Abstract
Sarcopenia, the age-related loss of muscle mass and strength, is a multifactorial condition that represents a major healthcare concern for the elderly population. Although its morphologic features have been extensively studied in humans, animal models, and domestic and wild animals, only a few reports about spontaneous sarcopenia exist in other long-lived animals. In this work, muscle samples from 60 healthy Podolica-breed old cows (aged 15–23 years) were examined and compared with muscle samples from 10 young cows (3–6 years old). Frozen sections were studied through standard histologic and histoenzymatic procedures, as well as by immunohistochemistry, immunofluorescence, and Western blot analysis. The most prominent age-related myopathic features seen in the studied material included angular fiber atrophy (90% of cases), mitochondrial alterations (ragged red fibers, 70%; COX-negative fibers, 60%), presence of vacuolated fibers (75%), lymphocytic (predominantly CD8+) inflammation (40%), and type II selective fiber atrophy (40%). Immunohistochemistry revealed increased expression of major histocompatibility complex I in 36 cases (60%) and sarcoplasmic accumulations of β-amyloid precursor protein–positive material in 18 cases (30%). In aged cows, muscle atrophy was associated with accumulation of myostatin. Western blot analysis indicated increased amount of both proteins—myostatin and β-amyloid precursor protein—in muscles of aged animals compared with controls. These findings confirm the presence of age-related morphologic changes in cows similar to human sarcopenia and underline the possible role of amyloid deposition and subsequent inflammation in muscle senescence.
Aging is a natural life process in all animal species 39 ; some of its most serious consequences are the effects on skeletal muscle. 43 Given the number of functions of muscle, such as deambulation and protein metabolism, alterations of contractile structures or metabolic pathways in skeletal muscle may severely impair animal health. 48 Moreover, in the elderly, the morphofunctional decline of the neuromuscular system is one of the most common causes of independence loss. 16,38
The word “sarcopenia” was used for the first time in 1989 by Irwin Rosenberg to state the age-related decrease of muscle mass. 12,43,44,45 At present, sarcopenia has been defined as the condition of muscle wasting strictly linked to aging, characterized by loss of skeletal muscle mass and function, which may affect both animals and humans, at different stages of severity 12,27,43 ; it is characterized by gradual loss of skeletal muscle mass resulting in decline of muscle function characterized by a decline in force-producing capacity and maximum velocity of shortening of muscle fibers. 48 These age-related alterations in skeletal muscle are attributed to a complex of factors involving neuromuscular junction, 26 muscle structure (architecture and fiber composition), and metabolism. 43,47,49 The loss of muscle mass and strength results from the progressive atrophy and loss of single muscle fibers, 23 along with loss of both types of motor units (slow and fast). 36 Several studies 36,55 indicate that, in sarcopenia, type II fibers (fast-twitch) seem to be more likely to undergo atrophy than type I fibres. Moreover, the progressive decrease of skeletal muscle quality is associated with fibrosis and intracellular deposition of non-contractile material. 37
The pathogenesis of these alterations are associated with specific biological processes, such as oxidative stress. 54 Oxidative metabolism generates reactive oxygen species (ROS), which accumulate over time and are responsible for damaging and altering cell components, particularly mitochondria and DNA sequences. 8,59 It has been reported that skeletal muscle is a tissue extremely sensitive to this process. 31
Mitochondrial dysfunction is also involved in the pathogenesis of degenerative diseases, predisposing skeletal muscle to the accumulation of β-amyloid precursor protein (AβPP). 50 The same mechanisms are reported in the human sporadic inclusion body myositis (s-IBM), the most common idiopathic inflammatory muscle disease in patients >50 years of age. 13,25,40 An intriguing feature of s-IBM muscle fiber phenotype is its similarity to Alzheimer disease, including accumulation of β-amyloid, phosphorylated tau, and several other proteins characteristic of Alzheimer disease. 3
Sarcopenia has been described in several animal species, from nematodes, flies, rodents, domestic and wild animals, and nonhuman primates to humans. 6,32,39,43,45,52
No information is available about sarcopenia in farm animals because of their short period of lifespan. Some exceptions are presented, such as Podolica breed, which is a dual-purpose bovine breed very common in the south of Italy that it is usually raised free outdoors in the mountains with a lifespan of about 15 years. In this article, we describe the age-related changes of skeletal muscle in Podolica cows to highlight a new model of sarcopenia for comparative studies.
Materials and Methods
Skeletal muscle samples from 60 elderly Podolica-breed cows (aged 15–23 years) and 10 young controls (3–6 years old) were studied by histochemistry, histoenzymatic procedure, immunohistochemistry, immunofluorescence, and Western blot.
The animals were randomly selected for this study during the arrival to the abattoir, and all old cows presented muscle atrophy, which is a clinical finding that can be observed in Podolica cattle >15 years of age.
Animals were subdivided in 3 groups (n = 20 each) and 1 control group (n = 10): Group 1: 15–17 years old (15 years, n = 7; 16, n = 7; 17, n = 6) Group 2: 18–20 years old (18 years, n = 7; 19, n = 7; 20, n = 6) Group 3: 21–23 years old (21 years, n = 6; 22, n = 8; 23, n = 6) Control group: 3–6 years old (3 years, n = 3; 4, n = 2; 5, n = 3; 6, n = 2)
Clinically, cattle showed moderate to severe muscle atrophy associated with weakness and difficulty in walking.
All cows were tested for several chronic diseases (brucellosis, tuberculosis, paratuberculosis, and leukosis) by serologic tests and skin test and then regularly slaughtered according to the European law 852/04 and 853/04 guidelines and procedures. For every animal of the study, a complete postmortem evaluation of carcass and organs was carried out by gross examination. No macroscopic alterations of adrenal and thyroid glands or other lesions commonly related to chronic wasting diseases were observed, including tuberculosis, paratuberculosis, leukosis, brucellosis, parasitosis (intestinal, pulmonary, or hepatic), renal disease, and neoplasia.
Muscle atrophy was evident after slaughtering, in old cows compared with young ones (Figs. 1, 2).
