Abstract
Sarcocystosis is a protozoal disease affecting a wide range of animals. The aims of this study were to characterize the following in sheep: (1) the muscle pathology in Sarcocystis infection, (2) the inflammatory infiltrate and its relationship to severity of infection, and (3) immune markers expressed by parasitized muscle fibers and parasitic cysts. Skeletal muscle samples from 78 sheep slaughtered in southern Italy were snap frozen and analyzed by histopathology, immunohistochemistry, and immunofluorescence. Polymerase chain reaction (PCR) and sequencing were used for Sarcocystis species identification. All 40 muscle samples tested were PCR-positive for Sarcocystis tenella. Histologically, cysts were identified in 76/78 cases (97%), associated with an endomysial infiltrate of lymphocytes and plasma cells. The T cells were predominantly CD8+, with fewer CD4+ or CD79α+ cells. Eosinophils were absent. Notably, sarcolemmal immunopositivity for major histocompatibility complex (MHC) I and II was found in 76/78 cases (97%) and 75/78 cases (96%), respectively, both in samples with and in those without evident inflammatory infiltrate. The number of cysts was positively correlated with inflammation. In addition, MHC I was detected in 55/78 cyst walls (72%), and occasionally co-localized with the membrane-associated protein dystrophin. The findings suggest that muscle fibers respond to the presence of cysts by expression of MHC I and II. The possible role of MHC I and II in the inflammatory response and on the cyst wall is also discussed.
Sarcocystosis (syn. sarcosporidiosis) is a parasitic infection caused by the Apicomplexa protozoa Sarcocystis spp. that affect a wide range of animals, including mammals, reptiles, and birds. Sheep are the intermediate hosts of 4 species, namely 2 microcyst species (S. tenella [syn. S. ovicanis] and S. arieticanis) and 2 macrocyst species (S. gigantea [syn. S. ovifelis] and S. medusiformis). 3
The consequences of sarcocystosis are different depending on the host (definitive, intermediate, or accidental) and the stage of disease. In intermediate hosts such as sheep, the infection is commonly asymptomatic, and the presence of muscular cysts is considered an incidental finding occurring in virtually all (close to 100%) sheep at the time of slaughter. 5,25,33,40 However, some species of Sarcocystis, particularly those having a canid as definitive host, may cause clinical signs including fever, inappetence, and reduced productivity in sheep. 25,40
Those signs have been explained with the release of interleukin-1 (IL-1), prostaglandin E2 (causing fever), and tumor necrosis factor–alpha (TNF-α) by the Sarcocystis-infected macrophages. These cytokines can cause inappetence, anemia, and suppression of the release of pituitary growth hormone (GH) as a potential reason for weight loss. 11 Less frequently, abortion and stillbirths 40 or central nervous signs, including acute myopathy, ataxia, paresis, and death, have been associated with sarcocystosis. 18,27 Acute disease is associated with the 2 phases of schizogony within endothelial cells of small blood vessels. 3 Apparently, the severity of clinical signs is dose-dependent in experimental disease. 36 Pregnant sheep infected with high doses of S. tenella gave birth early to small, weak lambs and developed myositis, myocarditis, and encephalitis after parturition. 23
It has been reported that the most known muscle lesion associated with Sarcocystis spp. infection in intermediate hosts, especially in cattle and occasionally described in sheep, is an eosinophilic myositis. 9,19,40 It has been speculated that cattle with eosinophilic myositis lesions are genetically predisposed to produce immunoglobulin E in response to Sarcocystis bradyzoite antigen and that eosinophilic myositis represents an abnormal response to the degeneration of the cyst, including a host-dependent, Sarcocystis-specific, type-I hypersensitivity. 15 Furthermore, eosinophilic myositis was experimentally induced by intramuscular injection of adjuvanted Sarcocystis antigen in 2 calves. 41 Lymphocytic endomysial infiltrate accompanied by mild degenerative muscle fiber changes in intermediate hosts has been rarely reported. 5 Furthermore, severe chronic muscle inflammation is very seldom reported in intermediate hosts, and clinically relevant cases are sporadic in horses. 39
Relatively little is known of the immunity induced by infection with Sarcocystis spp., but some authors indicate that protective immunity develops and that cell-mediated mechanisms are likely important. It seems likely that Sarcocystis spp. stimulates immune mechanisms similar to those induced by other Apicomplexa protozoa as Toxoplasma gondii and Neospora caninum. Immunity to one species does not appear to trigger protective immunity to another species. 10
Sarcocystis-related muscle pathology is much more severe and histologically different in accidental intermediate hosts. Recurrent outbreaks of muscular sarcocystosis among tourists visiting islands in Malaysia have focused international attention on the disease; 12 indeed, humans may become accidental intermediate hosts after ingesting sporocysts and can develop symptomatic disease including fever, myalgia, headache, arthralgia, vomiting, and chronic, mainly lymphoplasmacytic polymyositis. 16,38 Less frequently, Sarcocystis infection has been associated with eosinophilic myositis in humans as well. 1 There have also been sporadic reports of aberrant muscular sarcocystosis characterized by severe chronic symptomatic myositis in dogs, 4,37 cats, 20 and nonhuman primates. 14
Considering the striking variation in Sarcocystis-related muscle disease, in addition to our preliminary data of chronic muscle inflammation in infected ruminants and the report of an immune activation of the cysts, 37 our hypothesis is that Sarcocystis is not an inert parasite of muscle fibers but rather suggests that there is an active host-parasite interaction that plays a primary role in determining the clinical and pathological presentation of the disease. The aims of the study were to characterize the following in sheep: (1) the muscle pathology in Sarcocystis infection, (2) the inflammatory infiltrate and its relationship to the severity of infection, and (3) immune markers expressed by parasitized muscle fibers and parasitic cyst.
