Abstract
Captive cheetahs have an unusually severe progressive gastritis that is not present in wild cheetahs infected with the same strains of Helicobacter. This gastritis, when severe, has florid lymphocyte and plasma cell infiltrates in the epithelium and lamina propria with gland destruction, parietal cell loss, and, in some cases, lymphoid follicles. The local gastric immune response was characterized by immunohistochemistry in 21 cheetahs with varying degrees of gastritis. The character of the response was similar among types of gastritis except that cheetahs with severe gastritis had increased numbers (up to 70%) of lamina proprial CD79a+CD21– B cells. CD3+CD4+ T cells were present in the lamina propria, and CD3+CD8α+ T cells were within the glandular epithelium. Lymphoid aggregates had follicular differentiation with a central core of CD79a+/CD45R+ B cells and with an outer zone of CD3+ T cells that expressed both CD4 and CD8 antigens. MHC II antigens were diffusely expressed throughout the glandular and superficial epithelium. No cheetah had evidence of autoantibodies against the gastric mucosa when gastric samples from 30 cheetahs with different degrees of gastritis were incubated with autologous and heterologous serum. These findings indicate that T-cell distribution in cheetahs is qualitatively similar to that in other species infected with Helicobacter but that large numbers of lamina propria activated B cells and plasma cells did distinguish cheetahs with severe gastritis. Further research is needed to determine whether alterations in the Th1:Th2 balance are the cause of this more plasmacytic response in some cheetahs.
An unusually severe gastritis associated with multiple types of Helicobacter spp. is a major cause of morbidity and mortality in captive cheetah populations. 5,9–11,38,39,54 This reaction is unlike that seen in free-ranging cheetahs or other felid species (both domestic and zoo held) that usually only develop mild inflammatory lesions when infected with similar Helicobacter.20,24,25,27,29,30,40,44,49,54,58 The lesions in cheetahs occur predominantly in the gastric fundus and are characterized by florid lymphocyte and plasma cell infiltrates, infiltration of glandular epithelium, parietal cell apoptosis and, in more chronic cases, may include gland hyperplasia, goblet cell metaplasia, fibrosis and/or atrophy. 5,10,38,39 Because no single strain of Helicobacter has been associated with gastritis in captive cheetahs 54 and the magnitude of the immune response differs considerably between captive and free-ranging cheetahs infected with similar bacteria, the pathogenesis of cheetah gastritis is suspected to be attributable to the nature of the immune response rather than bacterial factors alone.
In contrast to cheetahs, humans infected with Helicobacter pylori develop superficial gastritis with epithelial cell degeneration and a mixed chronic–active inflammatory cell population within the antral (pyloric) region of the stomach. 8 Neutrophils, a characteristic component of the human inflammatory response, are associated with the presence of the cag pathogenicity island of H. pylori, 47,55 which has not been identified in Helicobacter spp.–infecting cheetahs. 54 The acquired immune response to H. pylori consists of both lamina proprial and intraepithelial lymphocyte (IEL) populations. 2,31 In humans with superficial gastritis, B cells are primarily present in lymphoid follicles, while T cells are more dispersed throughout the lamina propria. 31 Most CD3+ T cells within the lamina propria express CD4, while those in the intraepithelial compartments express CD8. 12,22,31 Most intraepithelial T lymphocytes also express the T-cell receptor α/β heterodimer that recognizes peptides in the context of the major histocompatibility complex (MHC) molecules. These inflammatory cells are present in areas where gastric epithelial cells have increased expression of MHC II molecules. 12 Cytokine profiles indicate that most of the T cells in H. pylori gastritis have a Th1 phenotype. 7,50
Immunophenotypic analysis of the gastric inflammatory response to Helicobacter has also been conducted in several animal models. The inflammatory response of H. pylori–infected domestic cats was similar to that in humans. 13,18 In these domestic cats, lymphoid follicles were composed primarily of IgM+CD45R+ B cells with scattered T lymphocytes (CD4+ and CD8+) surrounding the follicles. Diffuse lymphocytic infiltrates were predominately CD3+CD4+ T cells with small groups of CD4+ and CD8+ T cells present within the epithelium of the gastric glands. Similarly, CD1 mice experimentally infected with a CagA+VacA+ strain of H. pylori have increased numbers of CD4+ and CD8+ T cells within the cardia and corpus, as well as aggregates of B and T cells along the base of the lamina propria. 57 In contrast, gnotobiotic pigs experimentally infected with H. pylori have a predominance of CD8+ T cells within the lamina propria. 32
In addition to the cellular immune response, the humoral response to H. pylori may contribute to disease pathogenesis. 14,15,43 Approximately 89% of H. pylori–infected humans develop anti-Helicobacter antibodies that cross-react with the gastric mucosa, 41 and 25% of these individuals develop antibodies that bind to the canaliculi of parietal cells. 6 These autoantibodies have been correlated with the presence and degree of inflammation and with atrophy of the gastric glands. 43 Specifically, the binding of antibodies to parietal cell canaliculi was significantly correlated with H. pylori infection, the presence of fundic (and not antral) gastritis, increased numbers of periglandular and IELs, increased destruction of the fundic glands, and gastric atrophy. 14–16,42,43,51 This histologic pattern is most similar to that commonly seen in captive cheetahs, so it is possible that affected cheetahs may also develop Helicobacter antibodies that cross-react with the gastric epithelium.
