Abstract
During a screen for vascular phenotypes in aged laboratory mice, a unique discrete phenotype of hyaline arteriolosclerosis of the intertubular arteries and arterioles of the testes was identified in several inbred strains. Lesions were limited to the testes and did not occur as part of any renal, systemic, or pulmonary arteriopathy or vasculitis phenotype. There was no evidence of systemic or pulmonary hypertension, and lesions did not occur in ovaries of females. Frequency was highest in males of the SM/J (27/30, 90%) and WSB/EiJ (19/26, 73%) strains, aged 383 to 847 days. Lesions were sporadically present in males from several other inbred strains at a much lower (<20%) frequency. The risk of testicular hyaline arteriolosclerosis is at least partially underpinned by a genetic predisposition that is not associated with other vascular lesions (including vasculitis), separating out the etiology of this form and site of arteriolosclerosis from other related conditions that often co-occur in other strains of mice and in humans. Because of their genetic uniformity and controlled dietary and environmental conditions, mice are an excellent model to dissect the pathogenesis of human disease conditions. In this study, a discrete genetically driven phenotype of testicular hyaline arteriolosclerosis in aging mice was identified. These observations open the possibility of identifying the underlying genetic variant(s) associated with the predisposition and therefore allowing future interrogation of the pathogenesis of this condition.
Arteriolosclerosis, a small arterial or arteriolar subtype of arteriosclerosis, describes thickening of arterial walls with luminal occlusion, resulting in loss of elasticity. 10,11,19,28 Hyaline arteriolosclerosis (HAS) specifically describes expansion of the subintima/media by abundant glassy, eosinophilic, amorphous, proteinaceous material with effacement of normal structure. Extreme narrowing can cause ischemia. Although most commonly associated with renal (and glomerular) disease in humans with hypertension or diabetes mellitus, it can also be seen as an age-related change in normotensive individuals, most commonly in the spleen, pancreas, and adrenal, with relative sparing of the kidneys. 19 Impaired blood pressure homeostasis has been suggested as the underlying mechanism for the lesion regardless of etiology. 13 Immunofluorescence studies of frozen renal biopsies have shown that the hyaline material consists of inactivated complement 3b (iC3b) bound to hyaluronic acid, with variable IgM and other complement components thought to arise from new antigens in the iC3b. 11
Arteriolosclerosis has rarely been reported as an age-related finding in mice. 26 In 1 study, arteriolosclerosis was identified in 8% and 21% of aging virgin female BALB/cAnNBdf and RFM/Un mice, respectively. 9 Similar to humans, lesions occurred most commonly in the spleen, kidney, and uterus and less commonly in the heart, pancreas, and intestine. Clapp 8 reported a similar distribution of HAS in 14.1% of aged female RF/Un mice. Interestingly, in Figure 206 of that monograph, HAS was described in the intertubular arteriole (“spermatic artery”) of a single male mouse from a different study, with no further comment. Maita et al 18 describe an infrequent syndrome of “systemic arteritis” consisting of “marked thickening of the tunica media with a considerable amount of eosinophilic deposits” in large cohort of outbred ICR Crj:CD-1 mice. Lesions affected small to medium arteries, with frequent thrombosis and mild leukocytic infiltration. In addition to the ovary, lesions were observed in uterus, kidney, and heart. Mullink and Haneveld 20 included medial hyalinosis in a wide spectrum of arterial lesions observed in spontaneously hypertensive mice, but little detail was reported. Isolated testicular HAS has not previously been reported as a phenotype in aging mice or any other animal species.
During a screen for vascular phenotypes in aged laboratory mice, a new discrete phenotype of HAS of the intertubular arteries and arterioles of the testes was identified in several inbred strains. This investigation describes the frequency and severity by strain of testicular HAS in aging mice of 30 inbred and wild-derived mouse strains.
