Abstract
Pathologic features of 12 cats with naturally acquired systemic hypertension and concomitant hypertensive encephalopathy were analyzed. All cats demonstrated acute onset of signs localized to the forebrain and/or brainstem, including stupor, coma, and seizures. All cats had systemic hypertension, ranging from 160 to 300 mm Hg. Gross lesions were identified in 4 of 12 cases, including caudal herniation of the cerebrum and cerebellum, sometimes with compression of the rostral colliculus and medulla. Histologically, all cases featured bilaterally symmetrical edema of the cerebral white matter. Associated vascular lesions, especially arteriolar hyalinosis, were also observed. Concurrent lesions were chronic tubulointerstitial nephritis (11/12 cases), adenomatous hyperplasia of the thyroid gland (4 cases), hypertensive choroidal arteriopathy (6 cases), and left ventricular hypertrophy (5 cases). This study demonstrates that the typical histologic manifestation of spontaneous hypertensive encephalopathy in cats is bilaterally symmetrical edema of the subcortical cerebral white matter.
Keywords
Hypertensive encephalopathy is an acute neurologic syndrome described in cats associated with sudden and sustained increases in systemic blood pressure. 3,12,14 Clinical signs and pathological findings are similar to hypertensive encephalopathy in humans and an associated condition called posterior reversible encephalopathy syndrome (PRES). In both diseases, affected individuals present with seizures and neuroimaging findings consistent with prosencephalic white matter edema. 13 This same brain region is affected by hypertension in cats, with associated neurologic clinical signs (lethargy, seizures, stupor, and blindness). 3,12,14 Systemic hypertension in cats has numerous underlying causes, most commonly chronic renal disease and hyperthyroidism. 4,6,9 –11 Other less common causes include pheochromocytoma, diabetes mellitus, and hyperaldosteronism. 10,14 While previous studies reported neuropathologic findings in experimentally induced hypertension in cats, 3 no study has described these changes in spontaneously occurring cases. As such, this report describes the characteristic gross and histopathologic lesions of 12 spontaneous cases of feline hypertensive encephalopathy.
The Penn Vet Diagnostic Laboratory database was searched between 2000 and 2013 for feline cases with the following diagnoses: “white matter edema,” “corona radiata edema,” “cerebral edema,” and central nervous system (CNS) “arteriolar hyalinosis,” “arteriolosclerosis,” and “arteropathy.” Twelve cases met the inclusion criteria for the study. Cases were included if they demonstrated acute onset of clinical signs consistent with forebrain and/or brainstem dysfunction and had concurrent systemic hypertension (>160 mm Hg) as documented by Doppler flow ultrasonography and/or oscillometry. Neurologic examination was performed by board-certified veterinary neurologists (6 cases), board-certified emergency medicine clinicians (4 cases), or board-certified veterinary internists (2 cases), who localized clinical signs to specific brain regions. Cases were excluded if cats were exposed to other known causes for bilaterally symmetrical leukoencephalopathy, like toxins (bromethalin or hexachlorophen), nutritional deficiencies, Feline Immunodeficiency Virus (FIV) infection, hyperammonemia, or liver failure. Furthermore, no cases had documented administration of therapeutics that increase blood pressure (ie, nonsteroidal anti-inflammatory drugs, antidepressants, or immunosuppressants). Complete autopsies were performed in 11 cases, and 1 whole brain was submitted for evaluation.
Brain tissue was fixed for at least 24 hours in a 1:1 ratio of 10% neutral buffered formalin and 37% formalin stock solution. Five representative levels were evaluated histologically: frontal lobe including caudate nuclei and internal capsule, parietal lobe with diencephalon, mesencephalon, metencephalon (cerebellum and cerebellar peduncles), and myelencephalon (medulla rostral to obex). Paraffin-embedded tissues were sectioned (5 μm) and stained with hematoxylin and eosin (HE). Selected tissue sections were stained with periodic acid–Schiff (PAS) and luxol fast blue (LFB).
