Abstract
Tissues from 9 Göttingen minipigs, aged 7 weeks to 1 year, with clinically diagnosed thrombocytopenic purpura syndrome were examined microscopically. All pigs had a history of spontaneous cutaneous purpura that was generally accompanied by disseminated visceral hemorrhages. Hematologic abnormalities included anemia (8 out of 9 pigs) and thrombocytopenia (7 out of 9 pigs), with platelet counts consistently below 20,000/μ. Microscopically, degenerative vascular lesions with morphologic features of arteriosclerosis were present in all 9 pigs. Vascular lesions affected small- to medium-sized muscular arteries and arterioles in various organs and extraparenchymal tissues; vessels of the renal pelvis and coronary arteries were consistently involved. Microscopic lesions in small- to medium-sized muscular arteries consisted of neointimal proliferation, medial thickening, luminal stenosis, thrombosis, disruption and fragmentation of the internal elastic lamina, necrosis of the tunica media, and medial deposits of myxoid matrix material. Microscopic lesions in arterioles included concentric laminar thickening of vessel walls (onion-skin pattern), endothelial cell hypertrophy, smooth muscle cell vacuolation, necrosis of the tunica media, thrombosis, and partial to complete luminal stenosis. Arteritis and/or periarteritis were also noted in 4 out of 9 pigs. Additional microscopic lesions included membranoproliferative glomerulonephritis (3 out of 9), myocardial microinfarcts (4 out of 7), renal interstitial fibrosis (2 out of 9), extramedullary hematopoiesis (6 out of 9), and intracapillary hyaline thrombi (2 out of 9). Degenerative vascular lesions have not been previously described in Göttingen minipigs with thrombocytopenic purpura syndrome. The etiopathogenesis of both the vascular lesions and thrombocytopenic purpura syndrome is currently unknown.
Keywords
Introduction
A syndrome of immune complex thrombocytopenic purpura with a proposed immune-complement mediated pathogenesis has previously been described in a Danish herd of Göttingen minipigs. 2 Eleven cases were reported over a period of 3.5 years, in minipigs between the ages of 3.5 and 9.5 months. The syndrome was clinically characterized by extensive subcutaneous hemorrhages, marked thrombocytopenia, and anemia. Consistently observed microscopic lesions included interstitial hemorrhages in the skin and viscera, and membranoproliferative glomerulonephritis. This retrospective study describes unique vascular lesions in 9 Göttingen minipigs from the USA with a clinically similar hemorrhagic syndrome.
Materials and Methods
Tissues from 9 Göttingen minipigs with clinical histories of cutaneous purpura and thrombocytopenia were selected from the archives of the Animal Disease Diagnostic Laboratory, Purdue University. Cases were submitted for histopathologic examination during the period of 28 May 2004, and 10 June 2005. All pigs were from the same supplier, which practices barrier breeding in specific pathogen free (SPF) facilities, and biannual health monitoring according to the Federation of European Laboratory Animal Science Associations (FELASA) recommendations. Clinical histories, hematologic data, and gross necropsy findings were provided by submitting clinicians. Published hematologic reference ranges were used to evaluate hematologic data. 1
Tissue samples, fixed in 10% neutral buffered formalin, were routinely processed, embedded in paraffin, sectioned in 4–6 µm thick sections, and stained with HE. All available tissues were examined microscopically. Tissues submitted varied between the 9 pigs, with the following distribution: kidney (n = 9), liver (n = 9), lung (n = 9), spleen (n = 8), lymph node (n = 8), small intestine (n = 8), heart (n = 7), urinary bladder (n = 7), stomach/omentum (n = 6), skin (n = 6), colon (n = 3), thymus (n = 2), salivary gland (n = 2), adrenal gland (n = 2), mesentery (n = 2), pancreas (n = 2), esophagus (n = 2), seminal vesicle (n = 1), prostate gland (n = 1), testis (n = 1), epididymis (n = 1), urethra (n = 1), diaphragm (n = 1), uterus (n = 1), ovary (n = 1), trachea (n = 1), thyroid gland (n = 1), eye (n = 1), brain (n = 1), ganglion (n = 1), aorta (n = 1), and skeletal muscle (n = 1). Selected sections were additionally stained with periodic acid–Schiff (PAS), Brown and Brenn Gram stain, Verhoeff van Gieson (elastin), Alcian blue, and/or Masson's trichrome stains.
Immunohistochemical testing for porcine reproductive and respiratory syndrome virus (PRRSV) and porcine circovirus type 2 (PCV-2) was perfomed on formalin-fixed paraffin-embedded sections of kidney from each of the 9 pigs. The primary antibodies used were rabbit polyclonal anti-PCV-2 antibody (ISU31) diluted 1 ° 1,500 incubated at room temperature for 60 minutes, and a commercially available mouse monoclonal anti-PRRSV antibody (Rural Technologies, Inc., Brookings, SD) diluted 1 ° 800 incubated at room temperature for 30 minutes. For PRRSV, antigen retrieval was performed by pretreatment with proteinase K for 5 minutes. A labeled streptavidin–biotin detection system was used, and diaminobenzidine (DAB) was used as chromagen. Sections of lung tissue and spleen from confirmed cases of PRRSV and PCV-2, respectively, were used as positive controls.
Results
The reported clinical status and hematologic abnormalities of the 9 pigs in this study are presented in Table 1. The pigs ranged from 7 weeks to 1 year of age. Eight of the pigs (Nos. 2–9) had terminal cutaneous purpura. All of these pigs, except for pig No. 5, for which no hematologic data were available, also had severe thrombocytopenia. Platelet counts were ≤ 20,000/µl in all 7 pigs with thrombocytopenia, and ≤ 5,000/µl in 4 of the pigs (Nos. 2, 3, 7, and 9). One pig (No. 1) was euthanized for severe epistaxis that developed 6 weeks after recovery from an episode of cutaneous purpura. Anemia was present in all 8 pigs for which hematologic data were available; 3 of these pigs (Nos. 4, 6, and 9) also had macrocytosis.
Reported clinical features and hematologic abnormalities in 9 Göttingen minipigs with thrombocytopenic purpura syndrome.