Materials and Methods
Mice
The following 30 strains of inbred and wild-derived mice were used in a large-scale aging study, 32,35 and as part of a detailed histopathological analysis, vessels were examined for vascular phenotypes: 129S1/SvImJ, A/J, AKR/J, BALB/cByJ, BTBRT+tf/J, BUB/BnJ, C3H/HeJ, C57BL/10J, C57BL/6J, C57BLKS/J, C57BR/cdJ, C57L/J, CBA/J, DBA/2J, FVB/NJ, KK/HIJ, LP/J, MRL/MpJ, NOD.B10Sn-H2b/J (NOD; a congenic strain with the NOD genetic background but with a histocompatibility locus from a diabetes-resistant strain), NON/ShiLtJ, NZO/HlLtJ, NZW/LacJ, P/J, PL/J, PWD/PhJ, RIIIS/J, SJL/J, SM/J, SWR/J, and WSB/EiJ. All mice were obtained from The Jackson Laboratory (Bar Harbor, ME) at 6 to 8 weeks of age. Mice were divided into 3 groups. The longitudinal study (65 females and 35 males; 555–985 days of age) maintained mice until they became morbid or died naturally (ie, death related to age). Two groups of mice were used in cross-sectional studies to define onset of lesions at defined ages. Mice were euthanized and studied at approximately 12 (372–418 days) and 20 (606–663 days) months of age, respectively. Cross-sectional and longitudinal study groups were set up in parallel. The cross-sectional groups and moribund mice were euthanized by CO2 asphyxiation using methods approved by the American Veterinary Medical Association and complete necropsies were performed. 31 The mouse rooms were maintained on a 12-hour light/12-hour dark cycle and at an ambient temperature of 21°C to 23°C. Mice of the same sex (4 per cage) were housed in duplex polycarbonate cages (31 × 31 × 214 cm) on pressurized individually ventilated mouse racks (Thoran Caging System, Hazleton, PA) with a high-efficiency particulate air-filtered supply and exhaust. Mice were allowed ad libitum access to acidified water (pH 2.8–3.2) and fed pellets containing 6% fat (LabDiet 5K52; PMI Nutritional International, Bentwood, MO). Regular monitoring for viruses, bacteria, parasites, and microsporidium showed that the colonies were free of any infestation (http://jaxmice.jax.org/genetichealth/index.html). All protocols were reviewed and approved by The Jackson Laboratory Animal Care and Use Committee.
Tissue Fixation and Preparation
Complete necropsies were performed at the time of euthanasia. 31 Tissues were collected, fixed in Fekete’s acid-alcohol-formalin solution overnight, and stored in 70% ethanol until processing. Bones were decalcified overnight in Cal-Ex (Fisher, Pittsburgh, PA) and briefly rinsed in water before trimming. Tissues were processed routinely for histology, embedded in paraffin, cut into 6-μm sections, and stained with hematoxylin and eosin (HE). Additional serial sections were stained with periodic acid–Schiff (PAS), Masson’s trichrome, and Movat’s pentachrome stains.
Histopathologic Analyses
All slides were initially reviewed by the same experienced, American College of Veterinary Pathologists (ACVP) diplomate veterinary pathologist (J.P.S.) and the vascular lesions reevaluated by a second diplomate veterinary pathologist (T.K.C.). Physiological phenotyping data using the International Knockout Mouse Project protocols were generated from this same group of mouse strains and are freely accessible online through the Mouse Phenome Database (MPD, http://phenome.jax.org). 2,3 Vascular lesions were coded to the Mouse Pathology (MPATH) and Mouse Anatomy (MA) ontologies as previously described, 29,33 and anatomical location and pathological diagnoses were combined into the precomposed PAM ontology, which classifies lesions from MPATH by anatomical site using the MA ontology. 1 Overrepresentation was calculated using the Ontofunc and Func tools 14 as described previously. 1 We performed a hypergeometric test to establish the strains in which vascular lesions are overrepresented. The P value obtained indicates which strains and sex have disproportionately frequent vascular lesions of all types with respect to all the strains examined.
The frequency of HAS lesions was defined as the number of mice carrying diagnosed lesions by strain and sex. Lesions were also characterized by severity scores for each mouse (scores: 0, normal; 1, minimal; 2, mild; 3, moderate; 4, severe). Average scores of all affected mice per strain were obtained.
Results
Description of Phenotype
Lesions were essentially limited to the small (approximately 50–100 μm diameter) intraparenchymal (intertubular) muscular arteries and arterioles of the testes (Figs. 1–3). Lesion severity was semiquantitatively evaluated on the basis of number and size of vessels affected as well as degree of vascular lesions. In minimal lesions, rare small arteriolar walls were expanded by eccentric medial droplets of brightly eosinophilic hyaline proteinaceous material. These droplets coalesced to expand the wall and impinged on the lumen in mild and moderate lesions, with increasing numbers of vessels affected. In severe lesions, multiple arterioles and small arteries were markedly expanded by abundant medial hyaline material that compressed or obliterated the lumina. Lesions were segmental within arteries and were particularly prominent at arterial branch points. There were various degrees of mild to moderate concentric adventitial fibroplasia or fibrosis (Fig. 4), with occasional adventitial infiltrates of low to rarely moderate numbers of plasma cells, macrophages, and lymphocytes. Rarely, mice had overt leukocytic infiltration of intima/media (macrophages, viable and degenerate neutrophils, pyknotic nuclei).
