Abstract
An 11-year-old female miniature Dachshund dog presented with a solid, soft, gray mass on the hepatic lateral left lobe. Histologically, the mass consisted of neoplastic proliferation of cells with round nuclei and eosinophilic and vacuolated cytoplasm arranged in alveolar, trabecular, and solid patterns. Immunohistochemically, the neoplastic cells were positive for pancytokeratin (CK AE1/AE3), CK5, CK14, vimentin, Sox9, and myoepithelial markers (α–smooth muscle actin, p63, and calponin). The morphological and immunohistochemical findings indicated a diagnosis of myoepithelial carcinoma. We conducted immunohistochemical studies on other representative canine hepatic tumors. Although the myoepithelial phenotype was not observed in the hepatocellular carcinoma, some tumor cells in cholangiocarcinoma showed immunohistochemical features of myoepithelium, suggesting that some neoplastic cells in cholangiocarcinoma may have the potential to differentiate into myoepithelial cells. To our knowledge, this is the first report in veterinary medicine of a hepatic carcinoma with a myoepithelial phenotype.
Keywords
In dogs, myoepithelial tumors are most frequent in the salivary and mammary glands, and rarely in the third eyelid, but they have not been described in the liver. 1,7,9,10,12 Commonly diagnosed malignant hepatic tumors in dogs are hepatocellular carcinoma, cholangiocarcinoma, and neuroendocrine carcinoma (carcinoid). Only 2 hepatic tumors with the myoepithelial phenotype have been reported in humans; 1 case was a myoepithelial carcinoma, 6 and the other was an epithelial myoepithelial carcinoma. 16 Human hepatic myoepithelial carcinomas are regarded as most probably of biliary origin. 6
We encountered a hepatic carcinoma with a myoepithelial phenotype in a dog. To our knowledge, there is no other such report in a dog. We report the clinicopathologic findings of this case and compare its immunohistochemical phenotype with those of other hepatic carcinomas that are commonly observed in dogs.
A total of 6 hepatic tumors from dogs were used in this study. All specimens were submitted as biopsy samples to the Laboratory of Veterinary Pathology, Osaka Prefecture University. Case 1 was the myoepithelial carcinoma, cases 2 to 4 were cholangiocarcinoma, and cases 5 to 6 were hepatocellular carcinoma. The clinical and pathological data are provided in Supplemental Table S1 and immunohistochemistry data in Table 1. Case 1, an 11-year-old female miniature Dachshund dog with a history of polydipsia and polyurea for approximately 6 months, presented with a liver mass that was revealed by computed tomography. The liver mass was located on the lateral left lobe of the liver and was 9 × 7 × 7 cm, well defined, solid, soft, and gray.
Immunohistochemistry Findings in the Tumors Analyzed.
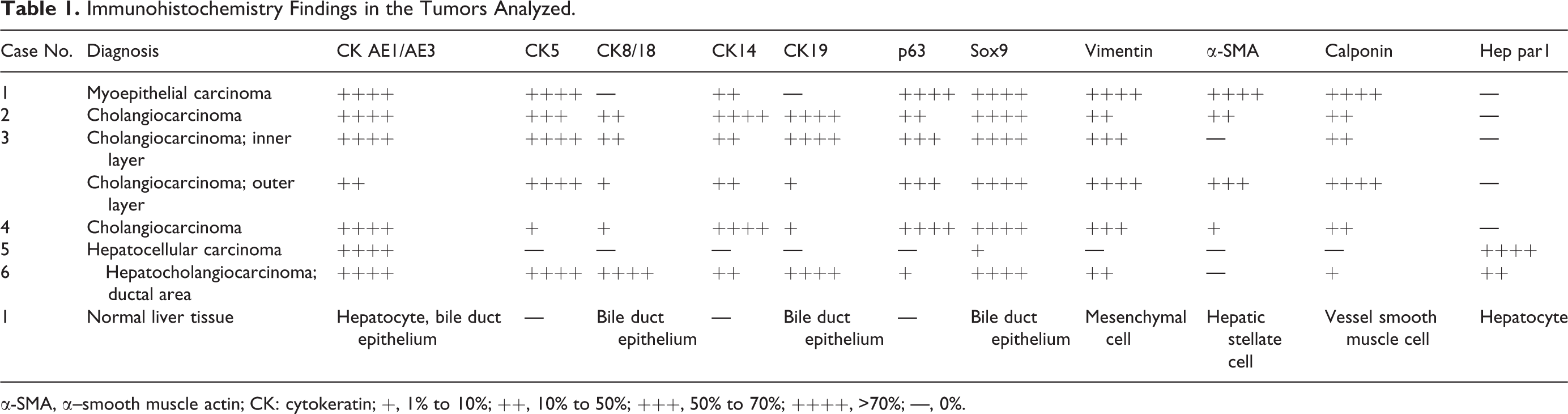
α-SMA, α–smooth muscle actin; CK: cytokeratin; +, 1% to 10%; ++, 10% to 50%; +++, 50% to 70%; ++++, >70%; —, 0%.