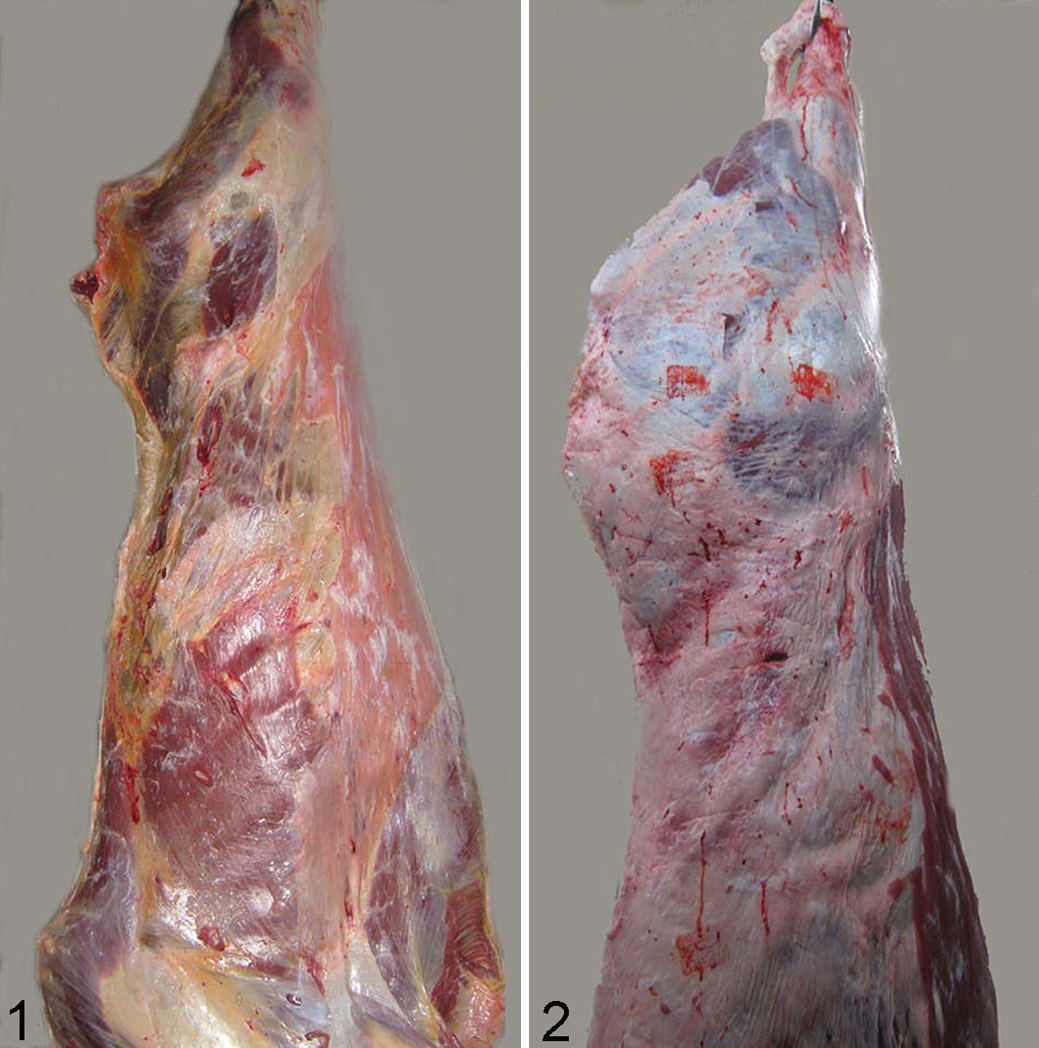
Immediately after slaughtering, skeletal muscle samples 1 × 1 × 1 cm in size from triceps brachii and semitendinosus were collected, snap frozen in liquid nitrogen as previously described, 19 and stored at –80°C.
For histologic and histochemical examination, sections were cut in a transverse plane at 10 µm with a cryostat (–20°C) and stained according to our routinely performed extensive laboratory stains. 41,43 Specifically, we performed (1) hematoxylin and eosin (HE) and Engel trichrome (ET) for a basic morphologic evaluation and mitochondria distribution;(2) reduced nicotinamide adenine dinucleotide tetrazolium reductase to observe distribution of mitochondria; (3) succinate dehydrogenase and cytochrome oxidase (COX) to evaluate activity and distribution of mitochondria; (3) nonspecific esterase for the evaluation of the neuromuscular junctions and lipofuscins; (4) ATPase at pH 9.4 and 4.3 for histochemical fiber type I and II subtyping, respectively; and (5) Congo red with fluorescence microscopy with an excitation filter suitable for fluorochromes such as Texas red for amyloid detection. 2 Approximately 20 fields at 20× magnification were evaluated for each section by 2 independent pathologists (A.C., O.P.) with a concordance rate of 95%.
Immunohistochemical analysis was performed according to the procedure previously described. 41,43 In brief, the cryosections where incubated with primary antibodies (diluted in phosphate-buffered saline) overnight at 4°C. After 2 washes in phosphate-buffered saline, MACH 1 mouse probe (Biocare Medical, LLC, Concord, CA, USA) was applied for 20 minutes at room temperature. After, MACH-1 Universal HPR-Polymer (Biocare Medical) was added for 30 minutes at room temperature. The reaction was detected by DAB chromogen diluted in DAB substrate buffer. Finally, sections were counterstained in hematoxylin.
The primary antibodies used were directed against mouse monoclonal β-amyloid 1-16 (6E10, 1:300, SIG-39300; Signet, Dedham, MA, USA), anti-human CD79α mouse monoclonal antibody (1:200, clone JCB117; Dako, Milan, Italy), anti-human CD3 polyclonal rabbit antibody (1:200; Dako), anti-bovine CD4 (1:200) and CD8α (1:200) monoclonal mouse antibodies (clones GC50A1 and CACT80C; VMRD, Pullman, WA, USA), and MHC I (1:300) monoclonal mouse antibody (clone H58A; VMRD). All the antibodies were established for the use on bovine tissue as described previously. 15
We used specific controls for each immunohistochemistry assay as follows: for antibodies directed against lymphocytes and plasma cell antigens, we used sections from bovine lymph nodes belonging to the archive of Veterinary Medicine Department of University of Naples Federico II. Blood vessel endothelial cells within sections were used as internal control for MHC I expression. For ß-amyloid, we used sections from brain of old chimpanzee with ß-amyloid deposition.
In the corresponding negative control sections, the primary antibody was either omitted or replaced with normal serum from the same species of primary antibody (rabbit or mouse).
To quantify the histologic and immunohistochemical findings, score systems were performed for each parameter; the percentage of atrophic fibers per section, the percentage of necrotic fibers per section, the percentage of ragged red fibers (RRFs)per section, the percentage of vacuolated fibers, the percentage of MHC I–positive fibers, and the percentage of 6E10 (amyloid)–positive fibers were scored as follows: 0 = absent/none, 1%–25% = mild, 26%–50% = moderate, and >50% = severe.
The atrophic fibers, being smaller than normal, were usually easy to identify given the variability and change in fiber size by simple microscopic inspection. Sometimes we used a measurement of the diameter of the smallest and largest fibers with an eyepiece micrometer to establish the range of sizes. 20
Necrotic fibers were detected by HE or ET as pale “liquefied” or hyaline-stained fibers. 20 RRFs were detected as coarsely granular fibers showing prominent peripheral accumulations of abnormal mitochondria, which stained bluish with HE and red with ET stain. 20
Predominantly angular atrophy, the presence of lymphocytic infiltrate, the presence of fiber-type grouping, and the presence of type II selective atrophy were scored as follows: 0 = absent and 1 = present.
Angular atrophic fibers were detected in cross sections stained with HE as fibers that had lost their polygonal shape and had angular profiles. 20 Fiber-type grouping was evaluated on ATPase stains at pH 4.3 and 9.4 as groups of fibers of the same type (>2 fibers) closer to each other and consistent with reinnervation of denervated muscle fibers from collateral nerve sprouting.
