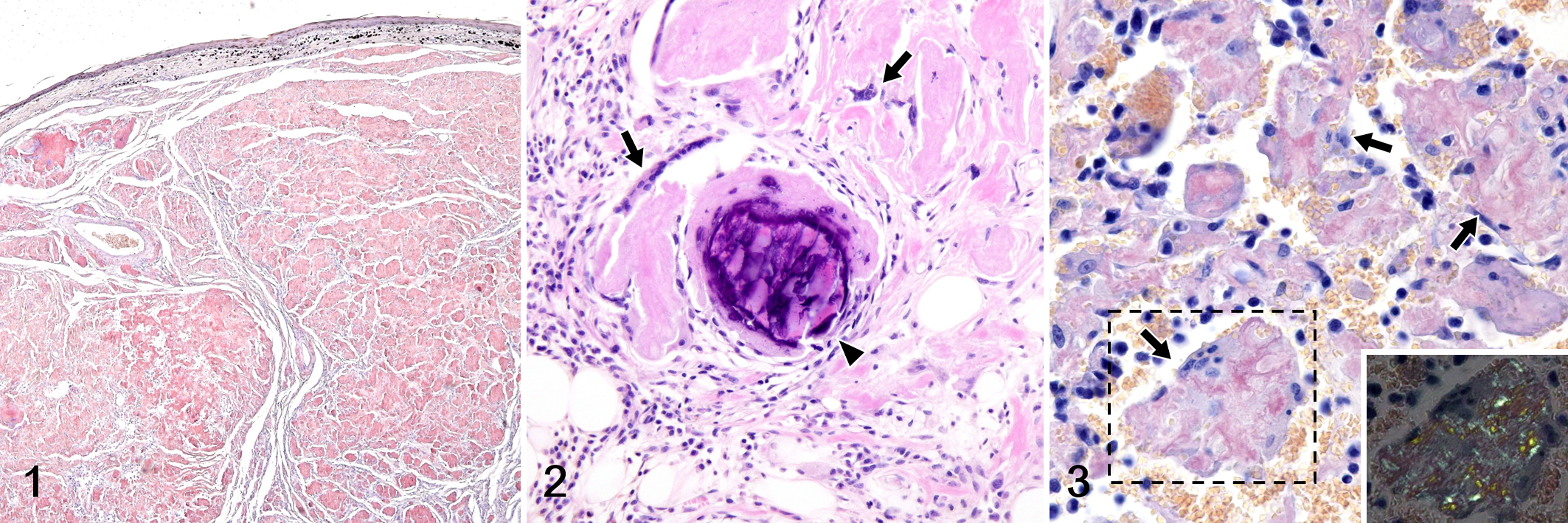
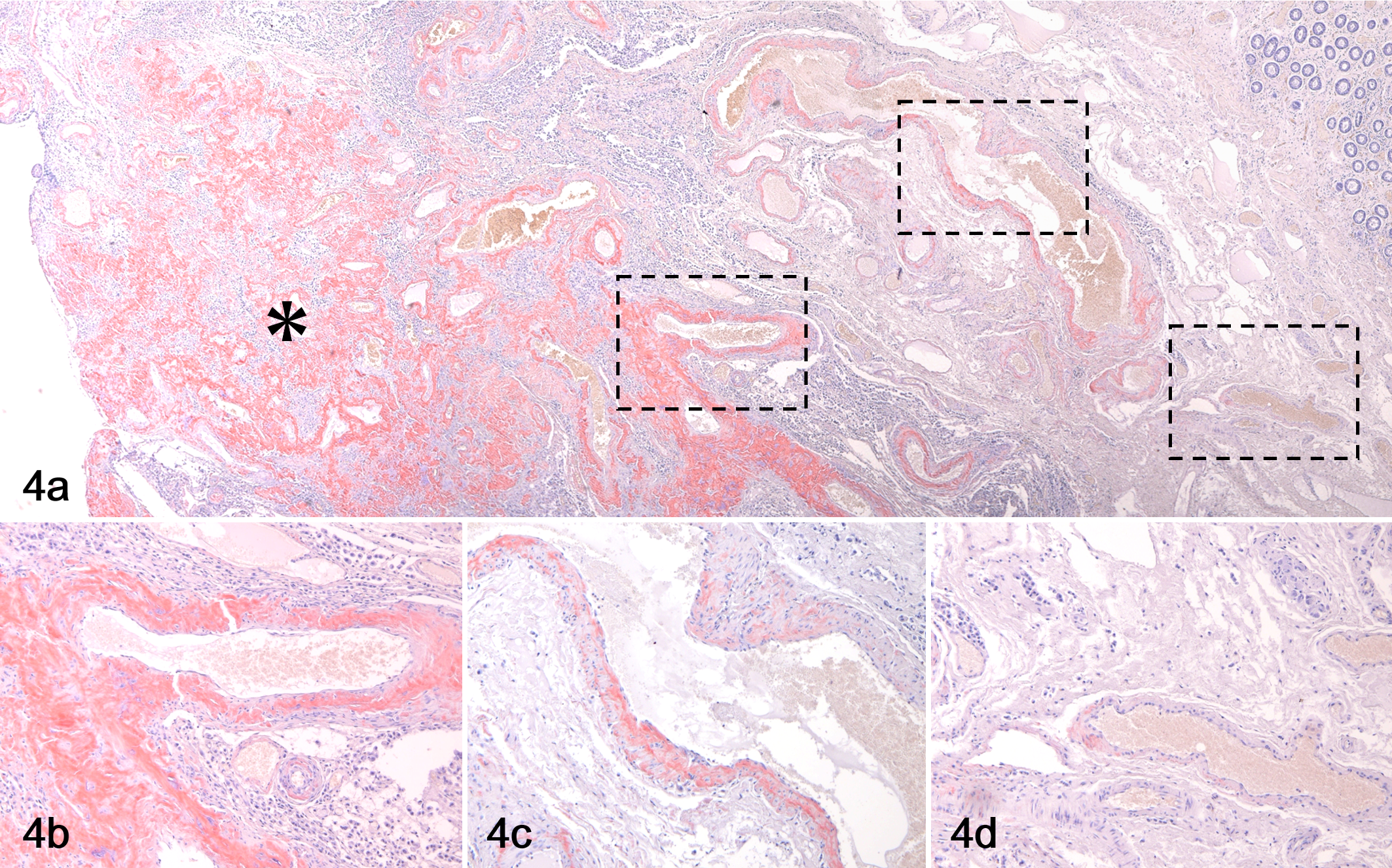
Vascular amyloid deposition, rectum, dog, case 4. (a) The vascular amyloid deposits are less severe with increasing distance from the lesion center (*). (b–d) Higher magnifications of the boxed regions in “a.” Congo red.
The effect of potassium permanganate pretreatment on the intensity of Congo red staining was determined. In 12 cases, the Congo red staining was resistant to potassium permanganate pretreatment. In contrast, in 5 cases, permanganate treatment unexpectedly resulted in a decrease in Congo red staining and a decrease or disappearance of green polarization (Table 2; Fig. 5).
Permanganate Resistance and Immunohistochemistry Data in 17 Cases of Plasmacytoma With Amyloidosis.
