Abstract
From 1994 to 2002, tissues from 61 prairie dogs were submitted to Northwest ZooPath for histopathology. Of these, 12 (20%) had hepatocellular carcinoma (HCC). Three were pets submitted from private veterinary practices. The others were submitted from zoos in the United States. All were adults, ranging from young adult to 7 years of age, with average age of 5.1 years. The most common clinical signs were weight loss, lethargy, palpable abdominal mass, and respiratory difficulty. All tumors were well-differentiated HCCs in which four histologic patterns were recognized. The trabecular pattern was predominant in nine tumors, and the pseudoglandular pattern was predominant in two tumors. The pelioid pattern was also represented in eight tumors. A papillary pattern was present in one case. In seven cases vacuolar change resembling lipidosis was present in the neoplastic hepatocytes of both primary and metastatic tumors. Anaplasia was mild to moderate in most tumors, but a marked degree of anaplasia was noted in the metastatic foci of the case with papillary differentiation. Metastasis to lung was noted in five cases. One of these also had metastasis to the spleen, and another had metastasis to heart and mediastinum. In two cases there was concurrent hepatitis and in two cases, cirrhosis. All tumors and nonneoplastic liver stained negatively for woodchuck hepatitis virus surface and core antigens, and orcein and Victoria blue positive staining of hepatocytes typical of hepadnavirus infection in humans and woodchucks was not present. HCC is apparently common in captive prairie dogs. The hepatic neoplasia observed in prairie dogs was similar to that associated with hepadnaviral infection in humans, wood-chucks, and ground squirrels, but no direct evidence of hepadnaviral infection was detected. The rate of metastasis in captive prairie dogs was higher than that reported in woodchucks.
Keywords
Introduction
Prairie dogs (Cynomys spp.) are members of the family Sciuridae, which includes ground squirrels, flying squirrels, and woodchucks. 7 Hepatocellular tumors are common in woodchucks (Marmota monax), 5,12,15,16 and have also been documented in Beechey's ground squirrel (Spermophylus beecheyi), 8 the arctic ground squirrel (S. parryii), 18 and Richardson's ground squirrel (S. richardsonii). 9,17 In these species hepatocellular carcinoma (HCC) is closely associated with infection by related hepatitis B–like viruses (hepadnaviruses).
Hepadnaviruses have a narrow host range and are difficult to propagate in vitro. 10 These viruses have been detected in humans, 10 woodchucks, 5 various species of squirrels, 8,9,17,18 and Pekin ducks. 2 Replication of hepatitis B virus (HBV) can be successfully achieved by transfecting hepatoma cell lines with cloned HBV DNA genomes. 10 However, the lack of culture systems permissive for hepadnavirus infection and the narrow host range of hepadnaviruses have hampered the understanding of viral pathogenesis and tumorigenesis. Infection is generally established based on serologic evidence of surface antigen or core antigen, DNA hybridization using Southern blot analysis of fresh liver, positive staining of hepatocytes in histologic section using orcein or Victoria blue, immunohistochemistry for hepadnavirus core or surface antigens in hepatocytes, or a combination of these techniques. 1,2,6,8,10,11,13,15–18
The association between HCC and hepadnavirus infection in sciurids is similar to that in humans, in whom infection with HBV is a major etiologic factor in hepatocarcinogenesis. 1,11,15,16 In humans there is a strong relationship between chronic HBV infection and development of hepatitis, cirrhosis, and HCC, especially in areas where HBV is endemic, such as China, Southeast Asia, and sub-Saharan Africa. The incidence of HCC in infected individuals is 100-fold higher than in uninfected persons. 1 HCCs of humans are primarily of trabecular pattern and have a moderate rate of metastasis. 3
HCC is common in woodchucks, with several early reports of natural infection and many subsequent reports of experimental hepadnavirus infection and tumorigenesis. 1,5,12 Woodchucks range throughout most of the Eastern and Midwestern United States and throughout Canada. Although a comprehensive seroepidemiologic survey for woodchuck hepatitis virus (WHV) throughout its host range has not been performed, infection is hyperendemic to the mid-Atlantic states. 16 Woodchucks infected at birth that do not clear the infection have a lifetime risk of developing hepatocellular adenoma (HCA) of essentially 100%, usually developing neoplasms by 3–4 years of age. 5,16 As for humans, the trabecular pattern of tumor growth is the most common morphologic variant of HCA in woodchucks. 12 HCC in woodchucks has a very low rate of metastasis. 12
A low prevalence of HCC associated with hepadnavirus infection has also been documented in Beechey's ground squirrels from the San Francisco Peninsula. The ground squirrel hepadnavirus (GSHV) was shown to be closely related to WHV. In this study of 75 wild-caught, long-term (5 years) captive squirrels, three animals, all more than 4.5 years of age or older, developed HCC. Two of these tumors had a trabecular pattern, and one had a medullary pattern. Metastasis was not reported. 8
During studies of seasonal obesity a high frequency of hepatic neoplasms was observed in captive Richardson's ground squirrels. Of 12 Richardson's ground squirrels examined thoroughly, seven had mild or moderate degrees of chronic portal hepatitis, and six (50%) had HCC. Neoplasia was observed in 1.5- to 3-year-old wild-caught and captive-bred squirrels raised in the laboratory but was not observed in wild-caught animals of any age that were not held in captivity, suggesting that factors in the captive environment may increase tumorigenesis. Hepatocellular tumors were primarily of the trabecular type, but pseudoglandular and pelioid variants were also observed. Metastasis was not reported. Serologic testing for hepadnavirus surface antigen, anticore antibody, and virion DNA, which recognize the GSHV of Beechey's ground squirrels, was negative, but Southern blot analyses of liver cell DNA demonstrated fragments that hybridized with a GSHV probe. 17 In a separate study of 40 wild-caught Richardson's ground squirrels from Alberta, Canada, 40% had serologic evidence of GSHV infection, and two had HCCs, indicating that infection can occur in the wild population. 9
Arctic ground squirrels (S. parryi kennicotti) in the Brooks Range of Alaska have also been observed to have a frequent occurrence of hepatic neoplasia. 18 Arctic GSHV, closely related to WHV and GSHV, has been shown to infect free-ranging squirrels in the Brooks Range based on nucleic acid hybridization of neoplastic hepatic tissue with hepadnavirus probes and in plasma by cross-reactivity using antibodies to hepadnavirus surface and core antigens. 18 In a study of captive squirrels, about one third of the aged animals maintained in captivity, including virus-infected as well as uninfected squirrels, developed hepatocellular adenomas, and the incidence of hepatocellular neoplasia was greater in virus-infected than in uninfected animals. 18
There have been few reports of hepatocellular neoplasia in prairie dogs, and these were limited to low numbers of animals. 14,19,20 Although hepadenavirus infection is suspected as a possible cause of the tumors, it could not be documented in these animals. The following report describes the incidence, morphology, and immunohistochemistry of HCC in black-tailed prairie dogs submitted to an exotic specialty diagnostic service.