To compare immunophenotypes among hepatic epithelial tumors, we examined 3 cholangiocarcinomas (cases 2–4), 1 hepatocellular carcinoma (case 5), and 1 hepatocholangiocarcinoma (case 6). All cases were treated by surgical excision.
In case 4, the mass had close contact with the omentum, which had multiple metastatic nodules. In case 5, the lung and spleen also had metastatic nodules. The other 4 dogs (cases 1, 2, 3, and 6) had no detectable macroscopic lesions other than the single hepatic tumors.
The tumor and normal liver tissues were fixed in 10% neutral buffered formalin, processed routinely, and embedded in paraffin wax. Sections were cut at 3 μm and stained with hematoxylin and eosin (HE). Sections were also subjected to immunohistochemistry (IHC) with primary antibodies specific for cytokeratin (CK) AE1/AE3, CK5, CK8/18, CK14, CK19, p63, Sox9, vimentin, α–smooth muscle actin (α-SMA), calponin, and Hep par1. 3 Details of the antibodies are summarized in Supplemental Table S2. After dewaxing and pretreatment, tissue sections were processed using a Histostainer (Nichirei Biosciences, Tokyo, Japan). Briefly, sections were treated with 5% skimmed milk in phosphate-buffered saline (PBS) for 10 minutes and allowed to react with primary antibodies for 1 hour. After incubation in 3% H2O2 for 15 minutes, the sections were incubated with horseradish peroxidase–conjugated secondary antibody (Histofine Simple Stain MAX PO; Nichirei Biosciences) for 1 hour. Positive reactions were visualized with 3,3′-diaminobenzidine (DAB Substrate Kit; Nichirei Biosciences). Sections were lightly counterstained with hematoxylin.
Tumor tissue from case 1 was also subjected to periodic–acid Schiff (PAS) reaction and reticulin silver impregnation stain (Watanabe’s method) for examination of the basement membrane.
Small pieces of formalin-fixed tumor sample from case 1 were postfixed in osmium tetrachloride, dehydrated through graded alcohols, and embedded in epoxy resin. Ultrathin sections were stained with uranyl acetate and lead citrate and examined with an electron microscope (H-7500; Hitachi, Tokyo, Japan).
In case 1, microscopically, the tumor was mostly encapsulated, but in some areas, the border was ill-defined and neoplastic cells infiltrated the surrounding hepatic tissues. The neoplastic cells were arranged in alveolar, trabecular, and solid patterns and separated by thin fibrous stroma that was demonstrable by PAS and reticulin silver impregnation (Figs. 1, 2). The neoplastic cells had round nuclei, eosinophilic and vacuolated cytoplasm, moderate cellular and nuclear atypia, and 45 mitotic figures per 10 high-power fields. Extensive necrosis was present at the center of the tumor.