Because of the high prevalence of gastritis and the unusual severity of the inflammatory response in captive cheetahs, more knowledge of their immune response was needed to better understand the pathogenesis of this naturally occurring disease. Toward this aim, infiltrating inflammatory cells in cheetah gastritis were characterized by immunohistochemistry, and the presence of autoantibodies against the gastric mucosa was evaluated.
Materials and Methods
Histology and Leukocyte Immunohistochemistry
This study utilized archived gastric fundus samples from 21 cheetahs representing the spectrum of histologic lesions in this species, and obtained at necropsy (n = 10) or by endoscopic biopsy during routine health examinations (n = 11). Because gastritis in cheetahs is multifocal 10,38 and to ensure accurate representation of the degree of gastritis from biopsy samples, a total of 10 biopsies were taken from the stomach using standard 2-mm flexible biopsy forceps. 5 Biopsy and necropsy samples were taken primarily from the fundic region because gastritis in cheetahs occurs primarily at this site. 10,38 Samples were obtained from visually abnormal regions or from random areas if no lesions were noted visually. Cheetahs (10 males, 11 females) ranged in age from 4 to 17 years, with an average age of 8 years and a median age of 7 years. The study cheetahs were free-ranging in Namibia (Nos. 1, 2) or housed at 8 facilities throughout North America (Nos. 3–21). Gastric samples from 14 cheetahs were fixed in 10% neutral buffered formalin and processed routinely for histopathology. Samples from the remaining 7 cheetahs were obtained fresh and embedded in OCT compound (Tissue Tek, Baxter Diagnostics Inc, McGaw Park, IL), snap frozen in either 2-methylbutane (cooled to freezing point in liquid nitrogen) or in 100% ethanol (cooled to freezing point on dry ice), and stored at –70°C until processing. All samples were sectioned at 5 μm and stained with hematoxylin and eosin to assess the severity of gastritis. The presence of Helicobacter sp. within the lesions was confirmed by Warthin-Starry silver stain and by polymerase chain reaction. Helicobacter spp. were previously determined to be Helicobacter heilmannii–like Helicobacter in all but 3 cheetahs (Nos. 3, 17, 21) that were infected with a H. pylori–like Helicobacter. 54
Immunohistochemistry was performed on 4- to 5-μm paraffin-embedded and snap-frozen sections using a modification of a 3-step labeled streptavidin method previously described. 3,45 Sections from formalin-fixed paraffin-embedded sections were steamed at 94°C for 30 minutes in citrate buffer, pH 6.6, for antigen retrieval. Snap-frozen sections were fixed in 100% acetone for 7 minutes and allowed to air dry. Modifications to the previously described procedure include addition of a blocking step for endogenous avidin–biotin using the Avidin–Biotin Blocking Kit (Vector Laboratories, Burlingame, CA) prior to incubation with normal serum. Normal horse serum was used for all antibodies with the exception of CD3 and CD45R, for which normal goat serum was used. A second blocking step for endogenous peroxidase—using the Peroxidase Blocking Reagent (Dako Corporation, Carpinteria, CA) for 5 minutes at room temperature—was added before the addition of the streptavidin label. Additionally, biotinylated horse anti-mouse or goat anti-rat secondary antibody was incubated for 30 minutes with 1% cheetah serum to block nonspecific binding to plasma cells. Antibodies (Table 1) developed for use against feline leukocyte antigens were provided by Dr P. F. Moore (Davis, CA) with the exception of CD3∊ (Clone CD3-12, Novacostra Laboratories, LTD, Newcastle Upon Tyne, United Kingdom) and CD79a (Clone HM57, Dako Corporation). These CD3∊ and CD79a antibodies are not feline specific but are cross-reactive. All the antibodies were validated for use in the cheetah by flow cytometry and site-specific binding in cheetah tissues (P.F.M., unpublished data). Antibodies against CD4, CD8α, CD8β, MHC II, CD21, and CD5 antigens could be utilized only in frozen sections due to antigen availability. Antibodies were used at a 1:10 dilution for the frozen sections and at the following dilutions in formalin-fixed sections: CD45R, 1:800; CD3, 1:5; CD79a, 1:25; and CD18, 1:20.