Materials and Methods
Prairie dog cases submitted to Northwest ZooPath, Snohomish, Washington, from 1994 to 2002 were reviewed for diagnosis of HCC. Signalment, history, and gross lesions were recorded. Cases of confirmed HCC were collated and reexamined microscopically by one of the authors (M. M. Garner). Tumors were classified based on microscopic tumor patterns, cellular features, degree of anaplasia, and presence of metastasis. Concurrent disease processes were also recorded.
For each case, sections cut from paraffin blocks containing HCC and nonneoplastic hepatic tissue were stained for hepadnavirus proteins using anti-WH core antibody (WHC) and anti-WH surface antibody (WHS) and the methods described previously. 6 Briefly, the hydrated sections were treated with polyclonal rabbit antibodies against WHC and against WHs. A biotin–streptavidin detection system (ZYMED Laboratories, Inc., San Francisco, CA) was used with biotinylated anti-rabbit immunoglobulin, peroxidase-conjugated streptavidin, and diaminobenzidine as chromogen. Sections were treated in parallel with normal rabbit serum instead of the antibody as negative controls. Sections also were stained using orcein and Victoria blue stains for detection of hepadnavirus surface antigen. 3,13
Results
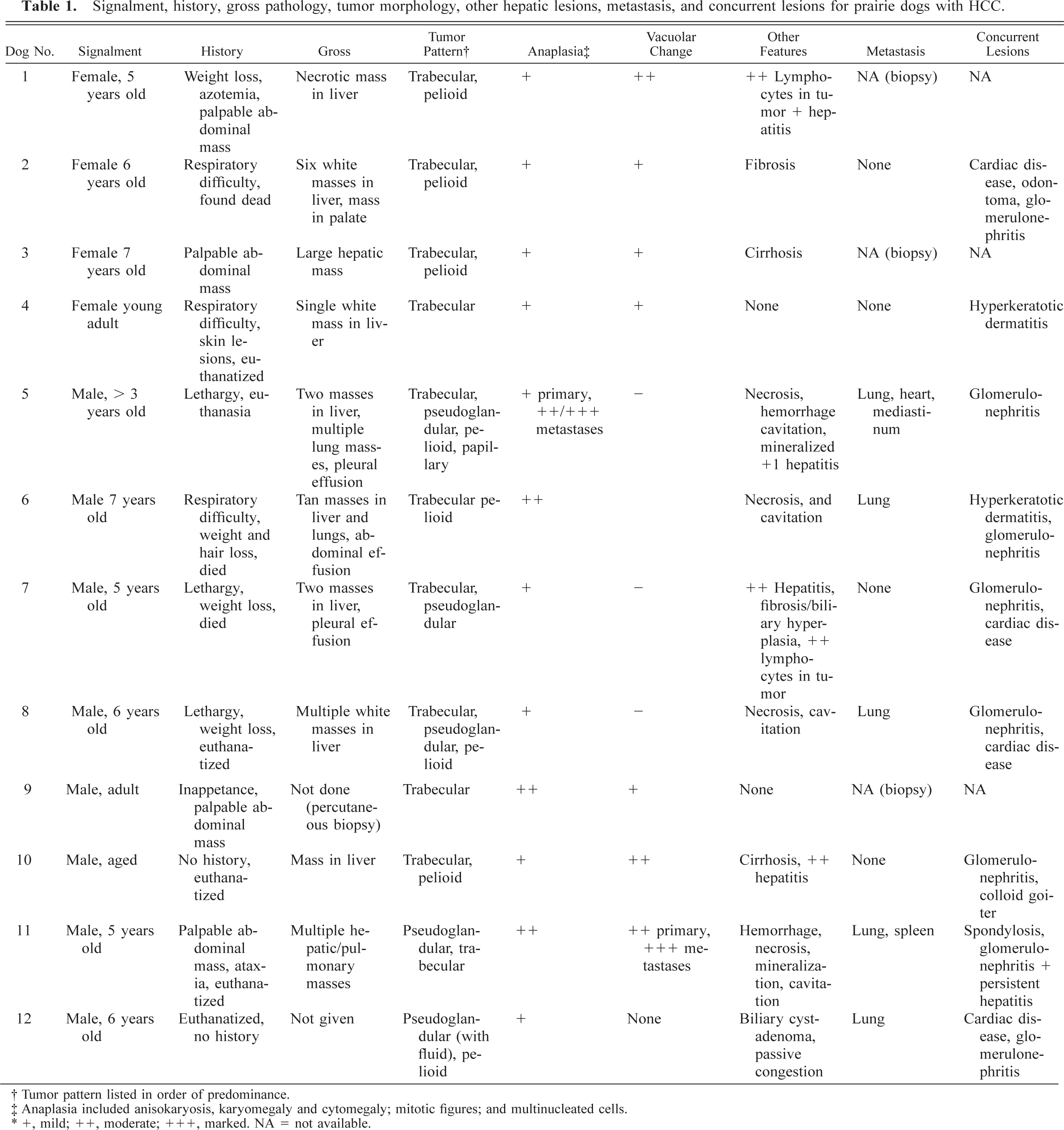
From 1994 to 2002, tissue sets that included liver from 61 prairie dogs were submitted to Northwest ZooPath for histopathologic evaluation. Of these, 12 (20%) had HCC. Three were pets submitted from private veterinary practices. The others were submitted from zoos in the United States. Signalment, history, gross pathology, tumor morphology, other hepatic lesions, metastasis, and concurrent lesions are summarized in Table 1. Prairie dog Nos. 1–4 were female, and prairie dogs Nos. 5–12 were male. All were adults, ranging from young adult to 7 years of age, with average age of 5.1 years for animals of precise known age. Prairie dog Nos. 4–5, 8, and 10–12 were euthanatized; dog Nos. 2, 6, and 7 died; and dog Nos. 1, 3, and 9 were alive at the time this manuscript was written. The most common clinical signs were weight loss (prairie dogs Nos. 1 and 6–8), lethargy (prairie dog Nos. 5, 7, and 8), palpable abdominal mass (prairie dog Nos. 1, 3, 9, and 11), and respiratory difficulty (prairie dog Nos. 2, 4, and 6). One or more masses were noted in various liver lobes of all animals. The masses were white to tan and sometimes cavitated or umbilicated (Fig. 1).
