Abstract
Regulatory T cells may serve as targets in cancer immunotherapy. A previous study showed that the chemokine CCL17 and the receptor CCR4 play roles in regulatory T cell recruitment in canine urothelial carcinoma. In this article, we show that the BRAFV595E mutation is associated with tumor-produced CCL17 and regulatory T cell infiltration in dogs with urothelial carcinoma. In comparison with healthy dogs, dogs with urothelial carcinoma showed increased CCL17 mRNA expression in the bladder and elevated CCL17 protein concentration in urine. Immunohistochemistry showed increased levels of Foxp3+ regulatory T cells in the tumor tissues of urothelial carcinoma. The density of Foxp3+ regulatory T cells was positively correlated with CCL17 concentration in urine, indicating that CCL17 is involved in regulatory T cell recruitment. Moreover, tumor-infiltrating regulatory T cells and urine CCL17 concentration were associated with poor prognosis in dogs with urothelial carcinoma. The number of tumor-infiltrating regulatory T cells, CCL17 mRNA expression, and urine CCL17 concentration in cases with BRAFV595E mutation were higher than those in cases with wild-type BRAF. In vitro, high CCL17 production was detected in a canine urothelial carcinoma cell line with BRAFV595E mutation but not in an urothelial carcinoma cell line with wild-type BRAF. Dabrafenib, a BRAF inhibitor, decreased CCL17 production in the cell line with BRAFV595E mutation. These results suggest that BRAFV595E mutation induced CCL17 production and contributed to regulatory T cell recruitment in canine urothelial carcinoma.
Keywords
Canine urothelial carcinoma, also known as transitional cell carcinoma, is a malignant tumor derived from transitional epithelial cells of the urinary tract. Although urothelial carcinoma accounts for 1% to 2% of all malignant tumors in dogs, it is the most common tumor affecting the urinary bladder. 11,32 It has a poor prognosis because of its high-grade muscle-invasive nature and frequent metastasis to regional lymph nodes or distant tissues (eg, lungs, bone, liver). 19 Previous studies have shown that a somatic point mutation in the BRAF gene (BRAFV595E) is present in over 70% of dogs with urothelial carcinoma. 7,29,30 BRAF is an isoform of RAF serine/threonine kinase, which belongs to the RAF/MEK/ERK mitogen-activated protein kinase (MAPK) pathway. The MAPK pathway is a key signaling pathway in cell proliferation and survival; therefore, aberrant activation of this pathway leads to uncontrolled growth in cancer. 8 However, little is known about the impact of the BRAFV595E mutation on disease development or progression in dogs with urothelial carcinoma.
Oncogenic mutations in the BRAF gene occur in 8% of all human cancers, with the majority harboring a BRAFV600E mutation. 6 In particular, the BRAF mutation is an important mutation in melanoma, accounting for approximately 50% of melanoma patients. 43 BRAFV600E leads to constitutive activation of the MAPK signaling pathway, resulting in reduced apoptosis, increased invasiveness, and increased metastatic behavior. 42 The BRAFV595E mutation observed in dogs is homologous to the human BRAFV600E mutation. Thus, it is possible that the BRAFV595E mutation plays a role in oncogenesis in canine urothelial carcinoma. Several drugs, such as encorafenib, vemurafenib, and dabrafenib, have been developed to selectively target the BRAFV600E mutation in humans. These BRAF inhibitors are approved by the Food and Drug Administration for the treatment of metastatic melanomas that express BRAFV600E. 3 Although the antitumor effects of BRAF inhibitors were initially believed to be via direct effects on tumor cells, subsequent analyses using biopsies from BRAF-mutated melanoma patients treated with BRAF inhibitors exhibited increased number of cytotoxic T cells. 50 Furthermore, BRAF inhibitors block infiltration of regulatory T cells (Tregs) into tumor tissues in murine models of melanoma. 20,39 These observations suggest that BRAF mutations influence the host immune response in the tumor microenvironment.
The role of the host immune response has been investigated in several types of cancers. Tumor-infiltrating lymphocytes have different effects on tumor progression. In both human and canine cancers, accumulating evidence suggests that infiltration of cytotoxic T cells in the tumor predicts a good prognosis, whereas infiltration of Tregs correlates with a poor prognosis. 1,2,10,13 –15,17,18,23,33,34,45 Tregs are an immune suppressive subset of CD4+ helper T lymphocytes expressing the interleukin (IL)-2 receptor alpha chain (CD25) and transcription factor forkhead box P3 (Foxp3). 47 Tregs play a role not only in suppression of immune response and inflammation but also in tumor progression, presumably by inhibiting antitumor immunity. A recent study by our group has demonstrated that the tumor-produced chemokine CCL17 induces Treg infiltration via the chemokine receptor CCR4 in dogs with urothelial carcinoma. 21 However, the mechanism underlying upregulated CCL17 production in canine urothelial carcinoma remains unclear.