For immunofluorescence stain, sections were dried at room temperature for 1 hour, preincubated with normal mouse serum diluted 1:10, and overlaid overnight in a humid chamber at 4°C with primary antibody (GDF8/11 [H109], sc 28910, dilution 1:50; Santa Cruz Biotechnology, Inc, Dallas, TX, USA) directed against myostatin. A TRITC fluorochrome-labeled rabbit anti-mouse secondary antibody was applied (1:50; Jackson Laboratories, Bar Harbor, ME, USA) on sections for 2 hours at room temperature. The slides were washed 3 times in phosphate-buffered saline and mounted under aqueous medium. For observation and photography, a laser-scanning confocal microscope (LSM-510; Zeiss, Oberkochen, Germany) was used.
For Western blot, the muscle tissues from elder cows and controls were cut at the cryostat at 20 µm and then lysed at 4°C in 200 µL of lysis buffer (Tris-buffered saline, 20mM Tris-HCl, pH 7.6, 140mM NaCl, 30mM sodium pyrophosphate, 5mM EDTA, 0.55% Nonided P40, 1% Triton X-100, 50mM NaF, 0.1mM Na3VO4, 1mM PMSF, 1mM benzamidine, 1mM iodoacetamide, 1mM phenanthroline). Protein concentration in the supernatant was determined by BCA protein assay (Pierce, Rockford, IL, USA), and lysates were adjusted to equivalent concentrations with lysis buffer. Aliquots of 30 µg of total muscle lysate were then separated on SDS-PAGE. Proteins were transferred to PVDF membranes that were blocked overnight at 4°C with 5% nonfat milk in TTBS (TBS with 0.05% Tween 20) and probed with primary antibodies anti-myostatin(GDF8/11 [H109], 1:1000, sc 28910; Santa Cruz Biotechnology, Inc) and mouse monoclonal β-amyloid 1-16 (6E10, 1:1000, SIG-39300; Signet), as well as horseradish peroxidase–conjugated secondary antibodies performed in blocking solution for 1 hour at room temperature. Immunoreactive bands were visualized by SuperSignal West Pico Chemiluminescent Substrate Kit (Pierce). The same blots were stripped and reprobed with anti-β-actin monoclonal antibody provided by Sigma-Aldrich (A3853, 1:2000) to confirm equal loading of proteins in all lanes. Quantification of immunoreactivity was performed by densitometric analysis via the Kodak Gel Logic 440 Imaging System (EastmanKodak Company, Rochester, NY, USA).
Statistical Methods
For measuring the association between age and studied parameters, data from immunohistochemistry and the morphology were compared with the gamma statistic G. 51 To establish the relation of present association, Somers d statistic was performed. 51 Data obtained from Western blots were analyzed with StatView software (Abacus Concepts, Piscataway, NJ, USA) by Student’s t test. Blots were revealed by ECL and autoradiography with ß-actin as a loading control. The autoradiographs shown are representative of 4 independent experiments. Bars represent the mean ± SD of 4 independent experiments. P values <.05 were considered statistically significant.
Results
Full results by group are shown in Supplemental Tables 1–3 (groups 1–3, respectively). HE staining of muscle samples from old cows showed variability in diameter size of muscle fibers and mild to severe fiber atrophy. The prevalence of fiber atrophy in studied groups was as follows: Group 1: 40% mild, 30% moderate, 30% severe Group 2: 20% mild, 30% moderate, 50% severe Group 3: 20% moderate, 80% severe
In the control group, no muscle fiber atrophy was observed.
There was a statistically significant positive association between age and presence of muscle atrophy (G = 0.7836; P < .001), and this relation was moderate (Somers d = 0.5956; P < .001).
Angular atrophy (Fig. 3) was observed in 90% of aged animals. The prevalence of angular atrophy in studied groups was as follows: group 1, 80%; group 2, 95%; group 3, 95%. There was a statistically significant positive association between age and presence of angular atrophy (G = 0.8108; P < .001), and this relation was small (Somers d = 0.3000; P < .001).
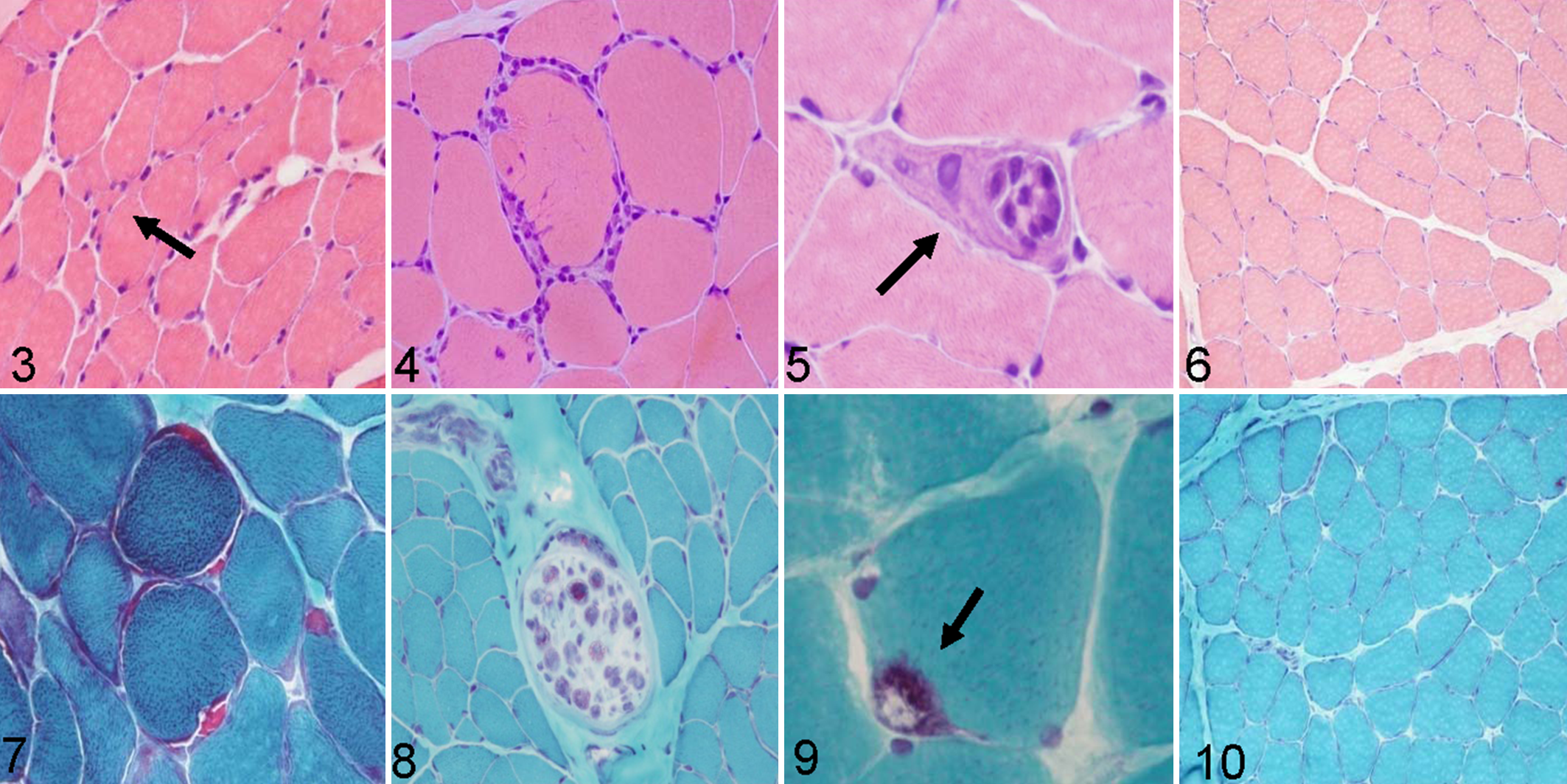
Presence of lymphocytic infiltrates (evidence of inflammation) was not observed on the samples of the control group. Lymphocytic infiltrates (Fig. 4) were observed in 40% of samples from aged animals. The prevalence of inflammation in studied groups was as follows: group 1, 25%; group 2, 35%; group 3, 60%. There was a statistically significant positive association between age and presence of inflammation (G = 0.6176; P < .001), and this relation was small (Somers d = 0.2889; P < .001).