Materials and Methods
Study Animals
The study included 78 skeletal muscle samples (semimembranosus or semitendinosus muscles) randomly selected from a larger population of 594 regularly slaughtered healthy sheep, 4 to 5 years old, collected in 1 year from 7 different areas of the whole Basilicata region of southern Italy. The 7 districts correspond to the districts defined by the national health service system (Azienda Sanitaria Locale). Sheep were serologically negative for brucellosis. Moreover, the absence of prion diseases was confirmed in all animals by performing the rapid test recommended by European law. The study did not require consent or ethical approval according to European Directive 2010/63/EU. The animals were slaughtered in strict accordance with European slaughter regulations (CE n° 1099/2009 of 24 September 2009) for the protection of animals at the time of killing (Ref. Official Journal of the European Union L 303/1). Permission to obtain the samples was granted from the owner of the abattoir and from the veterinary inspector responsible for the sanitary surveillance.
All muscles were immediately transported under refrigeration to the Laboratory of Comparative Neuromuscular Disorders of the Department of Veterinary Medicine of Naples. Samples of 1 x 1 x 1 cm were snap frozen in isopentane precooled in liquid nitrogen within 2 hours after sampling.
Sarcocystis Species Identification
DNA was extracted from a subsample of 40 frozen muscle samples by using the DNeasy Blood and Tissue Kit (QIAGEN, Hilden, Germany). A PCR for partial 18 S rRNA gene sequence of Sarcocystis spp. was performed using 10 µM of each primer reported by Yang et al (2001), 45 3 L (5′-CTAGTGATTGGAATGATGGG-3′) and 3 H (5′- GGCAAATGCTTTCGCAGTAG -3′) in a final volume of 50 μL, containing 4 µL of DNA, 0.2 mM of each dNTP (Sigma-Aldrich, Steinheim, Germany), 2.0 mM of MgCl2, and 1.25U of FastStart Taq DNA Polymerase (Life Technologies, Carlsbad, CA, USA). The thermal cycler conditions were: an initial denaturation step at 95°C for 10 minutes, followed by 40 cycles of 94°C for 40 seconds, 57°C for 1 minute, 72°C for 1 minute, and a final elongation step at 72°C for 7 minutes. The PCR products were detected on a 2% ethidium bromide–stained low melting agarose gel (Bio-Rad, Madrid, Spain). Then, the PCR products were purified by QIAquick PCR Purification Kit (QIAGEN) and sequenced. The obtained sequences, in both forward and reverse directions, were analyzed using the Chromas version 2.1 software and compared with 18 S sequences of Sarcocystis present in GenBank, using BLASTn system.
Histology and Immunohistochemistry
Frozen sections (8 µm thick) were stained according to our routinely performed laboratory stains. 30 Specifically, the following stains were performed: (1) hematoxylin and eosin (HE) and (2) Engel trichrome (ET) for a basic morphologic evaluation and mitochondria distribution; (3) reduced nicotinamide adenine dinucleotide–tetrazolium reductase (NADH-TR) to observe intermyofibrillar pattern and secondary distribution of mitochondria; and (4) succinate dehydrogenase (SDH) and (5) cytochrome oxidase (COX) to evaluate activity and distribution of mitochondria.
The number of cysts was counted in a whole section of 1 cm2 for each muscle sample. A scoring system was designed to assess the degree of fiber atrophy as follows based on assessment of 100 fibers at x200 magnification: mild (score 1), <10% atrophic fibers; moderate (score 2), 10% to 50% atrophic fibers; and severe (score 3), >50% atrophic fibers. 32 A scoring system was also defined for the number of lymphocytes and plasma cells based on light microscopy: no infiltration (score 0); mild infiltration (score 1), 5 to 25 lymphocytes/plasma cells per high-power field (HPF) (x400); moderate infiltration (score 2), 26 to 50 lymphocytes/plasma cells per HPF; and severe infiltration (score 3), more than 50 lymphocytes/plasma cells per HPF. 32 At least 10 fields at x400 magnification were evaluated for each section by 2 independent pathologists (T.B.P. and O.P.) under an optical microscope (Nikon E600; Nikon, Tokyo, Japan), with a concordance rate of 95%.
For immunohistochemistry (IHC), frozen sections (8 µm thick) were processed with the MACH1 Universal HPR Polymer Detection Kit (Biocare Medical LLC, Concord, CA, USA). Briefly, the sections were dried for 1 hour at room temperature and fixed in acetone at 4°C for 3 minutes; peroxide block was applied for 15 minutes at room temperature, and then the sections were incubated for 30 minutes with background sniper (Biocare Medical LLC). The primary antibodies were diluted in phosphate-buffered saline (PBS) and incubated overnight at 4°C. MACH1 mouse probe was applied for 20 minutes at room temperature. Horseradish peroxidase (HRP)–polymer was added for 30 minutes at room temperature. After every step of the procedure, the sections were washed in 0.01 M PBS (pH 7.2–7.4). The reaction was revealed by using 3,3’-diaminobenzidine (DAB) chromogen diluted in DAB substrate buffer. Finally, sections were counterstained in Carazzi’s hematoxylin. Primary antibodies were directed against major histocompatibility complex I (H58A, mouse monoclonal antibody, dilution 1:200; VMRD, Pullman, WA, USA), major histocompatibility complex II (H42A, mouse monoclonal antibody, 1:200; VMRD), CD3 (IS503, rabbit polyclonal antibody, 1:50; Dako, Milan, Italy), CD79a (HM57, mouse monoclonal antibody, 1:50; Dako), CD4 (17D1, mouse monoclonal, 1:50; VMRD), CD8 (PT36B, mouse monoclonal, 1:50; VMRD), and dystrophin ROD domain (DYS 1, clone Dy4/6D3, dilution 1: 50; Novocastra Laboratories Ltd, Newcastle Upon Tyne, UK).