Antibody Panel Used to Immunotype Inflammatory Cell Populations in Cheetahs With Gastritis
The proportions of individual lymphocyte subsets within the total inflammatory cell population of the sample were assessed semiquantitatively to determine the relative character of the inflammatory infiltrate regardless of absolute numbers of inflammatory cells: 0, no cells stain; +, < 30% of lymphocytes stain positively; ++, 30–70% of lymphocytes stain positively; +++, > 70% of lymphocytes stain positively. Separate scores were given for the intraepithelial and lamina proprial compartments as well as for lymphoid follicles along the base of the lamina propria.
Immunohistochemical Detection of Autoantibodies
To determine if cheetahs develop anti-Helicobacter antibodies that also bind to epitopes on the gastric mucosa, autologous and heterologous cheetah serum was tested for reactivity against cheetah gastric mucosa. 14 Fresh gastric fundus samples from 25 captive North American and 5 free-ranging Namibian cheetahs were obtained at necropsy (n = 3) or by endoscopic biopsy during routine health examinations (n = 27). All cheetahs were infected with Helicobacter. Seven of these cases were also utilized in the immunophenotyping experiment. To determine if the severity of gastritis determined the development of autoantibodies, samples were selected for varying degrees of gastritis based on previously described criteria 5,38 and included captive cheetahs with mild (n = 5), moderate (n = 9), and severe gastritis (n = 11), as well as 5 wild cheetahs without gastritis. Serum from 10 Helicobacter-infected cheetahs was used to determine if autoantibodies were present; 5 of these cheetahs had moderate to severe gastritis and 5 had minimal gastritis. Because antibodies against cheetah IgG are not available, serum antibodies were isolated and biotinylated using commercially available reagents (IgG Purification Kit and EZ-Link Sulfo-NHS-LC-Biotinylation Kit, Pierce Chemical Company, Rockford, IL). Binding of labeled serum to Helicobacter within the gastric mucosa was utilized as an internal positive control for the isolation and labeling of anti-Helicobacter IgG. To test antibody binding between autologous serum and