Abstract

The tuberculous granuloma is an evolutionarily conserved structure used by a wide range of species to control mycobacterial infections. It is likely that the early stage interactions between mycobacteria and host cells within this structure determine the long-term outcome of infection, potentially influencing whether or not the host develops clinical signs, becomes infectious, or responds to diagnostic tests. In this issue of Veterinary Pathology, Palmer et al 9 publish the results of important and timely research into the early stages of granuloma development in cattle experimentally infected with Mycobacterium bovis. This study replicates natural infection in deploying aerosol-generated infective droplets and uses detailed immunohistochemical and in situ hybridization techniques to probe the content of granulomas 15 and 30 days postinfection. In focusing on this early phase of infection, this research further elucidates the relative roles played by innate and acquired immunity to mycobacterial challenge and importantly highlights the link between the development of acquired immunity and necrosis within granulomas. This event favors the release of mycobacteria into the extracellular milieu and potentially their onward transmission through airways.
The authors describe how the presence of neutrophils is coincident with the development of necrosis within granulomas. Evidence suggests neutrophils play a somewhat nuanced role in the development of tuberculosis (TB) in humans and other species providing both innate protection during the very earliest phases of infection but also contributing to immunopathology in more longstanding disease. 3,12,13 Neutrophils are abundant in advanced cavitated lesions in human patients and also represent the predominant infected phagocyte in the airways in active human pulmonary TB. 6 Studies in neutrophil-depleted murine models suggest that these granulocytes contribute to tissue injury, and that a neutrophil-driven, interferon-inducible, whole-blood transcript “signature” in patients with active TB correlates with disease severity. 1,5,6,11 Alternatively, neutrophil influxes within a necrotic/cavitated lesion may reflect a nonspecific response to the necrosis rather than to extracellular mycobacteria per se. It would be anticipated that such an unchecked response would inevitably be deleterious to the host, possibly reflecting loss of control of infection or an inadequate compensatory response to reduced macrophage activity.
The heterogeneous and apparently autonomous nature of tuberculous granulomata within diseased human patients or cattle is well documented and may reflect their topographical pulmonary location. 4,7 Historical meticulous work by Medlar charting the distribution of tuberculous granulomas of varying morphology 8 in cattle found that potentially more infectious liquefactive, cavitating lesions were over-represented in subpleural dorsocaudal pulmonary regions (zone A1–2, Fig. 1). This parallels the finding of similar cavitating liquefied lesions in the dorsally located apical lung regions of human patients with postprimary tuberculosis. This begs the question why such dorsal locations favor this form of lesion development. For cattle, as distinct from humans, the dorsocaudal portion of the lung remains most elevated irrespective of whether the animal is standing or lying. In this pulmonary region, breathing amplitude and lymphatic drainage are reduced relative to lower locations, potentially facilitating the accumulation of inhaled infectious droplets. These droplets may have been inhaled from the exterior or have been generated from preexisting lesions within the animal’s lungs (ie, endogenous reinfection). 2 In this context, it has been demonstrated that Mycobacterium tuberculosis participates in the generation of aerosols within an infected lung through chemical interactions between mycobacterial cell wall mycolic acids, cord factor, and macrophage lipids. 10 The highly bioactive lipid compounds that result from this interaction facilitate aerosol production when macrophages lyse within airways and their released content is subject to the ongoing shear forces exerted by inspired and expired air. Cavitating, liquefied lesions may then develop when such aerosols deliver mycobacteria to “favorable” pulmonary locations. More elevated lung regions may also be subject to more limited immunological surveillance due to the reduced connectivity to lymph nodes. In addition to the potential effect of this factor on granuloma development, it may also affect the development of the immunological responses on which diagnostic tests are reliant. A further impact of more dorsally located pulmonary lesions is the increased mechanical stress on interlobular septa at this location given the lung has to support its own weight. This tension within septa would likely interfere with their participation in the encapsulation required to restrain lesion expansion.
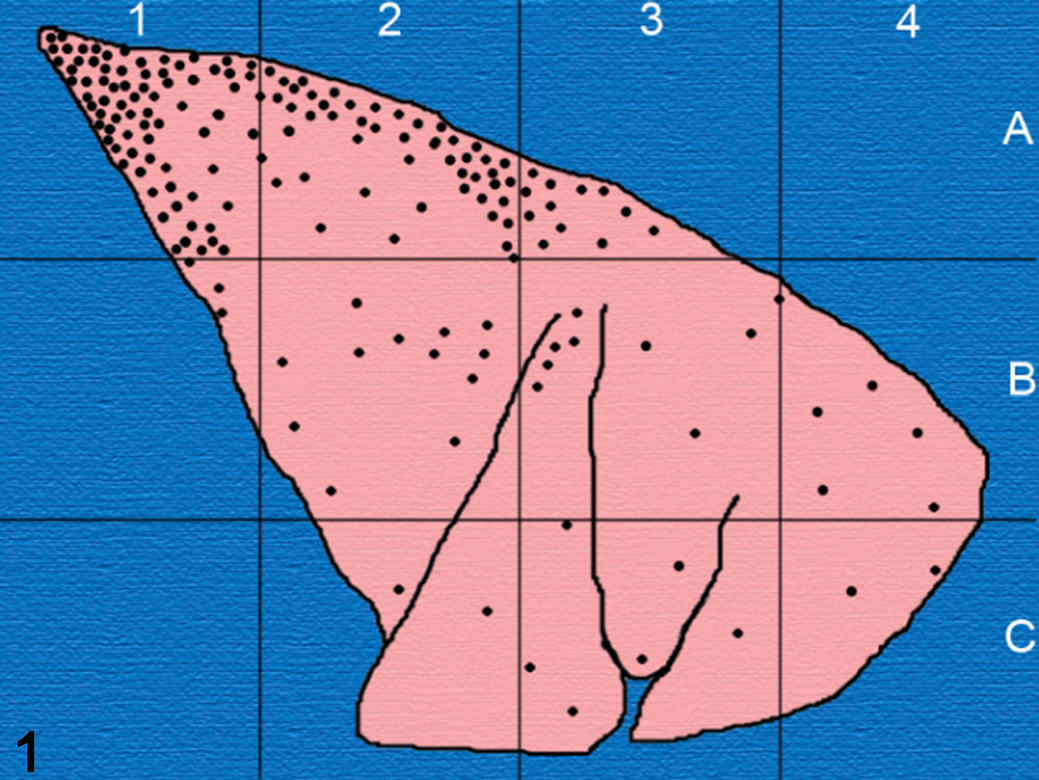
Distribution of cavitating tuberculous lesions in dorsocaudal pulmonary regions (zone A1–2) of cattle examined following abattoir slaughter. Adapted from Medlar. 8
It is only through detailed morphological and immunohistochemical analysis of tuberculous lesions in addition to charting their pulmonary and lymph nodal distribution that questions relating to their ultimate fate, and that of the host, will be answered. Pathology-based “animal-level” investigations such as those carried out by Palmer et al 9 are relatively infrequently employed in the context of bovine TB when compared to epidemiological and disease modeling studies focusing on herd-level factors. It is important that the animal-level element of disease investigation is not overlooked and that pathologists bring their insights and research methodologies to bear in attempting to explain features such as why only some animals within an exposed group develop disease, why there are variations in animal infectivity, and in the responses of infected cattle to diagnostic tests.
Footnotes
Acknowledgement
The author wishes to thank Mr. Brian Cloak for preparing the figure.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