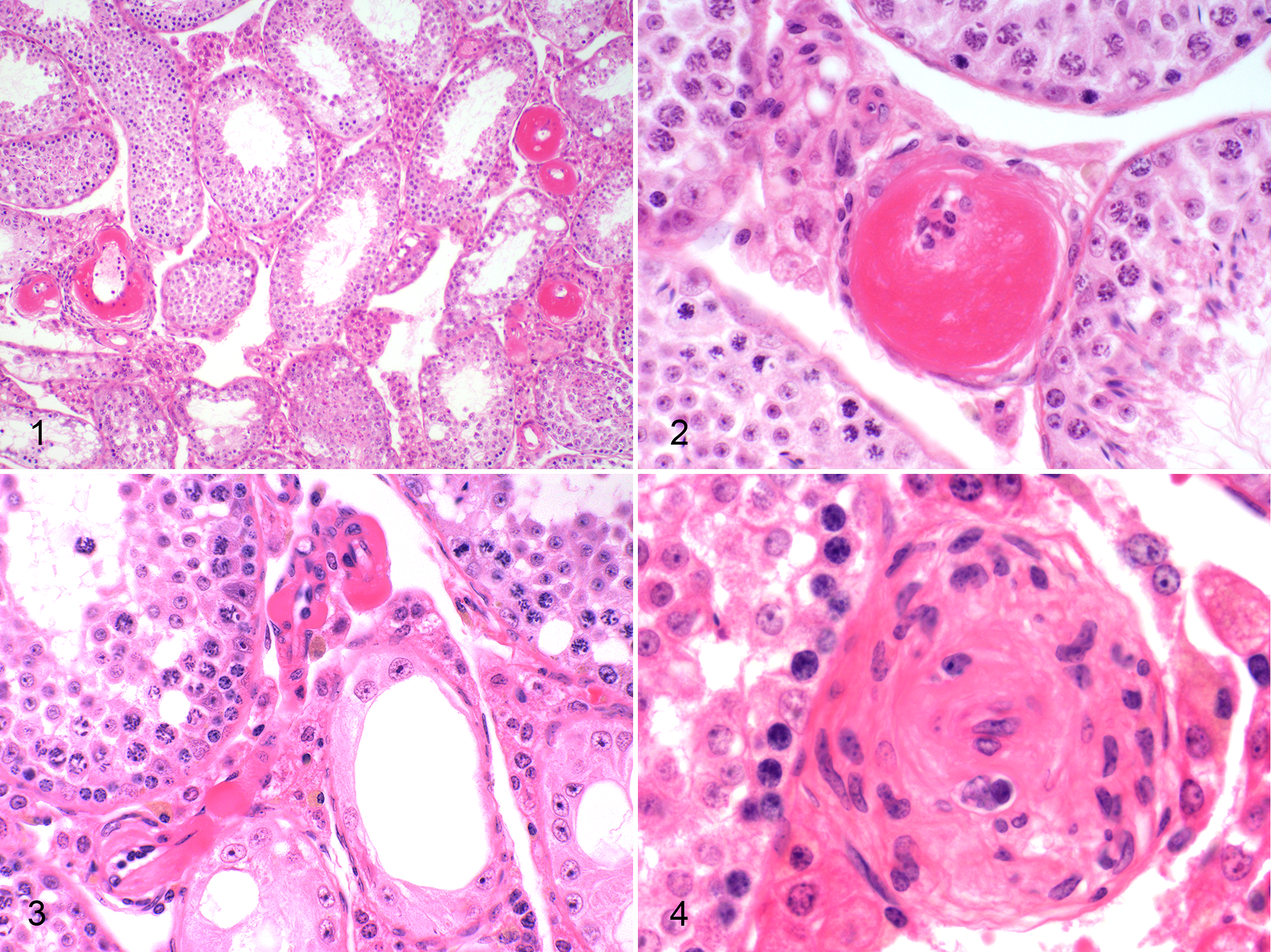
Arteriolosclerosis, testes, mice. Figure 1. Hyaline material expands the media of multiple medium and small intertubular arteries in a 20-month-old SM/J mouse. Unaffected arteries are present, demonstrating the segmental nature of the lesions. Hematoxylin and eosin (HE). Figure 2. Abundant hyaline eosinophilic material expands the media and compresses the lumen of a small intertubular artery of a 20-month-old SM/J mouse. HE. Figure 3. Sclerosis of the arterioles in a 20-month-old WSB/EiJ mouse. HE. Figure 4. Chronic lesion with florid adventitial fibrosis in a 20-month-old SM/J mouse. HE.