tissues, biotinylated antibodies from these 10 cheetahs were incubated with sections of their own gastric mucosa. To screen additional tissues for cross-reaction, biotinylated antibodies from 4 cheetahs—2 with severe gastritis and 2 with minimal gastritis—were incubated with heterologous gastric tissues from 20 other cheetahs with different degrees of gastritis.
Fresh gastric samples were embedded in OCT compound and snap frozen as previously described. Five-micrometer sections were fixed in 100% acetone for 7 minutes and allowed to air-dry. Endogenous peroxidase was blocked by incubation for 10 minutes in 3% H2O2 at room temperature. Endogenous avidin and biotin were then blocked using an Avidin-Biotin Blocking Kit (Vector Laboratories). Sections were incubated with 10% normal horse serum (Pierce Chemical Company) to reduce nonspecific binding. Sections were then incubated with biotinylated cheetah antibodies diluted 1:50 in phosphate buffered saline overnight at 4°C. Sections were labeled using an avidin–biotin enzyme complex (Streptavidin Peroxidase, Biogenex, San Ramon, CA) for 20 minutes at room temperature. Diaminobenzidine (Sigma, St Louis, MO) was used as a chromogen. Sections were counterstained with hematoxylin, dehydrated to xylene, and coverslipped. Negative controls were sections incubated with antibodies isolated and biotinylated from goat serum, a species that does not acquire Helicobacter infections. 21
Results
Histology
Cheetahs had varying severities, character, and distribution of gastritis throughout the sections of fundus (Figs. 1–6). Free-ranging cheetahs (Nos. 1, 2) were colonized with Helicobacter but had rare lymphocytes and no lymphoid follicles, neutrophils, or globule leukocytes (Fig. 1). Gastritis in captive cheetahs was classified into 1 of 4 forms: diffuse mild gastritis, diffuse mild with multifocally severe gastritis (Fig. 2), diffuse severe gastritis (Figs. 3, 4), and atrophic gastritis (Fig. 5). Two cheetahs (Nos. 3, 4) had diffuse mild gastritis characterized by small numbers of lymphocytes and plasma cells within the lamina propria, rare globule leukocytes, and some IELs. Cheetahs with diffuse severe gastritis (Nos. 14–17) had large numbers of inflammatory cells in both the