The percentage of muscle fibers with sarcolemmal positivity to major histocompatibility complex I and II (MHC I and II) was scored as follows: absent/none (score 0), 0%; mild (score 1), 1% to 25%; moderate (score 2), 26% to 50%; and severe (score 3), more than 50%. 29 MHC I positive cysts and the DYS 1 positive cysts were counted in a whole section of 1 cm2 for each muscle sample. The inflammation score was obtained by summing the scores for lymphocytic inflammation, MHC I, and MHC II. 32 For evaluation of MHC I and II, fibers directly adjacent to the inflammatory infiltrate were avoided, since positive endomysial inflammatory cells can make evaluation of sarcolemmal positivity difficult. The positive staining for MHC I and II varied from a continuous staining throughout the sarcolemma to a discontinuous sarcolemmal pattern. Endomysial blood vessels, which are normally positive for MHC I and II, were used as positive internal controls for immunohistochemistry with anti–MHC I and MHC II antibodies. Frozen section of normal control sheep lymph nodes was used as positive controls for immunohistochemistry with anti-MHC I, MHC II, CD3, CD79a, CD4, and CD8 antibodies. Sarcolemma was considered as positive control for immunohistochemistry with anti-DYS1 antibody. Histologically normal sheep muscle sections were used as negative control for all the antibodies except for anti-DYS1 antibody, for which frozen section of normal control sheep lymph nodes was used. For each antibody, a negative control section was also processed omitting or replacing the primary antibody with normal serum from the same species of primary antibody (rabbit or mouse).
Immunofluorescence
To determine if there was a co-localization between dystrophin and MHC I on muscle fibers and on parasitophorous vacuoles, immunofluorescence was carried out on 10 selected cases as follows. Cryosections were dried at room temperature for 1 hour, preincubated with normal mouse serum diluted 1:10, and overlaid overnight in a humid chamber at 4°C with primary antibody directed against Dystrophic ROD domain (DYS 1, clone Dy4/6D3, dilution 1:10; Novocastra Laboratories Ltd). A FITC fluorochrome-labeled rabbit anti-mouse secondary antibody was applied (1:50; Jackson Laboratories, Bar Harbor, ME, USA) on sections for 2 hours at room temperature. Slides were rinsed with PBS, and a second primary antibody directed against MHC I (H58A, mouse monoclonal antibody, dilution 1:10; VMRD) was applied overnight at 4°C. A TRITC fluorochrome-labeled rabbit anti-mouse secondary antibody was applied (1:50; Jackson Laboratories) on sections for 2 hours at room temperature. Slides were rinsed with PBS and mounted with a solution of 1-part glycerol/ 1-part PBS. For scanning and photography, a laser scanning microscope (LSM 510; Zeiss, Göttingen, Germany) was used with illumination at 488 nm and read using a 505 to 560 nm band pass filter. To exclude autofluorescence, serial sections of muscle were incubated with PBS omitting the primary antibody. Frozen section of normal sheep lymph nodes was used as positive controls for anti-MHC I antibody and as negative control for anti-DYS1 antibody. Histologically normal sheep muscle sections were used as positive control for anti-DYS1 antibody and as negative control for anti-MHC I antibody.
Statistical Analysis
Statistical analysis was performed using IBM SPSS Statistics 25.0 with a level of significance of .05. The following correlations were evaluated using the Spearman’s rank correlation coefficient: (1) the number of cysts vs lymphoplasmacytic infiltration score, inflammation score, MHC I score, MHC II score, and the number of MHC I–positive cyst wall; (2) the atrophy score vs lymphoplasmacytic infiltration score and inflammation score; (3) the number of MHC I–positive cysts vs MHC I score, MHC II score, and inflammation score; and (4) the number of DYS1–positive cysts vs MHC I score, MHC II score, and inflammation score.
Results
Sarcocystis Species Identification
DNA amplification of 18 S rRNA produced a 500 bp fragment for all 40 samples analyzed (Supplemental Figure S1). An identity of 100% was found with Sarcocystis tenella by BLAST alignment (GenBank accession number KC209734.1) for all samples.
Histology and Immunohistochemistry
Intrasarcoplasmic, round-to-oval, septate sarcocysts with a thin wall (<1 µm), containing a myriad of crescent-shaped bradyzoites (Fig. 1), were detected in 76/78 muscle samples (97%). Muscles infected by Sarcocystis contained a mean of 13 cysts/cm2 (95% confidence interval, 10.0–15.9) and, in rare cases, up to 70 cysts/cm2.
