Abstract
A 7-year-old female Labrador Retriever dog showed extreme muscular weakness, muscle wasting, dysbasia, and mild dysphagia. An elevated value of creatine kinase (335 IU/liter) in the serum was detected. Electromyographic findings included increased insertional activity, fibrillation potentials, and bizarre high-frequency repetitive potentials. Histopathologic examination of skeletal muscles revealed myofiber necrosis and phagocytosis, regeneration of myofibers, and perivascular, perimysial, and endomysial infiltrations of lymphocytes, macrophages and plasma cells. Immunohistochemical evaluation demonstrated that infiltrative cells in the early stage of myositis were CD8+ T-cells and that an increased expression of major histocompatibility complex (MHC) class I was apparent on the surface of nonnecrotic muscle fibers. In contrast, many CD3+ cells (T cells) and HLA-DR-positive macrophages and B lymphocytes were found in the severely affected areas. These results suggest that both expression of MHC class I and CD8+ T-cell infiltration may play an important role in initiation of myositis. These histopathologic findings resemble those reported in naturally occurring polymyositis in humans.
Inflammatory myopathies make up an uncommon, heterogeneous group of disorders characterized by immunologically mediated injury and inflammation of mainly the skeletal muscles.11 In humans, three relatively distinct disorders, dermatomyositis, polymyositis, and inclusion-body myositis, are included in this category.6 The diagnosis of myositis is based on clinical symptoms, electromyography, levels of muscle-related enzymes in serum, and histologic findings.4,11,16 Polymyositis differs from dermatomyositis by the lack of cutaneous involvement and from inclusion-body myositis by the absence of rimmed vacuoles within myofibers.6 Case reports of polymyositis in dogs have been sporadic and include canine masticatory muscle disorders,15 bilateral polymyositis restricted to extraocular muscles,5 and polymyositis associated with Leptospira australis infection.13 In humans, cytotoxic T cells and macrophages play an important role in the development of polymyositis.2,3,9 This article includes the first report in animals of evidence of involvement of CD8+ T cells in the early stage of polymyositis, as has been demonstrated in humans.
A 7-year-old female Labrador Retriever dog with a clinical history of extreme muscular weakness, muscle wasting, dysbasia, and mild dysphagia was brought to the veterinary clinic. Physical examination revealed generalized muscular atrophy, especially in the head, lumbar and femur regions. There were no specific painful areas noted by palpation, and spinal reflexes were normal. The results of routine blood biochemistry and hematologic examination were normal except for an elevated value of creatine kinase (335 IU/liter). The titers of serum antibody against Leptospira species such as L. canicola, L. icterohaemorrhagiae, L. autumnalis, L. hebdomadis, and L. australis and antinuclear antibody were negative.
Electromyographic examination was conducted under xylazine sedation (1.0 mg/kg intramuscularly). Bipolar exploring electrodes (NEC San-ei Co. Ltd., Tokyo) were inserted into the left quadriceps femoris and biceps femoris muscles, and the electrical activity was monitored and amplified at a time constant of 0.03 seconds by an amplifier (Oscilloscope Model 2317, NEC San-ei Co. Ltd., Tokyo).
Biopsy samples were taken from the masseter, temporalis, biceps brachii, triceps brachii, quadriceps femoris, and biceps femoris muscles, fixed in 10% neutral buffered formalin, dehydrated in a series of alcohols, embedded in paraffin, cut at 4 μm, and stained with hematoxylin and eosin (HE), phosphotungstic acid hematoxylin, and Masson's trichrome. Selected tissues of the same samples were embedded in OCT compound (Miles, Elkhart, IN), frozen in liquid nitrogen, and processed for immunohistochemical analysis using labeled streptavidin (Dako, Glostrup, Denmark). Specific antisera used in this study were anti-canine CD8 monoclonal antibody (Biosource, Camarillo, CA), anti-human CD3 polyclonal antibody (Dako), anti-human class I major histocompatibility complex (MHC) antigen monoclonal antibody (VMDR, Pullman, WA), and anti-human HLA-DR, α-chain monoclonal antibody (Dako). The HLA-DR antibody was used as a marker for macrophages and B lymphocytes. Sections were also stained with biotinylated lectin from Bandeiraea simplicifolia (BS-1) Isolectin B4 (Vector, Burlingame, CA), a marker of macrophages. Because of the lack of suitable immunologic reagents, it was not possible to identify CD4+ cells. The skeletal muscle tissues of 6- and 9-year-old mixed-breed dogs with no muscular disorders were used as controls.