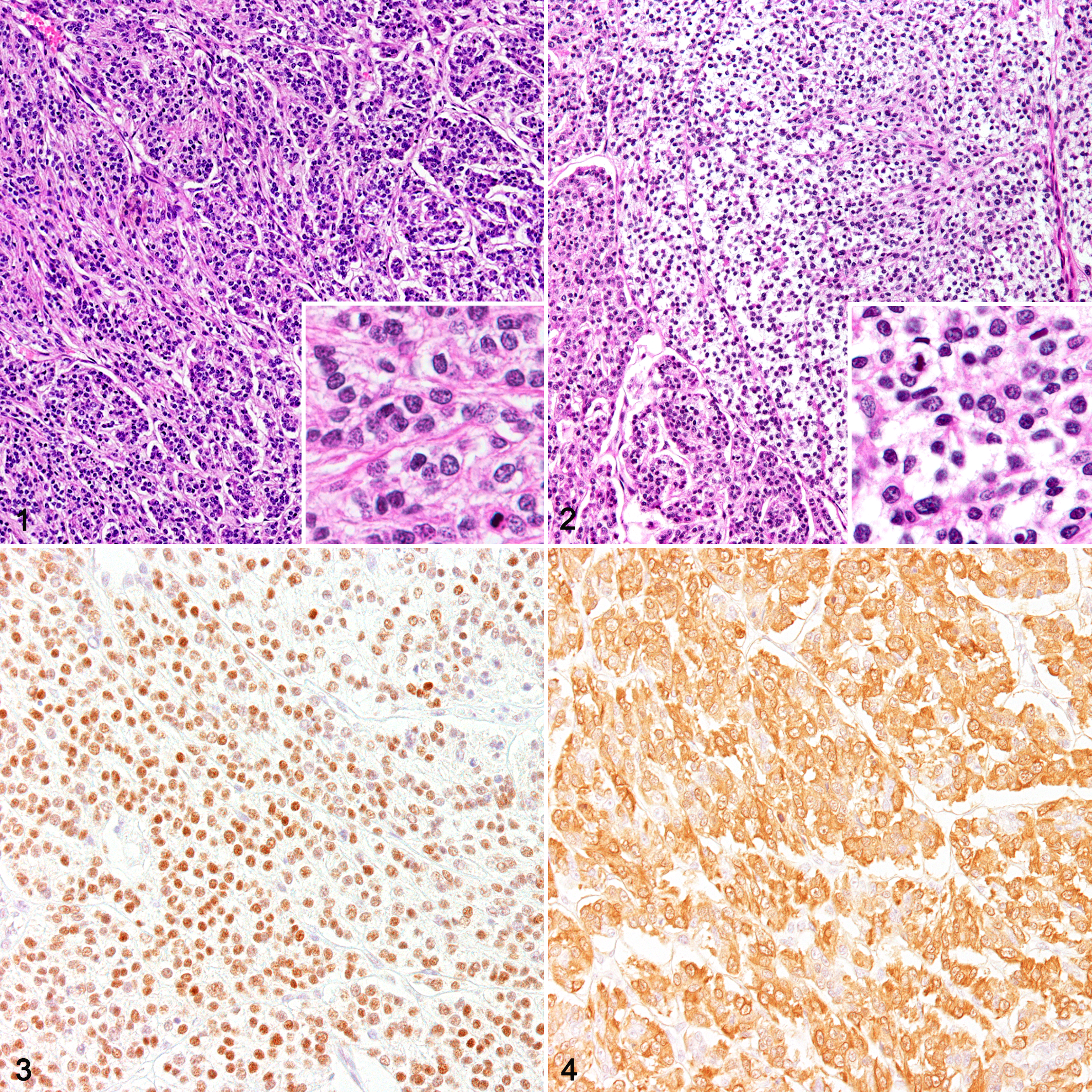
Myoepithelial carcinoma, liver, case 1.
By immunohistochemistry, the neoplastic cells were positive for pancytokeratin (CK AE1/AE3), CK5, CK14, vimentin, and Sox9 (Table 1). The neoplastic cells in the alveolar growth pattern reacted more strongly for CK5 and CK14 compared to the cells in the other areas. These neoplastic cells were also positive for myoepithelial cell markers α-SMA, p63 (Fig. 3), and calponin (Fig. 4) and negative for the hepatocyte marker Hep par1 and the bile duct epithelium markers CK19 and CK8/18. No difference in immunoreactivity was found between the clear cells and the eosinophilic cells. Ultrastructurally, small amounts of filaments were present in the cytoplasm, and neoplastic cells were closely attached to the basement membrane (Suppl. Fig. S1).
On the basis of these findings, we diagnosed this hepatic tumor as a myoepithelial carcinoma.