In the present study, we investigated the association of BRAFV595E mutation with CCL17 expression and Treg infiltration in dogs with urothelial carcinoma. We also examined the effect of BRAF inhibition on CCL17 production in canine urothelial carcinoma cell lines.
Materials and Methods
Ethical Statements
The Animal Care and Clinical Research Committees of the Veterinary Medical Center of the University of Tokyo approved the study protocol for urine and tumor tissue sampling from client-owned dogs. We obtained written informed consent from all dog owners. Use of dogs in these studies was approved by the Institutional Animal Care and Use Committee of the University of Tokyo. All experiments followed approved guidelines.
Dogs and Sample Collection
The signalment, clinical, and histological information of dogs with urothelial carcinoma enrolled in the study for histologic, gene expression, and protein expression analyses are provided in Supplemental Table S1. For immunohistochemistry, we used formalin-fixed, paraffin-embedded urothelial carcinoma tissues that were surgically removed from 24 dogs at the Veterinary Medical Center of the University of Tokyo. All dogs underwent total cystectomy. 36 No dogs received chemotherapy or radiation therapy before surgery. Lymph node involvement (iliac lymph node swelling) and lung metastasis were assessed by ultrasound, radiographic, and/or computed tomography examinations. Diagnosis of urothelial carcinoma was confirmed by histopathology. Histopathological findings according to the World Health Organization (WHO) classification and grade of tumors were reviewed by 2 pathologists with Japanese College of Veterinary Pathologists board certification (JKC, KU). 26,27 Survival time and current status (alive, deceased, or lost) of 24 dogs with total cystectomy were determined by medical record or interview. Overall survival was defined as the time from total cystectomy to the established cause of death of the animal at the end of the study (August 1, 2018). Normal canine bladder tissues were obtained from 10 healthy beagles via necropsy for other experiments and used as a control.
For gene expression analysis, fresh tumor tissues from 14 dogs with urothelial carcinoma were immediately submerged in RNA
For enzyme-linked immunosorbent assay (ELISA), fresh urine samples were collected by a urethral catheter from 47 dogs with urothelial carcinoma, 15 dogs with bacterial cystitis, 13 dogs with urolithiasis (calcium oxalate or struvite), 8 dogs with polypoid cystitis, and 17 healthy dogs, and stored at −80 °C until subsequent analysis.
Cells
We used canine cell lines derived from urothelial carcinoma with the BRAFV595E mutation, namely, Love, and urothelial carcinoma with wild-type BRAF, namely, K9TCC-Sh. 7,9 Love cells were grown in RPMI-1640 (Sigma), supplemented with 10% fetal bovine serum (FBS; Thermo Fisher Scientific) and 100 μg/ml penicillin and streptomycin (Sigma) at 37 °C under a humidified atmosphere with 5% CO2. K9TCC-Sh cells were grown in Dulbecco’s modified Eagle’s medium (Thermo Fisher Scientific), supplemented with 10% FBS (Thermo Fisher Scientific) at 37 °C under a humidified atmosphere with 5% CO2.
Immunohistochemistry
The expression of Foxp3 was examined by immunohistochemistry. 21 One paraffin-embedded 4-μm-thick section was used for immunohistochemistry in each case. Heat-induced antigen retrieval was performed by autoclaving the sections for 10 minutes at 121 °C in 10 mM sodium citrate buffer (pH 6.0). Endogenous peroxidase activity was blocked by incubation with REAL Peroxidase-Blocking Solution (DAKO) at room temperature for 10 minutes. Sections were blocked with 5% skim milk in Tris-buffered saline with 0.1% Tween 20 (TBST) at room temperature for 60 minutes, and then incubated with primary antibody rat anti-Foxp3 (1:400 dilution, clone FJK-16s, eBioscience) at 4 °C overnight. The cross-reactivity of the primary antibody with canine Foxp3 was confirmed in a previous study. 28 Samples were incubated with the secondary antibody biotin-labeled anti-rat IgG (Vector Laboratories) at 37 °C for 30 minutes, followed by HRP-labeled streptavidin (Dako) at room temperature for 30 minutes. Mesenteric lymph nodes were used as positive controls. Sections incubated with TBST only served as negative controls. The reaction products were visualized with 3,3′-diaminobenzidine. Cells with clear lymphocyte morphology and distinct nuclear immunolabeling for Foxp3 were evaluated as positive. Foxp3+ Tregs were manually counted in 10 representative fields (400× magnification: 0.238 mm2 per field) with the most intense positivity and >50% of tumor cells using ImageJ software. 22,37,38
Quantitative RT-PCR
We extracted total RNA from each canine bladder tissue using the RNAspin Mini RNA Isolation Kit (GE Healthcare). Two-step real-time reverse transcriptase-polymerase chain reaction (StepOnePlus; Applied Biosystems) was performed to examine CCL17 mRNA expression using THUNDERBIRD SYBR qPCR Mix (Toyobo) under amplification conditions of 95 °C for 10 seconds, followed by 40 cycles of PCR (95 °C for 5 seconds and 60 °C for 30 seconds). RPL13A and RPL32 were used as references.