Lesions in SM/J mice predominantly affected small arteries and, to a lesser extent, arterioles. By contrast, lesions in WSB/EiJ mice were essentially limited to arterioles (Fig. 2). Lesions were not present in any other organs, including kidney, pancreas, and spleen. No other vascular lesions (eg, polyarteritis nodosa [PAN], medial mineralization, hyaline glomerulopathy, atherosclerosis) were present in these mice. In 2 SM/J mice, there was mild to moderate HAS of a few renal arcuate arteries. One WSB/EiJ mouse also had focal HAS in the splenic red pulp. Other than these exceptions, lesions were not present in any other organs/sites.
Hyaline material was weakly to intensely PAS positive (Fig. 5). In a few 2-year-old SM/J mice, there were scattered lesions consistent with hyperplastic arteriolosclerosis, with concentric layers of plump hypertrophic smooth muscle cells in a loose (onionskin-like) matrix, highlighted by PAS staining (Fig. 6). By Masson’s trichrome, hyaline material varied from intensely bright red (fibrinoid) to blue and fibrillar (fibrosis) (Fig. 7), and there was occasional adventitial fibrosis (Fig. 8). By Movat’s pentachrome, there was fragmentation or loss of the internal elastic lamina, with accumulation of the hyaline material in the subendothelial space.
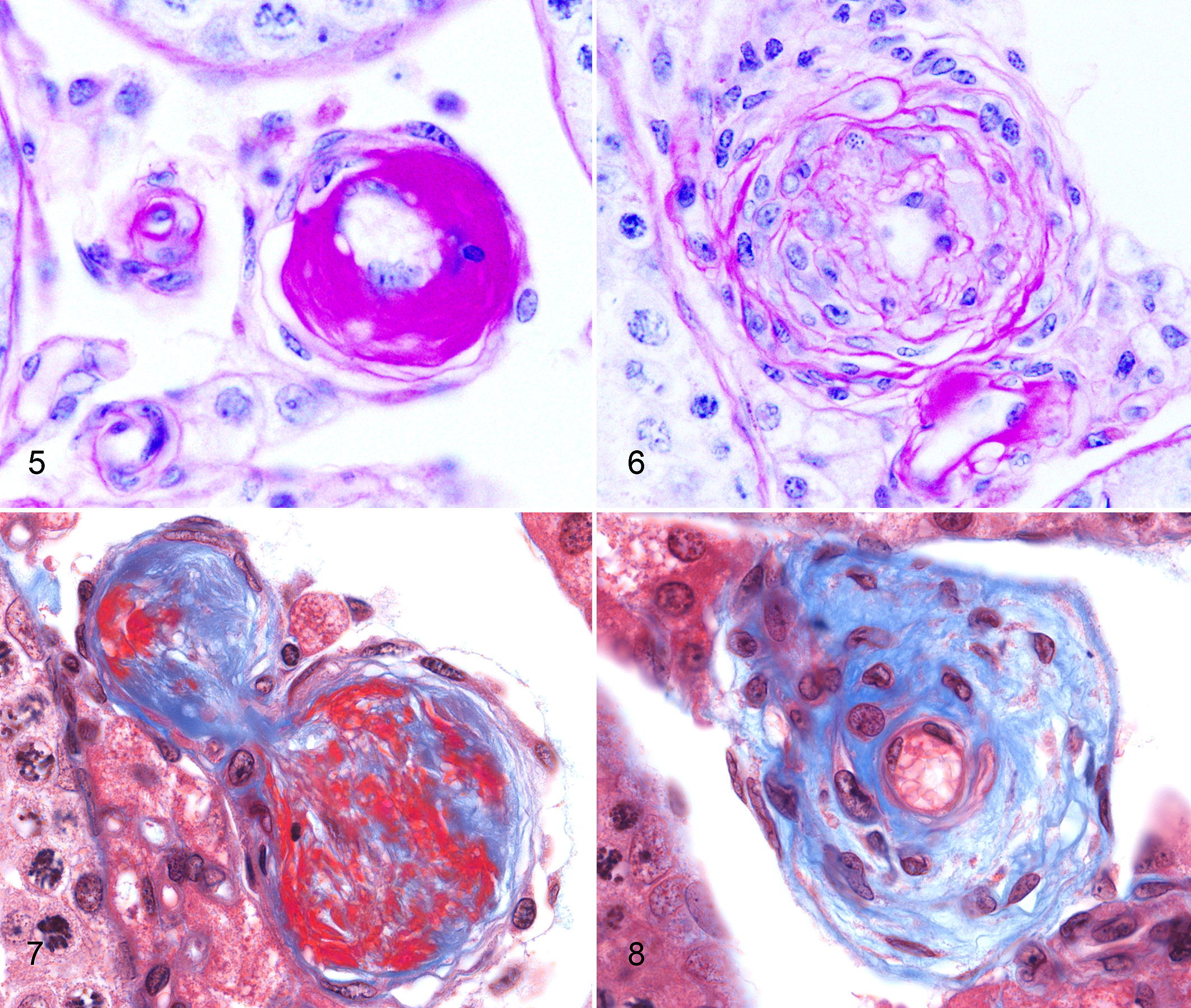
Arteriolosclerosis, testes, mice. Figure 5. Periodic acid–Schiff (PAS) positive hyaline material expands the arterial tunica media in a 20-month-old SM/J mouse. PAS. Figure 6. Hyperplastic smooth muscle in the media of a small testicular artery of a 20-month-old SM/J mouse. PAS. Figure 7. Abundant medial fibrinoid material (bright red) admixes with fibrillar collagen (blue) in a 12-month-old SM/J mouse. Masson’s trichome. Figure 8. Different arterial segment from the same mouse showing mature diffuse medial and adventitial fibrosis and demonstrating the variable nature of lesions within adjacent vessels. Masson’s trichrome.