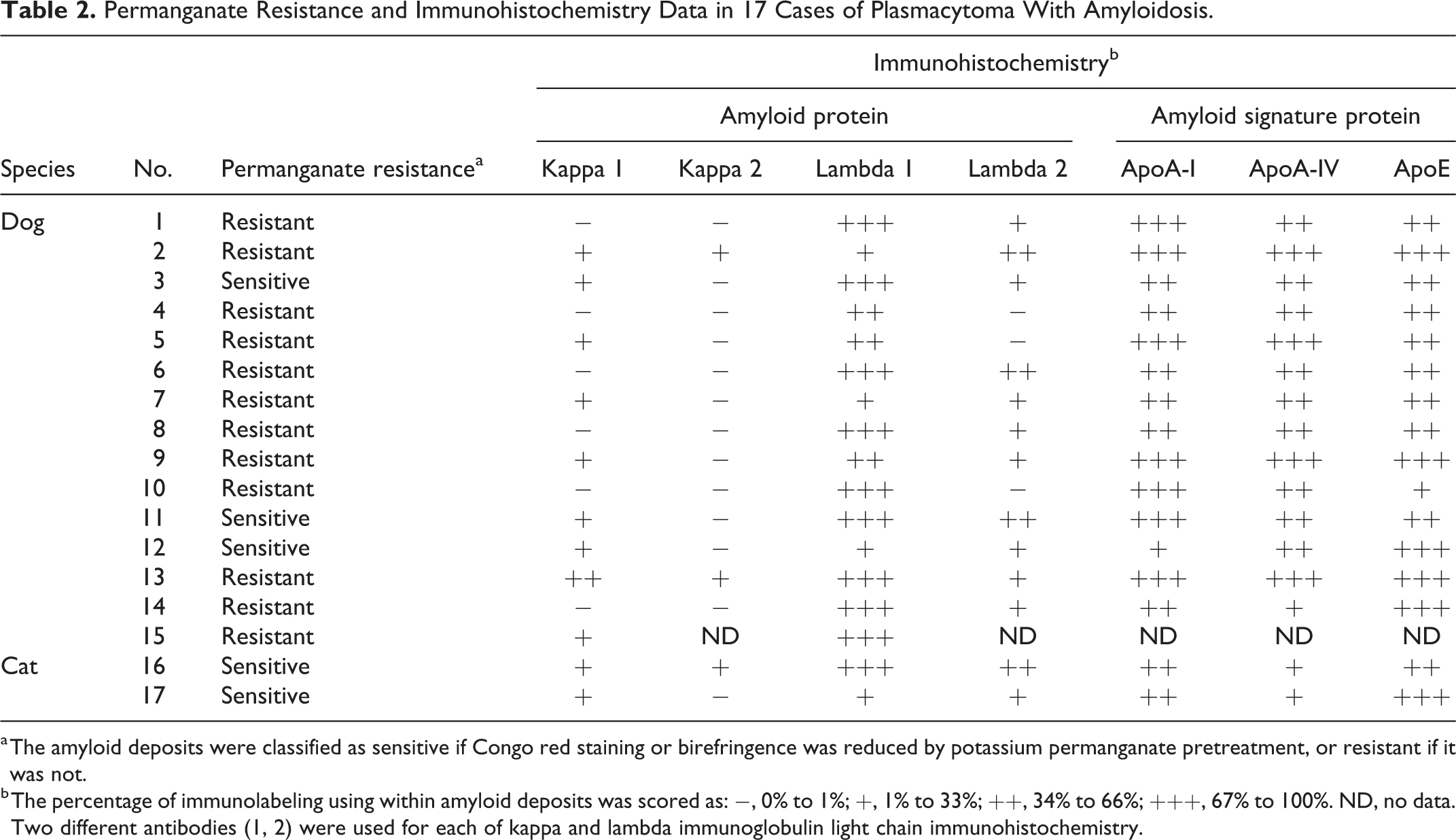
a The amyloid deposits were classified as sensitive if Congo red staining or birefringence was reduced by potassium permanganate pretreatment, or resistant if it was not.
b The percentage of immunolabeling using within amyloid deposits was scored as: −, 0% to 1%; +, 1% to 33%; ++, 34% to 66%; +++, 67% to 100%. ND, no data. Two different antibodies (1, 2) were used for each of kappa and lambda immunoglobulin light chain immunohistochemistry.
