Abstract
Bovine tuberculosis due to Mycobacterium bovis is a zoonotic disease classically carried by cattle and spilling over into humans primarily by the ingestion of milk. However, in recent decades, there have been many endemic geographic localities where M. bovis has been detected infecting wildlife reservoirs, limiting the progress toward eradication of this disease from cattle. These include cervids in North America, badgers in Great Britain, feral pigs in Europe, brushtailed possums in New Zealand, and buffalo in South Africa. An overview of these wildlife hosts will provide insight into how these reservoirs maintain and spread the disease. In addition, the authors summarize the pathology, current ongoing methods for surveillance, and control. In many instances, it has proven to be more difficult to control or eradicate bovine tuberculosis in wild free-ranging species than in domesticated cattle. Furthermore, human influences have often contributed to the introduction and/or maintenance of the disease in wildlife species. Finally, some emerging themes regarding bovine tuberculosis establishment in wildlife hosts, as well as conclusions regarding management practices to assist in bovine tuberculosis control and eradication in wildlife, are offered.
Bovine tuberculosis (BTB) due to Mycobacterium bovis is a zoonotic disease classically thought of as a disease primarily of cattle. It has one of the widest host ranges of all the M. tuberculosis complex organisms and can readily spill over into humans or a variety of domestic and wild animals. In the past, control of M. bovis was directed at controlling the spread of disease within domestic cattle by whole-herd annual skin testing, slaughter surveillance, and monitoring the movement of animals between herds. In addition, the pasteurization of milk is an important element in preventing spread of the disease to humans. In recent decades, our recognition of disease transmission at the interface between humans, domestic animals, and wildlife has increased. Once a disease becomes endemic in a wildlife population, disease control becomes a much more challenging issue.
Several examples demonstrate critical issues associated with wildlife reservoirs of BTB across the world, involving different host species in different ecological and environmental conditions. These include cervids in North America, mustelids and swine in Europe, marsupials in New Zealand, and bovids in Africa. In these instances, it has been assumed that livestock served as the initial source of infection for different wildlife species, which were able to maintain the disease in their free-ranging populations until contact between livestock and infected wildlife allowed the disease to be transmitted back to the livestock. Table 1 provides a summary overview of the following wildlife reservoir examples. The differences in the specifics of these interspecies outbreaks and issues associated with the control of these outbreaks in wildlife species provide insight into the maintenance, transmission, and control of this important zoonotic pathogen.
Summary of Key Features of Bovine Tuberculosis in Wildlife Reservoirs.
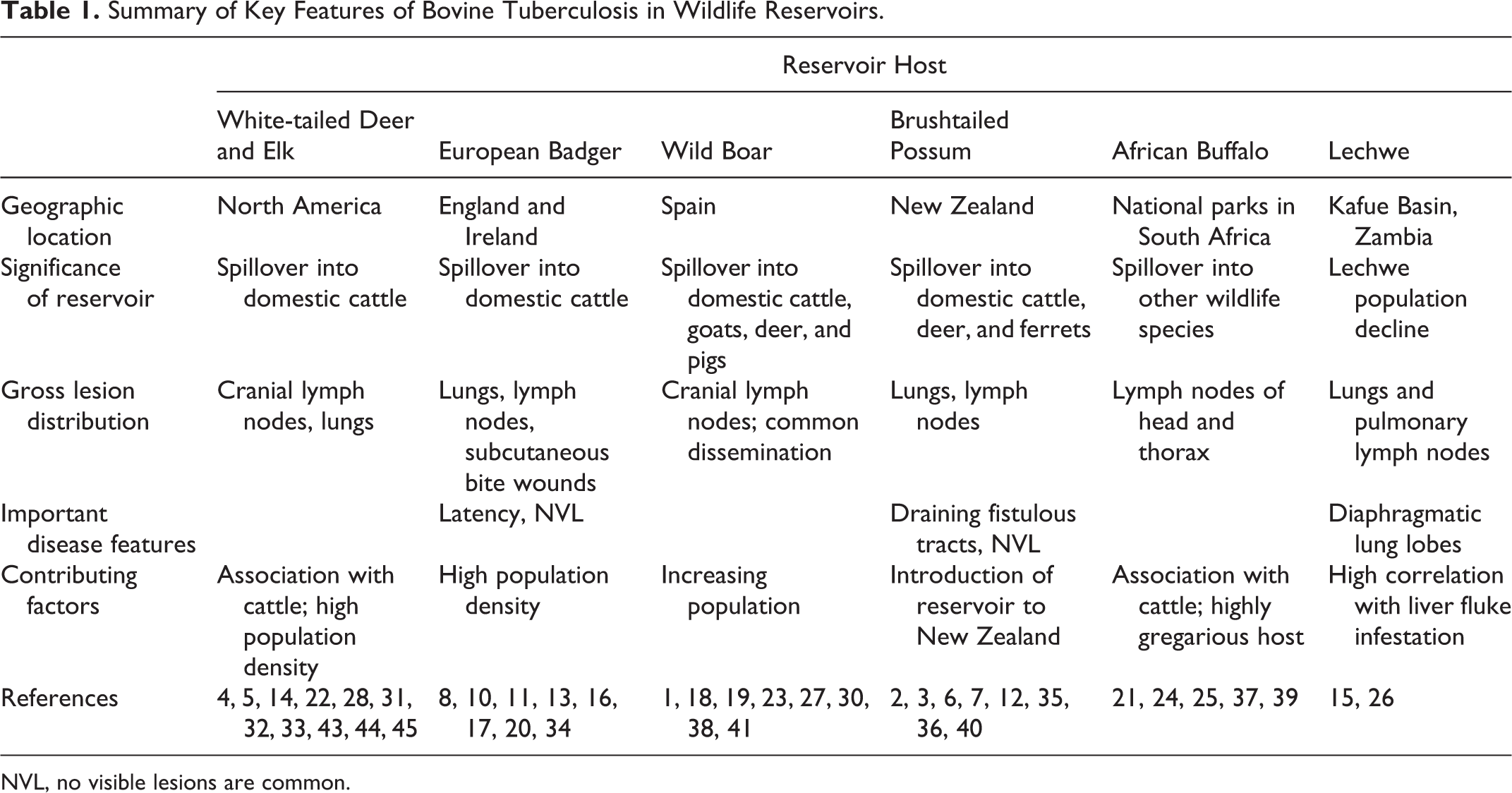
NVL, no visible lesions are common.
North American Examples
History and Reservoir Hosts
Known wildlife reservoirs of M. bovis in North America include white-tailed deer (Odocoileus virginianus) in both Michigan and Minnesota, United States, and both elk (Cervus canadensis) and white-tailed deer in Riding National Park, Manitoba, Canada.