Signalment, history, gross pathology, tumor morphology, other hepatic lesions, metastasis, and concurrent lesions for prairie dogs with HCC.
† Tumor pattern listed in order of predominance.
‡ Anaplasia included anisokaryosis, karyomegaly and cytomegaly; mitotic figures; and multinucleated cells.
∗ +, mild; ++, moderate; +++, marked. NA = not available.
Liver; prairie dog No. 11. Coalescing nodular foci of HCC occupy and deform most of the hepatic lobes. Bar = 2.0 cm.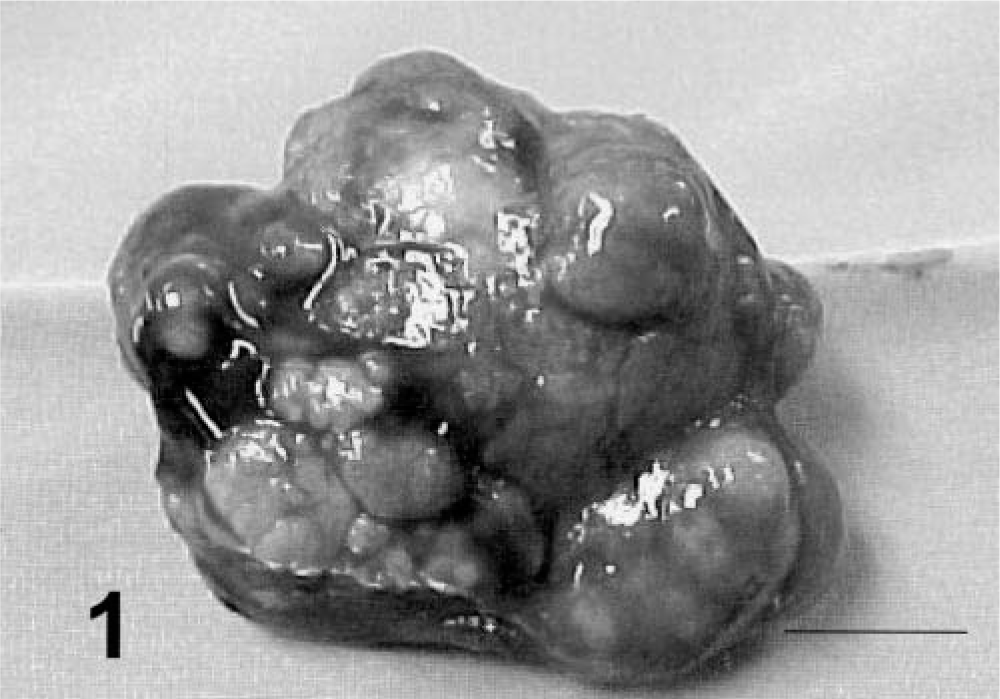
All HCCs were well differentiated, and determining the cell of origin was not difficult even for metastatic lesions. Four patterns were recognized: trabecular/solid, pseudoglandular, pelioid, and papillary. The trabecular/solid pattern was predominant in 9 dogs (Nos. 1–4 and 6–10), including all four female and six of eight male dogs. In these tumors neoplastic cells formed compact cords reminiscent of hepatic cords but with less organization and no clear association with hepatic sinusoids. Cord distinctions were unclear in some areas of these tumors, and neoplastic cells were arranged in solid sheets (Fig. 2).