Ages ranged from 8 to 16 years (mean and median age of 12 years) and breeds included domestic shorthair (9 cases) and domestic longhair, Persian, and Turkish Angora (1 case each) (Suppl. Table S1). All cases had systemic hypertension with systolic blood pressure measurements ranging between 160 and 300 mm Hg (mean, 228.75 mm Hg; median, 230 mm Hg). Blood pressure readings were taken within 1 day of or on the day of death and were concurrent with neurologic signs (Suppl. Table S1).
Clinically, neurological signs were localized to the prosencephalon and/or posterior fossa (brainstem and cerebellum). Eight cats demonstrated cranial nerve deficits consisting of 1 or multiple of the following: absent menace response, absent pupillary light reflex, anisocoria, and blindness. Six cats had altered mentation ranging from dull to comatose. Five animals developed 1 or multiple seizures or seizure-like episodes. Circling occurred in 3 cases.
Gross brain lesions were recognized in 4 cases, 3 of which had cerebral edema with loss of gray/white matter distinction. Two cases demonstrated caudal displacement or herniation of the cerebellum with variably evident symmetrical compression of the mesencephalon, particularly the rostral colliculi and medulla. One cat had focal cerebellar hemorrhage with caudal, rightward deviation of the vermis. The gross lesion in the fourth animal consisted of focal hemorrhage within the caudal medulla.
Histologically, all cases demonstrated bilaterally symmetrical, regional, to diffuse cerebral white matter edema in the corona radiata of the frontal and/or parietal lobes and was often most severe at the dorsal aspect of white matter tracts (Fig. 1a). Other commonly affected areas included the internal capsule in 9 cats and centrum semiovale in 3 cases. At low magnification, cerebral edema was characterized by bilateral regional pallor of subcortical white matter with blurring of white/gray matter distinction, visualized via LFB staining (Fig. 1b).