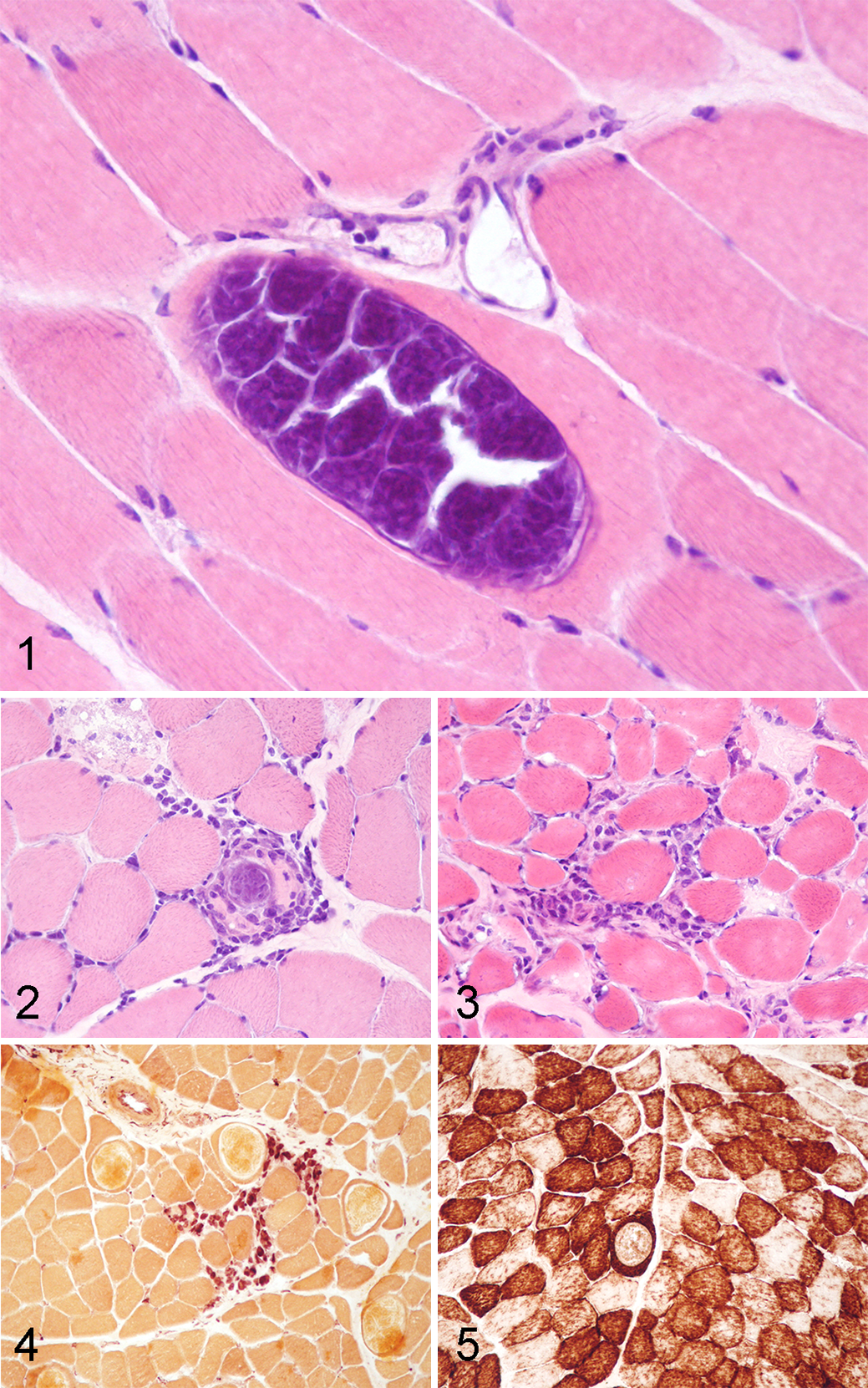
The lymphoplasmacytic inflammation was absent in 23/78 cases (30%) and was scored as mild in 32/78 cases (41%), moderate in 20/78 cases (26%), and severe in 3/78 cases (4%). The inflammatory infiltrate was usually organized in multifocal endomysial foci, occasionally centered around parasitized and nonparasitized fibers, and seldom arranged in perivascular cuffs (Figs. 2, 3). Furthermore, lymphocytes were present within morphologically normal parasitized and nonparasitized muscle fibers. Multifocally, in about 30% of cases, lymphocytes and plasma cells were admixed with esterase-positive macrophages (Fig. 4) or invaded necrotic muscle fibers (sarcoclastosis). Notably, eosinophils were never observed in any of the samples.
Increased variability in fiber size was a prominent finding in almost all cases (66/78; 85%) and was classified as mild in 41/78 cases (53%), moderate in 23/78 cases (30%), and severe in 2/78 cases (3%). In most of the inflamed cases (25/78; 32%), mitochondrial abnormalities such as central cores and the presence of “moth-eaten” fibers were observed using the histoenzymatic stains COX (Fig. 5), NADH, and SDH.
Sarcolemmal immunopositivity for MHC I (Fig. 6) was detected in 76/78 cases (97%), both in samples with evident inflammatory infiltrate and in cases without visible inflammation. It was scored as mild in 27/78 cases (35%), moderate in 32/78 cases (41%), and severe in 17/76 cases (22%). Similarly, MHC II (Fig. 7) was positive in 75/78 cases (96%) and classified as mild in 38/78 cases (49%), moderate in 26/78 cases (33%), and severe in 11/78 cases (14%).