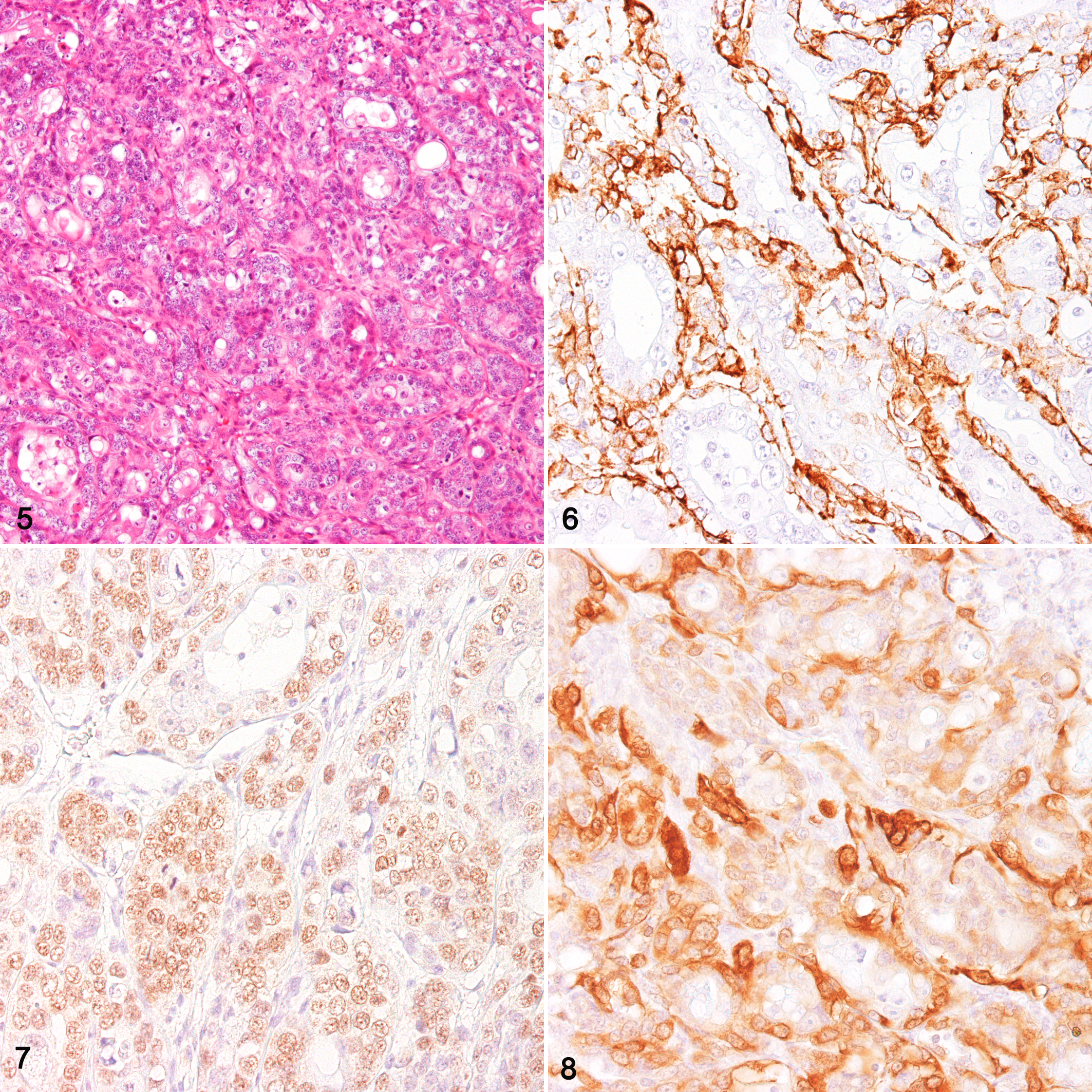
The normal liver tissue showed no immunoreaction for the myoepithelial markers α-SMA, p63, and calponin (Table 1). In the 3 cholangiocarcinoma cases (cases 2–4; Fig. 5), neoplastic cells were positive for CK AE1/AE3, CK5, CK8/18, CK14, CK19, Sox9, and vimentin. Some of the tumor cells were also positive for α-SMA (Fig. 6), p63 (Fig. 7), and calponin (Fig. 8). In cases 2 and 3, p63- or calponin-positive cells were more localized in the outer layer of the neoplastic tubules than in the inner layer. They showed characteristics that were similar to those of case 1, except for the positivity for CK19 and CK8/18.
Cholangiocarcinoma, liver, case 2.
In the hepatocellular carcinoma (case 5), the tumor cells were positive for CK AE1/AE3, Sox9, and Hep per1 and negative for CK5, CK8/18, CK14, CK19, p63, vimentin, α-SMA, and calponin; there were no cells showing a myoepithelial phenotype. In the hepatocholangiocarcinoma (case 6), there were ductal structures positive for CK19 and CK8/18, and parts of them were infrequently positive for p63; however, no cells were positive for α-SMA or calponin, suggesting that hepatocholangiocarcinomas have no cells with a myoepithelial phenotype.
