Abstract
P-glycoprotein (P-gp), which is encoded by the multidrug resistance gene (MDR-1); alpha fetoprotein (AFP); and vascular endothelium-associated antigens are well-known markers for human and canine hepatic diseases. We obtained liver tissues from 5 dogs with hepatocellular carcinoma (HCC) and 12 dogs with cirrhosis, and we performed histopathologic and immunohistochemical evaluations using anti-P-gp, anti-AFP, anti-CD31, and anti-CD34 antibodies. P-gp was expressed at higher levels in HCC than in cirrhotic livers (P < .01), and was most commonly localized in biliary canaliculi and small ductuli. AFP was localized mainly in the cytoplasm in HCC (P < .01) and in a few cases of cirrhosis. In both HCC and cirrhosis, the AFP-positive cells were morphologically similar to normal hepatocytes and showed an even cytoplasmic distribution of AFP. The endothelial markers CD31 and CD34 were used to investigate vascular distribution. CD31 was expressed strongly in the portal area and parenchyma in HCC, but it was rarely observed in the parenchyma in cirrhosis. CD34 expression could not be detected in both HCC and cirrhosis. This study constitutes the first comprehensive study of P-gp, AFP, and endothelial markers in canine HCC and cirrhosis. The importance of these markers in HCC and cirrhosis in dogs was demonstrated and provides a more accurate basis for a definitive diagnosis of HCC and cirrhosis in dogs.
Keywords
Introduction
Canine hepatocellular carcinoma (HCC) arises from the uncontrolled proliferation of hepatocytes. 4, 5, 21 The causal mechanisms underlying this disease are not fully understood but are thought to involve environmental factors. Exposure to carcinogens or cancer-causing chemicals, including fungal toxins, food additives, pesticides, dyes, and plants, may increase the risk of cancer development. 4, 6, 10 Viral infections have been associated with hepatic cancer in humans, but they have not been identified in dog tumors. 4 HCC may develop as a single (solitary) growth, multiple nodular growth, or many diffuse small growths. 4, 5, 21 It is the most common malignant hepatic neoplasm in dogs older than 10 years and is slightly more common in males than females. 5, 21
Liver cirrhosis is a condition characterized by irreversible damage of the liver that in humans and dogs mostly results from chronic hepatitis. 29 Chronic hepatitis can result from infection, hepatotoxins, breed-related inherited progressive liver diseases, metabolitic problems and autoimmunity, gradually progressing to irreversible cirrhosis characterized by bridging fibrosis, inflammation, and nodular regeneration. 29 Hepatocellular necrosis, activated hepatic stellate cells, and inflammation mediators are important causes of this process. 29
In this study, we demonstrated the presence of P-glycoprotein (P-gp) and alpha fetoprotein (AFP) in HCC and cirrhosis of the liver as tumor-associated antigens. We also investigated the expression of the endothelial cell markers CD31 and CD34 to determine the degree of vascular distribution in HCC and cirrhosis.
HCC is a malignant tumor with resistance to chemotherapy that is thought to result from enhanced expression of P-gp. P-gp is a marker for multidrug resistance and is the primary cause of resistance to chemotherapy. 9, 12 P-gp is a transmembrane glycoprotein encoded by multidrug resistance gene (MDR-1) and functions as an adenosine triphosphate–dependent drug efflux pump, reducing intracellular drug cytotoxicity. 9, 18, 19, 21, 23 This protein is also present in normal tissues, including the jejunum, liver, kidney, colon, and adrenal glands. 3, 21, 27 High levels of P-gp are expressed in tumor cells derived from organs normally expressing P-gp.
AFP is an oncofetal antigen normally produced by the liver and yolk sac during embryogenesis but not produced in normal adult tissues. AFP increases in the adult liver in response to liver regeneration or tumorigenesis, so it is a useful marker for liver disorders and cancers in adults. 11, 13, 14, 24
Angiogenesis is crucial for tumor growth and metastasis. Metastasis in HCC occurs through vascular and lymphatic invasion, with vascular invasion being more common. 4, 27 As angiogenesis is the growth of new blood vessels from preexisting vessels, it is important to know the existing vascular distribution in disease. Tumor vascularity in HCC and cirrhosis has been evaluated using endothelial cell markers, such as CD31 and CD34 in humans. 16, 20, 26, 27 These markers can provide a useful tool for diagnosis of HCC and cirrhosis in dogs as well.