superficial and deep regions of the lamina propria, as well as abundant IELs. Inflammatory cells consisted predominately of lymphocytes and plasma cells with variable numbers of large globule leukocytes (Fig. 6). In 2 cheetahs (Nos. 16, 17), the inflammation extended into the submucosa. Disruption of normal glandular structure, loss of parietal cells (many of which were sloughed and within gland lumens), and necrosis were also present (Fig. 4). Intraglandular neutrophils were present in some cases with severe gastritis (Nos. 6–8, 12, 14, 17) but also in 1 cheetah with mild gastritis (No. 3). In all these cases, neutrophils were a minor component of the inflammatory cell infiltrate. The majority of the cases in this study (Nos. 5–13) had a mix of the mild and severe forms. In these cases, the majority of the examined sections had mild gastritis with multiple severely affected areas that had all the features noted in the diffuse severe type of gastritis. Four cheetahs (Nos. 18–21) had atrophic gastritis characterized by large lymphoid aggregates at the base of the lamina propria, mucosal atrophy, and variable lamina proprial fibrosis. In these cases, small numbers of inflammatory cells were present within the lamina propria, but IELs were less prominent than in the other types of gastritis. Lymphoid aggregates were also present within some cases of severe gastritis. Three cheetahs had superficial mucosal erosions (Nos. 8, 13, 17). Glandular hyperplasia was noted in 3 cases (Nos. 13, 14, 17) and metaplasia in 2 cases (Nos. 11, 18). There was no apparent age difference in the type of gastritis, although small numbers in some groups precluded meaningful statistical evaluation. All degrees of gastritis were present in male and female cheetahs with the exception of mild gastritis, which was only present in 2 males.
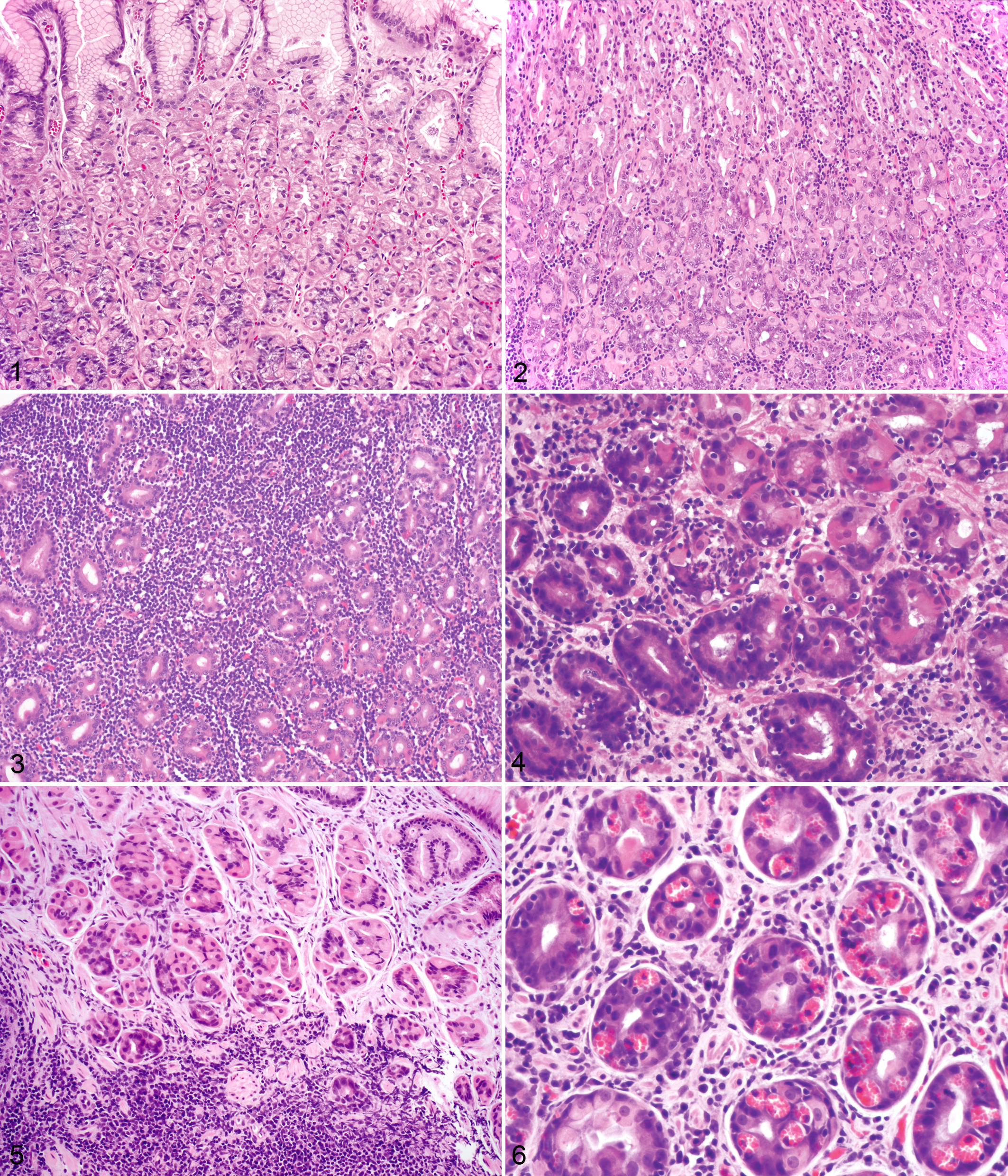
Stomach; cheetah No. 1. Stomach of a free-ranging cheetah that lacks inflammatory cell infiltrates despite colonization with Helicobacter. HE.
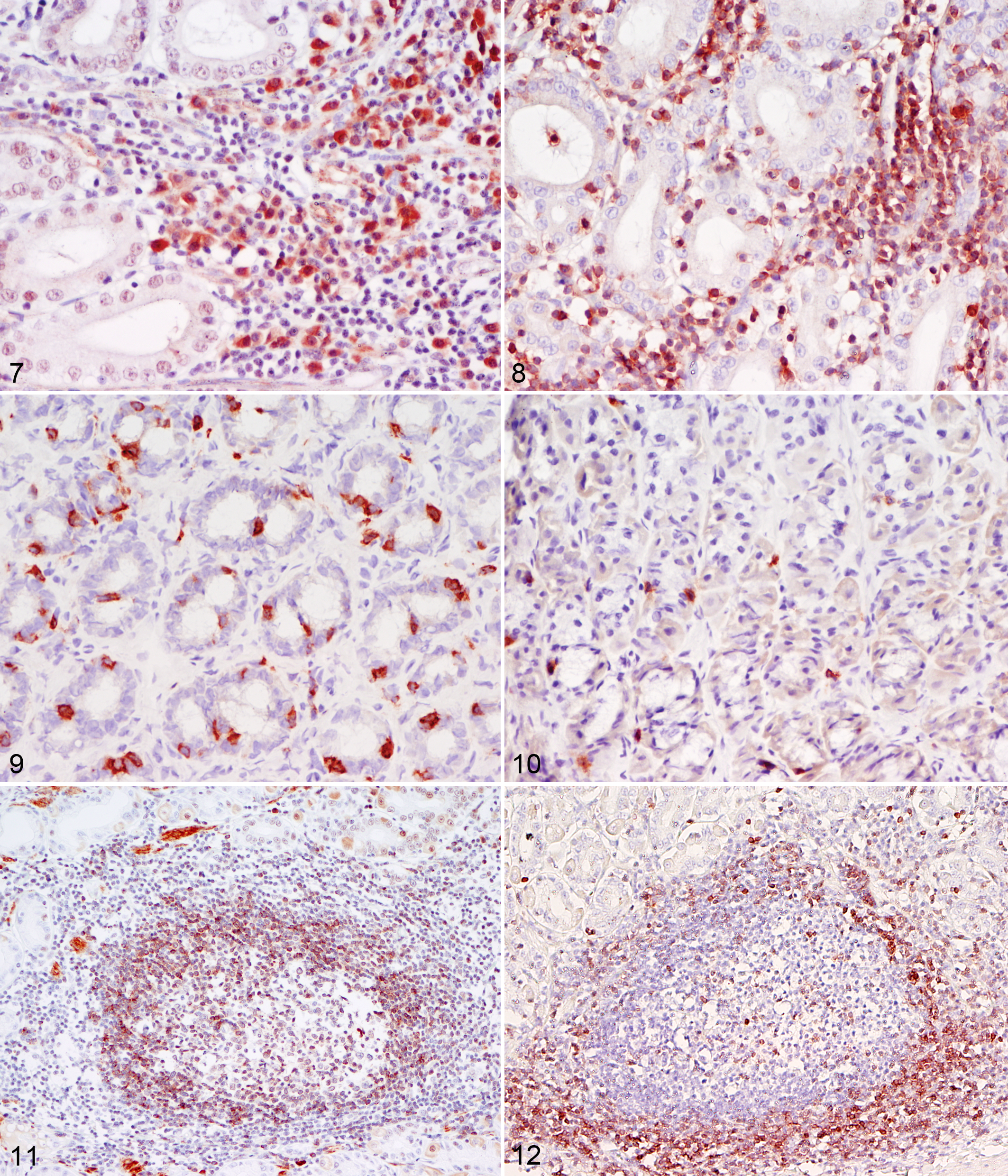
Stomach; cheetah No. 17. Large numbers of CD79a+ B cells and plasma cells (red immunostaining) within the lamina propria in severe gastritis. AEC and hematoxylin counterstain.