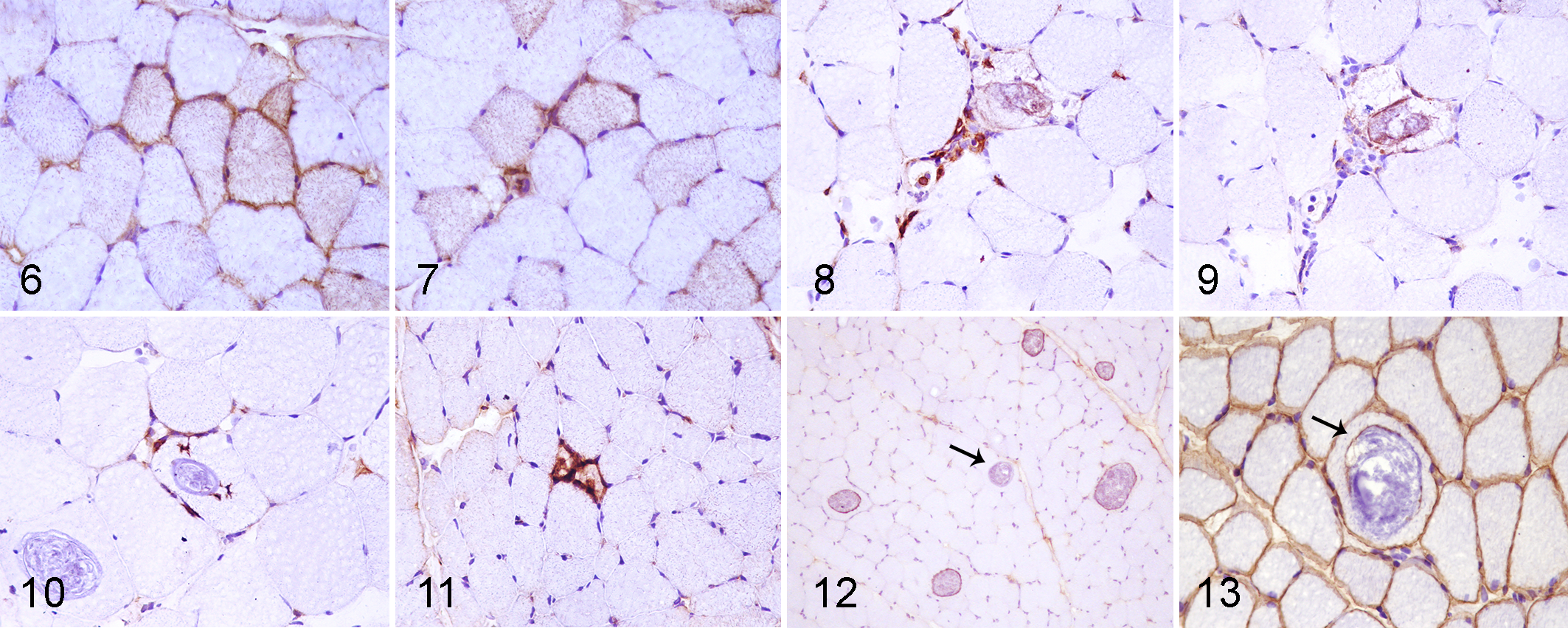
The predominant lymphocytic populations were CD3+ and CD8+ with lesser numbers of CD4+ cells (Figs. 8, 9). T lymphocytes were predominantly CD8+ in 44/78 cases (56%; on average, 80% of CD8+ and 20% of CD4+). In 10/78 cases (13%), CD4+ and CD8+ lymphocytes were in an equal proportion (50% of CD8 and 50% of CD4). In only 1/78 cases (1%), CD4+ lymphocytes were more numerous than CD8+ lymphocytes (40% of CD8 and 60% of CD4). Furthermore, CD8+ cells were present within non-necrotic parasitized and nonparasitized fibers (Figs. 10, 11). CD79+ cells were also present in 15/78 cases (19%).
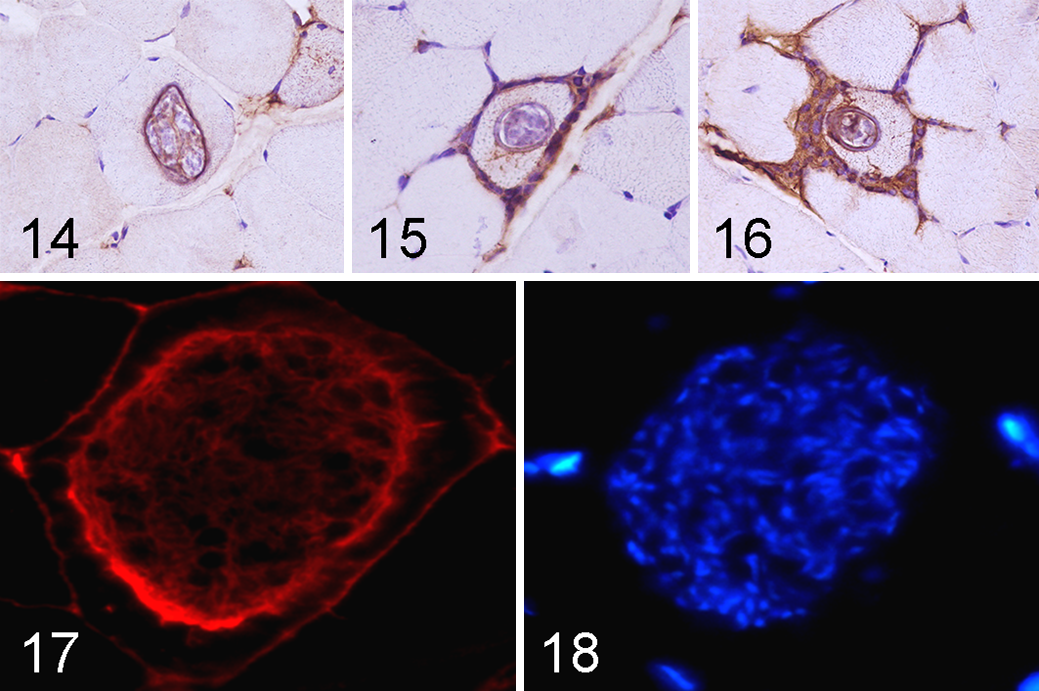
In 55/78 cases (72%), MHC I was also expressed on the wall of the cysts (Figs. 12, 13); 3 different staining patterns of MHC I immunopositivity were observed: (1) In most cases, only the cyst wall was immunopositive (Fig. 14) (about 70% of parasitized muscle fibers had this pattern); (2) parasitized muscle fibers occasionally had sarcolemmal immunopositivity without cyst-wall positivity (Fig. 15) (about 5% of parasitized muscle fibers had this pattern); (3) in few cases (about 5% of parasitized muscle fibers), and especially those with a prominent inflammatory infiltrate, both the sarcolemma and the cyst wall were immunopositive (Fig. 16). In the remaining 20% of parasitized muscle fibers, both the sarcolemma and the cyst wall were immunonegative for MHC I.
In 18/78 cases (23%), a small percentage of cysts (less than 10% of the cysts in the section) expressed DYS1 on the cyst wall.