The present study evaluated the histopathologic features and cellular localization of P-gp, AFP, and endothelial antigens in canine HCC and cirrhosis. The results from this study characterize important factors for the diagnosis and treatment of HCC and cirrhosis.
Materials and Methods
Selection of canine patients
Liver specimens were obtained from 17 dogs suspected of having liver diseases among dogs hospitalized or under treatment at the Veterinary Medical Teaching Hospital of Konkuk University (Seoul, Korea) during the years 2002–2005. Based on the canine patient files and results of hematoxylin and eosin (HE) staining, histopathologic analyses were done in accordance with the classification of tumors in domestic animals. 4 Of the liver specimens, 5 cases were HCC, and 12 cases were cirrhosis. Also, pathologists from the veterinary diagnostic center of the University of Nebraska (Lincoln, NE) examined them thoroughly.
Preparation of tissues for immunohistochemistry
Liver specimens obtained were fixed in 10% neutral-buffered formalin and subsequently embedded in paraffin. Serial 4-μm sections were acquired from each paraffin block.
For immunohistochemical analysis, a set of monoclonal and polyclonal antibodies were used. Primary antibodies included anti-mouse P-gp (BioGenex Laboratories, San Ramon, CA), rabbit polyclonal anti-human AFP (DakoCytomation, Fine Life Science Co., Seoul, Korea), anti-mouse CD31 (DakoCytomation, Fine Life Science Co.), and anti-mouse CD34 (DakoCytomation, Fine Life Science Co.). Also, we used two-step Envision system-AP (LSAB kit; DakoCytomation, Fine life Science Co.) and Envision system-HRP (LSAB kit; DakoCytomation, Fine Life Science Co.). Antibodies were used at a 1 : 300 dilution of P-gp, 1 : 100 of AFP, and 1 : 50 of CD31 and CD34. Tissue sections were first treated with 3% hydrogen peroxide (H2O2) solution for 20 minutes at room temperature, followed by phosphate-buffered saline (PBS) washing three times. Then they were immersed in a Coplin jar containing heat-induced epitope retrieval solution, 10 mM citric acid monohydrate buffer of pH 6.0. After heating in a microwave oven (high power) for 20 minutes, the slides were cooled to room temperature and washed in PBS three times. After three 5-minute washes with PBS, sections were incubated in a blocking solution of 5% normal goat serum in PBS for 30 minutes at room temperature. The slides for detection of P-gp were not treated with goat serum. Subsequently, sections were incubated with primary antibodies for 2 hours at room temperature. For secondary antibodies, we used Envision system-HRP for P-gp, CD31, and CD34, and Envision system-AP for AFP. We applied them to all slides for 40 minutes at room temperature and washed them with PBS. Next, the slides were incubated with substrates for Envision system-HRP and Envision system-AP until the desired staining intensity developed, and then the color reaction was stopped by washing in deionized water twice. The slides were counterstained with Harrison's hematoxylin and coverslipped. As a negative control, additional sections of specimens were processed with the omission of primary antibody.
Double-labeling immunohistochemistry for simultaneous detection of AFP and P-gp
Double-labeling immunohistochemistry (IHC) technique of a single tissue section was used for co-expression analysis of AFP and P-gp. The sections were treated to remove the endogenous peroxidase activity by incubation in 3% hydrogen peroxide (pH 7.4) for 20 minutes and then washing with PBS three times, followed by boiling in a Coplin jar containing heat-induced epitope retrieval solution, 10 mM citric acid monohydrate buffer of pH 6.0 in a microwave oven (high power) for 20 minutes. After cooling and washing with PBS, nonspecific antibody binding was blocked by the incubation with 5% normal goat serum (Sigma Chemical Co., St. Louis, MO) for 20 minutes at room temperature. The slides were then incubated overnight at 4°C with polyclonal rabbit anti-human AFP at a dilution of 1 : 200. The sections were washed three times in PBS and successively incubated with Envision system-AP for 30 minutes at room temperature. After washing with PBS, slides were incubated with substrates for Envision system-AP until color development was seen. The color reaction was stopped by washing the slides in deionized water. For the detection of P-gp, the slides were incubated for 2 hours at room temperature with anti-mouse P-gp (1 : 300 dilution in PBS). Subsequently, the slides were washed in PBS and incubated with Envision system-HRP for 40 minutes. After washing three times with PBS, color reaction was developed with DAB (Zymed Laboratories Inc., San Francisco, CA) and stopped by washing the slides in deionized water. Finally, the slides were counterstained with Harrison's hematoxylin and mounted for light microscopy.