White-tailed deer host in Michigan, USA
White-tailed deer are a cervid species, which are herbivores eating grass, weeds, shrubs, twigs, nuts, and lichen. They congregate in small family groups with a dominant adult female (doe), a yearling daughter, and that season’s fawns; several additional females may be associated as well. Mature males (bucks) generally occur as isolated individuals or in small groups, primarily interacting with does during breeding season. Only the males develop antlers. They are highly variable in size, ranging from a 25- to 215-kg adult weight. Deer are most active at dawn and dusk. 30 Starting in 1994, with the discovery of a single M. bovis lesioned white-tailed deer carcass, endemic tuberculosis has been found in wild deer over a 7-county area that continues to spill back into domestic cattle 18 years later. 44
Although the overall prevalence of BTB in deer has been relatively low (<5%) over time, there are small geographic locations where the prevalence of BTB is significantly higher, forming hot spots of infection. 32 The core area of endemic infection identified by the Michigan Department of Natural Resources (MDNR), designated Deer Management Unit 452, encompasses the highest prevalence area of tuberculosis in the state and has contained over 95% of all positive deer found since 1995. 34 This area comprises primarily deciduous forests, coniferous swamps, and rolling fields, which are all habitats frequented by white-tailed deer. 33 This area consists of approximately 1500 km2 of both privately owned and public lands. Much of the private lands in this area have been used as hunt clubs, which restrict public hunting and allow for the private landowners to lower hunting pressure on the deer. Frequently, these hunt clubs also practice various types of land management or supplemental feeding practices designed to increase deer numbers, help deer survive over winter, and promote the growth of larger deer for hunting purposes. 33,44 Supplemental feeds vary from widely broadcast corn or grains to huge piles of pumpkins, sugar beets, carrots, or grain. All of these activities have resulted in higher than normal biological carrying capacity on these properties, resulting in high deer population densities. It is now believed that these high densities, restricted hunting, and close congregation of deer at supplemental feeding sites have played an important role in increasing BTB prevalence in the wild deer population. 5,33
Michigan has a relatively small herd of free-ranging elk (800–1200 individuals) whose range overlaps with the endemic deer zone, and small numbers of M. bovis–infected elk have been detected through hunter submissions (less than 0.3% prevalence). 33 While elk or wapiti (Cervus elaphus), like deer, are cervids and herbivores; they have not been shown to be a maintenance host in Michigan but rather appear to serve as occasional spillover hosts with limited BTB lesions. 33
Deer were believed to be originally infected with BTB through exposure to domestic cattle that had high prevalence of this disease throughout the state during the years prior to World War II. Beef cattle are predominant in this region of Michigan and tend to be less intensively raised than dairy cattle. This has led to more interaction through direct and indirect cattle-deer contact on pastures in the endemic area.
Pathology
Typical gross pathology consists of multiple caseogranulomas with purulent centers in affected lymph nodes or lungs (Figs. 1, 2); these often grossly resemble abscesses since they frequently possess a soft caseous core. The medial retropharyngeal lymph nodes are the most frequent cranial lymph nodes to be infected in deer. 14 Approximately 30% to 35% of M. bovis–infected deer will exhibit spread to the thoracic viscera. Thoracic lesions commonly consist of multiple pulmonary granulomas widely disseminated throughout the lungs and often extending to both the visceral and parietal pleural surfaces. True dissemination of BTB lesions into multiple body cavities and numerous tissues has been only rarely encountered and generally occurs in juvenile fawns less than 6 months of age. The Diagnostic Center for Population and Animal Health laboratory was one of the first to conduct major wild deer surveillance for tuberculosis in the United States and so developed the methods and published the initial findings; 14 these methods have since been accepted and used in many different states and Canadian provinces for similar surveillance programs in wild cervids.
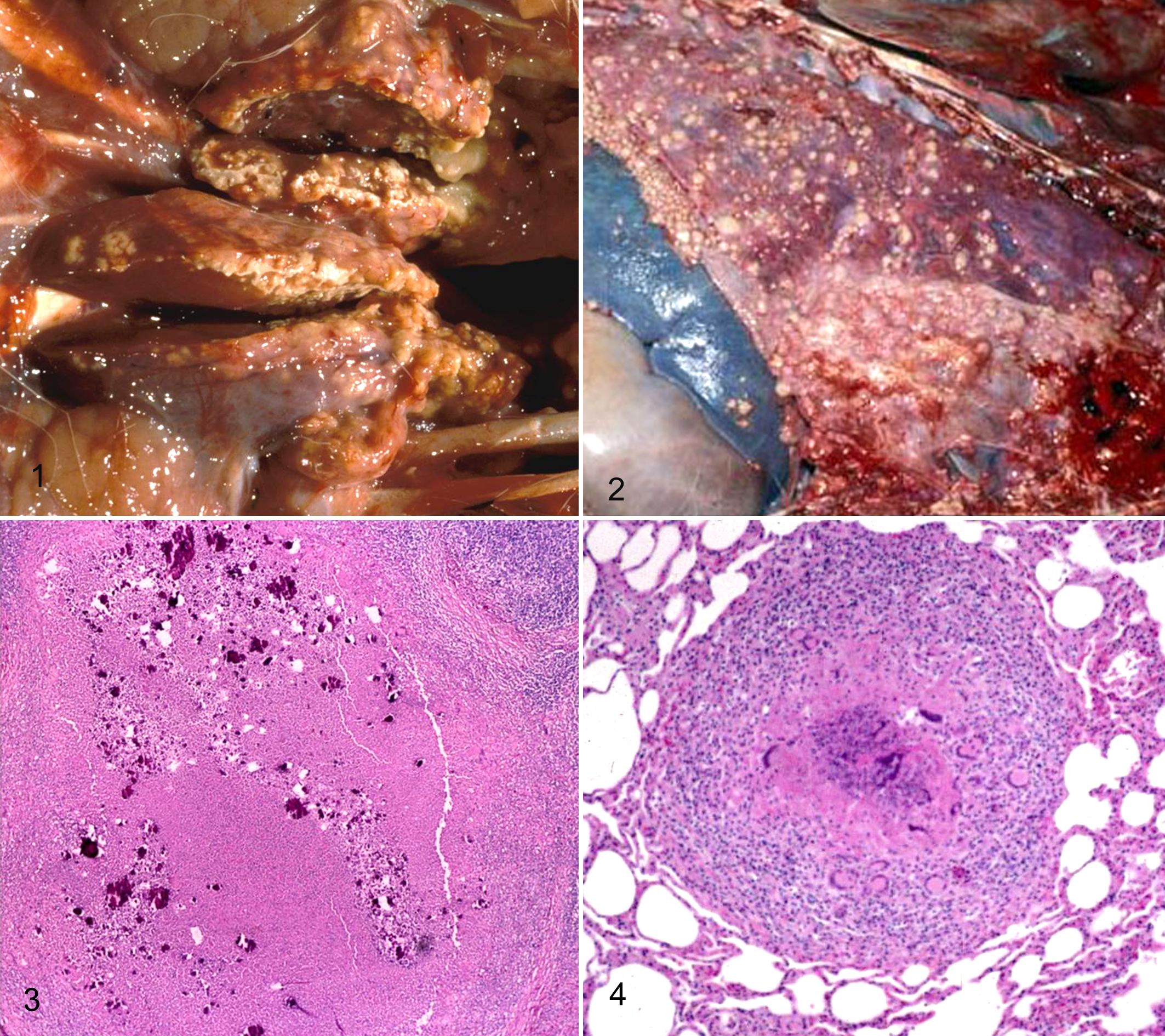
Histologically, both the lymph node and pulmonary lesions are true caseogranulomas with an outer layer of lymphocytes, histiocytes, and multinucleated giant cells, surrounding a central necrotic core that often contains large foci of partial mineralization (Figs. 3, 4). 14 Neutrophilic infiltrates are not a common characteristic of the inflammatory reaction with bovine tuberculosis in wild deer in Michigan. Radiating club-like Splendore-Hoeppli material has never been associated with a single positive case of M. bovis infection in Michigan deer; this is typically associated with other bacteria, including Actinomyces, Nocardia, or Staphylococcus species and various fungal infections. The presence of either suppurative inflammation or Splendore-Hoeppli material is an indicator of a lesion not being associated with M. bovis infection. 14 Acid-fast bacilli range from rare to uncommon and are most commonly present within the cytoplasm of histiocytes or multinucleated giant cells; however, sometimes small groups of these bacilli are encountered free within the central necrotic debris. 14