Frequency and Severity of Lesions by Strain
Hyaline arteriolosclerosis lesions were not noted in females of any strain in any organ. Overall lesion frequency in males was highest in the SM/J (0.9) and WSB/EiJ (0.73) strains (Table 1 and Fig. 9). Lesions were more severe in SM/J than in WSB/EiJ, and in both strains, the lesion severity increased with age from 12 to 20 months (2.6 to 3.77 and 1.33 to 2.09, respectively). HAS lesions appeared sporadically in individual males of other strains, including 129S1/SvlmJ (n = 5/42), A/J (1/32), BUB/BnJ (2/22), DBA/2J (1/24), and FVB/NJ (1/26). No lesions were present in mice from BALB/cByJ (n = 31 males), BTBRT+ tf/J (23), C3H/HeJ (29), C57BL/10J (32), C57BL/6J (39), C57BLKS/J (44), C57L/J (32), KK/HIJ (30), LP/J (37), MRL/MpJ (30), NON/ShiLtJ (28), NZO/HlLtJ (12), NZW/LacJ (27), PL/J (19), PWD/PhJ (25), RIIIS/J (32), SJL/J (11), or SWR/J (19) strains. Overrepresentation analysis of complete screening data, coded using the MPATH and MA ontologies, 33 excluding testicular HAS, showed an overall excess of vasculitis (polyarteritis nodosa) in females of BUB/BnJ and in males of NZO/H1LtJ (P = .008 and P = .0006, respectively). Compared with the distribution of HAS across these strains, this suggests that factors predisposing to the testicular lesions in SM/J and WSB/EiJ mice do not seem to produce an overall tendency to vasculitis/vascular lesions in other organs or in other strains, indicating distinct genetic predisposition for this lesion. Lesions were present at high or low frequency in strains from all mouse family tree groups except Group 3 (Japanese and New Zealand inbred strains).