Immunofluorescence
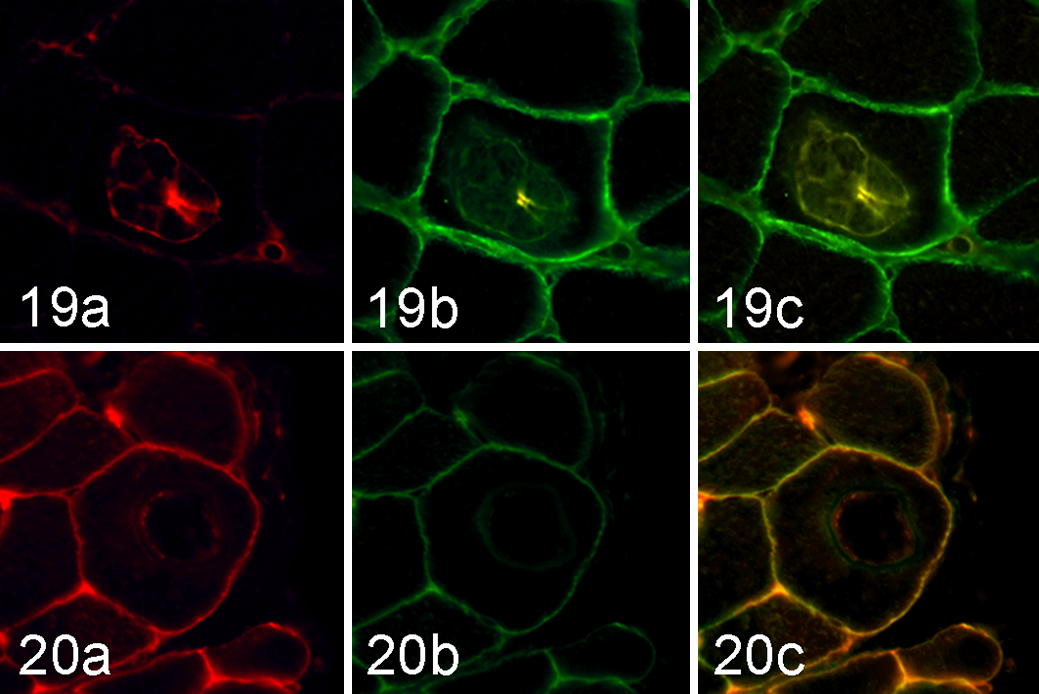
To explore a possible colocalization between the sarcolemmal-associated protein DYS1 and MHC I, immunofluorescence was performed on those 18 cases that had DYS1 positivity on the cyst wall using immunohistochemistry (Figs. 17, 18). A colocalization between MHC I and DYS1 was observed in 9/18 cases (50%) (Figs. 19, 20). No positivity was detected in the negative control sections (normal sheep lymph nodes for DYS1 and normal sheep skeletal muscle for MHC I) and in sections incubated with PBS without primary antibodies.
Statistical Analysis
There was a low positive correlation between the number of cysts and the inflammation score (r s = 0.329; P < .01) (Fig. 21), MHC I score (r s = 0.356; P = .01), and MHC II score (r s = 0.265; P < .05). There was a low positive correlation between the number of cysts and the number of MHC I–positive cyst walls (r s = 0.773; P < .001). Despite a positive trend, the number of cysts and the lymphoplasmacytic infiltration score were not significantly correlated (r s = 0.199; P = .08).
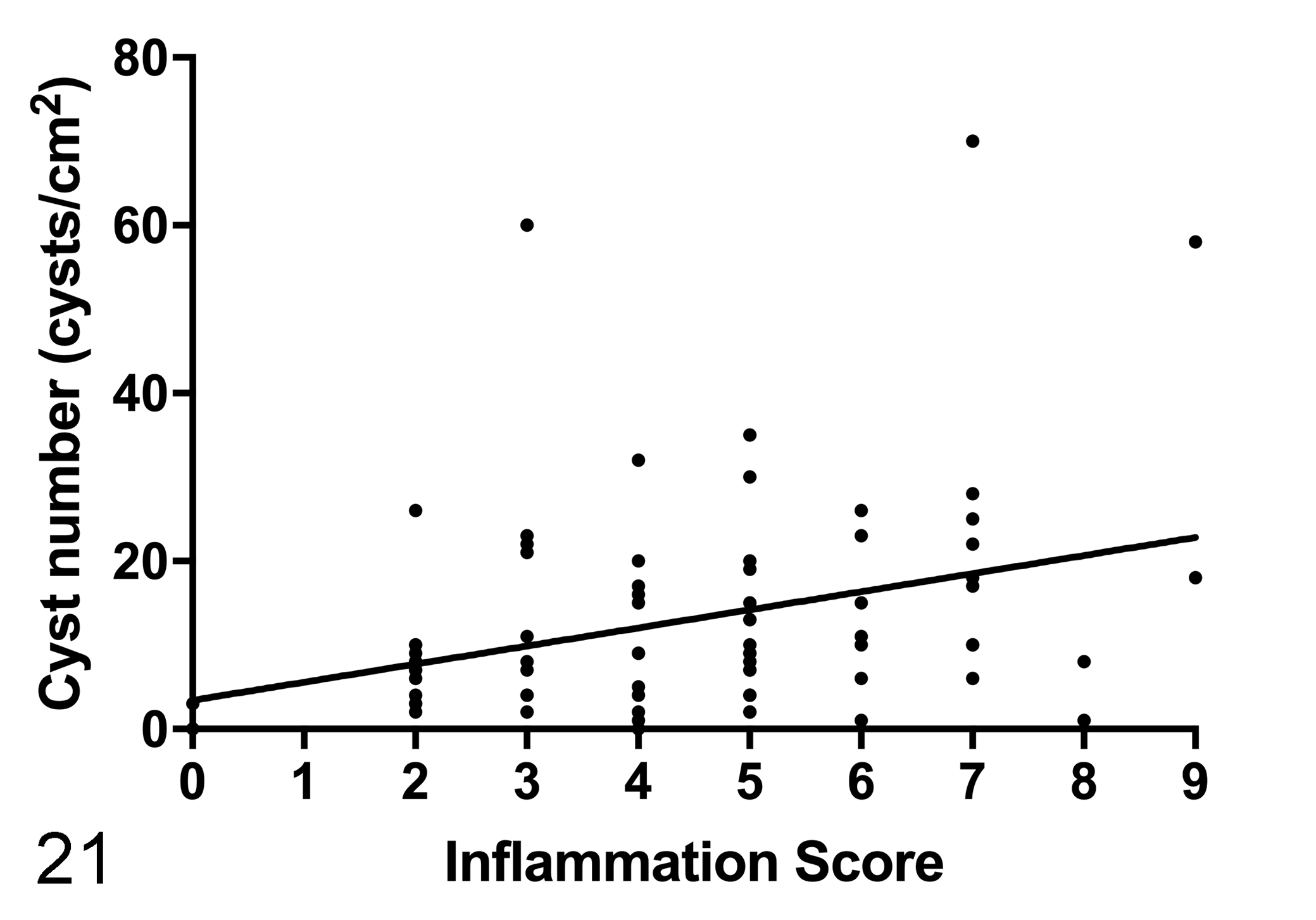
The atrophy score was not correlated with the lymphoplasmacytic infiltration score (r s = 0.186; P = .102) or inflammation score (r s = 0.139; P = .224).