Digital and statistical analysis
Digital images were acquired using Olympus DP70 digital camera and Olympus microscope (BX51; Mellville, NY) and transferred to Image Pro Plus software (Media Cybernetics Inc., Bethesda, MD) for analysis of area and intensity. For morphologic study, cross-sectional images of the liver stained with IHC were captured at 200× magnification, and circumferential traces were used to calculate the positive areas.
All statistical values in figures and text are expressed as means ± SEM. Statistical differences between groups were evaluated using a Sigma test. P ≤ 0.05 was considered to be statistically significant.
Results
Histopathology and immunostaining of tumor-associated antigens, AFP, and P-gp in HCC and cirrhotic liver
In the dogs with HCC examined in the present study, the tumor caused significant enlargement of the liver and several different types of hepatic nodules. There was no evidence of cirrhosis. Microscopically, HCC cells resembled normal hepatocytes to a variable extent. The cytoplasm of the cells contained large vacuoles containing fat. Also, clear cell change due to abundant glycogen was observed in the cytoplasm. By comparison, examination of cirrhotic livers showed extensive destruction of hepatocytes with condensation of stroma and cholestasis. Connective tissue staining demonstrated an increase of fibrous tissue in the portal tract (data not shown).
Figure 1 shows greater levels of AFP and P-gp in HCC than in cirrhotic livers. AFP was found primarily within the cytoplasm of hepatocytes in both HCC and cirrhotic livers. A significantly higher level of AFP was observed in HCC than in cirrhotic liver (P < .01), despite the high degree of variability in the number of AFP-containing cells in 5 HCC dogs (Fig. 1a). P-gp was expressed at high levels in HCC compared with cirrhosis (P < .01), and it was more intense in tumor cells than in surrounding normal hepatocytes (Fig. 1b).
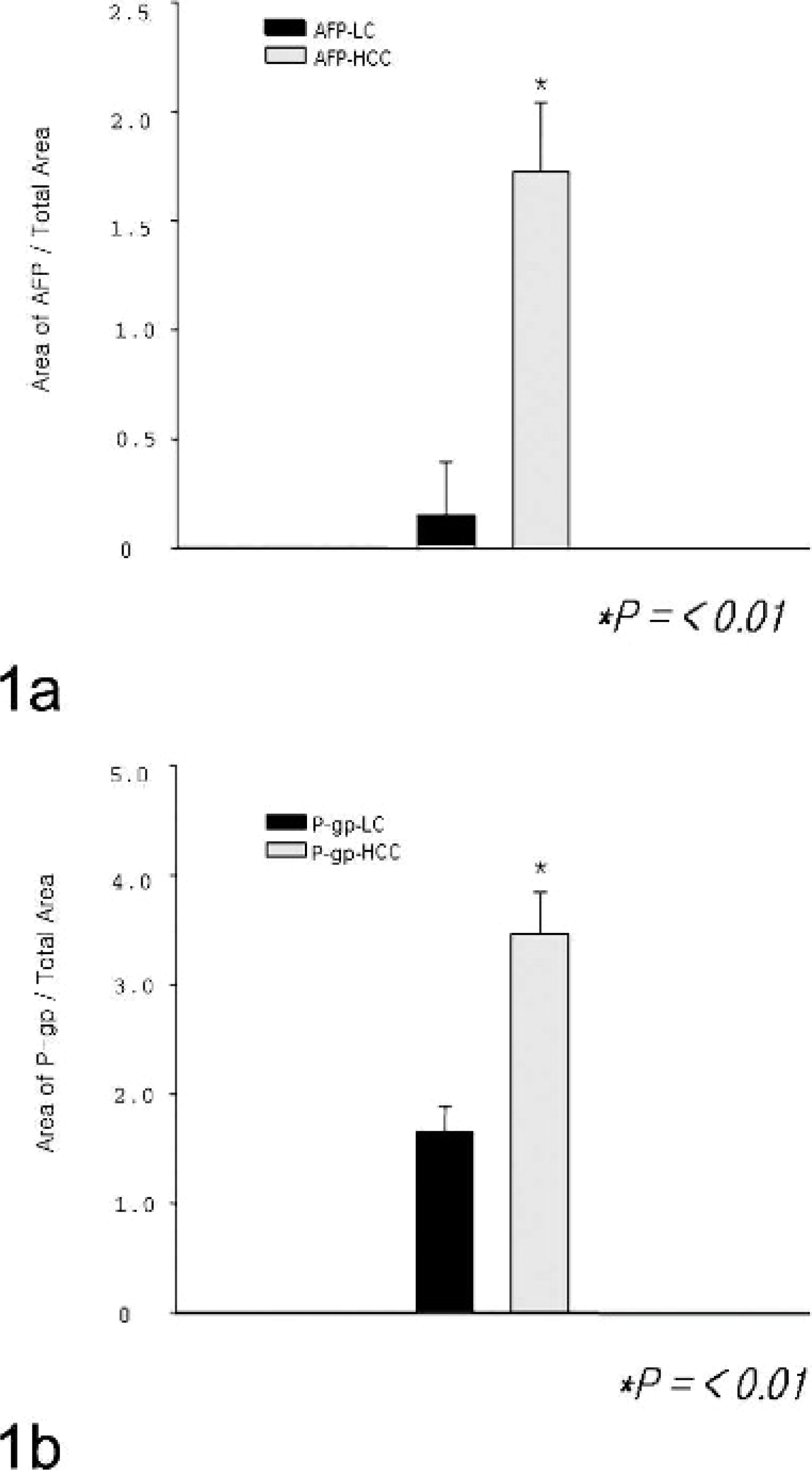
Immunologic staining of AFP expression in HCC and cirrhotic livers. Tissue preparations were stained with anti-human AFP antibody, and immunoreactive areas were quantified using Image Pro Plus software. Quantification is shown in histograms. Significantly higher levels of immunoreactivity were observed for AFP in the HCC (n = 5) canines than in cirrhotic liver (n = 12) canines. ∗ P < .01 compared to HCC and cirrhotic liver group.
AFP-positive cells were morphologically similar to hepatocytes and showed an even distribution of AFP in the cytoplasm (Fig. 2a). Most of the cells that contained vacuoles did not react with AFP, which was consistent with glycogen or lipid. By comparison, the proportion of AFP-positive signals was significantly lower in cirrhotic livers and tended to be obscure or weak (Fig. 2b). As for P-gp, in HCC, high expression of P-gp was evident in the homogenous basophilic cell hepatocytes but was not evident in either the clear vacuolated cells containing glycogen or lipid or in the eosinophilic cell types (data not shown). Major sites of P-gp localization were the biliary canalicular surface of hepatocytes and the small biliary ductules in HCC (Fig. 3a) and cirrhotic livers (Fig. 3b).