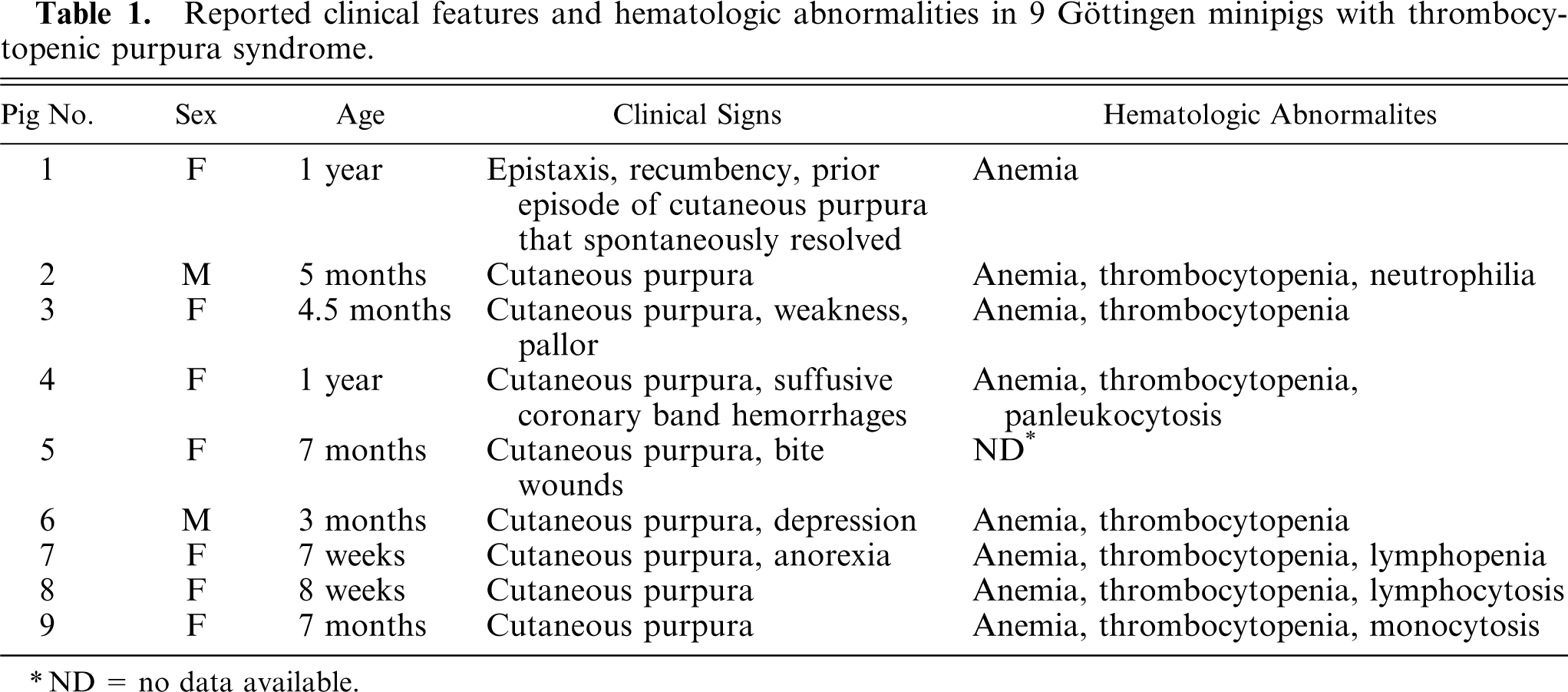
ND = no data available.
Disseminated hemorrhage was noted in all pigs with thrombocytopenia. Macroscopic hemorrhages most commonly affected the skin, heart, urinary bladder, intestine, kidney, and lung, but were also seen in various other tissues. Hemorrhage was severe and transmural in the urinary bladder of 4 out of 7 pigs (Nos. 2, 3, 6, and 7), and in the hearts of 5 out of 7 pigs (Nos. 3, 4, 5, 7, and 8). Effusions were not reported in any of the pigs.
Microscopically, hemorrhages were typically interstitial and often surrounded arterioles and small muscular arteries. Small aggregates of extracellular and/or intrahistiocytic hemosiderin pigment were often present within or adjacent to hemorrhagic foci. Extramedullary hematopoiesis (EMH) was a consistent microscopic finding in 6 out of 8 pigs with both anemia and thrombocytopenia (Nos. 2–7), and was seen in various locations, including hepatic sinusoids, splenic sinuses, alveolar capillaries, and the propria-submucosa of the urinary bladder.
Microscopic vascular lesions affecting arterioles and small- to medium-sized muscular arteries were detected in 9 out of 9 pigs, and were most consistently observed in the renal pelvis and heart. The morphology of vascular lesions varied between individual pigs, and is summarized in Table 2. Arteries and arterioles of the renal pelvis were affected in 9 out of 9 pigs. Epicardial and/or intramural coronary arteries were involved in 4 out of 7 pigs (Nos. 1, 5, 8, and 9) for which heart was examined. Vessels with varying degrees of degeneration were often observed in a single animal. Vascular lesions were most severe and chronic in pig No. 1, which had reportedly recovered from a purpuric episode approximately 6 weeks prior to death.
Range of microscopic lesions in arterioles and small- to medium-sized muscular arteries in 9 Göttingen minipigs with thrombocytopenic purpura syndrome.

Degenerative microscopic lesions observed in the small- to medium-sized muscular arteries included asymmetric thickening of vessel walls, eccentric to circumferential neointimal proliferation, medial thickening, partial to complete luminal stenosis, thrombosis, disruption and fragmentation of the internal elastic lamina, and focal medial deposits of myxoid, alcian blue-positive matrix material (Figs. 1, 2). In arterioles, the most striking microscopic lesion was concentric laminar thickening of vessel walls (onion-skin pattern), with accumulation of amphophilic matrix material between layers of hyperplastic and vacuolated smooth myocytes (Fig. 3). Affected arterioles had partial to complete luminal stenosis that was sometimes accompanied by thrombosis. The duration and severity of vascular lesions varied between tissues in a single animal, as well as between individual pigs. In the 2 pigs with mildest vascular lesions (Nos. 6 and 7), degenerative arteriolar changes consisted of smooth muscle cell vacuolation, medial thickening, and endothelial cell hypertrophy, and were restricted to arterioles of the renal pelvis (Fig. 4). These arterioles were not surrounded by hemorrhage.