Surveillance
Surveillance testing has been based on voluntary contributions of deer and elk heads or whole carcasses by hunters, so it is primarily a passive surveillance program. The MDNR collects hunter-harvested deer and elk heads at roadside check stations, based on predetermined quotas established for each county based on tuberculosis (TB) prevalence, numbers of deer, and hunter willingness to cooperate. Deer head submission is voluntary, whereas elk head submission is mandatory; there is no charge to individuals who submit their animal remains for tuberculosis examination. Part of the target quotas established are based on a 95% confidence of detecting tuberculosis at a level of 0.2% prevalence. 44 Heads collected at check stations are then transported to the Diagnostic Center for Population & Animal Health, Lansing, Michigan, for testing. Testing includes examination of the cranial lymph nodes, which are serially sectioned, and those lymph nodes with lesions are collected for routine histopathology, acid-fast staining, and mycobacterial culture. 14 Through the end of 2011, approximately 195 000 deer had been processed through the surveillance program, and a total of 705 BTB-positive individual deer were identified (S. M. Schmitt, personal communication, 2012).
Although initially, significant progress was made in decreasing the overall BTB prevalence in deer and controlling annual spillover into cattle herds, there has been a plateau reached in prevalence of just below 2% over the past 9 years. 34 Simply applying increased hunting pressure and outlawing supplemental feeding and baiting have not been effective in accomplishing further reductions in disease prevalence.
Control methods
A multiagency task force was created to set management and policy guidelines based on the best available science, educate the public, perform research to resolve questions that lacked sufficient information, and work toward the common goal of bovine tuberculosis eradication in the state of Michigan. 44 Methods of control currently used in Michigan include increased hunting pressure to reduce the total deer numbers to a level approximately half of what was previously present in Deer Management Unit (DMU) 452. 44 This has resulted in lower deer concentrations, thereby reducing deer-to-deer lateral transmission of bovine tuberculosis. Methods to increase hunting pressure have included adding additional early or late hunting seasons; encouraging antlerless (female) deer harvest, which reduces the reproductive potential of the population; and giving virtually unlimited year-round hunting permits to local landowners. 33 Deer density in the endemic BTB area began at an estimated 20 deer per km2 in 1995, was reduced to a low of 10 deer per km2 in 2004, and subsequently increased to 14 deer per km2 in 2008. 34
A second major measure has been the outlawing of baiting and supplemental feeding of deer, practices that result in unnatural concentrations of large numbers of deer and encourage disease spread by lateral transmission. A third method was to encourage farmers in the endemic area to improve biosecurity around their cattle facilities, to prevent direct cattle-to-deer contact or to fence off feed storage and feeding areas from deer to prevent contamination of feeds with M. bovis. 33
However, despite significant time and resources directed toward bovine tuberculosis control and eradication from Michigan’s deer, progress has slowed based on the annual deer BTB prevalence data having leveled at around 2%. In Michigan, deer are a highly valued resource to hunters and landowners alike, and massive culling or depopulation of deer is simply not publicly acceptable. 34 In addition, recent economic recession has resulted in reduced funds being available to supply resources for disease surveillance, research, and paying for compliance personnel to enforce feeding bans. 5,34
White-tailed deer host in Minnesota, USA
Bovine tuberculosis infection in white-tailed deer in Minnesota was identified after detection of the disease in a cattle herd decades after the state had been declared BTB free in 1971. The first confirmed tuberculosis-infected cattle herd was detected in July 2005, from Roseau County in extreme northern Minnesota. 4 This area of the state is rural, with limited numbers of predominantly small- to medium-sized cattle herds and agricultural crop farming, as well as suitable natural habitat for wild white-tailed deer. A bovine tuberculosis management zone of approximately 1500 km2 and a smaller bovine TB core area of 425 km2 were subsequently established to help manage the disease outbreak. 4
Pathology
Detailed pathology for the gross and histologic lesions of the Minnesota deer has not been published; however, the lesions should be very similar to those described in Michigan deer.