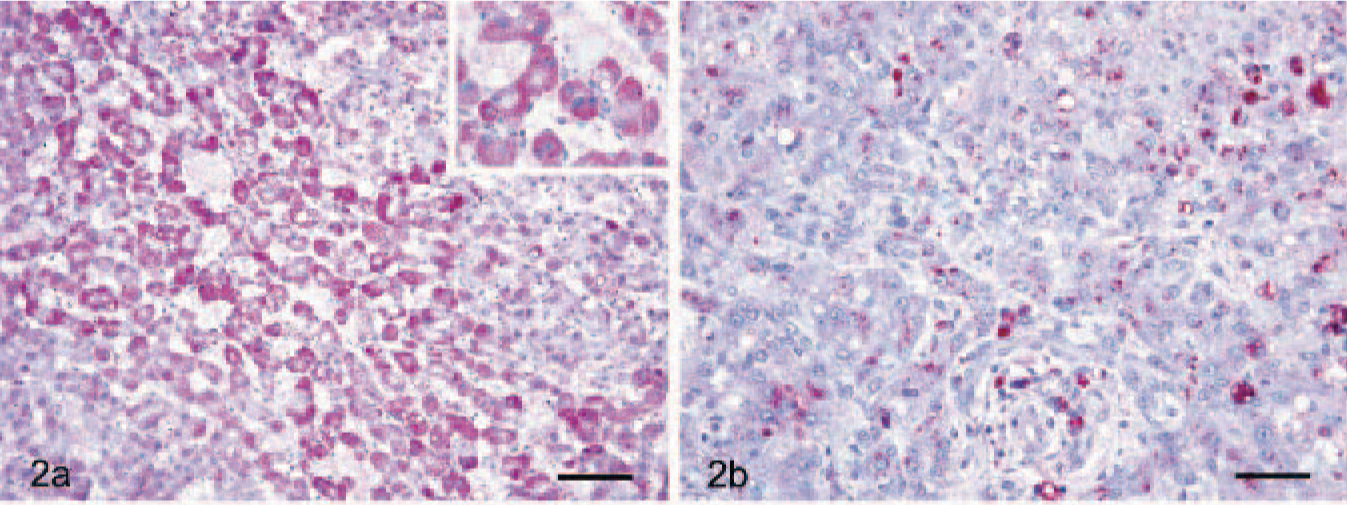
Dog. HCC and liver cirrhosis. Fig. 2a. Liver. HCC. Strong AFP expression in the cytoplasm of neoplastic hepatic cells. Formalin-fixed section labeled with the polyclonal antibody rabbit anti-human AFP. Envision system-AP; HE counterstain. Bar = 55 μm. Inset. Higher magnification of AFP-positive hepatic cells. Hematoxylin counterstain. Bar = 30 μm. Fig. 2b. Cirrhotic liver. Weak AFP expression in the cytoplasm of hepatic cells. Formalin-fixed section labeled with the polyclonal antibody rabbit anti-human AFP. Envision system-AP; hematoxylin counterstain. Bar = 30 μm.