Necrotic muscle fibers (Fig. 5) were observed in 80% of samples from aged animals. The prevalence of fiber necrosis in studied groups was as follows: group 1, 5%; group 2, 55%; group 3, 90%. In the control group, no necrosis of muscle fibers was observed (Fig. 6) . There was a statistically significant positive association between age and presence of necrotic muscle fibers (G = 0.8904; P < .001), and this relationship was moderate (Somers d = 0.4606; P < .001).
RRFs (Fig. 7) and demyelinated nerve axons (Fig. 8) were observed in ET-stained sections. They were observed in 70% of samples from aged animals. The prevalence of RRFs in studied groups was as follows: group 1, 50% mild; group 2, 80% mild; group 3, 60% mild and 20% moderate. There was a statistically significant positive association between age and presence of RRFs (G = 0.6860; P < .001), and this relation was moderate (Somers d = 0.3933; P < .001).
Vacuolated fibers (Fig. 9) were observed in 75% of samples from aged animals stained with ET. Presence of rimmed vacuoles within muscle fibers was not observed on the samples of the control group (Fig. 10). The prevalence of vacuolated fibers in studied groups was as follows: group 1, 65% mild; group 2, 50% mild and 15% moderate; group 3, 50% mild and 40% moderate. There was a statistically significant positive association between age and presence of vacuolated fibers (G = 0.7065; P < .01), and this relation was moderate (Somers d = 0.4628; P < .001).
Succinate dehydrogenase stain showed increased staining in muscle fibers (i.e. ragged blue fibers indicting mitochondrial proliferation) in a percentage of cases similar to RRFs observed with ET stain (Fig. 11) and compared with control (Fig. 12).
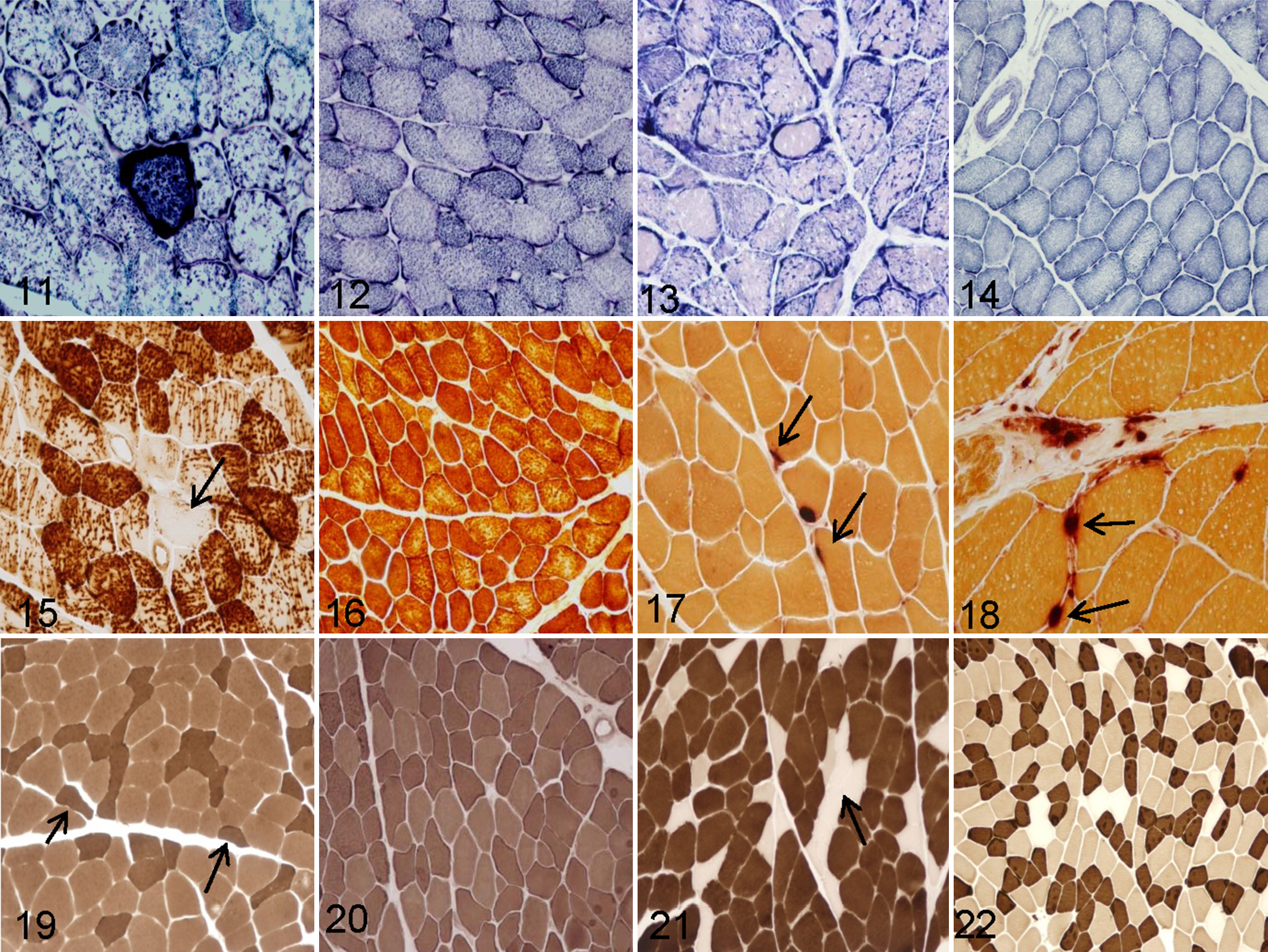
Abnormal internal architecture of the myofibers was studied with the nicotinamide adenine dinucleotide tetrazolium reductase stain, which showed moth-eaten fibers with only a few studied cases (Fig. 13; control, Fig. 14).
All the studied samples from aged animals showed alterations of the cytochrome C oxidase enzyme activity, such as moth-eaten fibers, subsarcolemmal mitochondrial collection, and partially or totally negative fibers (Fig. 15), which were observed in 60% of samples. The prevalence of COX-negative fibers in studied groups was as follows: group 1, 45% mild; group 2, 50% mild and 10% moderate; group 3, 50% mild and 25% moderate. In muscle samples from control group animals, no alteration of cytochrome C oxidase enzyme activity was observed (Fig. 16). There was a statistically significant positive association between age and presence of COX-negative fibers (G = 0.6375; P < .01), and this relation was moderate (Somers d = 0.3850; P < .001).