Myoepithelial cells are usually found in exocrine glands, and they exist between the luminal epithelium and the basement membrane. Myoepithelial cells exhibit a dual epithelial and smooth muscle phenotype and can contract and expel the secretions of exocrine glands. Normal bile ducts have no myoepithelial cells. 11 Myoepithelial carcinomas are composed of a pure population of myoepithelium. 2,9,10,12 Myoepithelial neoplasms are relatively rare. Myoepithelial carcinomas in the salivary glands usually contain a proliferation of spindle-shaped, plasmacytoid, or epithelioid cells, with immunohistochemical characteristics of myoepithelium. 9
Histologically, the cytoplasm of the tumor cells in case 1 varied from clear to eosinophilic, yet all of the neoplastic cells showed a myoepithelial immunophenotype. Tumor cells in salivary gland myoepithelial carcinomas are often supported by a basement membrane. 8 In case 1, we observed a close relationship between tumor cells and basement membrane histologically and ultrastructurally. In dogs, myoepithelial tumors occur commonly in the salivary glands but also in the mammary glands. 5 Canine mammary glands have myoepithelial cells that are positive for CK5, CK14, p63, α-SMA, calponin, and vimentin. 13 It has been reported that neoplastic myoepithelial cells in canine complex carcinomas in a mammary gland have immunophenotypes that are similar to those of normal myoepithelial cells. 13 This is similar to our findings regarding case 1.
In another study, a canine salivary malignant myoepithelioma showed immunohistochemical reactivity for CK AE1/AE3, muscle-specific actin and myosin, and focal positivity for CK 14. 2 Neoplastic cells in case 1 were also positive for both cytokeratin and myoepithelial markers. In one reported human case of a primary hepatic myoepithelial carcinoma, the neoplastic cells were positive for CK AE1/AE3, α-SMA, vimentin, and p63 and negative for Hep par1 and CK19, 6 mostly similar to that of the neoplastic cells in case 1.
The normal canine liver tissue showed no immunoreaction for myoepithelial cell markers. Similarly, it was reported that there were no cells positive for p63 in the normal human liver. 8 Hence, myoepithelial cells are not a likely origin of neoplastic cells in case 1.
Immunohistochemically, case 1 showed positivity for epithelial markers—especially low-differentiated cell markers. In addition, neoplastic cells in this case were positive for a foregut marker (Sox 9) and negative for cholangiocyte markers (CK19 and CK8/18). These results suggest that neoplastic cells in case 1 may have the immunophenotype of undifferentiated cholangiocytes. The expression of myoepithelial markers (α-SMA, p63, and calponin) in case 1 also implies myoepithelial differentiation.
In canine mammary gland, it was hypothesized that there are 3 subpopulations in the mammary myoepithelial basal cell lineage: progenitor cells, intermediary myoepithelial cells, and terminally differentiated myoepithelial cells. 13 That study indicated that the progenitor cells were positive for CK5, CK14, p63, and vimentin. The intermediary myoepithelial cells were positive for α-SMA and calponin, and the terminally differentiated myoepithelial cells were positive for vimentin, α-SMA, and calponin. Interestingly, the progenitor cells showed the potential to be luminal epithelium. 4,13 –15 Our present immunohistochemical findings demonstrated that case 1 tumor cells correspond to the abovementioned intermediary myoepithelial cells in the mammary gland basal cell lineage. This suggests that case 1 tumor cells were relatively undifferentiated but more mature than progenitor cells.
We also observed the expression of myoepithelial markers in canine cholangiocarcinomas, a finding that has not been previously reported. Some neoplastic cells in cholangiocarcinomas may have the potential to differentiate into myoepithelial cells. Given the presence of neoplastic cells with immunohistochemical positivity for myoepithelial markers but not mature cholangiocyte markers in case 1, we speculate that the neoplastic cells in this tumor probably originated from undifferentiated bile duct epithelium and underwent myoepithelial differentiation.
In conclusion, we report an unusual canine hepatic carcinoma with the myoepithelial phenotype, plus the presence of neoplastic cells with myoepithelial phenotypes in 3 cholangiocarcinomas. To our knowledge, this is the first report of a hepatic myoepithelial neoplasm in the veterinary literature. The accumulation of additional cases would be useful to further our understanding of the precise pathogenesis of this tumor.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985819854439 - Hepatic Myoepithelial Carcinoma in a Dog: Immunohistochemical Comparison With Other Canine Hepatic Carcinomas
Supplemental Material, DS1_VET_10.1177_0300985819854439 for Hepatic Myoepithelial Carcinoma in a Dog: Immunohistochemical Comparison With Other Canine Hepatic Carcinomas by Yuka Tsuji, Mizuki Kuramochi, Takeshi Izawa, Hideo Akiyoshi, Jyoji Yamate and Mitsuru Kuwamura in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