Surveillance
The initial BTB-positive beef cattle herd had a 1.2% BTB prevalence and was depopulated. 4 Four hundred seventy-four hunter-harvested wild deer from within a 25-km radius of the initial positive cattle farm were sampled by the Minnesota Department of Natural Resources, and 1 BTB-positive deer was identified. 4 Surveillance was initially instituted at the statewide level to determine the extent of the BTB infection in cattle. In 2006, 1554 cattle herds throughout the state and no positive herds outside of the local area were identified; however, 2 additional positive cattle farms were found locally in northwest Minnesota. 4 Deer surveillance for BTB in 2006 consisted of 4000 individuals sampled, with 5 additional individuals positive. The location of both positive cattle herds and the wild deer was used to develop the bovine tuberculosis management zone and inner core areas to assist in focusing disease management efforts. Subsequent testing of cattle and wild deer in the management zone led to the purchase and depopulation of 12 cattle farms, and 27 total wild white-tailed deer were found to be BTB infected over a 3-year period. No additional cattle herds or wild deer were found to be positive during surveillance conducted from 2009 through 2011. 4
Control methods
Extensive supplemental feeding and baiting of deer for hunting purposes, which has been associated with the maintenance and spread of BTB in Michigan deer, was not part of the longtime culture of deer hunters in Minnesota, having been previously outlawed throughout the state since 1991. 5 However, recreational feeding of deer was banned in northwest Minnesota in November 2006. 5 In an attempt to decrease deer density and thus reduce lateral transmission of BTB in the bovine TB core area, both aerial and ground sharpshooting of deer was conducted during the winters of 2007 through 2010. This removed approximately 2600 deer from the core area, including 14-BTB positive individuals. 5 In addition, expanded deer hunting in the bovine TB management area, through the addition of early and late season hunts, and increased bonus permits were issued. Landowners in the area were provided with deer harvest permits, which allowed for shooting of deer outside the normal hunting seasons by local residents. Wild deer density in the affected region was reduced by approximately 55% through the combination of increased public hunting and use of sharpshooting. 4
In addition to measures directed toward reduced deer density and congregation due to feeding, a voluntary cattle buyout program for cattle farms located within the bovine TB management area was initiated in 2007. This eventually resulted in the elimination of 46 cattle farms with the removal of 6200 cattle. 4 Farms that remained in the area were required to fence stored feed and winter cattle feeding areas to minimize contact with wild deer. This was aided by the state providing 90% cost share toward the total cost of the new fencing. 4 These measures provided for decreased total numbers of cattle within the area to become potentially infected with BTB, as well as increased biosecurity to prevent the transmission of disease from deer to cattle.
The rapid coordinated control measures by the Minnesota Departments of Agriculture and Natural Resources appear to have controlled the spread of the BTB infection. No new cases of infected cattle or deer have been detected since 2008, 4 and the state was reclassified as BTB Accredited Free by the US Department of Agriculture (USDA) in October 2011 (B. Hahn, personal communication, 2011). This particular case may be best summarized as preventing the establishment of a BTB wildlife reservoir from developing, rather than eradication of an established BTB wildlife infection. 4 In any event, Minnesota did a commendable job responding both quickly and decisively in eradicating bovine tuberculosis from its wild deer.
Elk and white-tailed deer in Riding Mountain National Park, Manitoba, Canada
Riding Mountain National Park in Manitoba, Canada, has had ongoing M. bovis in elk (C. elaphus) since the first detected case in 1992, with lesser numbers of white-tailed deer reported infected since 2001. 46 We have previously encountered the elk as a herbivore that grazes on grasses and lives in either forest or prairie habitats. Elk are notably larger in size than deer (ranging from 75–340 kg) and are more gregarious, forming herds of just a handful to dozens or hundreds of individual animals. Similar to deer, only male (bull) elk develop antlers. These herds tend to seasonally migrate to follow available forages. 30 The Riding Mountain National Park was created in 1930 and encompasses approximately 3000 km2 of boreal forest and prairie. 29 Portions of the park include public recreation areas and an Indian reservation (less than 3% of the total area), and the vast majority of the park is managed as wilderness (greater than 97% of the total area) with limited public access and no public hunting of deer or elk. The park is surrounded by agriculture on all sides, consisting of forage crops, grain farming, and extensive cattle production and grazing. 29 Cattle grazing actually extended into the park until it was banned in 1970. 23 The initial BTB infection began with infected cattle, which were allowed to graze within the park during the 1950s to 1960s, and has been maintained in multiple cattle herds that have bordered on the park since that time. 23
Pathology
A total of 41 culture-positive elk and 11 culture-positive deer have been described in surveillance conducted from 1991 through 2010. 45 Of the BTB-positive elk, 61.3% had gross lesions in the head, whereas 48.4% had gross lesions in the lungs. 45 The most common sites for gross lesions in BTB-positive elk included the tonsils, retropharyngeal lymph nodes, and the lungs. Grossly, these lesions varied from caseopurulent to granulomatous lesions. Histologically, lymph node lesions tended to be well encapsulated, either single or multifocal nodules, whereas lung lesions were more disseminated. 45 Gross lung lesions were 4 times more common in males (bulls) than in females (cows), but there was no significant lesion variation by sex for other sites such as lymph nodes. 45
Surveillance
Formal surveillance programs did not begin in the Riding Mountain area until 1997, although occasional BTB lesions have been found sporadically in elk since 1992. 45 The surveillance programs include hunter-killed passive surveillance using animals taken from outside the park, active surveillance based on antemortem blood testing conducted within the park from 2002 through 2012, ground-based active culling during several short periods, and targeted surveillance of opportunistically provided individuals (road kills, predation, winter kills). 45 Hunter-supplied samples generally have consisted of heads and/or lungs. The other forms of surveillance typically have provided whole carcasses for complete necropsy examination. These more complete necropsies have followed specific specimen collection protocols and included histopathology, Ziehl-Neelsen acid-fast staining, mycobacterial culture, and polymerase chain reaction (PCR) identification of isolates. 45
Passive hunter surveillance, conducted only on wild cervids that had foraged outside the park and could be legally hunted, has revealed a 0.4% disease prevalence in elk and 0.2% prevalence in deer. 29 Active surveillance has involved antemortem testing of wildlife, including some of the newer serologic-based assays, including multiantigen print immunoassay (MAPIA) and lateral flow test. 29,43 These active surveillance methods used within the park have revealed a combined prevalence of 3.6% in elk and deer. 29
Control methods
An Interagency Task Group for Bovine Tuberculosis was formed to establish surveillance eradication and management procedures directed at BTB in both wildlife and agricultural animals. Management measures to prevent spread of tuberculosis have included barrier fencing around cattle hay storage yards, elk population reduction through extended hunting seasons and reduced predator (wolf) control, making supplemental feeding and baiting of elk illegal, and controlled burns to improve elk habitat within the park to discourage elk ranging off-site into neighboring cattle feeding operations. 23 Current success of these methods in reducing disease lesions, especially in younger age classes of elk, and decreasing spillover BTB into deer and domestic cattle suggests that disease prevalence is declining and that progress is being made toward eradication. 45