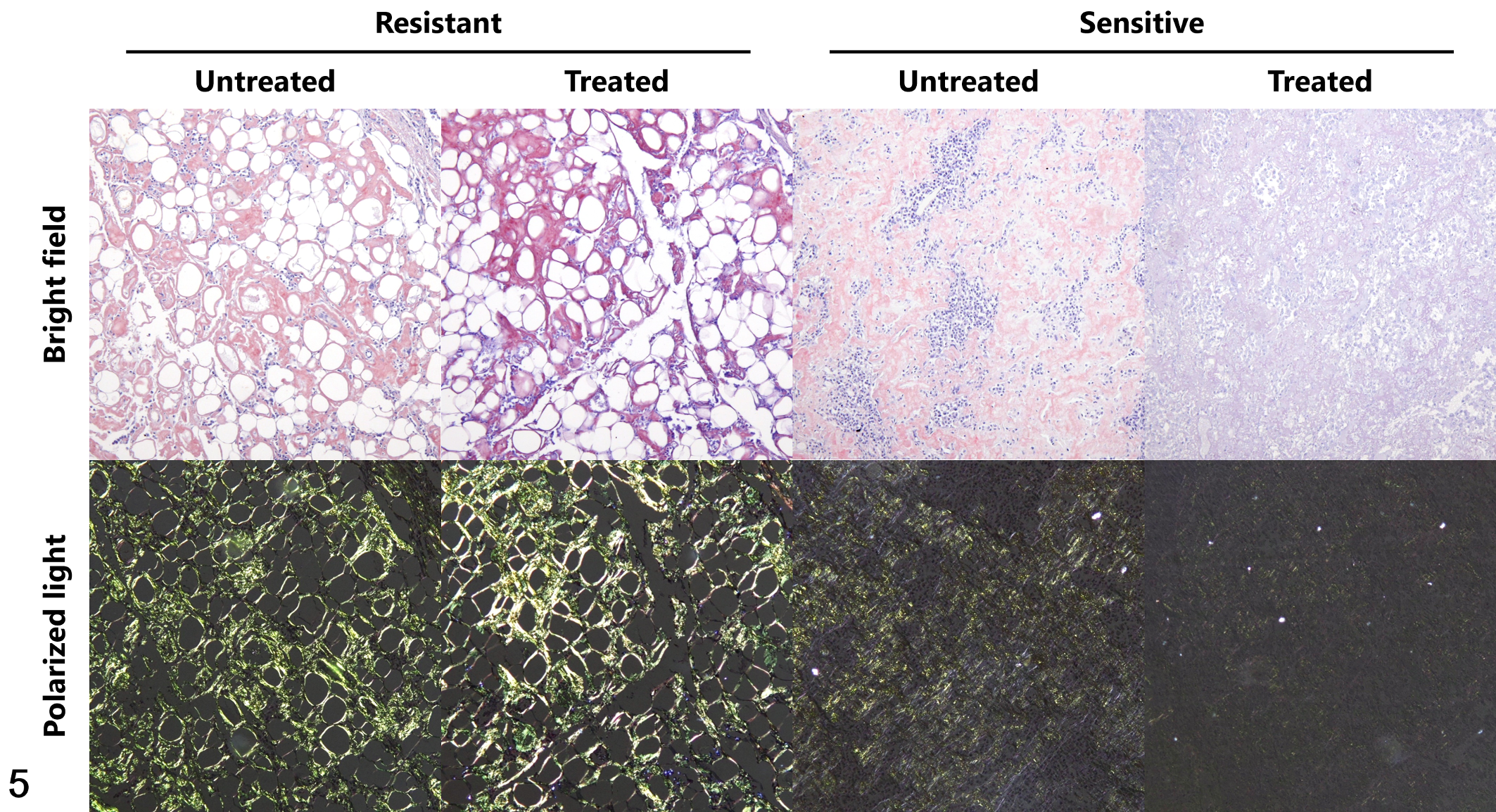
Localized AL amyloidosis, skin, dogs. Congo red staining with or without potassium permanganate pretreatment (treated and untreated, respectively). In case 7 (Resistant), the amyloid deposits retained Congo red staining and green birefringence after potassium permanganate treatment. In contrast, in case 11 (Sensitive), Congo red staining decreased and green birefringence was abolished by potassium permanganate treatment.
Immunohistochemistry
Table 2 shows the results of IHC for kappa and lambda light chains. AL-lambda amyloidosis was diagnosed in 6 cases because they were confirmed to be positive only for the lambda light chain. Since the remaining 11 cases were positive for both the kappa and lambda light chains, a diagnosis could not be reached. While IHC was performed using 2 antibodies for each of the kappa and lambda light chains, 7/11 and 3/17 cases were positive only for one antibody, respectively. The immunolabelling of amyloid deposits generally corresponded with immunolabelling of the neoplastic cells. In the positive control, all anti-kappa and anti-lambda antibodies reacted with normal canine and feline splenic plasma cells (data not shown).