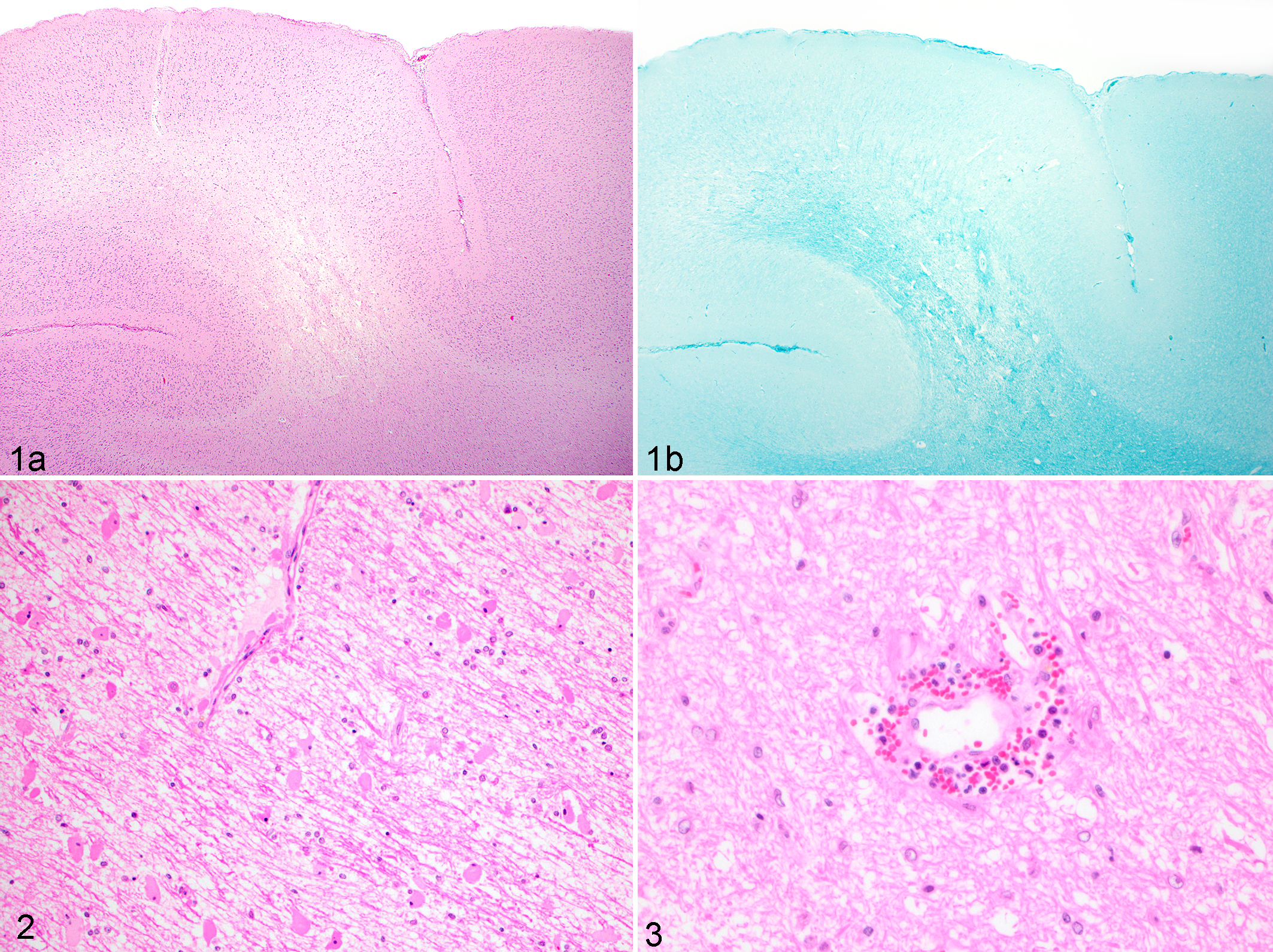
Hypertensive encephalopathy, brain, cat.
At higher magnification, there was variably severe separation of myelin fibers and marked perivascular expansion with clear space. Edema was accompanied by subjectively increased numbers of glial cells, generally approximate to lesion severity and characterized mainly by activated Alzheimer type II astrocytes (characterized by large nuclei with dispersed chromatin) with fewer microglia. In 3 of 12 cases, scattered reactive gemistocytic astrocytes were also present (Fig. 2). Within regions of edema and gray matter, small arteries were frequently lined by hypertrophied endothelium and sometimes surrounded by a thin cuff of erythrocytes mixed with modest numbers of lymphocytes and neutrophils. Within the adjacent neuroparenchyma, there were numerous Alzheimer type II astrocytes (Fig. 3). The presence of Alzheimer type II astrocytes was attributed to cerebral edema and hypertension, as hyperammonemia was not present in any of the cases. Six cases presented mild multifocal acute perivascular hemorrhage. The most severely injured arteries demonstrated changes consistent with arteriolosclerosis and hyalinosis. Walls were smudged, disrupted, indistinct, and sometimes contained hypereosinophilic hyaline, PAS-positive droplets (Fig. 4a,b). The most conspicuous lesions were present in deep penetrating arteries of the cerebrum. In addition, minimal spongiform change was noted within cerebellar white matter in 5 of 12 cases. Sections of mesencephalon were without significant microscopic lesions.