Frequency and Severity of Testicular Arteriolosclerosis by Age and Strain.a
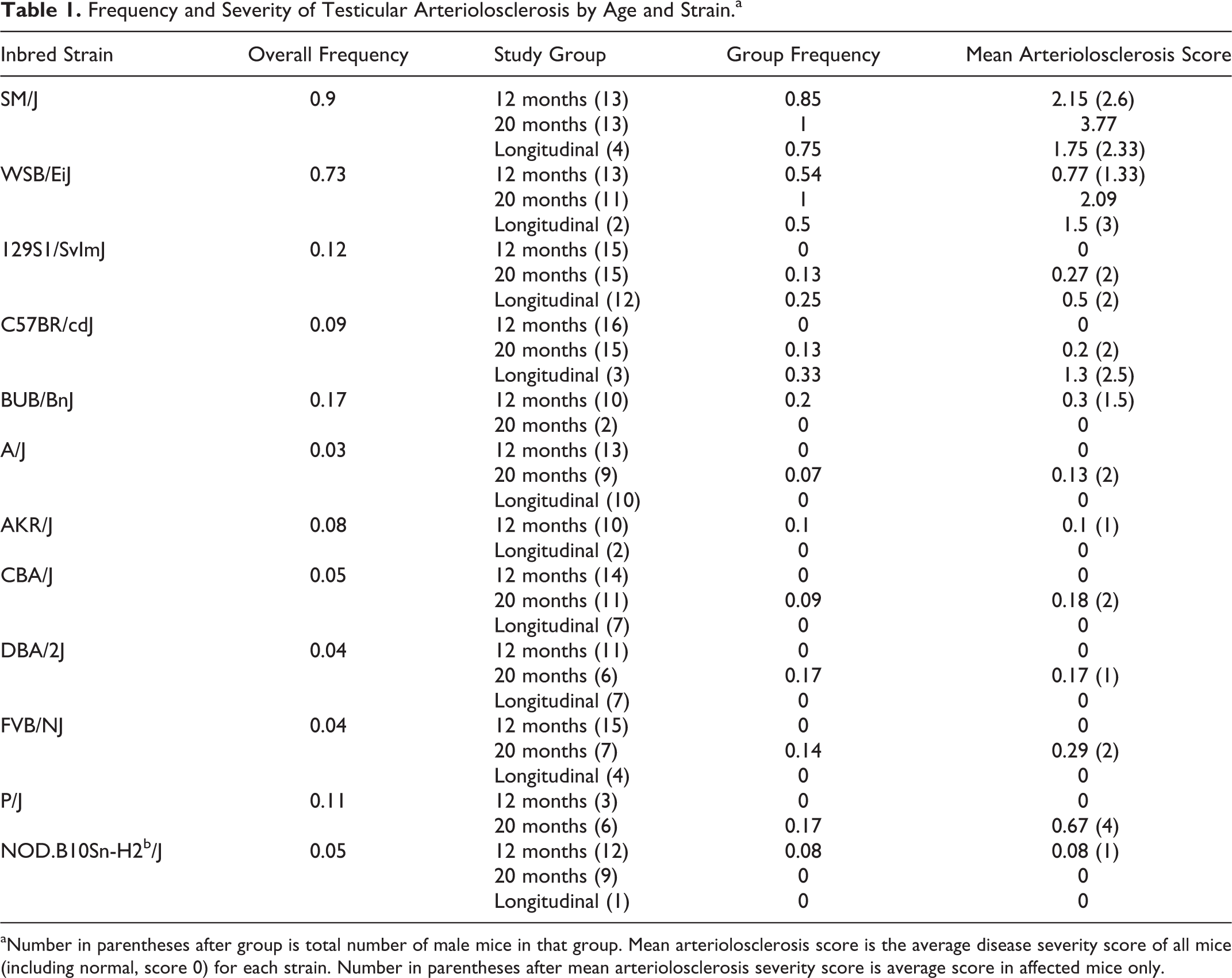
aNumber in parentheses after group is total number of male mice in that group. Mean arteriolosclerosis score is the average disease severity score of all mice (including normal, score 0) for each strain. Number in parentheses after mean arteriolosclerosis severity score is average score in affected mice only.