The dog gradually showed mild to severe atrophy of systemic skeletal muscles and loss of body weight (6.6 kg) over the course of 1 year and was euthanatized because of poor prognosis and completely necropsied. Tissue sections were processed for routine histology and immunohistochemistry with the same procedure used for the biopsied tissues.
A prolonged burst of activity associated with needle electrode insertion was prominent in the left quadriceps femoris and biceps femoris muscles. The burst included polyphasic potentials of variable amplitude and duration followed by prolonged discharge of electrical activity after cessation of needle movement. Fibrillation potentials with biphasic negative–positive waves were recorded in non-end-plate regions. The fibrillation also included bizarre high-frequency repetitive potentials. These abnormalities were distinct in the left quadriceps femoris muscle (Fig. 1).
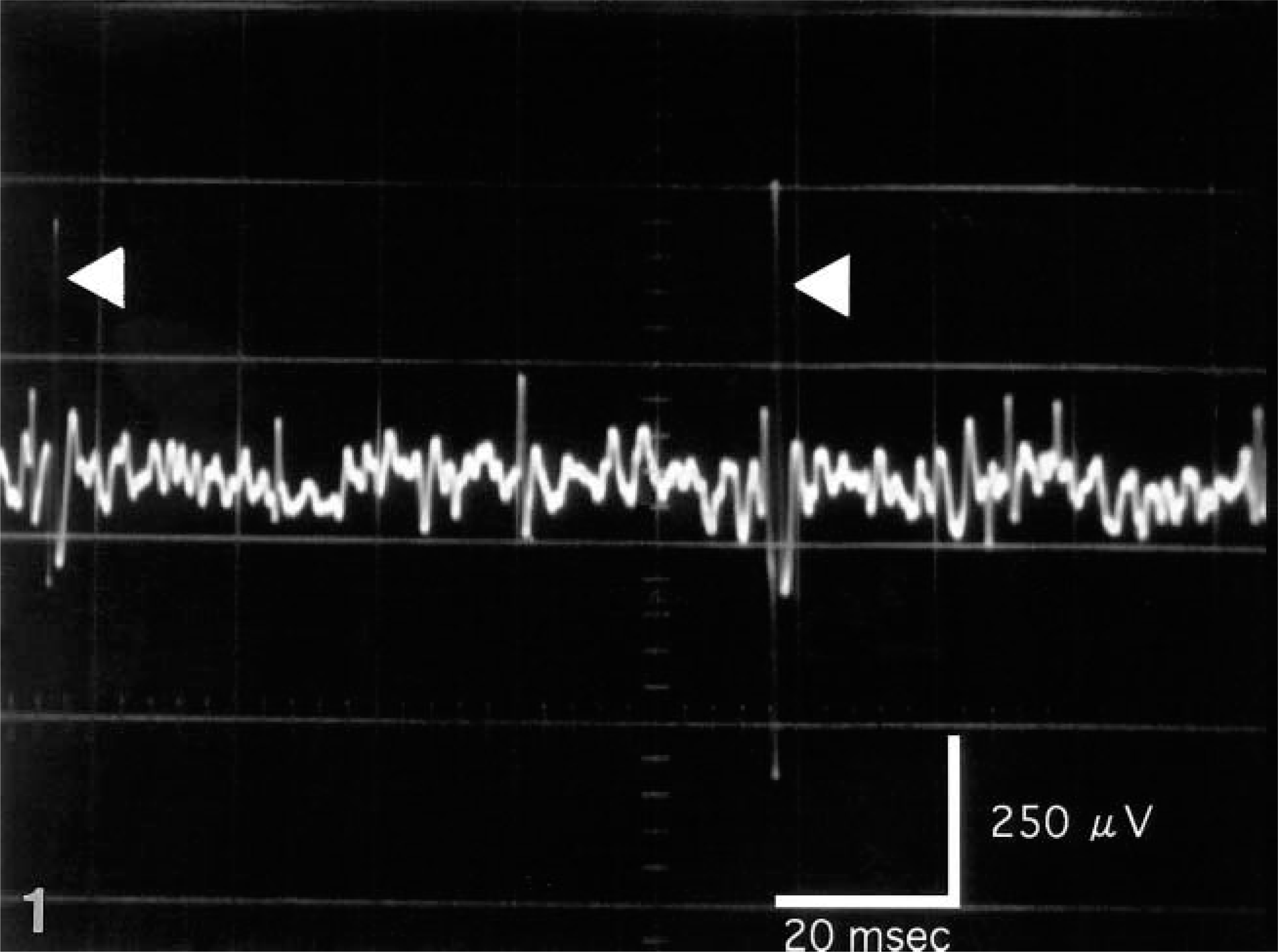
Electromyograph. Left quadriceps femoris muscle; dog. Fibrillation potentials with biphasic negative–positive waves were recorded in non-end-plate regions. The fibrillation included bizarre high-frequency repetitive potentials (arrows).