Eurasian Badgers in England and Ireland
History and wildlife host
The Eurasian badger (Meles meles) are mustelids, weighing commonly from 10 to 16 kg, but older males may weigh over 30 kg. 30 They are omnivores that eat a variety of vegetation, as well as birds, reptiles, amphibians, insects, and small mammals; earthworms can be a prominent portion of their diets. 8 Badgers are well adapted to digging and live in multiple animal-extended family groups, in multitunnel burrows known as setts. Badger social groups vary from 2 to 3 individuals in low-density areas to 8 to 20 individuals in high-density areas. 8 They commonly emerge from their burrows after sundown. Badgers range throughout most of Europe, including the British Isles, as well as parts of Asia. 30 The badger was first found to be infected with M. bovis in southwestern England in 1971 and in County Cork, Ireland, in 1974. 16 Since the 1950s, cattle testing and slaughter to eradicate BTB had made steady progress in decreasing disease prevalence, until the 1970s. Since that time, both southwestern England and portions of the Republic of Ireland have developed areas of increased BTB cattle outbreaks and longtime persistent infections in specific cattle herds of unknown origin. 11 Current evidence suggests that contact between cattle herds and BTB-infected badgers is a major factor contributing to the perpetuation of infection in some regions. 13,16,21
Pathology
Pathology associated with tuberculosis in badgers is centered on the respiratory system, with over 50% of the lesions restricted to the lungs and another 35% of infected badgers exhibiting lymph node involvement. 16 These lesions are typical caseogranulomas, although lung lesions vary from nodules, to finely disseminated miliary lesions, to widespread lobar pneumonia. Lymph nodes when affected are enlarged and edematous, and they may contain pale yellow to white areas of caseogranulomas. Extrathoracic forms of the disease often affect the kidneys, resulting in elongate radial lesions. 16 Lesions in the liver or spleen appear as pale sawdust-like disseminated lesions. Superficial subcutaneous tuberculous abscesses are commonly associated with bite wounds in badgers. 16 No visible gross lesions (NVL) appears to be a relatively common manifestation in infected badgers, which still have significant rates of positive M. bovis cultures from either their lungs or lymph nodes. One 10-year survey for M. bovis found an 80% prevalence of NVL in infected badgers in Gloucestershire. 16 At least during early stages of tuberculosis, badgers appear to have a containment phase of the infection that may last for years, similar to latency in humans, which is known to last for decades. 16
Surveillance
No current national surveillance program exists due to the ongoing debate over badger culling. Long-term prevalence of BTB infection in badgers in endemic regions of England has been reported as approximately 4%. 11 Unfortunately, each published survey has had variation in the postmortem techniques and the intensity and length of the examination. Detailed postmortem examination of all commonly affected lymph nodes and other visceral organs is required for an accurate diagnosis of BTB in badgers. Even when a detailed and time-consuming examination is used, many badgers will still have no visible lesions detected. 8 Infection in badgers can spread rapidly following bite-origin inoculation, resulting in a mean survival time of only 117 days, whereas badgers that receive respiratory origin infections have a longer mean survival time of 491 days. 16 However, by whichever route infection is acquired by the badger, the infection may progress to disseminated disease or may lay dormant for an extended period of time. Some animals may be able to resolve BTB lesions based on the presence of fibrotic resolving lesions. 8
Granulomas associated with BTB in badgers are composed predominantly of epithelioid macrophages, with lesser numbers of lymphocytes. 8 These granulomas are highly cellular, with limited areas of necrosis, mineralization, or fibrosis. The granulomas usually contain acid-fast bacilli, which are most commonly present within necrotic debris. Histologic features found in cervid hosts but rare in badgers include the presence of multinucleated giant cells and large areas of caseation, cavitation, or abscess formation. 8
Control methods
A variety of control programs have been used in Great Britain for the control of BTB. Traditional testing and slaughter of positive cattle has been practiced since the 1930s, with steady decrease in BTB prevalence in cattle until the 1970s. Once badgers were determined to be an important reservoir host over 30 years ago, badger culling became the management control method of choice. Setts with higher badger population densities (6–15 adults/km2) tend to correlate with higher BTB infection rates and higher spillover rates into domestic cattle. 11 Badgers tend to shed M. bovis either in their sputum (resulting in spread through bite wounds or contaminated feed) or through the feces (which contaminates the environment). 16,19 However, public sentiment has gradually turned against the culling of badgers in England. Therefore, badger culling continues only in the Republic of Ireland, whereas England, Wales, and Northern Ireland currently do not use this control method. 8
Recent large-scale badger culling studies have yielded conflicting results. In southwest England, the Randomised Badger Culling Trial (RBCT) was conducted from 1998 through 2005. 17,35 This involved 10 triplicate study areas; in one area, badgers were not culled; in the second area, only reactive badger culling in response to cattle herds with outbreaks of BTB was practiced; and in the third area, proactive culling of badgers was practiced regardless of cattle herd status. Each of the trial sites measured approximately 100 km2. Proactive culling of badgers did reduce cattle BTB prevalence by 17% within the culling sites, but BTB prevalence in cattle herds in adjacent sites increased by 29%. 35 In addition, reactive culling of badgers resulted in increased cattle BTB prevalence by 29% on trial sites. A proposed explanation for these results is that culling resulted in social perturbation of badger populations, resulting in increased badger movement, increased badger-to-cattle interactions, and geographic spread of BTB. 17,35 This interpretation of the results remains controversial; however, the Independent Scientific Group on BTB has recommended not using badger culling based on this study. Conversely, an independent study conducted in the Republic of Ireland, known as the Four Areas Project (FAP), found badger culling to be an effective control measure to reduce cattle BTB prevalence. 13,35
Since badgers are now generally protected, and culling may or may not result in decreased disease prevalence in cattle, there has been a major emphasis on development of vaccines for BTB control in badgers. Since these are subterranean animals, oral vaccine baits have been the preferred route for study. A number of different vaccine types are being investigated, but the longstanding bacillus Calmette-Guerin (BCG) vaccine, which has been in use in humans for over 100 years, seems to have received the most support and interest to date. 8,10 One experimental vaccine trial used the intradermal BCG vaccine in badgers; when these badgers were subsequently challenged with BTB, the vaccinated animals had increased postexposure life spans and reduced shedding of M. bovis bacilli. 8 Oral bait vaccines are considered the best way to vaccinate badgers in the wild; a large-scale field trial using oral vaccine baits is currently under way in Ireland. 8
Although a bovine tuberculosis control program is in place, and significant progress is being made with regard to researching the badger as a wildlife host, as well as investigation of oral vaccine baits, the lack of public support for badger control continues to slow progress on control and eradication of the disease. Also, conflicting results from several recent epidemiologic studies on badger culling have not helped clarify the situation. 13,35