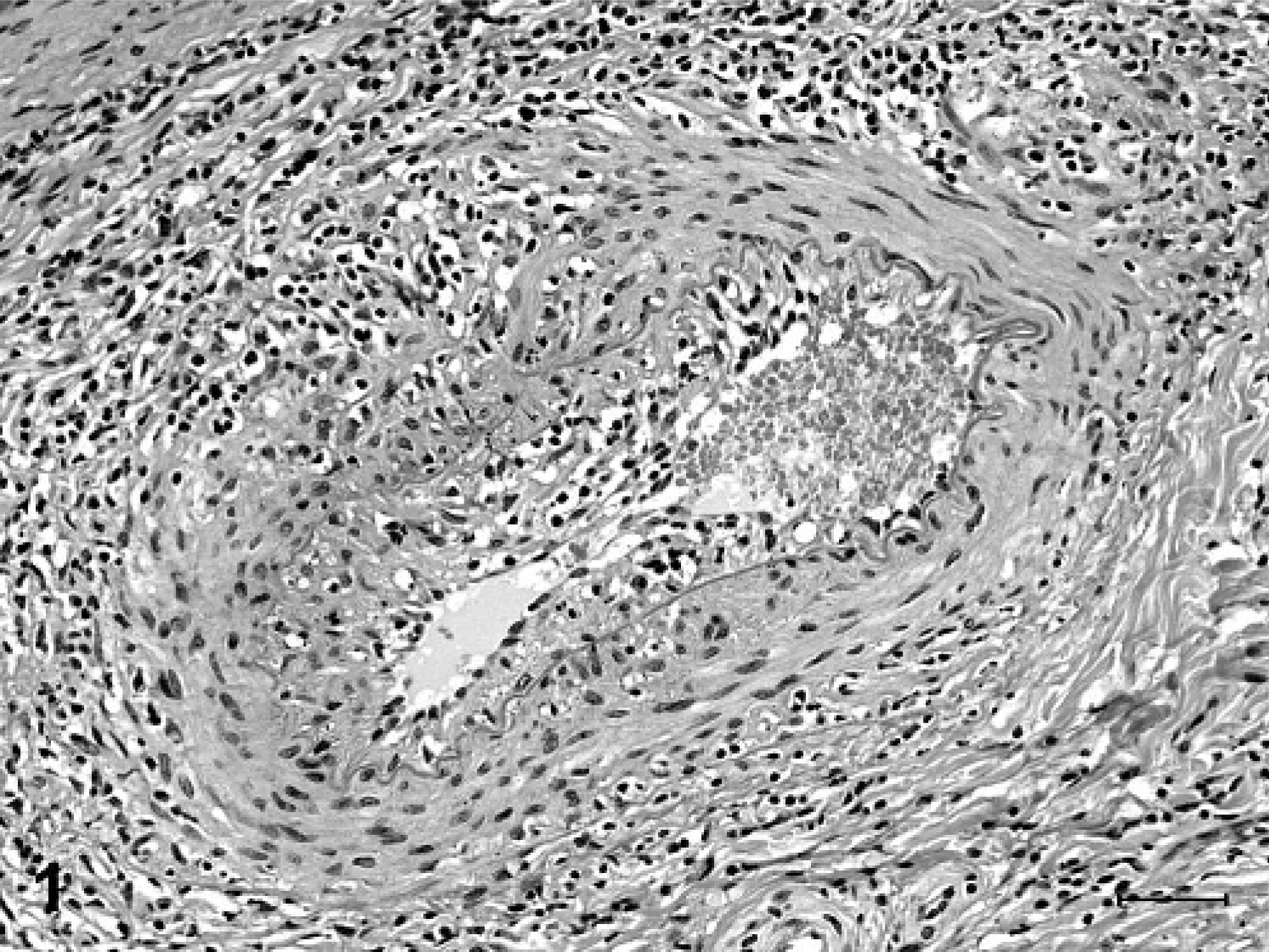
Spleen, muscular artery; pig No. 9. The vessel wall is asymmetrically thickened by neointimal proliferation and expansion of the tunica media by amphophilic myxoid material. The vessel wall is surrounded and multifocally infiltrated by lymphocytes. HE. Bar = 50 µm.