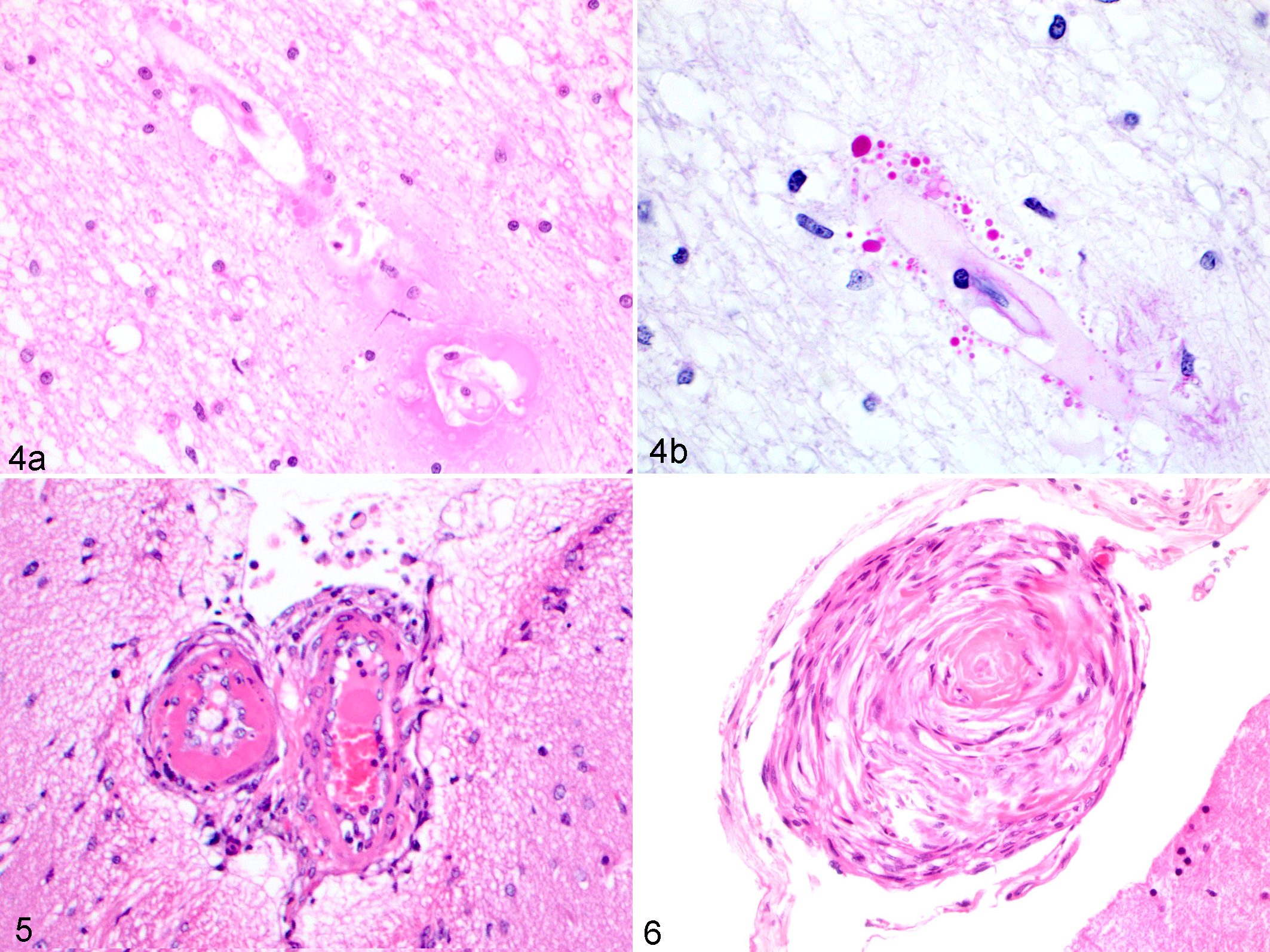
Vascular changes involving leptomeningeal arterioles were identified in 9 of 12 cases. Of these, multifocal arteriolar hyalinosis was the most consistent lesion (8 cases), characterized by mural thickening and replacement by deposits of hyaline material (Fig. 5).
Two cases exhibited infrequent hyperplastic arteriolosclerosis with characteristic “onion-skinning” fibrosis of vessel walls (Fig. 6). One of the cases (cat No. 10) had the highest recorded blood pressure (265 mm Hg) in the study. The other case (cat No. 9) had the longest reported history of hypertension (several months prior to death). In 5 cases, there were mild leptomeningeal perivascular inflammatory cell infiltrates of lymphocytes, histiocytes, plasma cells, and fewer neutrophils.
Concurrent lesions in nonnervous tissues were recognized, indicative of common comorbidities. Eleven of 12 cats had chronic tubulointerstitial nephritis ranging from mild to severe. Adenomatous hyperplasia of the thyroid gland was histologically diagnosed in 4 of 12 animals, all of which also had chronic tubulointerstitial nephritis. Of the 6 cases in which eyes were histologically examined, 4 had lesions of hypertensive choroidal arteriopathy (hyalinosis, mural thickening, and/or fibrinoid vascular necrosis). Five of 12 cases had cardiomegaly, with concentric hypertrophy of the left ventricular myocardium. In 2 of these cases, there was concurrent hyperthyroidism, and in 1 of the cases, cardiac myofibers were hypertrophied without disarray. In 1 case, cardiac myofibers were histologically normal, but intramural myocardial arteries demonstrated hyalinosis. No significant histologic lesions were found in the other 2 cases of cardiomegaly.