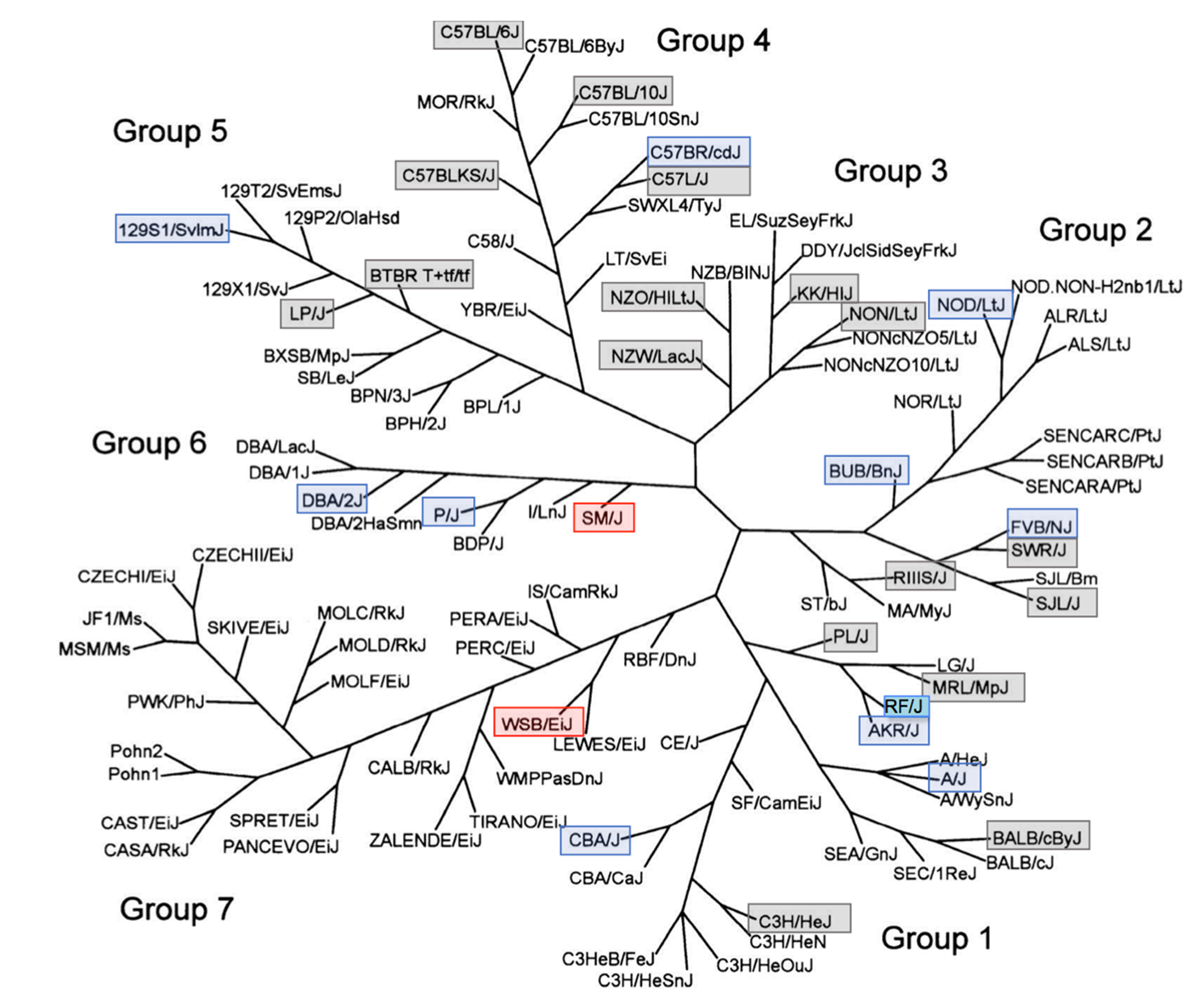
Testicular arteriolosclerosis by mouse strain. Adapted from Petkov et al. 25 Used with permission. The phenotype of testicular hyaline arteriolosclerosis is present with varying frequency in multiple genetically divergent and distinct inbred mouse strains. Red boxes delineate strains with high frequency, blue boxes for strains with low frequency, and gray boxes for strains negative for the phenotype. The PWD/PhJ strain (negative, not shown) is in Group 7. The RF strain in Group 1 (turquoise) was previously described to develop these lesions, but no frequency was reported.
Discussion
The frequency of arteriolosclerosis—often subsumed under the general condition of small vessel disease (SVD) 4 or, more accurately, hyaline arteriolar sclerosis—in inbred strains of laboratory mice has not been systematically reported previously. Here we show that the lesion is almost uniquely present in the testis of 2 inbred strains of mice and increases in frequency as these male mice age. Hyaline arteriolosclerosis has not been reported previously in the intratesticular arterial system of rodents, 7 as discussed above, but age-associated testicular HAS occurs in humans. 30 As with all types of vascular lesions, extratesticular HAS seem to have a more uniform anatomic distribution and much lower frequency in strains other than SM/J and WSB/EiJ; the resulting implication is that its localized occurrence in the testicular vasculature of SM/J and WSB/EiJ mice has a specific genetic component.