Liver; prairie dog No. 4. HCC, trabecular/solid pattern. Note the cords and sheets of neoplastic cells, the lack of sinusoids, and the hepatoid appearance of the cells. Note the normal liver in the margin (∗). HE. Bar = 260 µm. Inset: Note the slightly enlarged nuclei and enlarged hyperchromatic nucleoli and mitotic figure (arrowhead). HE. Bar = 100 µm.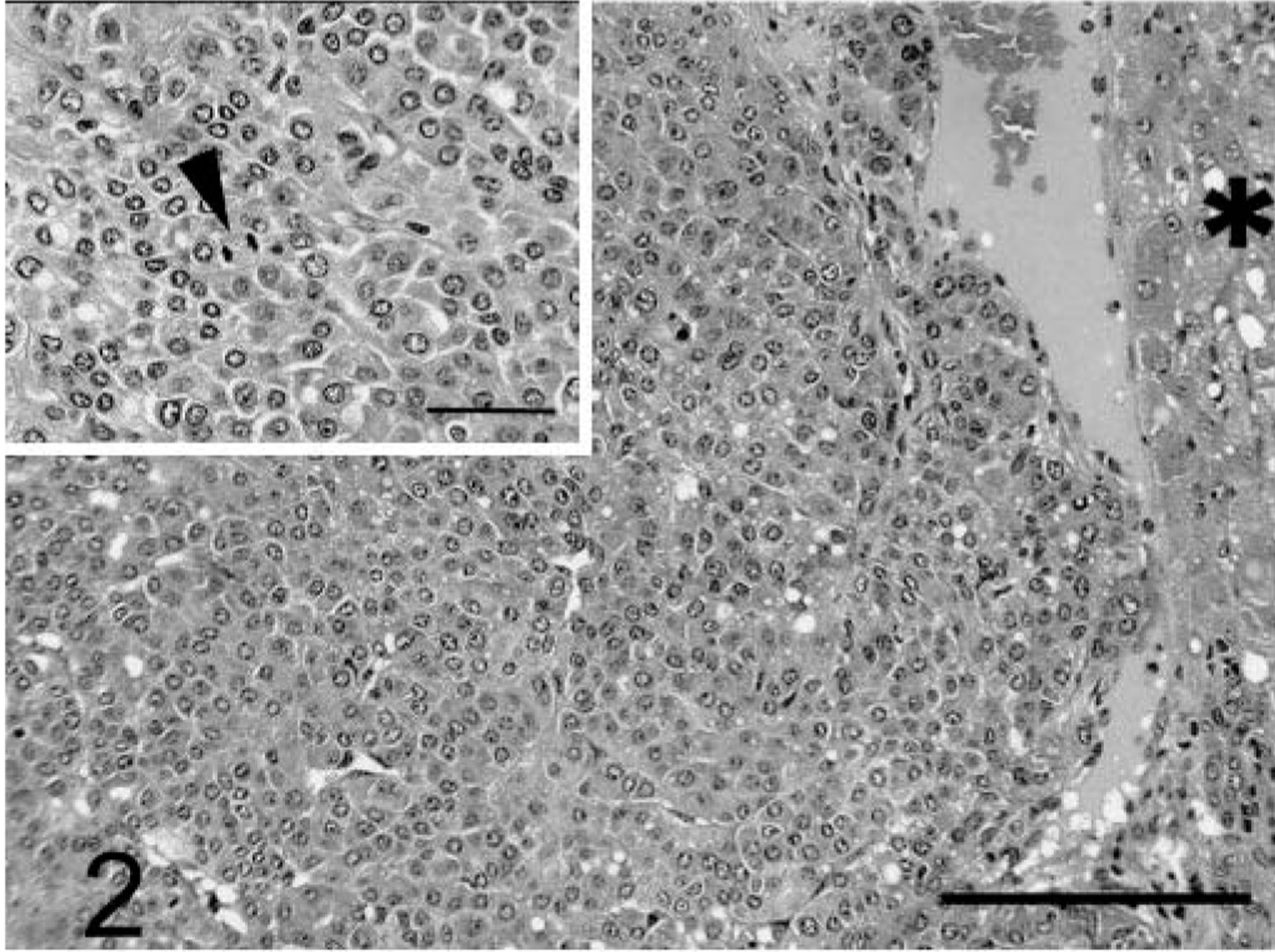
The pseudoglandular pattern was predominant in two of eight male dogs (Nos. 11 and 12), was a minor component in three of eight male dogs (Nos. 5, 7, and 8), and was not seen in the female dogs. This pattern consisted of glandlike formations of neoplastic cells, usually interspersed with solid or trabecular patterns. Cells lining the lumina were identical to those in the adjacent solid trabecular pattern. Pseudoglandular lumina were up to 150 µm in diameter and usually were clear, although rarely they contained scant pale basophilic fluid (Fig. 3).
Liver; prairie dog No. 12. HCC, pseudoglandular pattern. Note the glandlike formations (G) interspersed with trabecular pattern (T). HE. Bar = 720 µm. Inset: Note the low cuboidal line glandular lumina, which in some areas are apparently contiguous with small foci of trabecular tumor (arrowheads). HE. Bar = 100 µm.
The pelioid pattern was not a predominant pattern in any of the cases but was represented in three of four female dogs (Nos. 1–3) and five of eight male dogs (Nos. 5–6, 8, 10, and 12). This pattern occurred within the trabecular/solid pattern and comprised blood-filled channels up to 300 µm in diameter, lined on their luminal surfaces by cells identical to those in the adjacent trabecular/solid tumors (Fig. 4).