This case series describes the characteristic histological changes of spontaneous feline hypertensive encephalopathy. Similar to what was previously described in experimentally induced hypertension in cats, 3 histologic findings included separation of myelin sheaths by vasogenic edema in the cerebral white matter principally affecting the corona radiata and, to a lesser extent, the internal capsule and centrum semiovale. Interestingly, the few cases with grossly discernible lesions corresponded to higher blood pressure recordings.
Review of the clinical history of these cases revealed that hypertensive encephalopathy frequently occurred coincidentally with other metabolic/degenerative diseases that cause systemic hypertension, most commonly chronic renal disease and hyperthyroidism. There was only 1 case for which concurrent disease was not diagnosed. This may represent a case of essential or primary hypertension, which has been reported in up to 20% of hypertensive cats. 10
Both chronic renal disease and hyperthyroidism are common diseases in aged cats. A 50% prevalence of chronic renal disease has been demonstrated in adult feline populations, and it is estimated that over 10% of aged cats develop hyperthyroidism. 2,7,15 While the causative mechanism of hypertension differs (activation of the renin-angiotensin-aldosterone system [RAAS] vs cardiac and vascular compensatory response to vasodilation by tri-iodothyronine [T3]), the rise in systemic and intracranial pressure that occurs in both diseases results in neurologic clinical signs. Hypertensive encephalopathy is therefore an important differential diagnosis for neurologic signs in cats with chronic renal disease and hyperthyroidism.
In humans, the pathogenesis of hypertensive encephalopathy is thought to arise from an acute increase in blood pressure leading initially to arteriolar vasoconstriction and disruption of the blood-brain barrier, subsequently resulting in vasogenic edema. 13,14 Compensatory cerebral arteriolar vasodilation can then exacerbate cerebral edema. 13 With persistent hypertension, endothelium is damaged, and release of inflammatory cytokines can occur, further increasing permeability and edema. In addition, PRES is a condition in humans characterized by edema of the cerebral white matter in a pattern similar to that in hypertensive cats in this case series. The pathogenesis of PRES is unknown, although more than half of cases are associated with renal disease. 8
Regional edema of the subcortical white matter was noted grossly and histologically in these cases and confirmed with LFB staining. This histochemical staining pattern may also represent demyelination associated with edema. In humans, leukoencephalopathy and myelin loss can occur with hypertensive encephalopathy. 5,8 This is proposed to be secondary to chronic vascular obstruction and edema, but the exact pathogenesis of myelin loss is still undetermined. Another entity described in humans, known as osmotic demyelination syndrome, also results in cerebral edema and myelin loss and is associated with renal disease and hyponatremia. 8 The association with renal disease is similar to the feline cases here, although none of our cases were hyponatremic. Electron microscopy could be performed to determine whether white matter pallor represents edema alone or concurrent demyelination.
Arteriolar hyalinosis and arteriolosclerosis were a consistent vascular change within edematous areas, although associated hemorrhage was not always present. In many of these cases, hyaline vascular changes were evident in other organs, including eyes, kidneys, and heart, although only rarely with hemorrhage. This is in contrast to hypertensive encephalopathy in dogs and humans, in whom lacunar infarction and intracerebral hemorrhage are a common feature of cerebral vascular changes associated with hypertension. 16,17 This difference in lesions associated with hypertensive vascular changes could reflect the difference in cerebral vascular supply. Feline cerebral vascular supply to the brain is mainly by the maxillary artery (after branching from external carotid artery), while canine cerebral perfusion is supplied by the basilar and internal carotid arteries. 1
In summary, this study characterized typical histologic manifestations of spontaneous hypertensive encephalopathy in 12 cats. The consistent histologic lesion was bilateral vasogenic edema affecting the cerebral subcortical white matter within frontal and parietal lobes with arteriolar hyalinosis.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985819849500 - Neuropathology of Spontaneous Hypertensive Encephalopathy in Cats
Supplemental Material, DS1_VET_10.1177_0300985819849500 for Neuropathology of Spontaneous Hypertensive Encephalopathy in Cats by Molly E. Church, Bradley J. Turek and Amy C. Durham in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