The muscles in the regions of the head, lumbar vertebrae, and femur were severely atrophic and showed whitish discoloration. Atrophic changes in muscles of the trunk, brachial region, tongue, esophagus, and heart were not prominent. All other tissues were grossly unremarkable.
The seven biopsied skeletal muscles showed loss of myofibers and myofiber regeneration characterized by central nuclei, nuclear chains and basophilic cytoplasm. Also present were perivascular, perimysial, and endomysial infiltrations of lymphocytes, macrophages, and plasma cells and fibrosis and fatty tissue replacement (Fig. 2). Immunohistochemical analysis revealed that inflammatory cells in perimysial sites in the early stage of myositis were labeled by antibody against canine CD8 (Fig. 3) and CD3. Few cells showed a positive signal for HLA-DR antibody and stained with Isolectin B4. The majority of nonnecrotic muscle fibers showed specific staining for class I MHC partly on the surface membrane (Fig. 4), whereas there were few positive reactions around the muscle fibers in two control cases (Fig. 5). In the most severely affected areas, infiltrative cells were stained by the antibodies against human CD3 and HLA-DR and stained with Isolectin B4 (data not shown).1 There was no intense expression of class I MHC in the severe inflammatory lesions of muscle because the muscles were completely affected and necrotic.
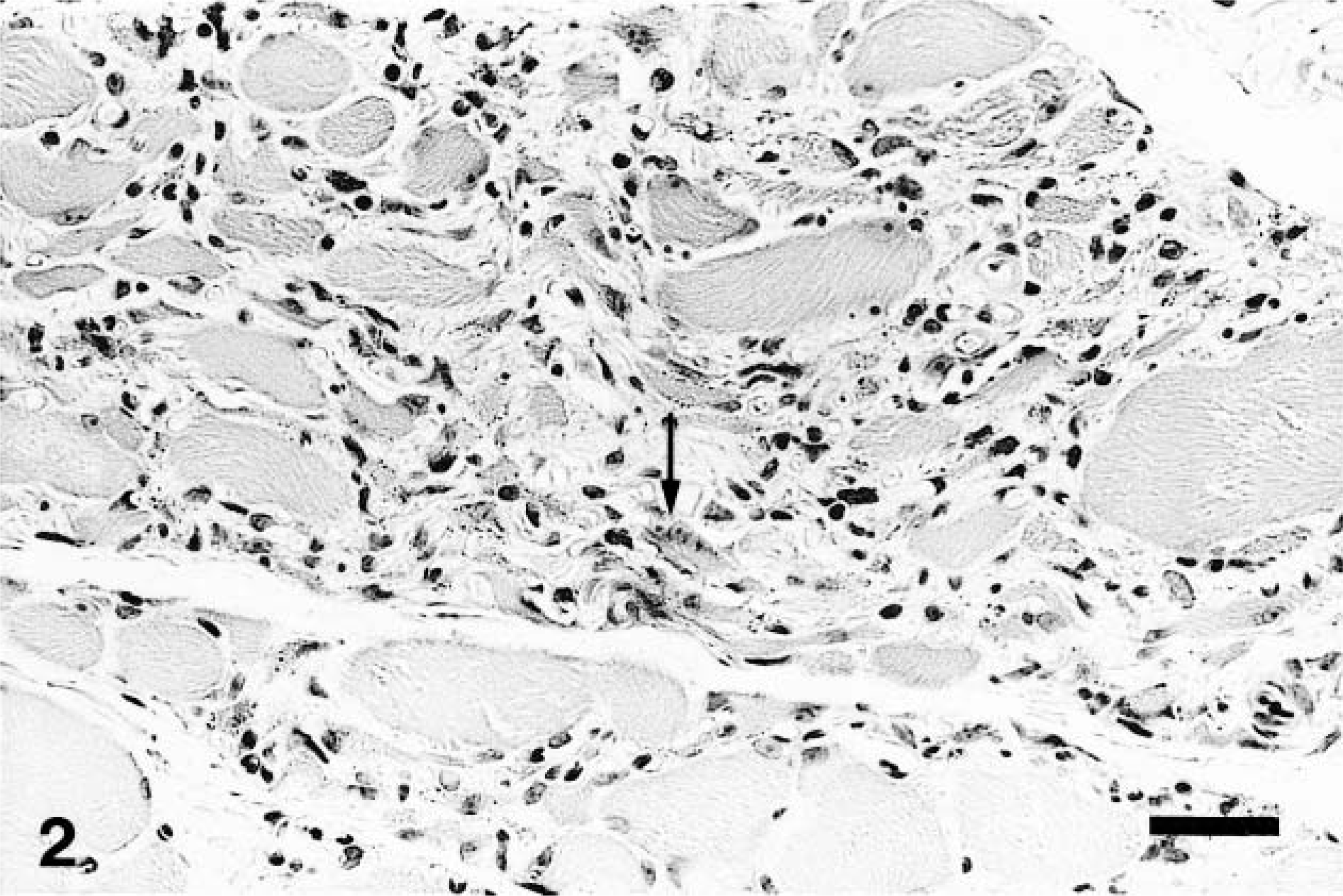
Quadriceps femoris muscle; dog. Note marked loss of skeletal muscle fibers, endomysial infiltrations of lymphocytes and macrophages, and regenerative myofibers (arrow). HE. Bar = 29.4 μm.
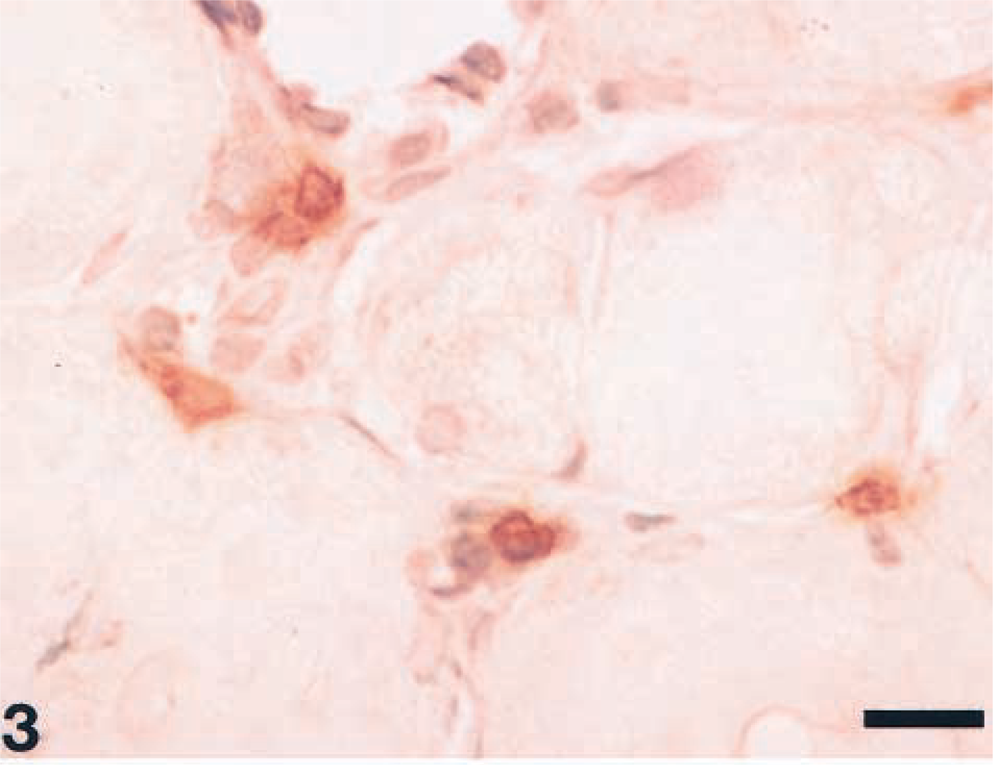
Biceps brachii muscle; dog. Note perimysial infiltration of cells with positive reaction to canine CD8 in the early stage of inflammation. Labeled streptavidin biotin, methyl green counterstain. Bar = 22.1 μm.