Liver; prairie dog No. 10. HCC, pelioid pattern. Note the blood-filled channels (arrows) interspersed with adjacent trabecular/solid tumor. HE. Bar = 860 µm. Inset: Note the lumen of the channel filled with blood (L) and delineated by neoplastic cells. HE. Bar = 90 µm.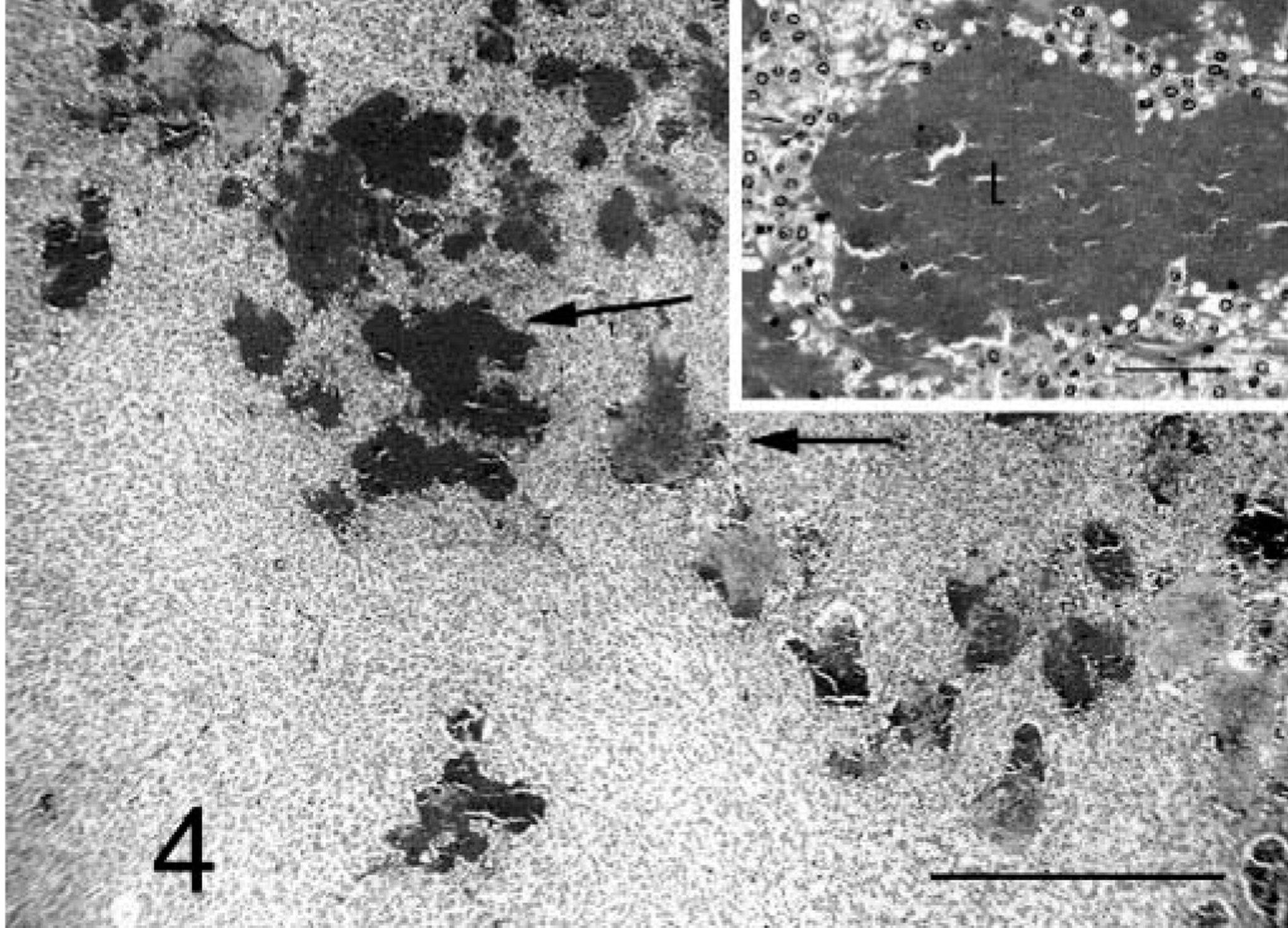
The papillary pattern was present only in male prairie dog No. 5 and, although not a predominant pattern, was present in both primary and metastatic foci. The pattern consisted of papillary formations of neoplastic cells that sometimes merged with cords or solid formations of the tumor (Fig. 5).
Liver; prairie dog No. 5. HCC, papillary pattern. Note the papillary formations of neoplastic cells. HE. Bar = 360 µm. Inset: Cells have moderate to marked anisokaryosis. Occasional multinucleated cells. HE. Bar = 90 µm.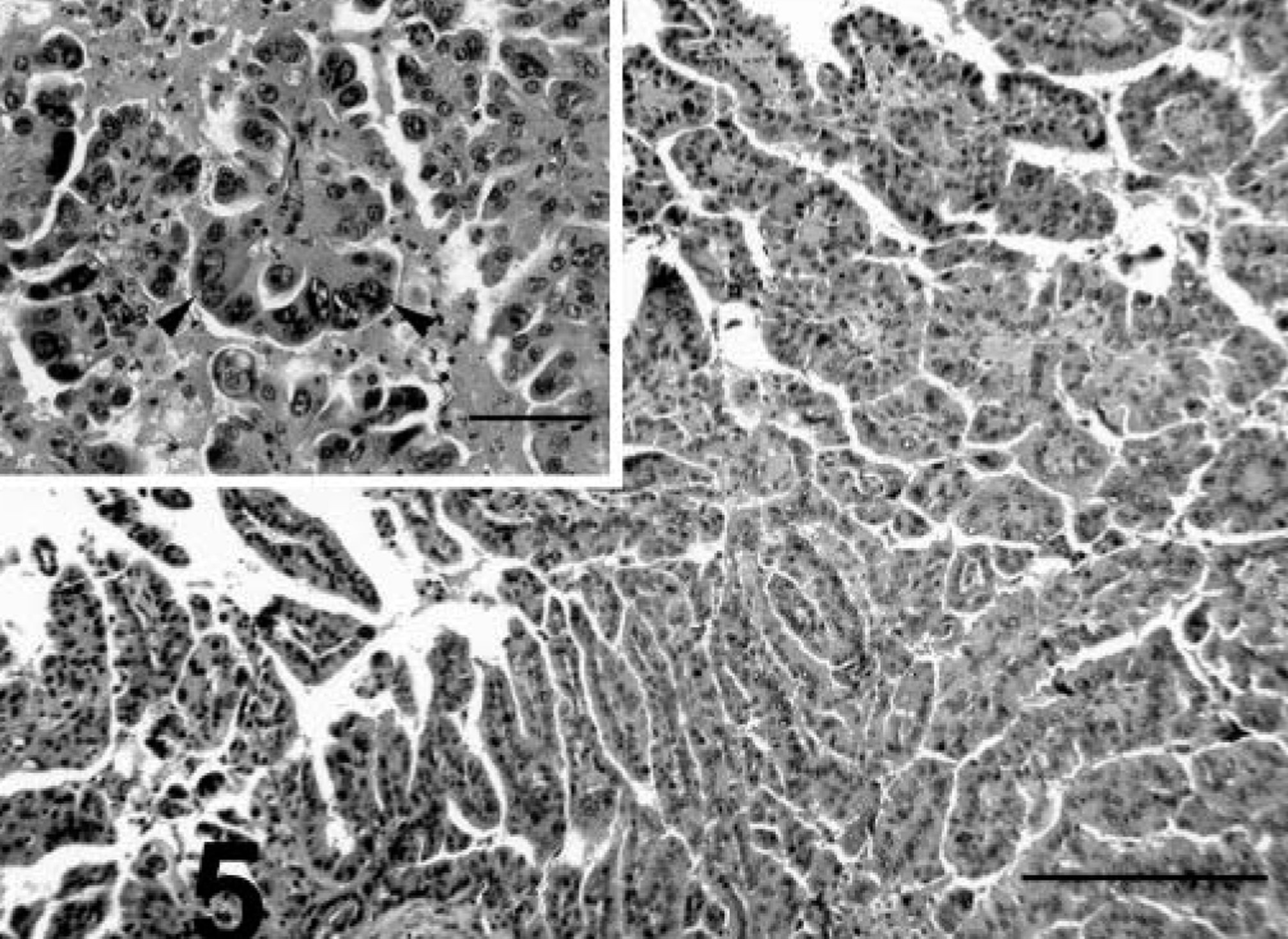
Spleen; prairie dog No. 11. Note the nodular focus of metastatic HCC. This is a trabecular/solid variant with fatty change in neoplastic cells. HE. Bar = 385 µm.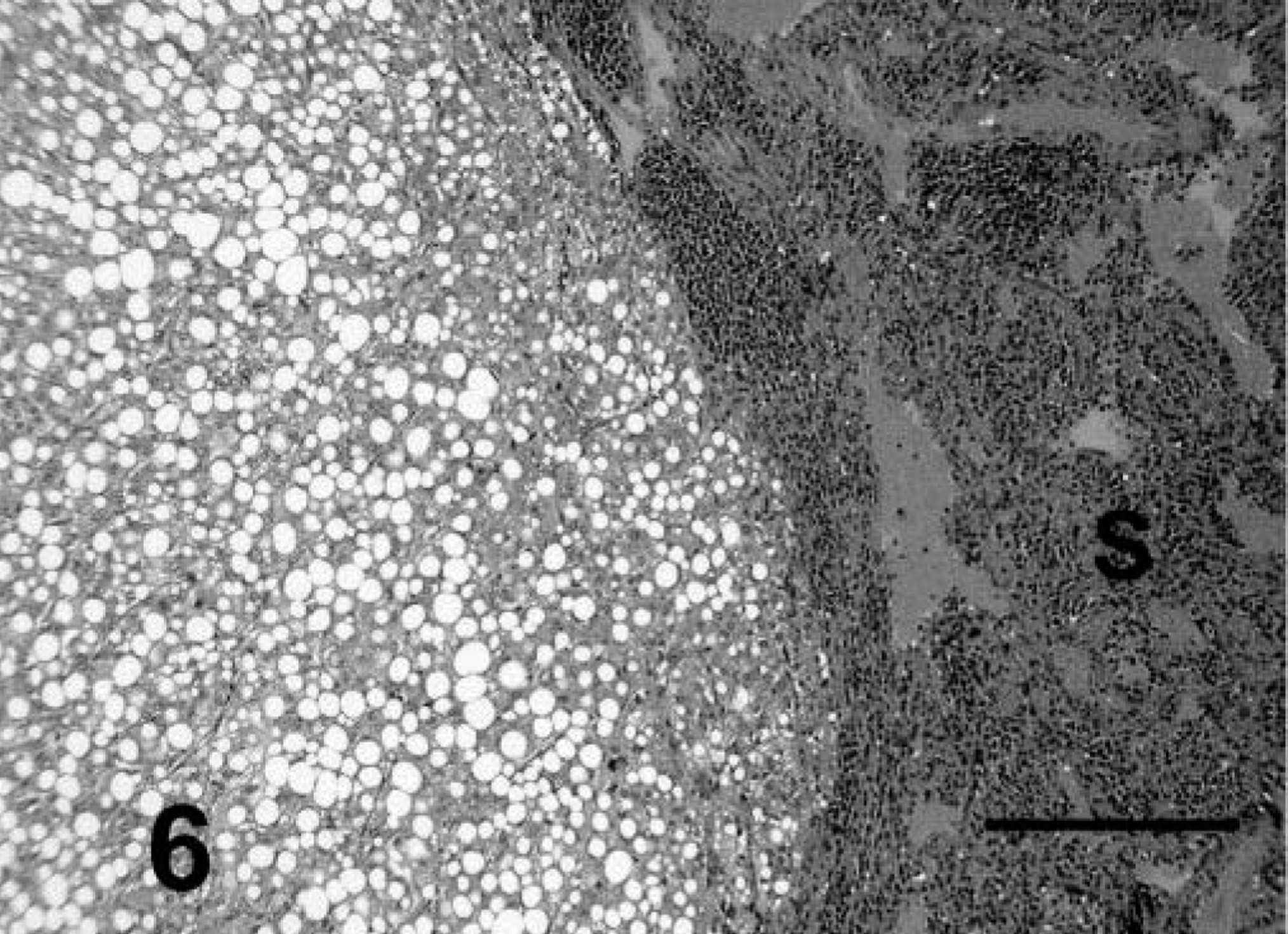
Lung; prairie dog No. 5. Note the nodular focus of metastatic HCC within pulmonary parenchyma (L). This is a trabecular/pseudoglandular variant with fatty change in the cells of the trabecular regions. HE. Bar = 400 µm.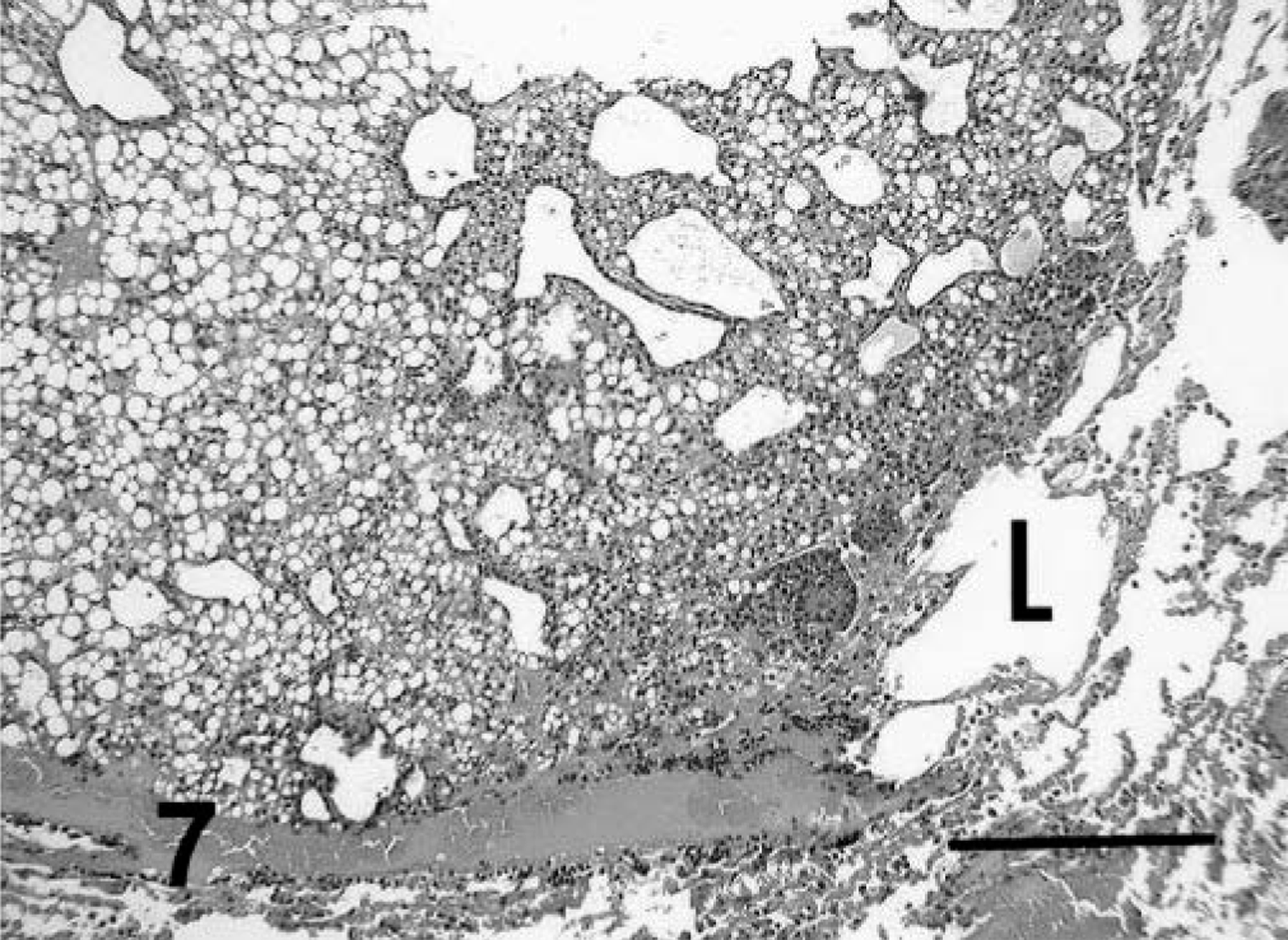