35
Table 1 lists the primer pair sequences. All primer sets exhibited >95% efficiency, which was determined by using a 10-fold dilution series from representative cDNA samples. Nuclease-free water was used as the negative control. Cycle threshold (Ct) values that indicate points of threshold intersects with the amplification curve of PCR were determined using StepOne Plus v2.3 software (Applied Biosystems). All samples were examined in duplicate, and the mean ΔCt was calculated. The expression level of mRNA (relative quantity) was calculated using 2−ΔCt, resulting in evaluation of carcinoma tissues as the
Primer Pair Sequences Used for RT-PCR.
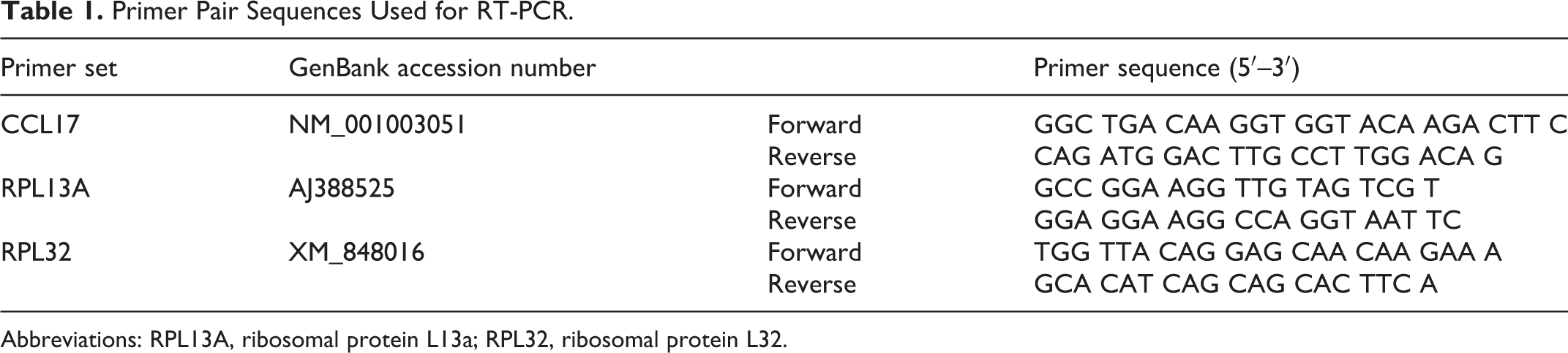
Abbreviations: RPL13A, ribosomal protein L13a; RPL32, ribosomal protein L32.
Detection of BRAFV595E Mutation
BRAFV595E mutations in all dogs and cell lines were examined by digital PCR assay, as previously described. 9 Genomic DNA was isolated from urine sediments and cells with the DNeasy Blood and Tissue Kit (QIAGEN), according to the manufacturer’s instructions. Digital PCR was performed using ProFlex Base (Thermo Fisher Scientific) and 2× Quantstudio3D Mastermix (Thermo Fisher Scientific). The probes specific for wild-type BRAF and BRAFV595E mutation genes were labeled with 4,7,2′-trichloro-7′-phenyl-6-carboxyfluorescein (VIC) and 6-carboxyfluorescein (FAM), respectively. Probe and primer sequences were as follows: wild-type probe_VIC, 5′-TAGCCACAGTGAAATC-3′; mutation probe_FAM, 5′-CCACAGAGAAATC-3′; BRAF forward, 5′-CATGAAGACCTCACAGTAAAAATAGGTGAT-3′; BRAF reverse, 5′-TGGGACCCACTCCATCGA-3′. PCR conditions were as follows: 10 minutes at 96 °C, 39 cycles of 60 °C for 2 minutes and 98 °C for 30 seconds, and 2 minutes at 60 °C. Raw data were obtained and analyzed using the Quantstudio 3D Digital PCR system (Thermo Fisher Scientific) and the Quantstudio3D Analysis Suite software (Thermo Fisher Scientific).