There was a low positive correlation between the number of MHC I–positive cysts and the MHC I (r s = 0.359; P = .001), MHC II (r s = 0.277; P < .05), and the inflammation scores (r s = 0.463; P < .001). There was a low positive correlation between the number of DYS1–positive cysts and the MHC I (r s = 0.251; P < .05) and inflammation scores (r s = 0.320; P < .01) but not the MHC II score (r s = 0.213; P = .061).
Raw data are reported in Supplemental Table S2.
Discussion
Although Sarcocystis is widely regarded as a “silent” muscle pathogen, it can be associated with a wide range of disease manifestations, ranging from localized forms (eosinophilic myositis), to acute and peracute systemic febrile disease and abortion during the endothelial schizogony phase, as well as clinically relevant polymyositis in accidental intermediate hosts. 3 These different clinical manifestations are partly imputable to different phases of the parasite lifecycle and partly to the degree of host immunity. 3
We identified a variable degree of chronic lymphoplasmacytic infiltrate in 71% of muscle samples of asymptomatic sheep naturally infected with S. tenella, and the percentage of cases with inflammation would be even higher if sarcoplasmic expression of MHC I and II (in about 97% of cases) were considered to be a more sensitive indicator of inflammatory myopathy. 17,30 The inflammatory infiltrate showed mainly a multifocal endomysial disposition with rare perivascular cuffs and some lymphocytes infiltrating nonnecrotic parasitized and nonparasitized muscle fibers. Lymphoplasmacytic infiltrate associated with muscular sarcocystosis in appropriate intermediate host is occasionally mentioned in the scientific literature; 3,18 however, an accurate incidence analysis and histopathological characterization of this inflammatory change have not been documented so far.
Experimentally, Sarcocystis tenella can induce muscle inflammation in sheep. 21,26 In our cases, the degree of muscle inflammation was positively correlated with the number of cysts, confirming a statistically significant relationship between the etiological agent and the histological and immunohistochemical markers of muscle inflammation. 32,34 It should be noted, however, that there was only a weak relationship between cyst number and inflammatory infiltrate based on hematoxylin-eosin stain, but the evaluation of MHC I and II expression revealed a strong relationship between cyst number and muscle inflammation.
Immunohistochemical evaluation of MHC I and II expression in skeletal muscle is used for diagnosis of idiopathic inflammatory myopathy. 17 MHC I and II are not constitutively expressed on muscle fibers but are induced during various inflammatory myopathies. 30 In a transverse histological section of muscle, there may be no inflammatory cells, but a multifocal inflammatory infiltrate may be present a few microns away. Considering their elongated shape, the same myofibers present in a section without inflammatory infiltrate could be exposed to an inflammatory microenvironment a few microns away. This condition can trigger the expression of MHC I and II on the sarcolemma and/or in the sarcoplasm of the entire myofiber and allows us to detect muscle inflammation even on sections without obvious inflammatory infiltration. 30,31 However, since the distribution of cysts is not always homogeneous within a muscle group, when parasites are not visible in histopathological sections, the presence of Sarcocystis cannot be immediately ruled out in case of muscle inflammation.
Overall, our results indicate that chronic inflammation in ovine muscular sarcocystosis is likely underestimated. The presence of inflammatory infiltrate, mainly consisting of CD3/CD8–positive T lymphocytes, together with the strong sarcolemmal immunopositivity to MHC I, suggests that parasitized muscle fibers are not merely inert “victims” of intracellular parasitism but, as in other myopathies, might play an active role in antigen presentation and in stimulating inflammatory response. 7,29,31 If so, this common protozoal disease should be included in the differential diagnosis of polymyositis in many animal species, including humans; furthermore, it should be excluded before considering idiopathic and immune-mediated myopathy in the diagnosis of muscle inflammation.
Expression of MHC class I and II antigens on the surface of muscle fibers and the CD8-positive T cells infiltrating morphologically “healthy” muscle fibers have already been described in other inflammatory myopathies associated with infectious/parasitic agents (eg, the protozoa Toxoplasma, Leishmania, Theileria equi, and Babesia caballi) 22,29,32 and immune-mediated myositis of humans, dogs, and horses. 6,8,32,44 Indeed, increasing evidence suggests that myositis formerly considered “idiopathic” in both humans and dogs might in fact be related to underlying systemic parasitic and infectious diseases triggering immune-mediated muscle damage. 6,8,29 For those reasons, it is possible that these 2 entities (infective and immune-mediated myositis) might be considered, at least partially, overlapping.