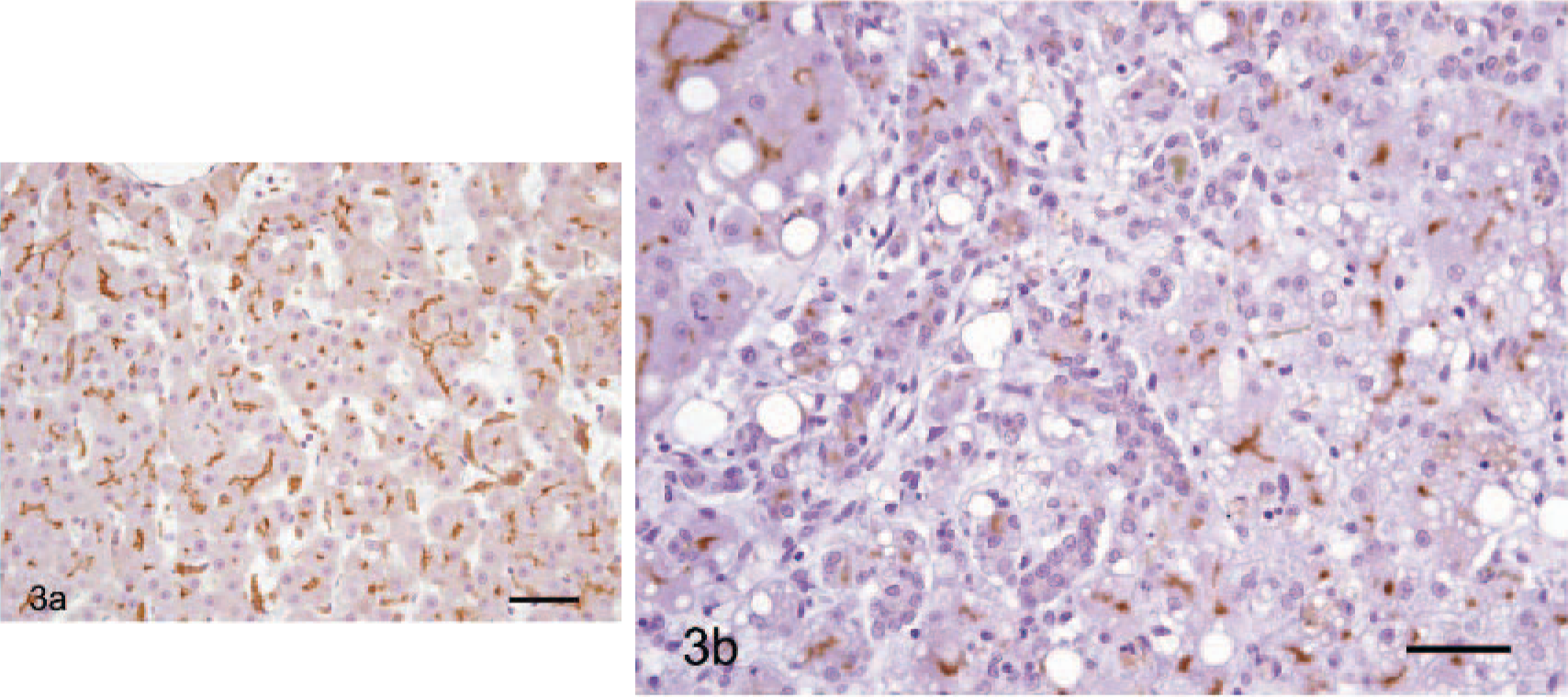
Dog. HCC and cirrhotic liver.
Double-labeling IHC for AFP and P-gp
Co-expression analysis of AFP and P-gp in neoplastic hepatic cells using double-labeling IHC revealed that only the areas consisting of homogenous basophilic cell types showed both AFP and P-gp expression (Fig. 4).
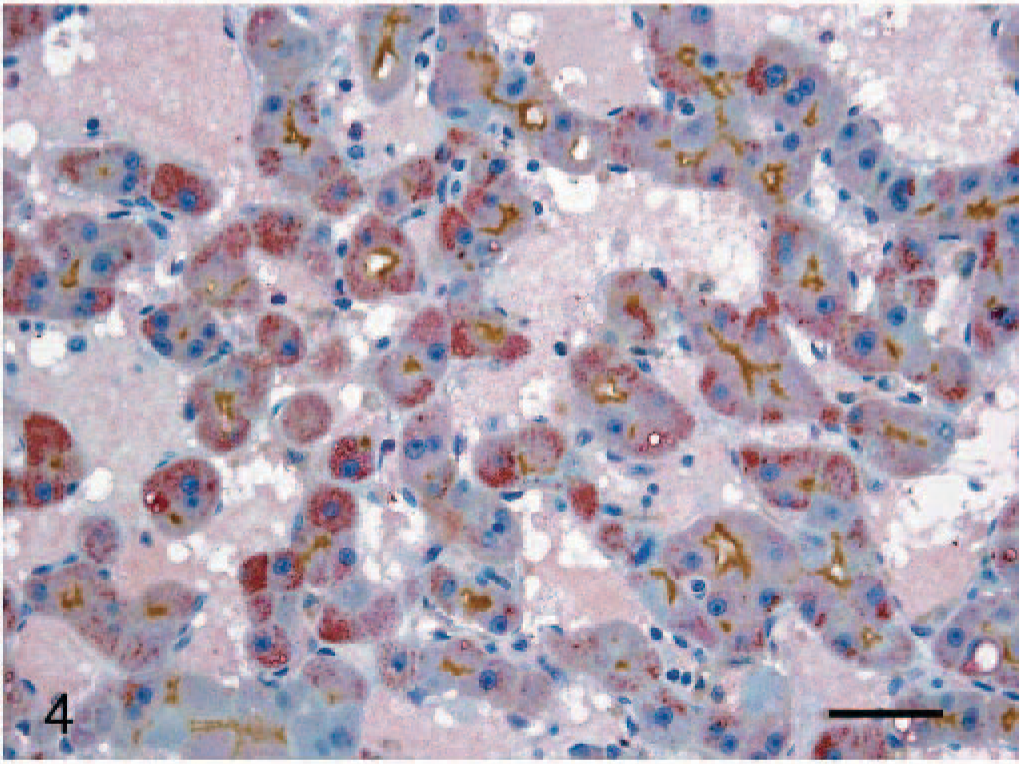
Liver. HCC. Strong co-expression of AFP and P-gp in the cytoplasm of hepatic cells. Formalin-fixed section double labeled with the polyclonal antibody rabbit anti-human AFP and monoclonal antibody anti–P-gp. Envision system-AP and Envision system-HRP; hematoxylin counterstain. Bar = 30 μm.