Considering IHC for the amyloid signature proteins, all cases had positive reactions for ApoA-I, ApoA-IV, and ApoE to various degrees (Table 2).
LMD-LC-MS/MS
Lambda light chain was detected by proteomic analysis in all 17 cases, and no amyloidogenic proteins other than the lambda light chain were detected; all cases were thus diagnosed as AL-lambda amyloidosis (Fig. 6). Of the 3 types of amyloid signature proteins in dogs (ie, ApoA-I, ApoA-IV, and ApoE), ApoA-I was the most frequently detected (9 of 15 cases), and ApoA-IV and ApoE were detected in 1 and 3 cases, respectively. The Mascot scores of the amyloid signature proteins were lower than that of the lambda light chain. Amyloid signature proteins were not detected in cats.
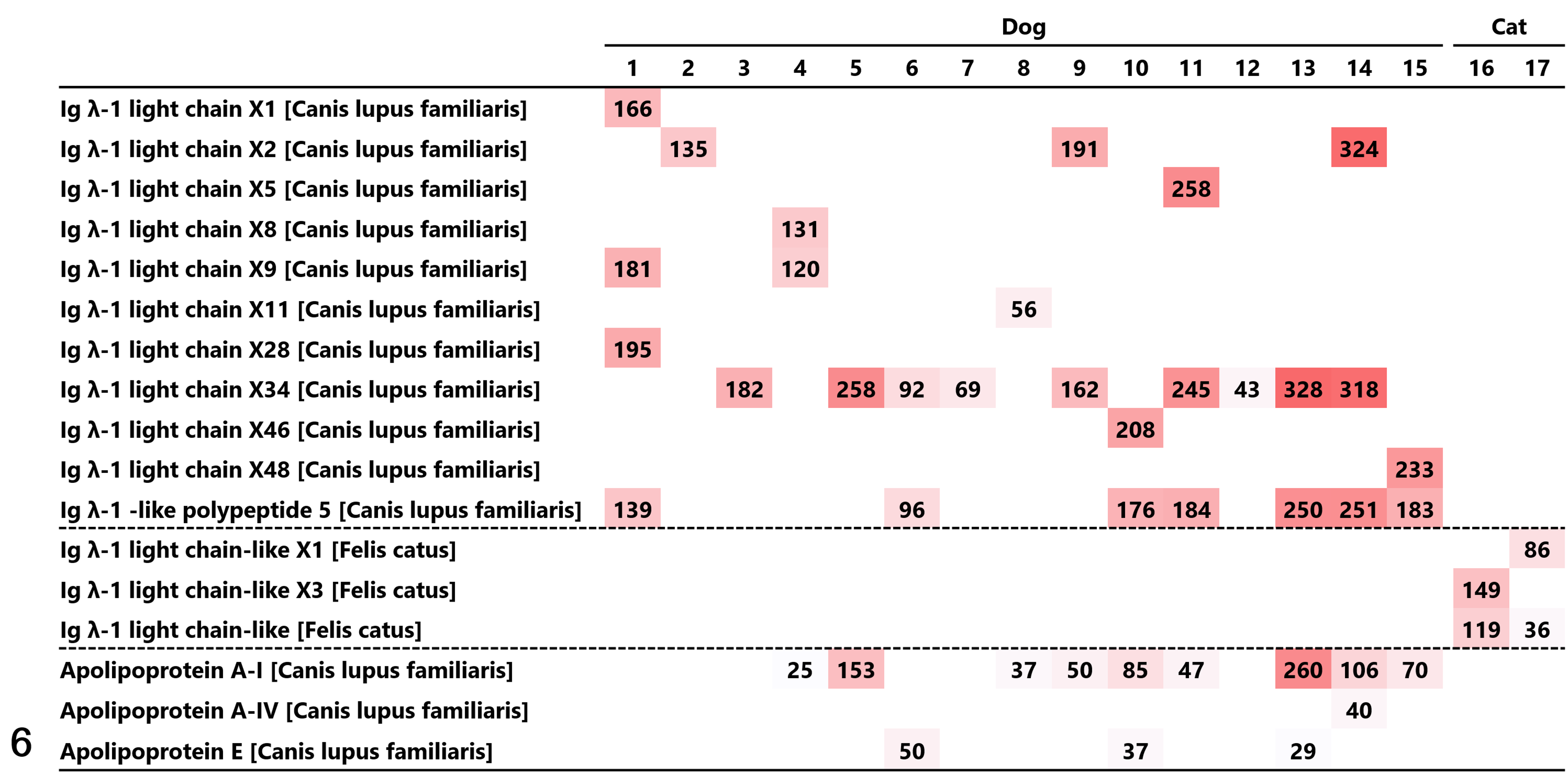
Proteomic analysis of the amyloid deposits in 17 plasmacytomas from dogs and cats. Among the proteins detected by Mascot analysis, the amyloidogenic proteins and amyloid signature proteins are listed. Each Mascot score is presented with a heat map depending on the size of the Mascot score. Immunoglobulin lambda light chain was detected in all 17 cases, and no amyloidogenic proteins other than lambda light chain were detected.
Discussion
In this study, all 17 cases were diagnosed as localized AL amyloidosis. Calcification was observed in 3 of 17 cases, and multinucleated giant cells were observed in all 17 cases, as can be found in human localized AL amyloidosis. 18,21,29 Calcification is thought to be caused by dystrophic calcification due to tumor cell death in AL amyloidosis associated with plasmacytoma. 7,21 It has been suggested that multinucleated giant cells may be implicated in amyloid formation. 29 It is also thought that apoptosis of neoplastic plasma cells may be induced by a cytotoxic effect of the aggregates produced in this process, thus forming an amyloidoma or tumor-like mass of amyloid with few neoplastic cells. 22,29 In this study, nodular amyloid deposition formed in 15 cases, and plasma cells were rarely observed in 3 cases. The histological features of localized AL amyloidosis in dogs and cats shown in this study are similar to those in humans in many ways, suggesting a similar pathogenic mechanism of localized AL amyloidosis in humans and animals.