SM/J mice were developed by MacArthur from 7 stocks and were selected for small body size. 17 SM/J mice are susceptible to atherosclerosis when fed a high-fat diet but maintain a normal high-density lipoprotein level (http://www.informatics.jax.org/external/festing/mouse/docs/SM.shtml). Interestingly, SM/J mice are reported to be difficult breeders. The relationship between the observed arterial disease and poor reproductive performance remains unexplored. However, there was no direct relationship between observed degeneration of the seminiferous tubules and arterial lesions present in examined mice. WSB/EiJ mice are particularly long lived, but SM/J males are regarded as having an intermediate life span (median life span 783 vs 871 days for WSB/EiJ), mitigating against any argument that the lesions are simply age dependent. 35 The SM/J strain is related to C. C. Little’s DBA and related strains (Group 6) derived from Mus musculus domesticus. 25 There was a low frequency of lesions in the only other Group 6 strains examined, DBA/2J and P/J.
Watkins star line B (WSB/EiJ) mice were derived from wild M. m. domesticus mice trapped on Maryland’s Eastern Shore by Michael Potter in 1976. 27 Notably, WSB/EiJ contains an allele of R2d2 that is subject to meiotic drive favoring its transmission and shows no evidence of introgression from other subspecies. 5 SM/J (Group 6) and WSB/EiJ (Group 7, wild-derived strains) are not closely related strains, suggesting that any shared genetic predisposition to HAS either was present in common ancestral M. m. domesticus mouse populations and subsequently lost from or otherwise suppressed in related strains or more likely arose spontaneously and independently in these 2 lineages and became fixed through inbreeding. 25,34 Notably, testicular HAS lesions were absent from the PWD/PhJ strain, the only other member of Group 7 examined. Phylogenetic relationships suggest that the closely related LEWES/EiJ might be informative as it is of all the wild derived strains most closely related to WSB/EiJ. 25 The presence of low frequency of lesions and negative findings within multiple-strain groups, including within 2 closely related strains (eg, C57L/J and C57BR/cdJ, FVBN/J and SWR/J), may also argue for a multigenic mode of inheritance.
Human studies have indicated several genes that may be involved in predisposition to HAS, mainly in circumstances of normal or accelerated aging. Central nervous system (CNS) degenerative disease is often associated with vascular lesions. ABCC9 variants have been implicated in hippocampal sclerosis; 15,21 –23 HTRA1 variants are associated with cerebral autosomal recessive arteriopathy with subcortical infarcts and leukoencephalopathy (CARASIL), although in this case, HAS was also described in extracerebral, visceral sites, 16 and LMNA is implicated in Hutchinson-Gilford progeria syndrome. 24 A study of stroke predisposition associated a diabetes risk allele within JAZF1 with arteriolosclerosis, 6 and a variant in GNB3 is implicated in radial vasculature hypertrophy. 12 Of these candidates, only Lmna and Abcc9 have vascular phenotypes in null mice (listed as abnormal vascular smooth muscle physiology; Mouse Genome Informatics, accessed January 3, 2019), and none specifically report HAS, testicular or otherwise (Mouse Genome Informatics, accessed January 3, 2019). Mice with a null allele of Jazf1 have very recently been reported, but the only abnormal phenotype described is reduced circulating fasting insulin levels. There is no reported expression in the vasculature or the gonad of either sex (http://www.mousephenotype.org/data/genes/MGI:2141450#section-associations). Although this discrete phenotype of testicular HAS would be an ideal candidate for genome-wide association studies, current single-nucleotide polymorphism (SNP) coverage density of the SM/J strain is insufficient to robustly identify candidate genes. An in-depth interrogation of the genetics underpinning this phenotype therefore awaits better sequencing of this strain.
Because of their well-defined genetics, including the availability of inbred strains, combined with the accessibility of the testes to manipulation or even unilateral excision, this mouse model presents a unique opportunity to study the genetics and pathogenesis of age-related arteriolosclerosis, as well as the possible interaction with male infertility. Additional larger future studies, including younger mice, may further refine the pathogenesis of this phenotype.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: J.P.S., K.A.S., and V.E.K. have sponsored research recently completed or in progress with Biocon, Bioniz, Curadim, Takeda, and Theravance all of which are unrelated to this project. All other authors state no conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from the Ellison Medical Foundation for the generation and histopathological phenotyping of the aging mice and the National Institutes of Health (AG025707 for the Shock Aging Center) for maintaining the aging mouse colony. T.K.C. was a recipient of a North American Hair Research Society Mentorship Grant. The Jackson Laboratory Shared Scientific Services were supported in part by a Basic Cancer Center Core Grant from the National Cancer Institute (CA034196). R.H. and S.A. are supported from King Abdullah University of Science and Technology (KAUST) Office of Sponsored Research (OSR) under award URF/1/3454-01-01. P.N.S. acknowledges travel support from the Warden and Fellows of Robinson College, Cambridge.