Leukocyte Immunohistochemistry
The proportion of cells staining for each leukocyte antigen are listed in Table 2. In general, there was no difference in the proportion and distribution of inflammatory cell types among the different forms of gastritis. However, there were increased numbers of CD79a+ B cells and plasma cells in the lamina propria of the diffuse severe gastritis in comparison with the other types of gastritis (Fig. 7). In contrast to expression of CD79a—with the exception of rare cells in a single cheetah (No. 11)—expression of the B-cell marker CD21 was not detected.
Distribution and Score for the Relative Number Inflammatory Cells Within the Stomach of Cheetahs With Gastritis a
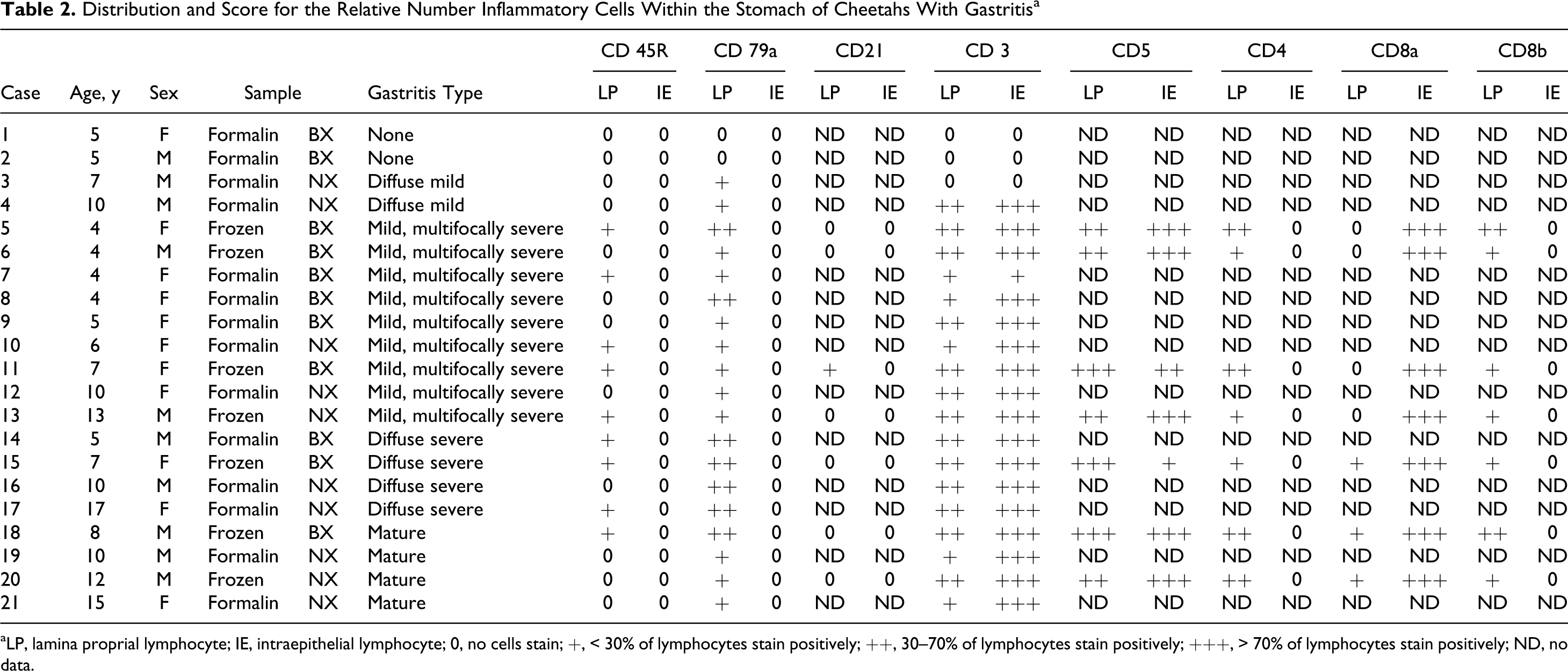
aLP, lamina proprial lymphocyte; IE, intraepithelial lymphocyte; 0, no cells stain; +, < 30% of lymphocytes stain positively; ++, 30–70% of lymphocytes stain positively; +++, > 70% of lymphocytes stain positively; ND, no data.