AL amyloidosis is the most common type of human systemic amyloidosis. 12,13,29 In contrast, systemic AL amyloidosis has not been reported in dogs and cats. In this study, severe vascular amyloid deposits were observed in the lesion center, and they became less severe with increasing distance from the lesion center. Similarly, human localized AL amyloidosis is accompanied by vascular amyloid deposition only in areas close to the lesion. 29 The vascular amyloid deposits in this study were also thought to originate from locally produced light chains rather than from systemic amyloidosis.
Congo red staining after potassium permanganate pretreatment is a classical method for differentiating AA amyloidosis from other amyloidoses. 27 In this method, it is thought that AA amyloid deposits lose affinity for Congo red after potassium permanganate treatment (sensitive), while other amyloid deposits including AL amyloid do not lose affinity (resistant). 27 However, in recent years, many exceptions for the susceptibility of amyloid to potassium permanganate treatment have been reported, and Congo red staining after potassium permanganate pretreatment has thus become obsolete as a diagnostic method for human amyloidosis. 15 In this study, 5 cases of AL amyloidosis showed a reduction or disappearance of Congo red staining after potassium permanganate pretreatment. If conventional criteria were used, these cases would have been misdiagnosed as AA amyloidosis. Furthermore, in some animal species, AA amyloid is resistant to potassium permanganate treatment. 26 Congo red staining after potassium permanganate treatment is still being used in veterinary pathology 2 ; however, its lack of accuracy as a diagnostic method for animal AL amyloidosis should be recognized.