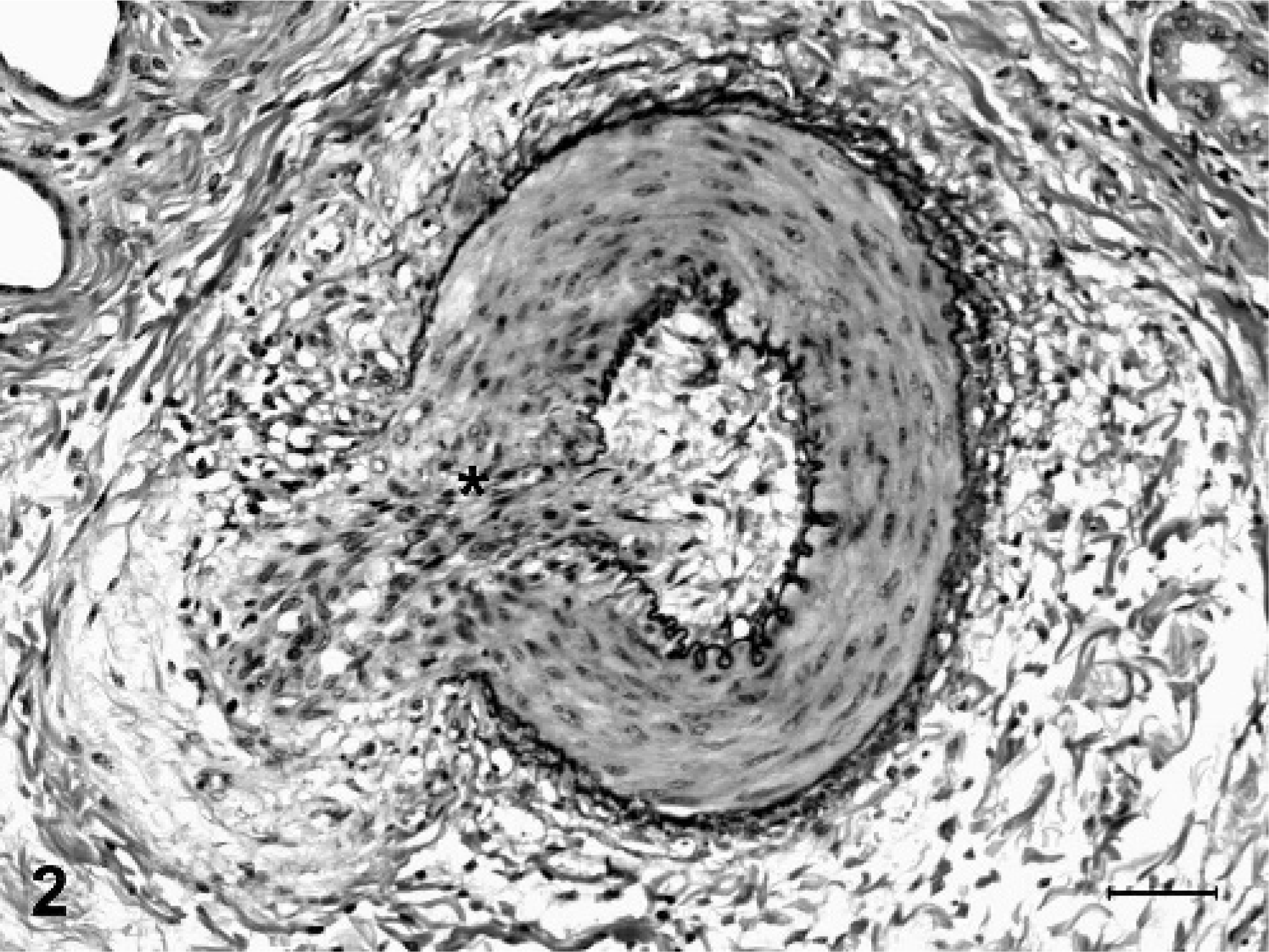
Kidney, muscular artery; pig No. 1. The lumen is obscured by neointimal proliferation, and there is segmental disruption of the tunic media (∗) and both internal and external elastic laminae. Verhoeff van Gieson's elastin stain. Bar = 50 µm.
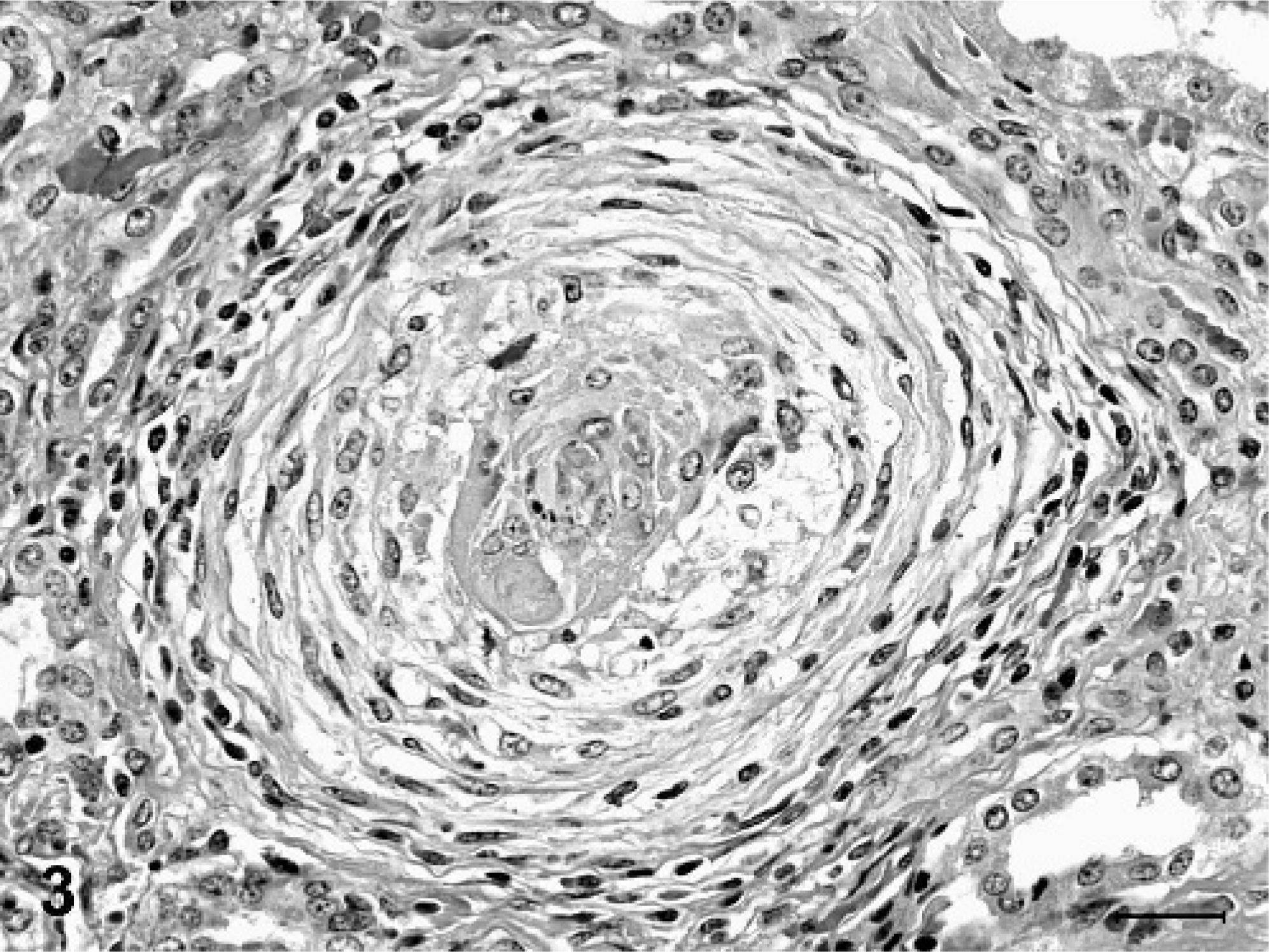
Kidney, arteriole; pig No. 1. The arteriolar wall is concentrically thickened with an onion-skin type pattern. Smooth muscle cells in the tunica media are vacuolated, and deposits of floccular amphophilic material sometimes alternate with the concentric layers. The lumen is narrowed and contains a small thrombus. The vessel is surrounded by a light perivascular infiltrate of lymphocytes, macrophages, and occasional eosinophils. HE. Bar = 25 µm.