Nonspecific esterase showed, in 80% of samples from aged cows, fewer and smaller neuromuscular junctions (Fig. 17) versus controls (Fig. 18). Percentage of small neuromuscular junctions in studied groups was as follows: group 1, 20%; group 2, 30%; group 3, 50%.There was a statistically significant positive association between age and presence of small neuromuscular junctions (G = 0.5851; P < .05), and this relation was small (Somers d = 0.2444; P < .05). Nonspecific esterase was suitable also for the identification of lipofuscin in almost all muscle samples of aged cows.
The ATP-ase stains performed at pH 4.3 and 9.4 was used to evaluate which type of muscle fibers undergoes atrophy. Type II fiber selective atrophy was not observed on the samples in control group but was observed on 40% of samples from aged animals (Fig. 19; control, Fig. 20). The prevalence of type II fiber selective atrophy in studied groups was as follows: group 1, 30%; group 2, 40%; group 3, 50%. No statistically significant association between age and presence of type II selective atrophy was observed (G = 0.4854; P = .053).
ATP-ase stains also revealed fiber-type grouping in 83% of samples from aged animals (Fig. 21), which was not observed on the samples in control group (Fig. 22). The prevalence of fiber-type grouping in studied groups was as follows: group 1, 65%; group 2, 85%; group 3, 95%. There was a statistically significant positive association between age and presence of fiber-type grouping (G = 0.8410; P < .001), and this relation was moderate (Somers d = 0.4056; P < .001).
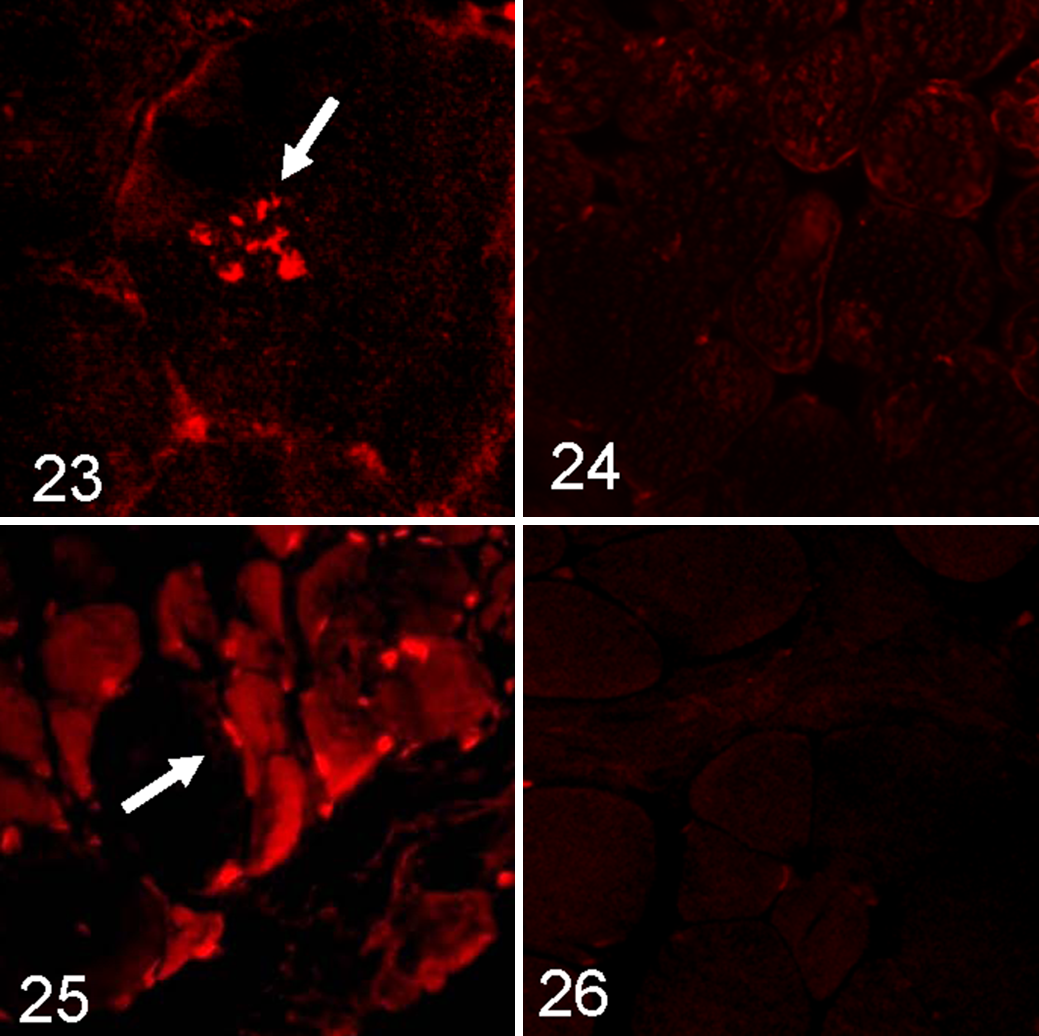
Multiple intrasarcoplasmatic deposits of amyloid identified by Congo red fluorescence visualized through Texas red filters were evident within approximately 30% of samples (Fig. 23; control, Fig. 24).
Immunofluorescence and Immunohistochemistry
Immunofluorescence showed accumulation of myostatin in fibers, mainly the atrophic ones (Fig. 25), in 50% of samples from aged animals. The prevalence of myostatin-positive fibers in studied groups was as follows: group 1, 10% mild; group 2, 20% moderate; group 3, 10% mild and 30% moderate. There was a statistically significant positive association between age and presence of myostatin-immunopositive fibers (G = 0.6364; P < .05), and this relation was small (Somers d = 0.2178; P < .05). No positive reaction on samples in control group was observed (Fig. 26).
Lymphocyte subtypes were identified in the samples based on staining patterns of antibodies against cell surface proteins (Figs. 27–30). In all cases, the predominant cell populations were positive for CD3+, mostly CD8+ (Fig. 27), and rare CD4+ (Fig. 29), with an endomysial, perimysial, and, in some cases, perivascular distribution. No inflammatory cells were observed in the control group (Figs. 28, 30). Scattered CD79α-positive cells were observed in only 2 cases of old cows.