Anaplasia was mild to moderate in tumors with trabecular/solid, pseudoglandular, and pelioid patterns and in the primary tumor with papillary pattern. Cells had typical hepatoid features and were cuboidal to polygonal, with moderate amounts of eosinophilic cytoplasm and central to slightly eccentric nuclei. Compared with normal hepatocytes, the cells usually had slightly enlarged nuclei and nucleoli, and nuclei were slightly more hyperchromatic. Examination of ten 400× fields revealed an average of slightly less than one mitotic figure per high-power field (Fig. 2). A marked degree of anaplasia was noted in the metastatic foci of prairie dog No. 5. Cells had moderate to marked anisokaryosis, and occasional binucleated or multinucleated cells were observed (Fig. 5). An average of four mitotic figures per high-power field was noted in the metastatic tumor. Prairie dog Nos. 1–4 and 9–11 had vacuolar change resembling lipidosis in neoplastic hepatocytes of primary and metastatic tumors. Affected cells had single, intracytoplasmic, large, clear, spherical vacuoles that displaced and occasionally compressed the nucleus. Prairie dog No. 5 rarely had eosinophilic intranuclear inclusions in neoplastic cells. Cavitation and necrosis were noted within the tumor in five cases. Metastasis was noted in five cases (42%) (Figs. 6–7). Lung was a site of metastasis for all these cases: one had metastasis of the spleen, and one had metastasis of the heart and mediastinum.