IHC for CD31 and CD34
Primary antibodies to CD31 and CD34 were used to detect vascular endothelium–associated antigens in HCC and cirrhotic livers. It was not possible to accurately evaluate CD34-positive cells, due to difficulties in identifying endothelial cells in HCC and cirrhotic livers. Expression of CD31 was generally observed in both HCC and cirrhotic liver tissues. CD31-positive cells were observed in vessels of the portal area and in the sinusoids of the hepatic parenchyma near the portal area in HCC (Fig. 5a). CD31 was stained as a circular pattern. In contrast, in cirrhosis, staining of CD31 was predominantly confined to vessels of the portal area (Fig. 5b).
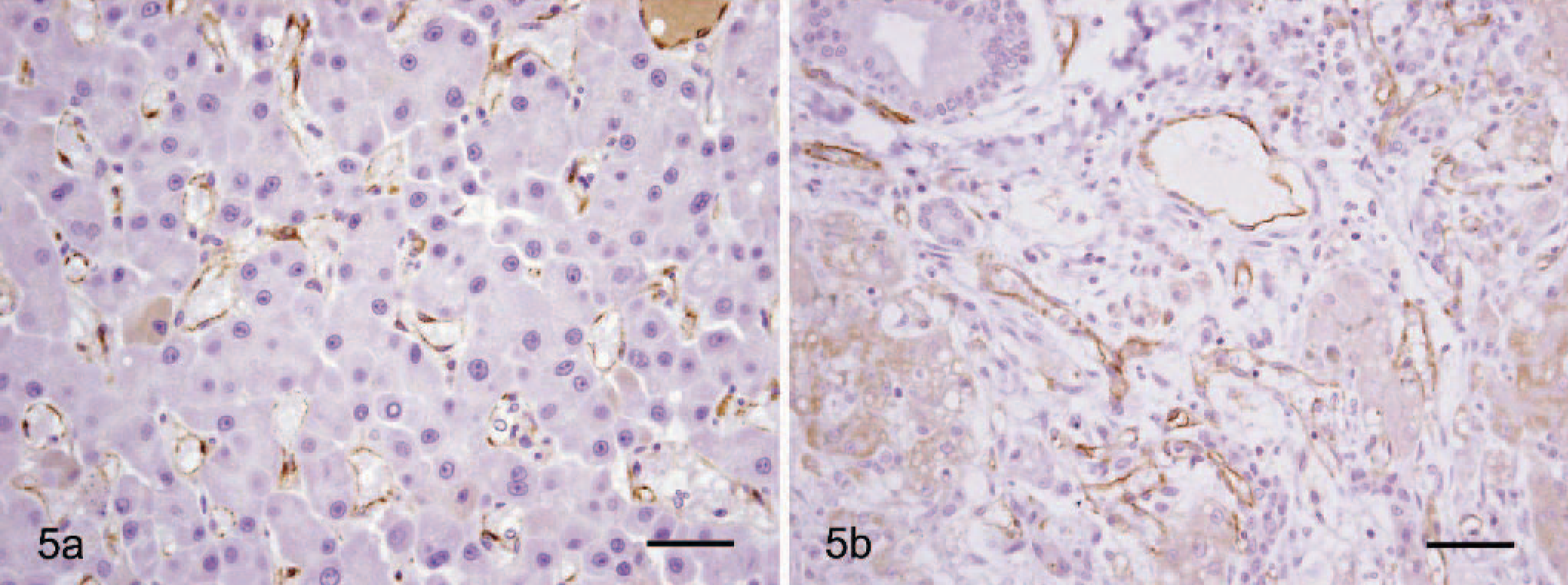
Dog. HCC and cirrhotic liver. Fig. 5a. Liver. HCC. CD31 expression in sinusoid lining cells of liver parenchyma. Formalin-fixed section labeled with the monoclonal antibody mouse anti-CD31. Envision system-HRP; hematoxylin counterstain. Bar = 30 μm. Fig. 5b. Cirrhotic liver. CD31 expression in large vessels of the portal area. Formalin-fixed section labeled with the monoclonal antibody mouse anti-CD31. Envision system-HRP; hematoxylin counterstain. Bar = 30 μm.