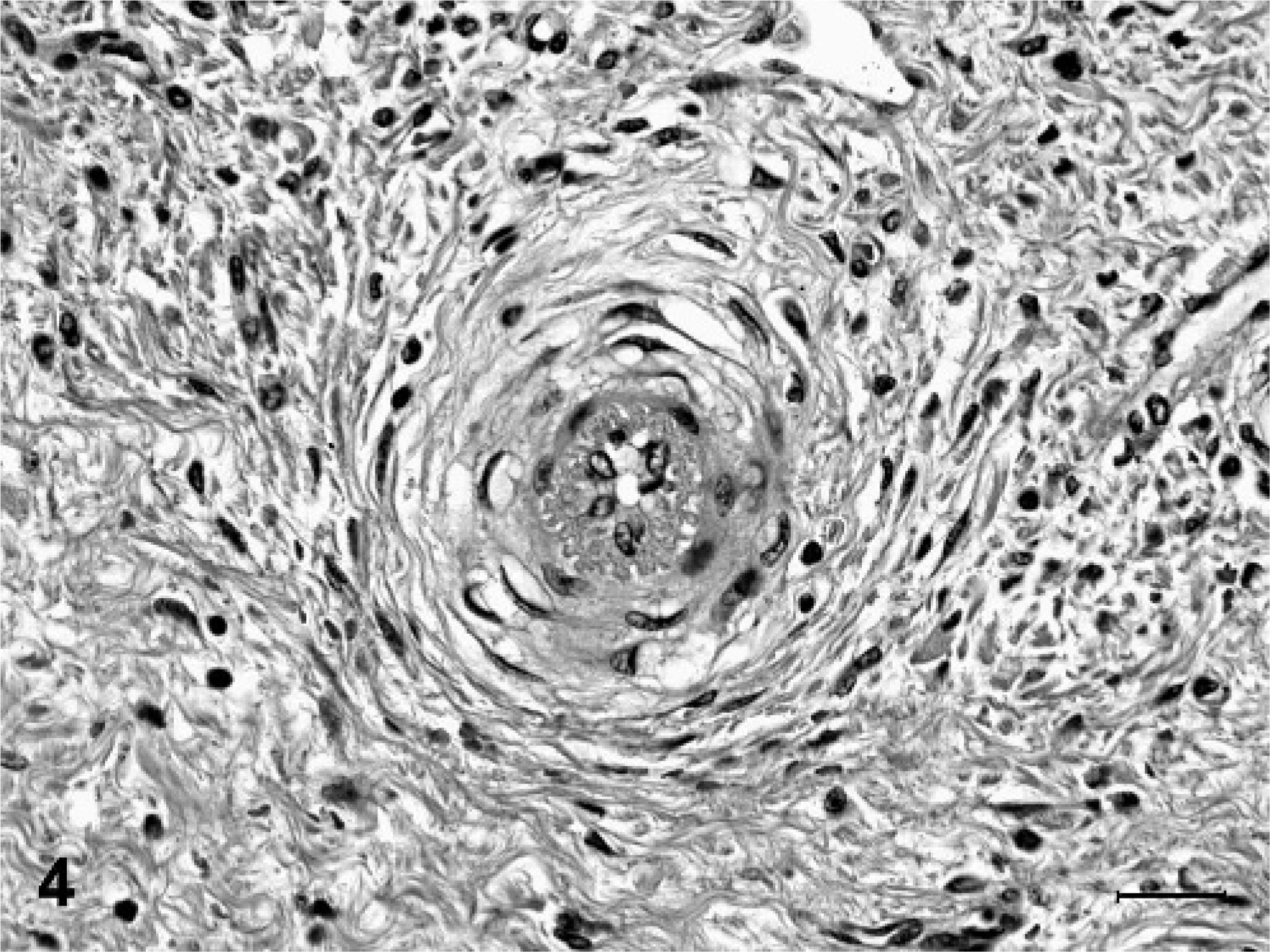
Kidney, arteriole; pig No. 7. The vessel wall has a concentric laminar pattern associated with medial thickening, vacuolation of smooth muscle cells, and increased size of endothelial cells. HE. Bar = 25 µm.
Less consistent findings in arteries and arterioles included focal to diffuse necrosis of the tunica media, thrombosis, and vascular or perivascular inflammation limited to small arteries and arterioles. Arteritis or periarteritis was seen in 4 out of 9 pigs (Nos. 1, 2, 5, and 9), and was characterized by mild to moderate accumulation of lymphocytes and macrophages, with fewer neutrophils and eosinophils (Fig. 5). Vascular inflammation sometimes occurred concurrently with necrosis of the tunica media (Fig. 6). Small intracellular or extracellular deposits of hemosiderin pigment were often admixed with perivascular inflammatory cells. Inflammatory vascular lesions were not limited to blood vessels with degenerative changes, and were not present in all examined tissues. Additionally, inflammation was not consistently observed in vessels with degenerative changes. Immunohistochemical tests for PRRSV and PCV-2 were performed on sections of kidney from each of the 9 minipigs. No antigen to either virus was detected in any of the minipigs, whereas strong immunoreactivity was observed in the positive control tissues.