In none of the sections of nonneoplastic liver available was there clear evidence of specific staining for hepadnaviral antigens using cross-reacting antibodies for WH core antigen or WH surface antigen. Similarly, orcein-positive cells or Victoria blue–positive cells typical of the “ground glass” cells containing HBsAg in humans or of WHsAg-positive woodchuck hepatocytes were not observed.
The most common concurrent, nonhepatic lesions were chronic glomerulonephritis in prairie dog Nos. 2, 5–8, and 10–12 and degenerative cardiac diseases in prairie dog Nos. 2, 7–8, and 12. Concurrent mild to moderate portal lymphocytic hepatitis was noted in 1, 5, and 7. Prairie dogs Nos. 1 and 7 had lymphocyte infiltrates within the tumors. Prairie dog Nos. 2 and 10 had concurrent cirrhosis. Moderate biliary hyperplasia and fibrosis were also noted in prairie dog No. 7. A biliary cystadenoma was present in prairie dog No. 12.
Discussion
Based on our study, HCC is a common cause of morbidity and mortality in prairie dogs, with this lesion present in 12 of 61 animals (20%) submitted over an 8-year period, for which liver was included in the submitted tissue set. The tumor was associated with progressive lethargy, wasting, and respiratory distress. The respiratory distress reported in the cases we have studied may have been due to pulmonary metastasis or diaphragmatic compression of the thoracic cavity by the expanding hepatic mass. Heart failure or odontoma, two other concurrent lesions seen in some of the study animals, also could potentially contribute to respiratory distress.