Biceps femoris muscle; dog. Note scattered increased expression of class I MHC on the surface membrane of some muscle fibers (arrows). Labeled streptavidin biotin, methyl green counterstain. Bar = 44.1 μm.

Biceps femoris muscle; dog (control). Note slight expression of class I MHC on the surface of the muscle fibers. Labeled streptavidin biotin, methyl green counterstain. Bar = 44.1 μm.
The histopathologic examination of necropsy materials revealed variably severe generalized muscle inflammation. The muscles in the regions of the head, lumbar vertebrae, and femur were most severely affected, whereas the inflammation was less prominent in the trunk, brachial region, tongue, and esophagus. Severe myositis with involvement of peripheral nerves (neuritis) was noted in some skeletal muscles such as the sternocephalis, scalenus dorsalis, intercostales, diaphragma, omotransversarius, trapezius, rhomboideus, teresmajor, gluteus medius, gluteus profundus, semitendinosus, sartorius, adductores and gastrocnemius. There were some foci of neurogenic grouping atrophy of myofibers in these sites. No inflammatory lesions were found in the skin or myocardium.
Hematologic, electromyographic, and histopathologic findings for this dog resembled those reported in humans with polymyositis.4,11,14,15 Electromyographical examination revealed increased insertional activity characterized by polyphasic potentials of variable amplitude and duration. This activity, which is highly characteristic of polymyositis, has been considered a result of alteration of the muscle membrane.14 The fibrillation potentials are most likely the result of segmental necrosis of muscle fibers.14 We used immunohistochemical techniques on frozen sections of mild lesions to identify cells in the early stage of myositis. CD8+ T cells were intimately associated with fewer macrophages, and increased expression of MHC class I was apparent on the surface of noninflamed muscle fibers. Previous immunohistochemical studies have demonstrated that intense expression of MHC class I and close interaction between cytotoxic T cells and macrophages consistently occur in humans with polymyositis.9 The expression of class I MHC on target cells is considered to be indispensable for the cytolytic action of cytotoxic T cells sensitized to a foreign molecule, such as a viral antigen that appears following infection of the target cell.10,12 There is ample evidence of damage to muscle fibers caused by cytotoxic T cells, which in general require class I MHC expression on target cells for the deleterious action.2,3,7,8 However, in humans, the factors or agents that trigger increased class I MHC expression have not been defined. Because of the lack of suitable immunologic reagents, it was not possible to identify cells. Therefore, we could not determine whether CD4+ T cells were present in the lesions.
Histopathologic findings in this case were characterized by severe development of myositis in the head, lumbar, and femur sites and by neuritis among the severely affected muscles. Expression of class I MHC was not demonstrable in these severe lesions because myocytes were completely necrotic. In a previous study of masticatory muscle myositis, there was a predilection for myositis with increased production of type 2M fiber-specific antibodies.15 Serum antibodies were not analyzed in the present study.
The results of this study demonstrated that both class I MHC expression on the surface of muscle fibers and participation of CD8+ T cells can play an important role in initiation of myositis. These findings may contribute to the elucidation of the pathogenesis of polymyositis in both humans and animals. Further study is required for the identification of the causative agents responsible for the intense expression of class I MHC and subsequent inflammation.