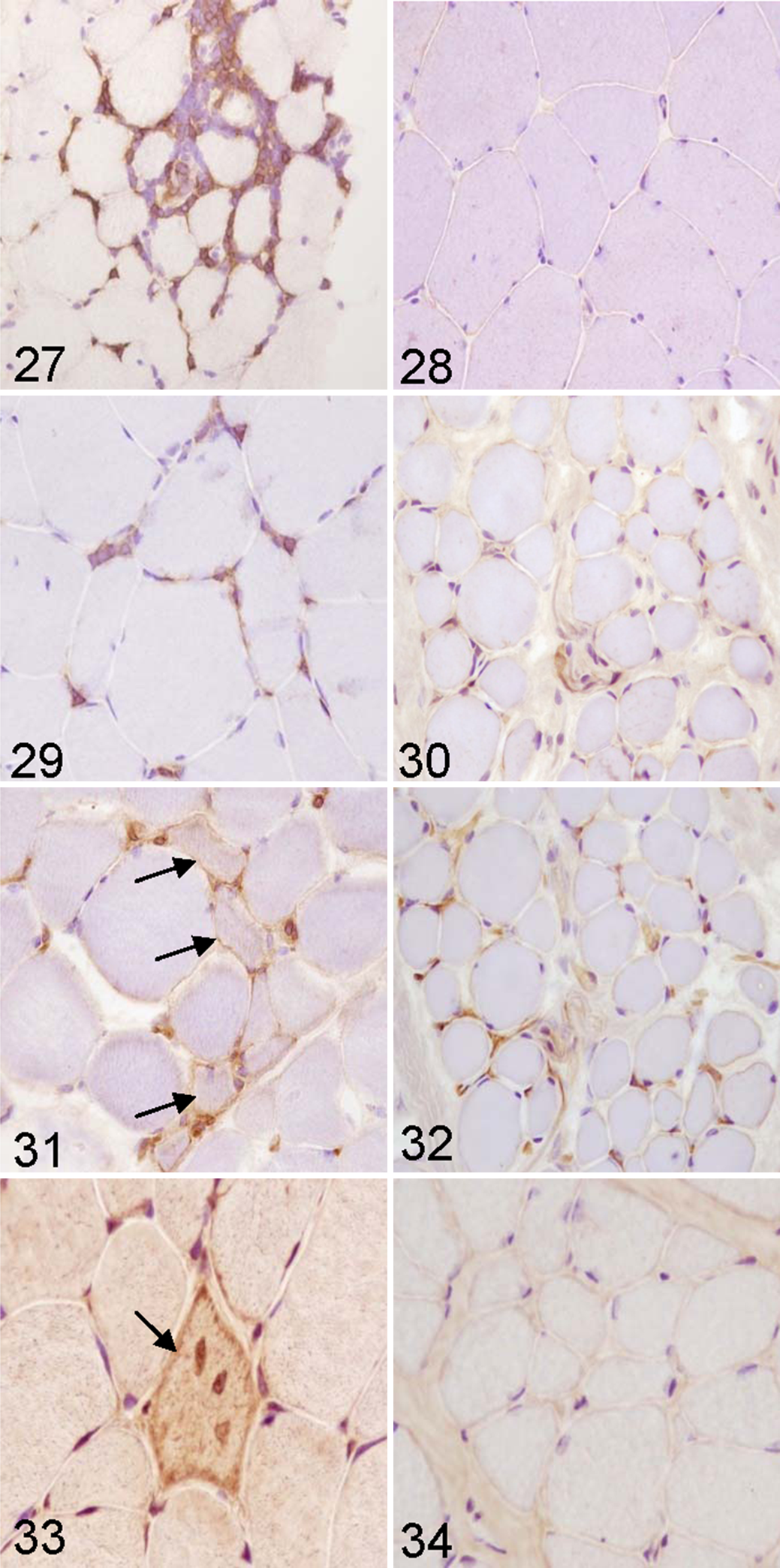
MHC I–positive fibers were observed in 60% of samples from aged animals (Fig. 31). The prevalence of MHC I–positive fibers in studied groups was as follows: group 1, 45% mild and 5% moderate; group 2, 40% mild and 20% moderate; group 3, 40% mild and 30% moderate. There was a statistically significant positive association between age and presence of MHC I–immunopositive fibers (G = 0.5114; P < .05), and this relation was moderate (Somers d = 0.3244; P < .01). In samples from control group, anti–MHC I showed positive reaction only on vessels of perimysium and endomysium (Fig. 32).
Positively stained fibers containing β-amyloid were observed in 30% of samples from aged animals (Fig. 33). The prevalence of 6E10-positive fibers in studied groups was as follows: group 1, 15% mild and 5% moderate; group 2, 5% mild, 20% moderate, and 5% severe; group 3, 5% mild, 20% moderate, and 15% severe. There was a statistically significant positive association between age and presence of 6E10 immunopositive fibers (G = 0.5075; P < .05), and this relation was small (Somers d = 0.2244; P < .05). Stain with primary antibodies 6E10 showed no positive reaction for β-amyloid on samples in control group (Fig. 34).
There was a statistically significant positive association between presence of 6E10-immunopositive fibers and presence of inflammation (G = 0.7172; P < .01), and this relation was moderate (Somers d = 0.4108; P < .001). However, no statistically significant association between presence of MHC I–immunopositive fibers and presence of 6E10-immunopositive fibers was observed (G = 0.4870; P = .079).
Western Blot
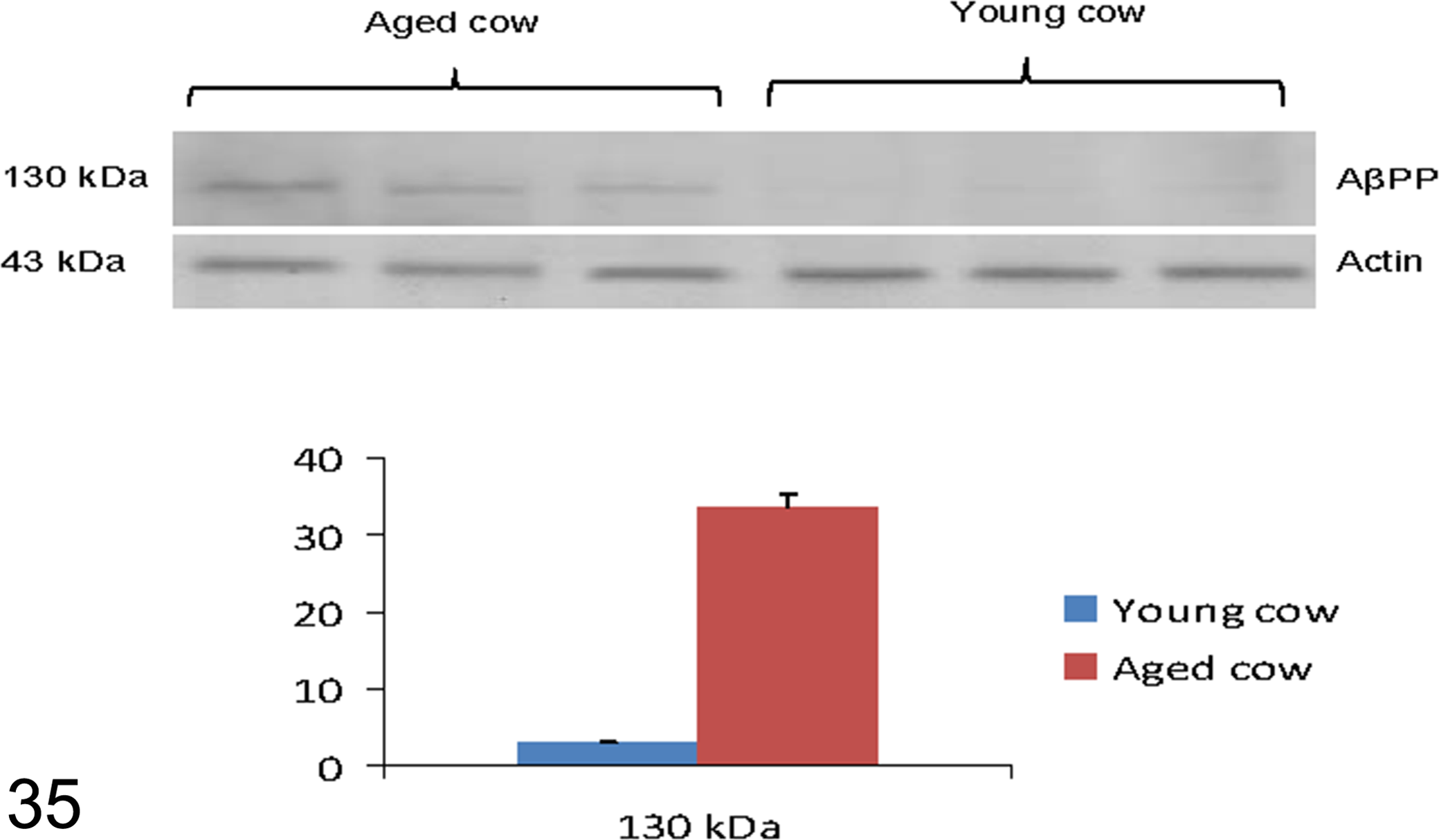
In the studied samples AβPP, recognized by primary antibody 6E10, was revealed as a strong band at 130 kDa (Fig. 35). In the same samples, the primary antibody GDF8/11 recognized 2 bands: one at 55 kDa, which corresponded to myostatin precursor protein, and the other at 28 kDa, myostatin dimer (Fig. 36).