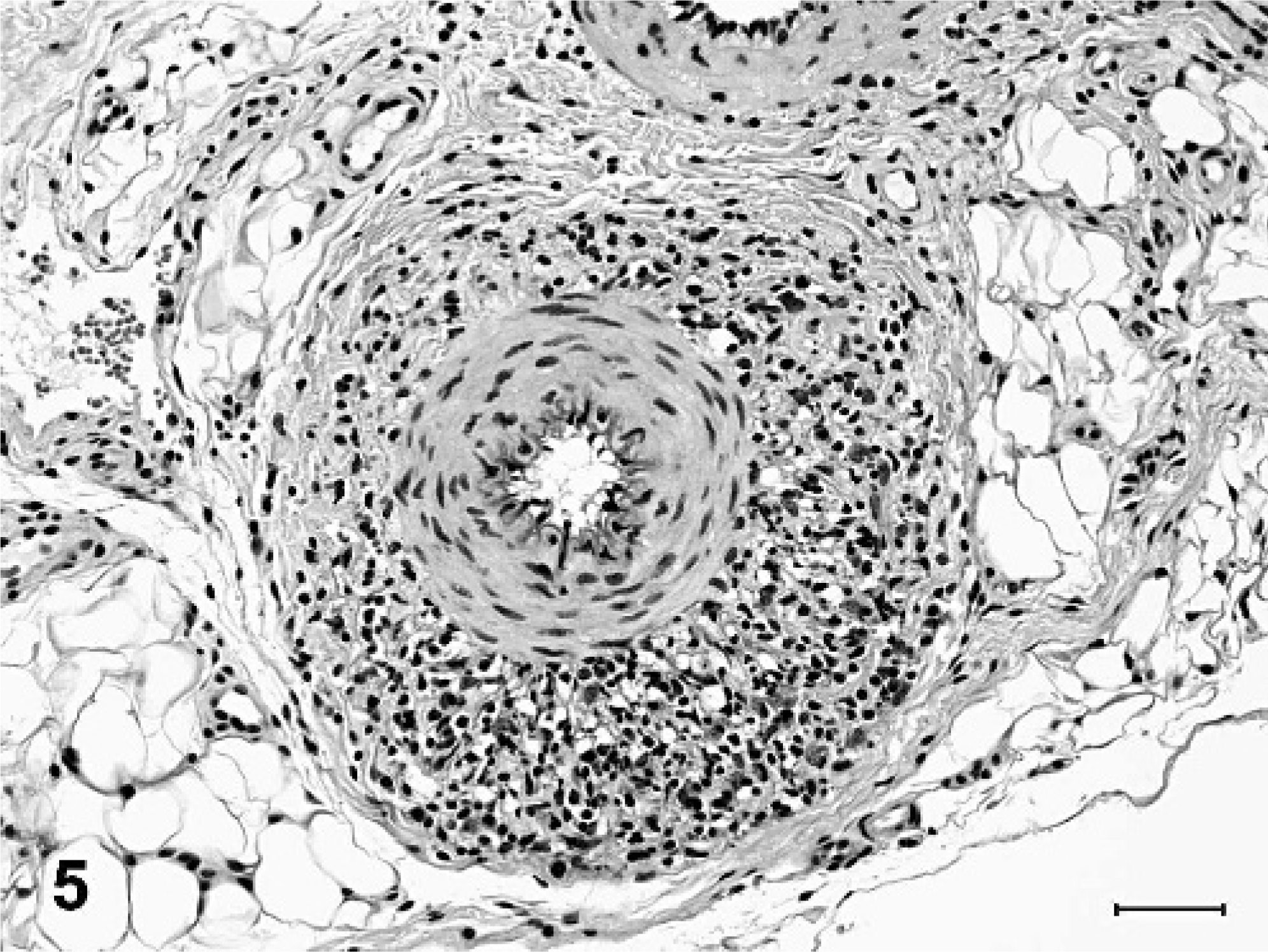
Omentum, small artery; pig No. 9. The adventitia is infiltrated with many lymphocytes, macrophages, and fewer eosinophils. HE. Bar = 50 µm.
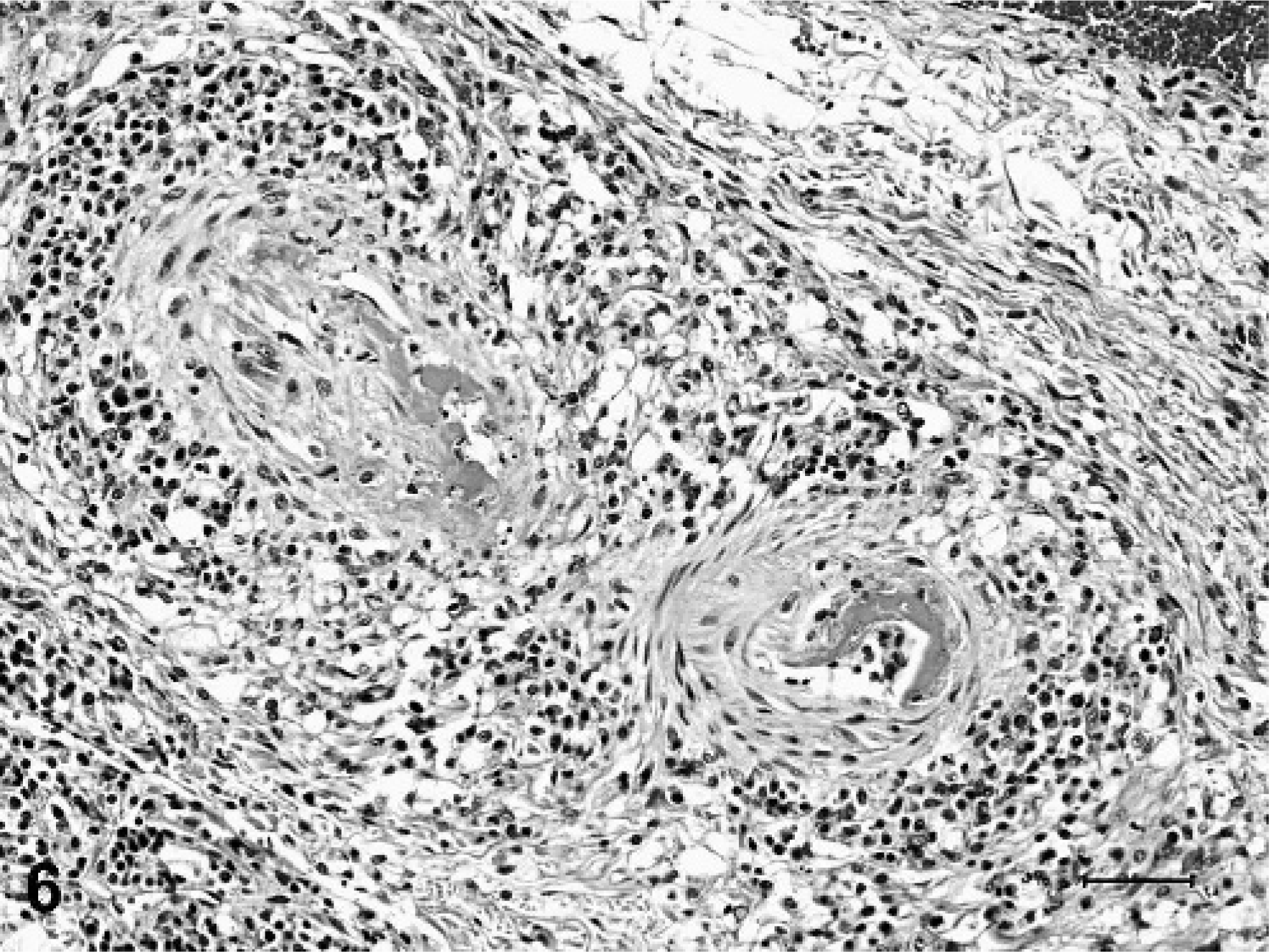
Kidney, arterioles; pig No. 5. The vessel walls are segmentally necrotic and replaced by hyaline eosinophlic material characteristic of fibrinoid change. Inflammatory cells infiltrate and encircle both arterioles. HE. Bar = 50 µm.