In this study, 11 of 17 cases could not be diagnosed by IHC. The difficulty of diagnosing amyloidosis by IHC has been reported in many studies, 9,16,17 and the causes were attributed to fragmentation during amyloid formation, the masking of antigens due to protein folding, and the presence of variable regions within the protein. 9,25 In veterinary pathology, anti-human antibodies are often used for IHC, and diagnosis may thus become difficult due to the differences between human and animal antigens. 11 In this study, IHC was performed using 2 antibodies for each of the lambda and kappa light chains, but in several cases, only one antibody showed an immunoreaction. For the diagnosis of animal AL amyloidosis using IHC, a combination of multiple antibodies would improve the sensitivity of the diagnosis. 10
All 17 cases were diagnosed as AL-lambda amyloidosis by LMD-LC-MS/MS even though some of them could not be diagnosed by Congo red staining after potassium permanganate treatment or IHC. Furthermore, even in the cases that were positive for both kappa and lambda light chains by IHC, only the lambda light chain was detected by LMD-LC-MS/MS. These results indicated that the LMD-LC-MS/MS method is effective for the definitive diagnosis of AL amyloidosis in dogs and cats. Since the immunoglobulin light and heavy chains have a large number of variations in the variable region, it is not easy to detect a variable region from protein databases in animals, 12,28 especially for species with incomplete or poorly annotated protein databases.
The results of IHC showed that all cases were positive for ApoA-I, ApoA-IV, and ApoE. However, LMD-LC-MS/MS detected ApoA-I, ApoA-IV, and ApoE only in 9, 3, and 3 cases, respectively, suggesting that IHC is more sensitive than mass spectrometry for detecting these proteins. In humans, the detection of amyloid signature proteins by mass spectrometry is considered to be evidence for the collection of amyloid deposits by LMD. 8,10,12,13 From the results of IHC, ApoA-I, ApoA-IV, and ApoE were considered to be amyloid signature proteins in dogs as they are in humans, but because these were not detected in the cat cases it is necessary to consider species differences in amyloid signature proteins when using mass spectrometry to diagnose amyloidosis.
The lambda/kappa ratio of immunoglobulin light chains produced by plasma cells is 91:9 in dogs and 92:8 in cats, 3 and AL-lambda amyloidosis has been reported to be the main type of AL amyloidosis in dogs and cats. 6,11,20 In this study as well, AL-lambda amyloidosis was dominant. Similarly, in human systemic AL amyloidosis, AL-lambda amyloidosis is found 2 to 3 times more often than AL-kappa amyloidosis; 14,16,29 this difference has been suggested to be due to the presence of amyloid-associated light chain lambda variable region germline genes. 1 These genes have also been indicated to be associated with the predominance of AL-lambda amyloidosis in dogs and cats. 30 However, in human localized AL amyloidosis, the frequencies of AL-lambda and AL-kappa amyloidoses have been reported to be the same. 22,29 In the future, it will be necessary to examine the differences in the developmental mechanisms of localized AL amyloidosis between humans and animals.
Currently, the diagnosis of animal AL amyloidosis is generally carried out by either IHC or Congo red staining after potassium permanganate pretreatment, and mass spectrometry is rarely used. However, these conventional methods have problems that may lead to a misdiagnosis. This study showed the superiority of LMD-LC-MS/MS to the other classical methods in the diagnosis of amyloidosis in dogs and cats by targeting AL amyloidosis, which is particularly difficult to diagnose. Unlike immunohistochemistry, which detects a small number of protein species on tissue sections, LMD-LC-MS/MS is capable of exhaustively detecting constituent proteins. Furthermore, the analytic method is not constrained by difference among animal species as it is for immunohistochemistry. LMD-LC-MS/MS will contribute to veterinary medicine as a reliable method for protein identification.
Footnotes
Acknowledgements
We are greatly indebted to Dr Yuki Maekawa, Dr Tomoyuki Harada, and Dr Soichiro Tsuda (FUJIFILM VET Systems Co, Ltd) for their cooperation in correcting samples and for useful information. The authors also thank Prof Ryuji Fukushima (Tokyo University of Agriculture and Technology) for providing normal sera of his dog and cat. We also greatly appreciate Prof Keiichi Noguchi (Tokyo University of Agriculture and Technology) for technical assistance with the LC/MS/MS analyses.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was partially supported by JSPS KAKENHI (Grant Numbers 17K17702 and 20K15660) and TUAT president’s discretionary funding to support the launch of next-generation research projects.