In all types of gastritis, the majority of inflammatory cells were T cells based on CD3 and CD5 staining (Fig. 8). Of the lamina proprial T cells, the majority expressed CD4, while smaller numbers expressed CD8α and CD8β (Figs. 9, 10). The intraepithelial T cells were exclusively of the CD8α subtype.
In cases of gastritis with lymphoid follicles, cells within the centers of these follicles expressed the pan-B-cell marker (CD79a) (Fig. 11), while the cells along the periphery of the aggregate were predominately T cells that expressed CD3, CD4, and/or CD8α and CD8β (Fig. 12). Rare cells within the centers of lymphoid aggregates expressed CD8α. Small numbers of inflammatory cells within the lamina propria and within the centers of lymphoid follicles expressed CD45R.
The majority of the cells within the lamina propria and smaller numbers of cells within the epithelium expressed CD18. The gastric epithelium (foveolar and glandular) strongly and diffusely expressed MHC II. Large globule leukocytes did not stain with any of the antibodies used.
Detection of Autoantibodies
Labeled antibodies isolated from cheetahs with differing degrees of gastritis bound to Helicobacter in both autologous and heterologous tissue sections. With the exception of nonspecific binding to luminal debris, no specific binding of autologous or heterologous serum to parietal or other epithelial cells was noted irrespective of the severity of gastritis. Labeled goat serum did not specifically bind to the cheetah gastric mucosa or to Helicobacter within tissue sections.
Discussion
The local immune response of cheetahs with generally mild Helicobacter-associated gastritis was characterized by abundant CD4+ T cells and fewer B cells (CD79a+) in the lamina propria and by CD8α+ T cells within the epithelium. Lymphoid aggregates and follicles were composed of CD45R+ B cells with scattered T cells in the germinal centers and around the periphery. This distribution of lymphoid cells is similar to that found in persistently H. pylori–infected humans, domestic cats, and CD1 mice, although the severity and location in the stomach differ. 12,13,22,52 In contrast, cheetahs with severe gastritis have larger numbers of CD79a+ B cells within the lamina propria. CD79a is expressed by all B cells and some plasma cells. None of the CD79a+ B cells also expressed CD21. Typically, CD21 is expressed on mature B cells and follicular dendritic cells but not on activated B cells or plasma cells. 19 These findings suggest that an important feature of severe gastritis in cheetahs is the presence of large numbers of activated B cells and plasma cells.
Two T-cell markers, CD3 and CD5, had a similar pattern of expression. There was also abundant expression of CD5 on IELs within the cheetah gastric mucosa. This is in contrast to previous reports in the domestic cat, mouse, rat, and human that have described decreased expression of CD5 in intestinal IELs. 17,28,35,48 It has been hypothesized that the decreased expression of CD5 in intestinal IELs is due to an absence of interaction between T cells and B cells in the intestinal epithelium. 56 Whether the relatively larger numbers of B cells within the mucosa of cheetahs with gastritis contributes to the increased expression of CD5 is uncertain, and further quantitative studies are needed to corroborate the descriptive findings of CD5 expression in cheetah gastric IELs.