It can be speculated that Sarcocystis can induce muscle damage and trigger muscle inflammation at least in 2 different ways: (1) by a direct damage to the cell or (2) by a stimulation of an immune-mediated myopathy. 32 The direct role of Sarcocystis in inducing cell damage and thus possibly stimulating MHC I expression may be related to the perturbance of endoplasmic reticulum (ER) homeostasis, that is, ER stress. ER stress 34 is reported in response to intracellular parasitism 13 and in human inflammatory myopathies. 24,43 In mammalian cells, ER stress activates the unfolded protein response, which is an elaborate signaling cascade activated to restore ER homeostasis and ensure cell survival.
In our samples, prominent mitochondrial abnormalities were detected in severely inflamed cases. Mitochondrial abnormalities are frequently associated with muscle inflammation, as reported in several human inflammatory myopathies where they are generally associated with damage to the mitochondrial DNA. 28,42 Mitochondrial damage can activate ER stress since the endoplasmic reticulum has intimate contact with mitochondria in specialized areas called mitochondria-associated ER membranes (MAM). It is thus conceivable that ER stress pathways and mitochondria also work together to cause myofiber damage in Sarcocystis myositis. ER stress itself has been associated with the activation of inflammatory responses in skeletal muscle: NF-κB, JNK, interleukin-6, and TNF-α have been shown to be activated by ER stress mechanisms. 24
A novel and interesting finding of our work is the evidence of MHC I on the parasitophorous vacuole of Sarcocystis. To the authors’ knowledge, the same finding has been described only once in the literature, in a severe case of myositis in a dog that was an accidental intermediate host of Sarcocystis spp., 37 where it was interpreted as an active involvement of this parasite’s structure in the pathogenesis of muscle inflammation. In Apicomplexan parasites, the parasitophorous vacuole membrane (PVM) derives from the host cell plasmalemma, whose biochemical composition undergoes significant changes as the intravacuolar parasite grows. In particular, the host cell proteins are progressively and selectively excluded from the PVM, while those of the parasite are incorporated. As a result, the changed PVM does not becomes fusigenic for host cell lysosomes 2 and is protected from host immunity. In this study, scattered cysts expressed the sarcolemmal-associated protein dystrophin in 23% of cases, and dystrophin was colocalized with MHC I in half of these cases. This finding could be explained with an incomplete exclusion of host cell protein from the parasitophorous cell membrane; how this finding could influence the inflammatory machinery is unknown.
The MHC I immunostaining pattern on parasitized myofibers displayed at least 4 different combinations: cyst-only immunopositivity (70%), sarcolemma-only positivity (5%), both sarcolemma and cyst wall positivity (5%), and no staining of either structure (20%). The number of MHC I–positive cysts was statistically correlated to the overall MHC I positivity score, to the inflammation score, and to the number of cysts present in the sections. The timing of MHC I positivity of the cyst wall relative to that of sarcolemmal MHC I positivity is not known. The reasons underlying the different combination staining patterns could be interpreted with 2 hypotheses as follows, which are not mutually exclusive. First, MHC I might be present on the cyst membrane because it was present on the sarcolemma at the time of the formation of the PVM. This would imply the expression of MHC I on the sarcolemma before that fiber was parasitized. So, the cysts that express MHC I could be more recent than those that do not express it. Further studies are needed to understand if these cysts lose the expression of MHC I and other sarcolemma-associated proteins (eg, dystrophin) over time. Second, as already suggested, 37 the expression of MHC I on the cyst membrane might indicate an active immune response against the parasitized muscle fibers. This may be the consequence of cyst degeneration, which is considered the most relevant factor triggering inflammation in muscular sarcocystosis. 37
An interesting aspect of our study is the absence of eosinophils in muscle of Sarcocystis-infected sheep. These data seem to contrast with older studies 9,40 that reported eosinophilic myositis in Sarcocystis-infected sheep. Usually, eosinophils are associated with immune responses to helminthic parasites and seem not to be implicated in the immune response against protozoa. 35 However, several lines of evidence support the involvement of eosinophils during infection with different protozoa in the early stage of the infection. 35 We speculate that the direct interaction of bradyzoites with the immune system of the host can elicit a Th2 response with chemotactic activity for eosinophils. This might happen in the early stage of the infection with Sarcocystis spp. or in those sporadic cases where the parasitic cyst content (bradyzoites and their products) is exposed to the extracellular environment by rupture of the sarcolemma and cyst membrane.
Conclusions
We characterized an inflammatory myopathy concomitant with muscle sarcocystosis in sheep. This condition is characterized by infiltration of mainly CD8+ T lymphocytes and by the sarcolemmal immunopositivity for MHC I and II, and the inflammation is positively correlated with cyst number. The occasional MHC I immunopositivity of the cyst wall was identified. The present data are of value in analyzing host-parasite interactions in this protozoan infectious disease, and studies of the disease in sheep may inform the immunopathogenesis of other idiopathic inflammatory myopathies.
Supplemental Material
Combined_supplemental_materials-Pagano_et_al - Muscular Sarcocystosis in Sheep Associated With Lymphoplasmacytic Myositis and Expression of Major Histocompatibility Complex Class I and II
Combined_supplemental_materials-Pagano_et_al for Muscular Sarcocystosis in Sheep Associated With Lymphoplasmacytic Myositis and Expression of Major Histocompatibility Complex Class I and II by Teresa Bruna Pagano, Francesco Prisco, Davide De Biase, Giuseppe Piegari, Maria Paola Maurelli, Laura Rinaldi, Giuseppe Cringoli, Serenella Papparella and Orlando Paciello in Veterinary Pathology
Footnotes
Author Notes
Teresa Bruna Pagano and Francesco Prisco contributed equally to this work.
Acknowledgements
We thank Raffaele Ilsami for the excellent technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