European Wild Boars in Spain
History and wildlife hosts
The European wild boar (Sus scofa) is widely distributed throughout Eurasia and Africa and subsequently has been introduced by humans to many other areas, as well as giving rise to our domestic pigs. These medium-sized mammals may weigh up to 350 kg, possess a simple stomach, and are omnivores that consume leaves, roots, bulbs, tubers, fruit, earthworms, reptiles, birds, eggs, rodents, and carrion. Their ability to root with their snouts and tusks allows them to bypass many types of barrier fencing. 30 The first reported cases of M. bovis in wild boars in Spain occurred in 1991. 18 Pigs have long been recognized as an animal that is readily susceptible to M. bovis, but it has generally been considered as a spillover host with inapparent infection limited to the cranial lymph nodes. 28,31 However, recent evidence from the Iberian Peninsula now implicates wild boar as a significant wildlife reservoir of BTB. 28 Evidence to support this includes common genotypes of M. bovis present in both wild boars and domestic animals; high prevalence of the disease in boars even where disease in domestic animals is not present; presence of widespread tuberculosis lesions, including the lungs and thoracic lymph nodes, suggesting these animals can shed the organisms; and juvenile wild boars tending to have disseminated lesions in multiple anatomical sites. 24,28 Matters are further complicated by the presence of BTB-infected badgers, red deer (Cervus elaphus), and fallow deer (Dama dama), which may also serve as wildlife reservoir hosts, but the boar appears to be the most important host at present. 20 Although wild boars are present throughout Spain, BTB prevalence in cattle herds and population densities of boars are highest in central and southern Spain in the semiarid Mediterranean habitats. 20
Pathology
Feral pigs are known to be highly susceptible to BTB and in fact have been used as sentinels to screen for environmental contamination by M. bovis both on the Hawaiian islands as well as in studies conducted in New Zealand. 31 In Mediterranean Spain, surveillance has shown that nearly 60% of wild boars had generalized TB lesions in more than one anatomical region. 24 Although the cranial lymph nodes, particularly the mandibular lymph nodes, were most likely to show grossly visible lesions of BTB, histologic evidence of infection has been found in 38% of lungs, 23% of livers, and 13% of spleens from infected animals. Large lesions in multiple anatomic regions were most frequently encountered in juvenile swine (12–24 months of age). 24
Diagnostic methods for identification of TB in hunter-harvested wild boar have recently been published. 42 This study indicates that mycobacterial culture in combination with histopathology is the most sensitive method to detect infection and that gross pathology, PCR of tissue, and acid-fast staining of smears or histologic sections all result in underestimating disease prevalence and numerous false-negative results. These findings are not unexpected to most pathologists experienced with the diagnosis of tuberculosis.
Surveillance
In many regions of Spain, Portugal, Italy, and the French Mediterranean island of Corsica, the numbers and population densities of wild boars and feral pigs are increasing, and these areas have correlations with increased prevalence of BTB in domestic cattle, goats, domestic pigs, and deer. 28,39 Such increases in swine population are associated with property fencing and increased feeding of wild swine, known factors to increase BTB prevalence in cervid species. 28
Surveillance of wild boars in Spain is based primarily on postmortem samples obtained from hunter-harvested animals. Surveillance in Donana National Park demonstrated that BTB prevalence in wild boars has increased by 100% between 1998–2003 and 2006–2007. 20 Another survey conducted over a 10-year period in south-central Spain found that the prevalence of BTB in juvenile wild boars at 11 geographic sites was increasing, whereas only 3 study sites had decreasing BTB incidence. 20 Even more worrisome is a recent report that over 50% of mandibular lymph nodes in wild boars that had gross lesions suggestive of tuberculosis were actually infected with Mycobacterium caprae—a closely related member of the Mycobacterium tuberculosis complex. 18 Therefore, wild boars appear to be a wildlife reservoir for more than one species of Mycobacterium that can spill over into domestic animals. Recently, studies have been conducted to evaluate several serologic assays to detect antibodies against both M. bovis and Mycobacterium avium subspecies paratuberculosis in wild boars. 1 These assays include both enzyme-linked immunosorbent assay (ELISA) and a dual-path platform test. Currently, a nationwide Wildlife Disease Surveillance Program is being designed to evaluate BTB and other disease conditions in a variety of wildlife species. 20
Control methods
As mentioned above, a nationwide surveillance program is being developed to gain further information on disease prevalence and trends in wildlife species throughout Spain. In addition, preliminary studies evaluating methods for delivery of oral BTB bait vaccines to juvenile wild boars have been conducted, as well as studies evaluating shedding of BCG from feces, internal organs, and environmental contamination. 20 At the present time, no centrally coordinated, integrated plan exists for the control and eradication of tuberculosis from wild boars in Spain. Therefore, prospects for control of bovine tuberculosis in this wildlife reservoir are not encouraging at the present time.
Brushtail Possums in New Zealand
History and wildlife hosts
The brushtail possum (Trichosurus vulpecula) is native throughout Australia and Tasmania and was introduced to New Zealand in the mid-1800s to start a fur industry. 30 Possums are arboreal marsupials that weigh between 1.3 and 5.0 kg. They are nocturnal and generally nest in tree cavities. Primarily herbivorous, they feed on shoots, leaves, grasses, flowers, fruits, and seeds, which are occasionally supplemented with insects. They appear to be solitary in nature, even in high-density populations that are present in some areas of New Zealand. Although their value for use as a fur species in New Zealand has greatly diminished, finding no natural predators, their populations have expanded and multiplied to the point where they have become an unwanted pest. 30
Ferrets (Mustela putorius furo) are another introduced wildlife species present in New Zealand with high population densities and high prevalence of tuberculosis. These animals were introduced from Europe in the 1880s. Ferrets are mustelids, which are nocturnal, and live in pastures or forest margins. They are carnivores whose diets include small mammals, birds, frogs, fish, and invertebrates. 30 However, their role as maintenance hosts and reservoirs for BTB has remained controversial. A recent study has shown that by significantly reducing the possum population in a local area, a corresponding rapid decrease in ferret BTB lesions by 80% for up to 5 years occurred in the same area. 3 Furthermore, the mesenteric lymph node is the most common site of infection, suggesting ingestion of infectious material, likely from carrion or prey, is an important source of BTB infection. 37 These studies would indicate that the ferret likely serves as a spillover host rather than a true reservoir host.
Pathology
Possums are highly susceptible to M. bovis infection, and once clinically ill, their survival times range from 3 to 14 months. 41 Although internal lymph nodes and lungs are commonly infected as in other wildlife species, the disseminated form of BTB commonly results in infection of subcutaneous lymph nodes in the inguinal and axillary regions, which frequently develop draining fistulous tracts, making them ideal for shedding the infectious organisms. 7 One study found that by tranquilizing healthy possums and placing them in pastures, their erratic movements simulated terminally ill possums with end-stage TB. 36 Furthermore, these simulated tuberculous possums aroused great interest and close inspection by cattle, oftentimes even progressing to sniffing and licking of the possums. 36 One can readily understand the implications of this behavior for the transmission of BTB to cattle.