In cheetahs, there was an imbalance in the types of CD8+ lymphocytes within the epithelium, with expression of the CD8α but not CD8β chain, suggesting that these gastric IELs express the CD8αα homodimer rather than the CD8αβ heterodimer. Although the CD8αβ heterodimer is the more common form of CD8 expressed by lymphocytes and the form described in humans with gastritis, 22 small intestinal IELs predominately express CD8αα in domestic cats, mice, rats, and humans. 23,28,33–35,48 It is unknown if the CD8αα homodimer is the most common form expressed by gastrointestinal IELs in cheetahs or if the CD8αβ heterodimer is more commonly expressed. The biological significance of CD8αα homodimer expression by gastrointestinal IELs is not well understood.
In cheetahs, there was diffuse expression of MHC II on the gastric epithelium and inflammatory cells throughout the stomach, as well as diffuse expression of the β2 integrin CD18 on inflammatory cells. Upregulation of MHC II molecules in foveolar and glandular epithelial cells has been a consistent feature of H. pylori infection in humans and mice. 12,57 Whether MHC II expression in cheetahs is a response to Helicobacter infection alone or the inflammatory response cannot be determined. Helicobacter infection is virtually ubiquitous in cheetahs worldwide, and few uninfected cheetahs have ever been documented. 40 Frozen sections from uninfected cheetah cases were not available for analysis. Evaluation of MHC II expression in wild cheetahs, which are infected with Helicobacter but rarely develop gastritis, 40,54 may help elucidate whether MHC II upregulation is a feature of infection or the inflammatory response.
Autoreactive antibodies have been reported in humans and mice with gastritis histologically similar to that noted in cheetahs. 14–16,37,41–43 Serum antibodies against Helicobacter (based on positive staining of the Helicobacter within tissue sections) were present in cheetahs; however, there was no evidence of autoreactive antibodies. This finding suggests that the severe inflammation and atrophy in cheetahs are not due to autoimmunity but to some other, possibly extrinsic, factor affecting the immune response.
Although the cellular constituents of the immune response in cheetahs were generally similar to that noted in other species, cheetahs with severe gastritis had abundant plasma cells noted by histopathology and more activated CD79a+ B cells within the lamina propria than other species. Studies of children with H. pylori–associated gastritis have found increased numbers of lamina proprial plasma cells; however, numbers were reported in comparison with uninfected controls rather than percentage of total infiltrating cells. 36 Some histologic differences have been noted between gastritis associated with H. pylori and that with H. heilmannii in humans; 52 however, no differences have been noted in lymphocyte subsets. 26 Experimental H. heilmannii infection of C57BL/6 mice, however, is associated with large numbers of IgA+ cells within the lamina propria. 46 Although the majority of cheetahs were infected with H. heilmannii–like Helicobacter, 3 cheetahs (Nos. 3, 17, 21) in the current study were previously determined to be infected with an H. pylori–like Helicobacter. 54 No specific type of gastritis was present in these 3 cheetahs, and no differences were noted among cheetahs with the same type of gastritis, despite differing Helicobacter. These findings suggest that, as with humans, 26 no qualitative differences in lymphocyte subsets are noted between the 2 infections in cheetahs.
It has been hypothesized that chronic stress may play a role in the pathogenesis of cheetah gastritis because captive cheetahs develop the most severe disease. In support of this hypothesis, significantly higher baseline cortisol concentrations have been documented in captive cheetahs. 53 Glucocorticoids have widespread and complex effects on the immune system, including alteration of cytokine expression and lymphocyte subpopulations, resulting in a Th1-to-Th2 shift. 1,4 In other species, the immune response to Helicobacter infection is primarily that of a Th1 (cell-mediated) dominant response, 7,50 although a mixed Th1–Th2 response has been identified in H. heilmannii–infected C57BL/6 mice. 46 Given the numbers of activated B cells within the stomachs of cheetahs with severe gastritis, further evaluation of cytokine expression levels and typing of Ig class within the stomachs of cheetahs is warranted to further characterize the immune response.
Footnotes
Acknowledgements
We thank the following institutions and their veterinarians for contributing tissues for this study: Cheetah Conservation Fund, Fossil Rim Wildlife Center, Honolulu Zoo, Kansas City Zoo, Knoxville Zoo, The Living Desert, Saint Louis Zoo, Sacramento Zoo, White Oak Conservation Center, Wild About Cats, Wildlife Safari, and the Zoological Society of San Diego. We also thank Stacy Schultz and Diane Naydan for technical assistance.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported, in part, by the Morris Animal Foundation (D03ZO-81). This publication has not been reviewed or endorsed by the Morris Animal Foundation, and the comments and views expressed do not necessarily reflect the views of the foundation, its officers, affiliates, or agents.