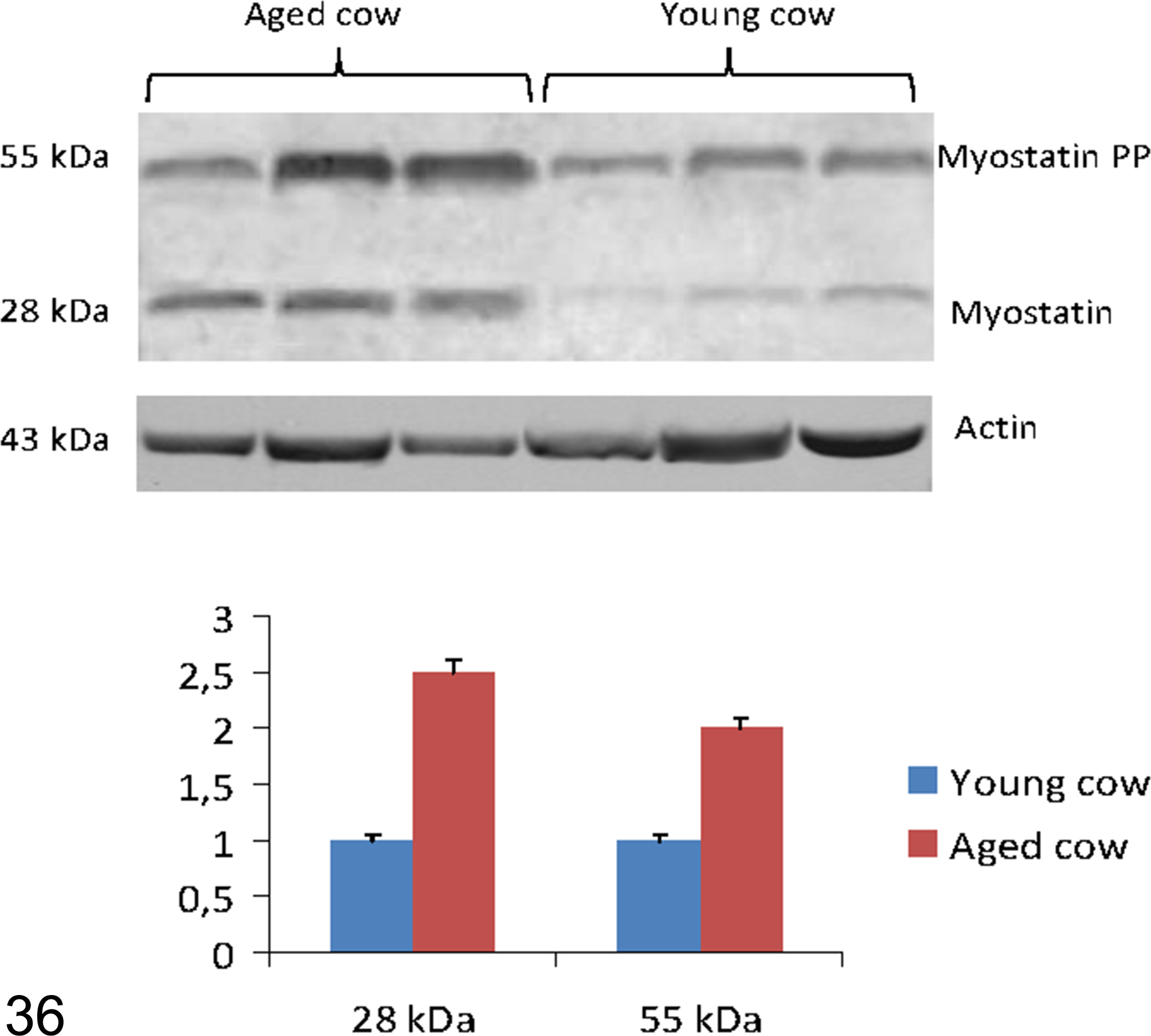
Western blot analysis for myostatin, based onGDF8/11, recognizing 2 bands: at 55 kDa, corresponding to myostatin precursor protein; at 28 kDa, myostatin dimer. Densitometric values of myostatin and its precursor protein (PP) show a significant increase in the older animals versus young controls. *P < .01 vs control.
The results, normalized for actin, showed a significant increase in the oldest animals compared with young controls (P < .01vs control) for AβPP (Fig. 35) and for myostatin and myostatin precursor protein (P < .01vs control; Fig. 36).
Discussion
Sarcopenia is a multifactorial condition that occurs in a variety of species, but only in humans and laboratory animals are extensive data available on its pathogenesis and morphologic features. 7,17,19,22,43 In this work, we describe for the first time some of the most important consequences of muscle aging in cows, suggesting new insights for a comparative approach on the study of this concerning healthcare.
Our most relevant results indicated a statistically significant positive association between (1) age and increasing percentage of COX-negative fibers,(2) age and increasing percentage of angular atrophic fibers and fiber-type grouping, (3) increase of sarcoplasmic deposits of β-amyloid/AβPP and presence of lymphocytic inflammation, and (4) age and lymphocytic inflammation associated with MHC I sarcolemmal expression.