The concept of NVL in infected wildlife is important in brushtailed possums. This problem is certainly not limited to possums but commonly occurs in badgers as previously described. In possums, it has been established that many individual animals in early stages of infection will show no gross lesions, but when pooled cultures of critical tissues, including lung and lymph nodes, are processed, many additional M. bovis–infected animals are found. 12 This is not surprising to experienced researchers working with tuberculosis, since BTB lesions start at the microscopic level and progress to form larger macroscopic lesions. One study found that 10.9% of possums surveyed had visible lesions, but when pooled cultures for M. bovis were conducted, the prevalence of infection increased by an additional 8.5%. 12 Although culturing nonlesioned tissues adds both time and expense to the diagnostic process, it is important not to declare geographic areas or wildlife populations tuberculosis free without microbiological confirmation.
Surveillance
The possum serves as the principal wildlife reservoir host for BTB in New Zealand, and common point prevalence rates of infection range from 1% to 10%, with an average prevalence of 5%, depending on the geographic location. 41 Throughout the 1970s and 1980s, bovine tuberculosis prevalence in domestic cattle and farm-reared deer climbed and disease spread over an increasingly wider area of the country. However, once the possum was recognized as the principal host and effective control methods were applied, the New Zealand TB prevalence in both domestic cattle and farmed deer has steadily declined. 2,6,41 For example, in one study, cattle herd prevalence of tuberculosis decreased from 2.4% in 1993 to 0.3% in 2004 following possum control efforts. 41
Control methods
Once the brushtail possum was found to be the primary reservoir of BTB in New Zealand, a massive culling program using trapping and poisoning with the pesticide 1080 (sodium fluoroacetate) was initiated. 2,6,41 When possum populations are reduced to levels of 40% or less of their previous uncontrolled state, the prevalence of BTB in possums, ferrets, cattle, and deer undergoes a precipitous decline. 2,6 One significant advantage that New Zealand has over other countries is that its primary wildlife reservoir host—the possum—is universally recognized as an introduced pest species, so there is minimal public outcry against the culling of these animals.
Another important difference in their control program is that the livestock industry members themselves fund and run the control efforts. New Zealand livestock owners direct their own program, fund its operation, and cooperate without receiving indemnity payments for BTB-infected cattle or deer. 41 This represents a basic difference from the situation in North America, where government-run eradication programs depend on monetary indemnities to gain livestock owner compliance. 5 New Zealand researchers are among the world leaders in the current study of oral bait BTB vaccines, which many believe is the future for wildlife tuberculosis control. 9
It appears that New Zealand is making significant gains in its efforts to control BTB in wildlife hosts, and their program is currently making steady progress toward controlling the disease.
African Buffalo and Lechwe in Parks in Africa
History and wildlife hosts
African buffalo (Syncerus caffer) and lechwe antelope (Kobus leche) have both been found to be important wildlife reservoirs of BTB in South Africa and Zambia, respectively. African buffalo living in the Kruger National Park and Hluhluwe-iMfolozi Park are particularly affected by endemic BTB. 26 Kruger National Park was initially established as a game reserve in 1898 and was subsequently expanded and became a national park in 1926. It encompasses 19 485 km2, extends 320 km from north to south, and is approximately 65 km from east to west. Kruger is home to 147 mammalian species, including approximately 27 000 African buffalo. 26 Hluhluwe-iMfolozi Park was established in 1895 and encompasses 960 km2. 26 These parks’ wildlife populations were confirmed positive for BTB for the first time in 1990 and 1986, respectively. 26 During the 1990s, drought may have stressed the buffalo, allowing subclinical BTB infections to progress and result in mortalities. 38 It appears that British colonists carried BTB to Africa in domestic cattle during the 1800s, and the disease subsequently spilled into various wildlife species. 25
African buffalo are highly gregarious herding animals that travel in herds of several hundred animals. These large ruminants weigh between 300 and 900 kg. They spend much of the day grazing on grasses or leaves or wallowing in mud. Each herd has a home range through which they move in a circuitous pattern throughout the year. 30
Lechwe antelopes are gregarious marsh antelopes that live in small groups of several dozen animals. They weigh between 50 and 300 kg. Like buffalo, the lechwe are ruminants and favor swampy tracts in which to graze. They also follow local seasonal movement patterns in response to environmental conditions. 30 Lechwe affected by BTB live in the Kafue Basin of central Zambia composed of flood plains surrounding the River Kafue. 27 Lechwe numbers in the early 1970s were estimated around 90 000 animals. 15 Surveys indicated that significant population declines in these antelope have been ongoing since the 1970s. 27
Other wildlife species known to be infected with BTB in South Africa’s national parks include lions (Panthera leo), cheetahs (Acinonyx jubatus), chacma baboons (Papio ursinus), greater kudu (Tragelaphus strepsiceros), leopards (Panthera pardus), hyenas (Crocuta crocuta), large-spotted genets (Genetta tigrina), warthogs (Phacochoerus aethiopicus), bushpigs (Potamochoerus porcus), and eland (Taurotragus oryx); however, most or all of these animals—with the possible exception of the kudu—appear to be secondary dead-end hosts at this time. 25
Pathology
A recently published survey of 19 African buffalo that tested positive by intradermal tuberculin testing and were culled from Hluhluwe-iMfolozi Park described the BTB lesions present. 22 Gross lesions in 80% of these buffalo were restricted to the retropharyngeal, bronchial, and mediastinal lymph nodes, whereas only 20% of tuberculous buffalo had gross lesions in their lungs. 22 This lesion distribution suggests that the primary route of infection in buffalo is by inhalation. Gross lesions consisted of single to multiple granulomas, with central caseation and mineralization. 22
Histologically, tuberculous lesions in buffalo lymph nodes contained a variety of inflammatory cells, including epithelioid macrophages, lymphocytes, and multinucleated giant cells. The degree of encapsulation of the granulomas, as well as amounts of necrosis and mineralization, varied by stage of the BTB lesion. 22 One common finding in buffalo BTB lesions was that acid-fast bacilli were only rarely observed (found in only 7 of 19 positive animals). The histologic features of BTB in buffalo appear similar to those reported for domestic cattle and North American cervids. 22
A survey conducted in 1971 of the BTB lesions in lechwe found that 36% of animals examined had gross lesions of tuberculosis. 15 The most common site of gross lesions was the lungs in 60% of BTB-infected lechwe, followed next by 40% of lechwe exhibiting lesions in pulmonary lymph nodes, with less than 20% of animals exhibiting gross lesions in the mesenteric lymph nodes. The diaphragmatic lung lobes had the highest incidence of pulmonary lesions. Pulmonary lesions consisted of granulomas with extensive central caseation and only light mineralization in most cases. Acid-fast bacilli were found microscopically in nearly all pulmonary granulomas. 15 Lymph node lesions in lechwe commonly exhibited heavy concentric rings of calcification. BTB prevalence was correlated with poor body condition scores in lechwe. In addition, BTB was also correlated with liver fluke infestation. No cases of BTB were found in juvenile animals younger than 18 months. 15
Surveillance