In the kidneys, vascular lesions were most often located in the pelvis (9 out of 9 pigs), and less frequently in the medulla. Arcuate arteries and cortical arterioles were affected in only 1 out of 9 pigs (No. 1) with renal lesions. This pig also had significant renal parenchymal atrophy, with striking loss of glomeruli and tubules in the cortex and medulla. Abundant fibrous connective tissue separating remaining glomeruli and tubules was demonstrated with Masson's trichrome stain. Many glomeruli were segmentally or globally atrophied, and most severely affected glomerular tufts were obliterated by acellular, eosinophilic, PAS-positive material (hyalinosis). Other glomeruli had features of membranoproliferative glomerulonephritis (MPGN), including thickened basement membranes and proliferation of mesangial cells with increased amounts of mesangial matrix. MPGN was also observed in kidneys of 2 other pigs (Nos. 8 and 9). In each of the 3 pigs with MPGN (Nos. 1, 8, and 9), variable numbers of renal tubules were distended with intraluminal protein casts. Pig No. 8 also had moderate, multifocal interstitial and periglomerular fibrosis.
Cardiac vascular lesions involving the epicardial and intramural coronary arteries were present in 4 pigs (Nos. 1, 5, 8, and 9). Small foci of myocardial coagulation necrosis, sometimes with myofiber mineralization and neovascularization were also observed in all 4 pigs. Two of the pigs (Nos. 1 and 5) also had chronic passive congestion of the liver, with loss of centrilobular hepatocytes.
Other tissues with vascular lesions similar to those described above included the urinary bladder (4 out of 7), skin (3 out of 6), gastrointestinal tract (3 out of 8), spleen (3 out of 8), lung (2 out of 9), lymph node (2 out of 8), omentum (2 out of 6), thymus (1 out of 2), mesentery (1 out of 2), ovary (1 out of 1), and prostate gland (1 out of 1). Arteriolar and venous thrombosis with hemorrhage and necrosis of adjacent tissue was sometimes seen in the skin, urinary bladder, and gastrointestinal tract of affected pigs. Two pigs (Nos. 2 and 9) had widespread hyaline thrombi in glomerular and alveolar capillaries, suggestive of disseminated intravascular coagulation (DIC) or other consumptive coagulopathy. Aside from the changes described above, capillary lesions were not a prominent finding in the other pigs.
Discussion
Severe thrombocytopenia and anemia were consistent findings in pigs with reported history of thrombocytopenic purpura syndrome. In all 7 pigs with terminal thrombocytopenia, platelet counts were low enough to cause spontaneous hemorrhage. Disseminated hemorrhage, typically involving the skin and various other visceral locations, was the most commonly reported macroscopic lesion. Microscopically, hemorrhages were usually interstitial and surrounded small arteries and arterioles, suggesting that some hemorrhages were directly attributable to vascular damage. Macrocytosis in 3 out of 8 pigs and microscopic demonstration of EMH in 6 out of 8 pigs with anemia and thrombocytopenia is suggestive of regenerative anemia, and possibly a longer course of illness. EMH is considered a normal microscopic finding in very young pigs. The ages of pigs with EMH in this study ranged from 7 weeks to 1 year, well beyond the age of normal EMH. Thus, EMH is considered to be a significant finding indicating at least a partial compensatory response to the severe anemia and thrombocytopenia.
Degenerative and proliferative vascular lesions were present to varying degrees in small- to medium-sized muscular arteries and/or arterioles in 9 out of 9 pigs with a reported history of thrombocytopenic purpura syndrome. Microscopic vascular lesions were consistently observed in small arteries and arterioles of the renal pelvis, but were also present in blood vessels of other organs, most notably the epicardial and intramural coronary arteries. The earliest vascular lesions observed in arterioles of the renal pelvis were endothelial cell hypertrophy, smooth muscle cell hyperplasia, mild medial thickening, and subtle onion-skin patterning of the vessel wall. Chronically affected arterioles had prominent concentric laminar thickening of the vessel wall, deposits of myxoid matrix, and partial to complete luminal stenosis. Microscopic lesions in muscular arteries included asymmetric thickening of vessel walls, neointimal proliferation, medial thickening, disruption and fragmentation of the internal elastic lamina, and deposition of myxoid matrix. Acute necrosis of the tunica media was sometimes superimposed upon these more chronic changes.