Discussion
Primary liver cancer is rare, comprising less than 2% of all cancers observed in canine species. 4, 5, 21 The most common primary liver tumor is HCC, a malignant tumor arising from hepatic cells. 5
In the present study, the markers P-gp, AFP, CD31, and CD34 were evaluated in canine HCC specimens and in cirrhotic liver without HCC using histopathology, IHC, and double- labeling IHC.
P-gp is normally expressed in tissues with excretory function, including the jejunum, kidney, liver, and adrenal gland. In these tissues, P-gp plays a role as an efflux pump for physiologic metabolites, thereby providing a protective mechanism against chemicals in diet. This capacity of P-gp leads to strong resistance to chemotherapy. 18, 22, 28
In this study, P-gp was more highly expressed in HCC than in cirrhotic liver, which is consistent with the known resistance of HCC to chemotherapy. In accordance with previous human studies, the overexpression of P-gp was demonstrated in all canine HCC specimens. 9, 12 The difference in the level of P-gp expression between HCC and cirrhotic liver tissue can aid in the diagnosis and treatment strategy of these diseases. The overexpression of P-gp is detected on the bile canalicular surface of hepatocytes, in the cells lining the luminal space and on the apical surface of small biliary ductules in both HCC and cirrhotic livers.
AFP is normally expressed in fetal hepatic tissue but is absent from adult tissue, with the exception of neoplasia, primarily of the liver. 15 AFP is present in all mammals with a significant degree of homology. 17
In our experiment, AFP was identified in the cytoplasm of hepatocytes in both HCC and cirrhotic liver. A higher proportion of positive cells was identified in HCC than in cirrhotic livers, and these cells were morphologically similar to normal hepatocytes. In HCC, AFP was located multifocally in the cytoplasm, whereas in the cirrhotic liver, expression of AFP was localized mainly to a single cell or a nest of cells in the periportal area of hepatic lesions. There was a high degree of variability in the number of AFP-positive cells among the dogs. A higher level of AFP expression was found in HCC than in cirrhotic livers, suggesting that AFP could potentially be used as a diagnostic marker for distinguishing between these diseases. Our results support previous studies that show increased levels of AFP in the blood serum, indicating liver damage. 1, 2, 11, 13, 14, 25
Double-labeling IHC used to determine whether AFP colocalized with P-gp in HCC and in cirrhosis revealed that HCC cells resembling normal hepatocytes expressed both AFP and P-gp. Most vacuolated cells filled with glycogen or lipid did not express AFP or P-gp.
As HCC is a rich, vascularized, malignant tumor, angiogenesis plays an important role in tumor formation and progression. 7, 8 Angiogenesis starts from preexisting vessels, and the endothelial cell markers CD31 and CD34 were used to ascertain the extent of vascular distribution in HCC and cirrhosis. CD31 expression was observed in vessels of portal area and in the sinusoid of liver parenchyma in HCC, whereas it was restricted at a high level to large vessels of portal area in cirrhosis. Unfortunately, CD34 could not be detected in HCC or cirrhotic tissues.
In conclusion, the present study represents the first comprehensive study of P-gp, AFP, and endothelial markers in canine HCC and cirrhosis liver tissues. Differential expression levels were demonstrated and provide the basis for definitive diagnosis of HCC and cirrhosis in dogs.
Footnotes
Acknowledgements
D.-N. Hwang contributed to this paper as the co-first author. We thank Ms. R.-H. Jang for excellent technical assistance and J.-Y. Yhee for preparation of photographs. We also thank Dr. Alan R. Doster, Veterinary Diagnostic Center, University of Nebraska, Lincoln, Nebraska, for critical review of hepatic tissues in this study. This study was supported in part by a grant for scientific animal research from the Ministry of Agriculture and Forestry (MAF) of Korea and in part by a grant from the Konkuk University research foundation. This report represents a thesis submitted by R. E. Tashbaeva to fulfill the requirements for an M.S. degree.