Surveillance for BTB in Kruger National Park has been ongoing for several decades. The first major survey was conducted in 1991–1992 and examined over 2000 buffalo that were culled from all known wild buffalo herds. 40 It was found that the disease prevalence was stratified by geographic location in the park. Southern herds had a BTB prevalence of 27.1%, central herds had a BTB prevalence of 4.4%, and northern herds had a BTB prevalence of 0%. 40 In 1998, a stratified, 2-stage cluster sampling method survey included necropsies of just over 600 buffalo and estimated BTB prevalence at 38.2% in southern herds, 16% in central herds, and 1.5% in northern herds. 40 This survey indicated an increasing BTB prevalence, particularly in the southern and central zone herds. It was also interpreted as evidence that BTB had been initially introduced to buffalo from infected domestic cattle grazing areas surrounding southeastern Kruger during the 1960s to 1980s. 40 Modeling predictions estimate that the overall prevalence of BTB in Kruger’s buffalo could reach 90% over the next 25 years. 38
Between 2004 and 2008, 119 Kafue lechwe were hunted by special permit and postmortem examinations conducted for BTB. This survey revealed 24.3% of lechwe had gross lesions consistent with TB, and 27.7% were mycobacterial culture positive. 27 This is in comparison with a 36% prevalence reported for BTB in the 1971 survey of lechwe. 15
Control methods
Control efforts to date include an electrified fence surrounding Kruger National Park to prevent buffalo from coming into direct contact with domestic cattle outside the park, although it has been noted that some species, including kudu and Lechwe antelope, can easily jump over this fence. 38 One method proposed to help control BTB in Kruger buffalo was to separate the southernmost herds from the northern herds by creating a buffalo-free zone by fencing across portions of the park. 26 This idea was never implemented due to the disruption in normal seasonal buffalo herd movements that would have resulted from such fences. Originally, much higher prevalence rates existed in buffalo in southern Kruger, but over the past 20 years, the infection has spread progressively northward so that nearly all buffalo herds are now affected by significant tuberculosis prevalence. 40
Presently in Hluhluwe-iMfolozi Park, buffalo are captured in corrals, and antemortem skin testing is conducted, with all skin reactors being culled. To date, the prevalence rate of TB in buffalo has been significantly reduced (below 10% prevalence) using this method. 38 No major control initiatives appear to have been instituted specifically for BTB in Kafue lechwe.
For the future, vaccination of buffalo appears to be the preferred method of control, and studies using BCG vaccine in a buffalo model system are currently ongoing. 26,38 Despite ongoing efforts to control bovine tuberculosis in southern African wildlife parks, the complex interaction of so many valuable wildlife species makes this an extremely difficult system to manage for BTB disease control. Progress is likely to continue in slow increments for the foreseeable future.
Conclusions
M. bovis has an extremely wide host range. Many different species can become reservoir hosts, including herbivorous species (deer, elk, African buffalo, lechwe) and omnivorous species (wild boars, badgers, possums). Human interference, through the introduction of the causative agent of BTB by way of infected cattle and poor biosecurity, which allows the mingling of domestic cattle with wildlife species; increasing encroachment and disturbance of the natural environment by expansion of agriculture; and supplemental feeding of wildlife, leading to abnormally high-density congregation of wildlife species, are all recurring themes in the development of wildlife reservoirs of tuberculosis.
The distribution of tuberculous lesions in the upper and lower respiratory tracts of the various wildlife species reviewed indicates that M. bovis may be spread by aerosol droplet infection, but contamination of feed sources and pastures, as well as omnivores feeding on infected carcasses, are additional important routes of BTB transmission in wildlife species. Reservoir hosts may carry and shed BTB for years with no clinical signs (cervids), and disease may progress slowly (buffalo) or rapidly (possums, juvenile feral boars, badgers). The common element in many of these wildlife hosts appears to be close family groups that spread the disease within the species and environmental or feed contamination leading to disease spread to other animal species.
Decreasing population densities of wildlife reservoirs of BTB and improving biosecurity to prevent interaction between wild and domestic animals help to control the lateral spread of the disease. However, in many situations we have examined, these methods are by themselves not sufficient to eradicate the disease. Other methods such as focal depopulation of wildlife species—if those species are not endangered or highly valued by the public—are additional strategies that can be successfully employed. Unfortunately, not all wildlife reservoir species are appropriate for such extreme control methods.
An emerging theme expressed by many of the researchers around the globe involved in the control and eradication of BTB is currently focused on the development of wildlife vaccines. Such vaccines must significantly reduce the shedding and spread of M. bovis, if not actually prevent the disease in that reservoir host. Additional concerns include how to distribute and gain widespread coverage of free-ranging animals, which in many cases means the development of oral bait-type vaccines. Other considerations include nontarget species ingestion of such bait vaccines and the ability to develop antemortem surveillance tests that can distinguish between vaccine response and true infection with wild-type M. bovis. This may mean that a genetically engineered vaccine is more acceptable than a more conventional BCG-based vaccine.
Improved diagnostics are a critical need to detect all infected individual animals, particularly in instances where individual animals are infected and exhibit subclinical disease or no visible lesions. Until new methods of localized geographic live capture of wild animals, combined with rapid antemortem testing allows for the release of BTB tested-negative animals, or euthanasia of positive BTB animals, further progress in disease prevalence reduction or eradication may not be possible. Equally important in wildlife species is the ability to test an animal at a single time point, instead of over several days, as with classic skin tests currently used in domestic cattle and captive cervids. Recently developed serologic assays, including the gamma interferon assay, the MAPIA, and the rapid test, are all attempts to provide such single point-of-contact assays, which could become valuable tools in surveillance of free-ranging wildlife species.
Regardless of the methods employed, the control of bovine tuberculosis once it establishes itself in a wildlife maintenance host population is generally a long-term commitment. Only in the case of Minnesota deer, in which the disease had not yet become firmly established, was the eradication campaign quickly successful. Furthermore, a coordinated approach including many public stakeholders, including wildlife personnel, legislators, livestock producers, hunters, and landowners, with widespread public support of the proposed control program is critical to successful control and eradication of BTB. It appears that BTB in its many forms remains an important disease in both domestic and wild animals and that its eventual control will remain an ongoing concern for many decades into the foreseeable future.
Footnotes
Acknowledgements
The authors acknowledge that this invited review originated from an oral presentation at the concurrent session on tuberculosis, which was originally presented at the Annual Meeting of the American College of Veterinary Pathologists on 7 December 2011, in Nashville, Tennessee.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