Ischemic lesions secondary to vascular damage occurred in the heart and kidneys of several pigs. All 4 pigs with coronary vessel lesions had myocardial microinfarcts characterized by coagulation necrosis, sometimes with myofiber mineralization and formation of granulation tissue. Renal interstitial fibrosis was seen in 2 pigs. The pig with the most severe and widespread vascular lesions (No. 1) had severe renal interstitial fibrosis with tubular atrophy and hyaline obliteration of glomeruli characteristic of malignant hypertension and end-stage glomerular disease.
It is unclear whether vascular degeneration precedes vascular inflammation, or alternatively, whether degenerative lesions represent sequelae of vasculitis. Vascular lesions in 4 out of 9 pigs were associated with vascular or perivascular inflammation and necrosis of the tunica media. The majority of inflammatory cells were lymphocytes and macrophages. Additionally, presence of hemosiderin pigment within and around damaged blood vessels is consistent with prior episodes of hemorrhage. Previous episodes of clinical or subclinical hemorrhage could have coincided with initial vascular injury and/or inflammation.
To date, there are no published reports of degenerative vascular lesions with morphologic features of arteriosclerosis in the Göttingen minipig. Two previous reports of spontaneous lesions in clinically healthy Göttingen minipigs described focal necrotizing vasculitis in various organs, including the stomach, kidney, lung, heart, and spleen, in approximately 5% of the examined pigs. 11, 21 Because case submissions were used in this retrospective study, control tissues from age-matched, clinically normal Göttingen minipigs were not available for examination. However, in the experience of 2 authors (K. A. Maratea and G. W. Stevenson), comparable vascular lesions have not been observed in tissues from Göttingen minipigs submitted for reasons other than thrombocytopenic purpura.
A syndrome of immune complex-associated thrombocytopenic purpura in sexually mature Göttingen minipigs with similar clinical features as the present series of cases has been previously described. 2 Necrotizing vasculitis was observed in several pigs; however, degenerative and/or proliferative vascular lesions with morphological features comparable to those described in the present study were not reported. The authors proposed an immune complex pathogenesis based on the immunohistochemical demonstration of IgM, IgG, and/or complement component C1q in glomeruli of some affected animals. However, these findings should be interpreted with caution, because both immunoglobulins and complement products may be nonspecifically entrapped within damaged vessel walls. Additional studies are necessary to determine whether immune complexes and complement products are present within the vascular lesions and glomeruli, or in the circulation. The current series of cases also suggests that this syndrome is not restricted to sexually mature animals, because 2 of the pigs were 7–8 weeks of age.
The distribution and morphology of vascular lesions observed in this series of Göttingen minipigs are distinct from naturally occurring degenerative and inflammatory vascular diseases of conventional swine. Aged swine can develop spontaneous atherosclerosis, which is morphologically characterized by eccentric intimal thickening and lipid accumulation that primarily affects elastic arteries and medium- to large-sized muscular arteries, including the coronary arteries. 14, 24 Lipid accumulation was not a feature of the coronary arterial lesions observed in this series of cases.
Inflammatory diseases of swine associated with arteritis include septicemic salmonellosis and erysipelas, classical swine fever (CSF), porcine reproductive and respiratory syndrome (PRRS), and porcine dermatitis and nephropathy syndrome (PDNS). 3, 14, 16, 17, 18, 22, 24 Most of these diseases cause systemic vasculitis characterized by acute necrosis of the tunica media in arteries and small arterioles, resulting in thrombosis and infarcts in the skin, kidneys, spleen, and other organs, in contrast to the degenerative vascular lesions that predominated in the Göttingen minipigs.
Severe arterial and arteriolar lesions with endothelial cell swelling, lymphohistiocytic inflammation, and necrosis of the tunica media that are localized to the renal pelvis and medulla have been described in pigs experimentally infected with PRRS virus. 4 PRRSV antigen has been immunohistochemically demonstrated within the vascular lesions of pigs with both natural and experimental PRRSV infections. 4, 18 The minipigs in this series of cases were barrier-bred in SPF facilities, and undergo biannual tests for a wide range of swine pathogens, including those listed above, based on FELASA recommendations. 5 Per the submitting veterinarian, the herd is negative for the above-mentioned pathogens, including PRRSV and PCV-2. Additionally, no immunoreactivity to either PRRSV or PCV-2 was detected immunohistochemically in sections of kidney from any of the nine minipigs.
In humans, the onion-skin pattern of arterial and arteriolar thickening observed in 3 out of 9 pigs is most often associated with primary or secondary malignant hypertension and thrombotic microangiopathic diseases. 6, 19, 20 Ultrastructurally, the hallmark onion-skin lesions are composed of concentric layers of smooth muscle cells and thickened, reduplicated basement membranes. 20 Progressive luminal occlusion, fibrinoid change, and acute necrosis are common sequelae, especially in the kidneys. 6 Malignant hypertension, defined as a rapid, severe elevation of blood pressure, generally with diastolic pressures greater than 110–130 mm Hg, is most often superimposed on preexisting, secondary forms of hypertension such as chronic renal disease, endocrine disorders, and vascular diseases. 6, 13, 20 Blood pressure was not reported in the current series of cases.
Miniature pig breeds developed for use in laboratory settings are widely used in biomedical research. Swine are a desirable large animal model of cardiovascular disease because the anatomy of the coronary vasculature and collateral circulation in swine is similar to that of humans, the heart weight ° body weight ratio is similar to humans, and swine have similar dietary habits and lipoprotein structures as humans. 7, 24 The Göttingen minipig has been used as an experimental model of atherosclerosis, coronary artery disease, diabetes mellitus, and metabolic syndrome, as well as in drug safety trials. 8, 9, 10, 12, 15, 23 The presence of vascular disease in 9 Göttingen minipigs with thrombocytopenic purpura syndrome is significant, because undefined vascular disease in these animals is likely to complicate the diagnosis of experimentally-induced vascular changes. Further studies are necessary to identify possible etiologic agents, clarify the association of vascular lesions with thrombocytopenic purpura syndrome, and to determine a possible relationship with similar human diseases.
Footnotes
Acknowledgements
We thank Dr. Ingeborg M. Langohr for critical review of the manuscript and Dr. John F. Van Vleet for helpful suggestions.