The irregular distribution of mitochondria evidenced by histochemical and histoenzymatic stains (ET, succinate dehydrogenase, and COX) and the presence of COX-negative fibers in many of the samples from elderly cows suggest that, even in this species, sarcopenia is characterized by mitochondrial abnormalities and dysfunctions. In humans, age-related mitochondrial disorders have been associated with the accumulation of mitochondrial DNA mutations mainly induced by increase levels of ROS with aging 1,31 that contribute to the pathogenesis of age-related muscle atrophy. 28 Tissues with limited mitotic capacity such as brain and skeletal muscle are very sensitive to the accumulation of ROS because of their high metabolic activity and the limited ability to counteract the ROS-induced damage with the generation of new cells. 29,33 Mutation and deletion of mitochondrial DNA created by excess ROS leads to the synthesis of defective mitochondrial proteins, decreased ATP production, and further generation of ROS. 30 This increase of ROS is involved in many age-related diseases, such as atherosclerosis, diabetes mellitus, and Alzheimer disease. 1,29,53
Another prominent finding in our study is the presence of neurogenic lesions, such as angular atrophy and fiber-type grouping. In humans and animal models, aging is associated with a gradual loss of motor neurons and neuromuscular junction dysfunction. 46 Even though the pathogenesis of neuromuscular junction impairment is still poorly understood, many factors seem to be primarily involved, such as mitochondrial alteration, age-related chronic inflammation, and neurodegeneration. 26 Previous studies demonstrated an age-related remodeling of motor units leading to progressive denervation, followed by reinnervation from remaining adjacent neurons 18 ; this aspect seems to be involved in the age-related loss of muscle strength and force-producing capacity. 19,47 Denervation of skeletal muscle results in atrophic angular fibers scattered at random or in small or large groups within a biopsy; adjacent intact motor nerve may sprout and reinnervate clusters of denervated fibers resulting in fiber-type grouping. 20 Thus, our results (fiber-type grouping) reflect the reinnervation process of muscle fibers.
Sarcoplasmic deposits of congophilic material, mostly immunoreactive to 6E10 antibody, were detected in 30% of samples and strongly correlated to lymphocytic infiltrates. Age-related β-amyloid deposits in skeletal muscle are typical of s-IBM, the most common muscle disease of older humans. 3,4 The characteristic features of s-IBM at light microscopy are the presence of vacuolated fibers detected on ET and identified as multiple or single foci of ß-pleated sheet amyloid visualized with Congo red fluorescence through Texas red filter associated with extracellular T-cell inflammatory infiltrates. 2,4 The pathogenesis of s-IBM is presumably very complex, but the accumulation of post translationally modified proteins, including β-amyloid, may be eliciting the T-cell inflammatory reaction. 57
A critical issue in human s-IBM and Alzheimer disease is the relationship among cytokines, amyloid, and chronic inflammation. In s-IBM, cytokines such as IL-1β derived from macrophages and T cells are in excess and colocalize with β-APP. Furthermore, evidence indicates that β-APP enhances IL-1β production and IL-1β upregulates β-APP and β-APP gene expression, supporting an interaction between amyloid and inflammatory mediators. 13 Until now, s-IBM was the unique opportunity to study this relationship because of the accessibility of muscle specimens from which β-amyloid can be extracted and monitored; as such, we can offer a new model to investigate this relationship. The immunogenicity of β−amyloid was recently studied in healthy elderly subjects and patients with Alzheimer disease and compared with young/middle-aged adults. 13 Strong β-amyloid reactive and HLA-restricted T-cell responses against the immunogenic β-amyloid 1–42 peptide were found in the elderly, suggesting that β-amyloid is presented as an antigen by antigen-presenting cells in the context of MHC–T-cell receptor interaction. 13
Aside from humans, s-IBM has been reported only once in a dog. 34 To our knowledge, this is the first report of age-related muscle deposits of amyloid; thus, its causative factors are unknown. However, a common pathogenetic mechanism can be presumed.
Furthermore, we observed age-related muscle inflammation, where lymphocytic infiltrates were observed in 40% of samples from aged animals and MHC I–positive fibers in 60%. There was a statistically significant positive association between age and presence of MHC I–immunopositive fibers (G = 0.5114; P < .05), and this relation was moderate (Somers d = 0.3244; P < .01).
What triggers T-cell activation in age-related bovine myositis still remains unclear. In human polymyositis and sIBM, there is evidence of an antigen-directed and MHC I–restricted cytotoxicity mediated by CD8+ T cells, as supported by the following: the cytotoxicity of endomysial T cells to autologous myotubes, the clonal expansion of autoinvasive T cells and the restricted usage of T-cell receptor gene families, the upregulation of costimulatory molecules, and the release of perforin granules by autoinvasive CD8 cells to lyse muscle fibers. 13
Upregulated cytokines, chemokines, and adhesion molecules enhance the transmigration of T cells from the circulation to the muscle. 13
Immunohistochemical detection of sarcolemmal MHC class I is considered a valid test for immune-mediated idiopathic inflammatory myositis in humans 14,56 and dogs, 42 in the presence or absence of cellular infiltration. Muscle fibers do not normally express MHC class I antigens, but in human inflammatory myopathies and canine masticatory muscle myositis, MHC class I sarcolemmal expression has been found to be a valid diagnostic marker for skeletal muscle inflammation. 41,42 MHC I expression has been correlated to the active role of muscle fibers in antigen presentation and in initiating and maintaining pathologic events in immune-mediated myositis. 21 The use of MHC I in bovine myositis has never been described so far; given our results, it could be considered as a diagnostic tool even in this species.
Skeletal muscle atrophy statistically correlated with age in old animals versus young controls. This is a typical histologic finding of sarcopenia in all species. Muscle atrophy reflects a condition of unbalanced protein metabolism that, in sarcopenia, has been associated with a chronic low-grade systemic inflammation (called inflammaging), 24,28 increased levels of muscle-impairing inflammatory cytokines such as TNF-α, age-related hormonal changes, and an imbalanced protein synthesis/degradation. 7,11,44,47,49
Furthermore, in western blot analysis of selected samples from more affected old cows, bands corresponding to myostatin precursor protein and myostatin dimer were significantly increased. Myostatin, one of the transforming growth factor β superfamily members, is a protein secreted by skeletal muscle that is considered a negative regulator of muscle growth during development and skeletal muscle mass during adulthood. 5 The inhibitory action of myostatin on muscle hyperplasia or hypertrophy has been widely confirmed by a phenotype expressed by its encoding gene in knockout mice. 35 The inhibition of myostatin activity (or the administration of compounds that interrupt its intracellular signaling pathways) has been proposed as a potential therapeutic treatment for sarcopenia. 58 Our data suggest that in cattle, myostatin is involved in the onset and progression of sarcopenia, in accordance with previous reports suggesting that myostatin and myostatin precursor protein may play a novel role in the pathogenetic cascade leading to selective type II muscle fiber atrophy. 60
Selective type II fiber atrophy, even if not statistically confirmed, was more frequently observed in older animals versus young controls. This aspect of muscle atrophy has been already reported in transgenic mice and human sarcopenia, given a greater susceptibility of these fibers to oxidative stress. 10,44,54
Conclusions
To date, this is the first report on age-related changes of skeletal muscle in cows. The morphologic changes associated with sarcopenia seem to have many aspects already described in humans and other species. 9,32,52,60 Results refer to muscle samples from only female cows; thus, possible sex-related differences should be further investigated in future studies. 61
More detailed studies on the molecular pathway involved in those alterations will show if cattle could be considered a spontaneous animal model for sarcopenia and other age-related muscle pathologies.
Footnotes
Acknowledgment
We gratefully thank Raffaele Ilsami for his technical support in histology.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this study was supported, by grant Movie of the POR RETE DELLE BIOTECNOLOGIE IN CAMPANIA and, in part, by University of Naples Federico II.