HCC commonly develops in HBV-infected humans, with 100 times greater prevalence of this tumor in infected than in uninfected people. 1 HCC is common in woodchucks, with several early reports of natural infection and many subsequent reports of experimental hepadnavirus infection and tumorigenesis. 1,5,12 Woodchucks infected at birth that do not clear the infection have a lifetime risk of developing HCA of essentially 100%. 5,16 The prevalence of HCC in ground squirrels may be lower. In a study of 75 captive California ground squirrels, three animals, all more than 4.5 years of age or older, developed HCC. 8 Forty wild-caught Richardson's ground squirrels were examined in one study, and two cases of HCC were found, both in adult animals. 9 Approximately one third of the captive arctic ground squirrels in one study developed benign hepatocellular neoplasms, and the incidence was higher in virus-infected animals. 18 It is possible that the prevalence of HCC in prairie dogs generally may be higher than that in ground squirrels but lower than that in woodchucks.
The morphologic features of HCC in prairie dogs closely resemble those of HCC in woodchucks, having trabecular/solid, pseudoglandular, and pelioid formations. 12 The trabecular pattern was the predominant form in our cases and resembles that described in other prairie dogs. 19,20 The trabecular pattern is also the most common pattern seen in humans 4 and in Beechey's and Richardson's ground squirrels. 8,17 The papillary pattern seen in prairie dog No. 5 appears to be an unusual variant of HCC in the family Sciuridae. The rate of metastasis of this tumor is low in hepadenavirus-infected woodchucks 12 and apparently has not been observed in other sciurids. 8,9,17,18 However, metastasis was noted in 42% of the prairie dogs in our study. These findings indicate that prairie dogs may have a higher rate of metastasis of HCC than other species, more in line with the moderate rate of metastasis seen in humans. 3
In woodchucks, Richardson's ground squirrels, and humans, various stages of hepatitis associated with hepadnavirus infection precede or are seen concurrently with the development of HCC. Lesions start as mild lymphocytic portal hepatitis but eventually progress beyond the limiting plate and are associated with bile duct proliferation and fibroplasia but rarely progress to cirrhosis. 12 Inflammation was not a consistent feature in the livers of prairie dogs in our study, although two had cirrhosis and one had biliary hyperplasia and fibrosis. The finding of mild portal inflammation in three cases, lymphocytes in tumors in two cases, and bile duct proliferation with fibrosis in one case are similar to those of woodchucks infected with WHV, although overt cirrhosis is apparently rare in woodchucks. 12 These changes, however, were not consistent findings associated with the development of HCC in our prairie dogs. Hepatic inflammation also was not reported as a feature in Beechey's or arctic ground squirrels with hepadnavirus infection or hepatocellular neoplasia. 8,18
Failure to detect hepadnaviral antigens in prairie dogs with HCC using reagent antibody to cross-reacting WHV antigens, namely WHcAg and WHsAg, does not rule out definitively the possibility that a hepadnavirus infected some of the prairie dogs in this series. The negative result may be due to the fact that the primary antibody was raised against woodchuck virus and does not cross-react with a prairie dog virus. Reagent antibody to GSHV was not available. Similarly, the absence of Victoria blue– or orcein-positive hepatocytes does not absolutely rule out hepadnaviral infection in prairie dogs, but such stain-positive cells are characteristic of chronic HBV and chronic WHV infection. It also is important to note that Snyder 15 did report orcein-positive hepatocytes in prairie dogs with HCC, suggesting the presence of hepatocytes containing cross-hepadnaviral surface antigen using that technique. Serum was not available from our cases, so serology for hepadnavirus surface and core antigen could not be performed. Fresh or frozen tissues were not available from our cases, so Southern blot analysis could not be performed.
The high frequency of HCC in captive prairie dogs is similar to that reported in several closely related members of the family Sciuridae. 5,8,9,12,15–18 In all these other sciurids with a high prevalence of HCC, evidence of hepadnaviral infection has been demonstrated. To further investigate the possibility of hepadnaviral infection in prairie dogs, it would be useful to conduct seroepidemiologic studies of captive prairie dogs from zoos that have experienced a high incidence of HCC. Southern analysis or polymerase chain reaction methods for detection of hepadnaviral DNA would be useful, as well as tests for viral surface antigen and for cross-reacting antibodies to hepadnaviral core and surface antigens. Fresh frozen hepatic tissue from prairie dogs with HCC also would be valuable in further molecular analyses for hepadnaviral nucleic acids and proteins.
Eight of the prairie dogs had mild glomerulonephritis. The cause for this condition could not be determined. Aging, low-grade sepsis, and parasite migration were considered possible contributing factors. The relationship of this lesion to the hepatic tumors is unknown. A retrospective review of the prairie dog submissions to Northwest ZooPath identified several cases of low-grade glomerulonephritis in prairie dogs that did not have liver tumors, suggesting that they are possibly unrelated events.
Conclusions
These prairie dog tumor cases were similar to HCC in woodchucks in that animals were aged, were of both sexes, had white to tan masses in various hepatic lobes, had variable morphologic tumor patterns for which the trabecular patter predominated, had tumors that were well differentiated with slight to moderate degree of anaplasia, and had occasional cavitation and necrosis. The cases were dissimilar to HCC of woodchucks in that metastasis seems to be a more common sequel in prairie dogs. Concurrent hepatitis may be less common than in humans, woodchucks, and Richardson's ground squirrels. The cause for the apparent high prevalence of HCC in prairie dogs submitted to Northwest ZooPath is not known; however, the similarities of this condition to that of hepadnavirus-related hepatocellular neoplasia in humans, woodchucks, and ground squirrels suggests that the condition in prairie dogs may have a viral etiology.
Footnotes
Acknowledgements
We acknowledge the expertise of Histology Consulting Services (HCS) in excellent slide preparation and thank Mary Ascenzi of the Department of Clinical Sciences, College of Veterinary Medicine, Cornell University, Ithaca, New York, for special histochemical and immunohistochemical stains. We also thank the following institutions for submission of cases and for follow-up collaborations: Southwest Animal Hospital (Beaverton, OR), Riverside Animal Hospital (Jacksonville, FL), Sno-Wood Hospital (Woodinville, WA), and the animal health departments of the San Francisco Zoo, Woodland Park Zoo, Dallas Zoo, Los Angeles Zoo, Fort Worth Zoo, and Detroit Zoo.