Cell Proliferation Assay
The effect of a BRAF inhibitor, dabrafenib, on cell proliferation was determined in canine urothelial carcinoma cell lines by using a sulforhodamine B (SRB) assay. 48 Love and K9TCC-Sh cells were seeded at optimized densities of 3.5 × 102 and 2.0 × 102 cells per well in 96-well plates, respectively. After initial incubation for 24 hours, the cells were exposed to dabrafenib (Selleck) at varying concentrations (0.01 nM to 100 μM) for 24 hours. As a control, the same amount of DMSO (dimethyl sulfoxide) was added. Cells were fixed with 5% cold trichloroacetic acid solution (FUJIFILM Wako Pure Chemical) at 4 °C for 60 minutes, stained by 0.058% SRB (Sigma) in 1% acetic acid at room temperature for 30 minutes, and solubilized in 10 mM unbuffered Tris solution. The absorbance was measured at 520 nm by using the iMark Microplate Reader (Bio-Rad). The samples were examined in triplicate, and 3 independent experiments were performed.
ELISA
Love and K9TCC-Sh cells were seeded in serum-free medium at optimized densities of 1.2 × 105 and 6.5 × 104 cells per well in 6-well plates, respectively. After initial incubation for 6 hours, the culture medium was discarded. The cells were further incubated in fresh serum-free medium with 1 to 10 μM dabrafenib (Selleck) for 24 hours. The supernatants of the culture media were collected and immediately frozen at −80 °C until subsequent measurement.
Canine CCL17 protein concentrations in urine and culture medium samples were measured using the Canine TARC/CCL17 ELISA Kit (Cusabio), according to the manufacturer’s protocol. The CCL17 concentration in the medium was normalized by the cell count number and expressed as pg/105 cells. Urine creatinine concentration was evaluated using the LabAssay Creatinine Kit (FUJIFILM Wako Pure Chemical), and urine CCL17 concentration was expressed as pg/mg of creatinine.
Western Blotting
Love and K9TCC-Sh cells were seeded in serum-free medium at optimized densities of 7.4 × 105 and 4.0 × 105 cells per well in 90-mm dishes, respectively. After initial incubation for 6 hours, the culture medium was discarded, and the cells were further incubated in fresh serum-free medium with 1 to 10 μM dabrafenib (Selleck) for 60 minutes. Cells were lysed using RIPA buffer containing protease and phosphatase inhibitor cocktail (Roche Diagnostics). The cell lysates were centrifuged at 14 000 rpm at 4 °C for 10 minutes. Protein content was measured by a BCA Protein Assay Kit (Thermo Fisher Scientific). Protein samples (10 μg) were loaded in SDS-polyacrylamide gel electrophoresis and transferred onto polyvinylidene difluoride membranes (Bio-Rad). The membranes were blocked with 5% skim milk in TBST at room temperature for 60 minutes, and then incubated with primary antibodies, rabbit anti-ERK (1:1000 dilution, Cell Signaling), rabbit anti-Tyr 202/Thy204-phophorylated ERK (1:2000 dilution, Cell Signaling), and mouse anti-β-actin (1:10000 dilution, Merck) at 4 °C overnight. The membranes were washed with TBST and were incubated with the secondary antibody, horseradish peroxidase-conjugated anti-rabbit or anti-mouse IgG antibody (1:10000 dilution; GE Healthcare), at room temperature for 60 minutes. Proteins were detected using the Immobilon Forte Western HRP substrate (Merck), and luminescence was captured using an imaging system (ChemiDoc Image Lab, Bio-Rad).
Statistical Analysis
We used JMP Pro version 14.2.0 (SAS Institute) for statistical analyses. Intergroup comparisons were assessed using the Mann-Whitney
Results
Clinical and Pathological Findings in Dogs With Urothelial Carcinoma
A total of 47 dogs with urothelial carcinoma were included in this study. Of these, 24 dogs were evaluated and categorized by histopathology (Supplemental Table S1). Based on the WHO classification, 26 6 (25%) tumors were classified as papillary and noninfiltrating, 11 (45.8%) as papillary and infiltrating, and 7 (29.2%) as nonpapillary and infiltrating. There were no tumors classified as nonpapillary and noninfiltrating. Based on the histologic grade, 27 3 (12.5%) tumors were classified as low-grade and 21 (87.5%) as high-grade. Among the 24 dogs used in immunohistochemical assessments, iliac lymph node involvement was observed in 5 (20.8%) dogs. No distant metastasis (to the lung) was identified in any of the cases at total cystectomy.
BRAFV595E mutation in all dogs used in this study were examined by digital PCR assay. 9 Of the 47 dogs with urothelial carcinoma, the BRAFV595E mutation was detected in 36 (76.6%) cases (Supplemental Fig. S1 and Supplemental Table S1). The BRAFV595E mutation was not detected in any normal dogs or dogs with nonneoplastic urinary tract diseases (Supplemental Table S1).
CCL17 Expression Is Elevated in Dogs With Urothelial Carcinoma
Our study had shown increased CCL17 expression in dogs with urothelial carcinoma. 21 We first examined CCL17 mRNA expression in the bladder and CCL17 concentration in the urine of another cohort. CCL17 mRNA expression in urothelial carcinoma increased by approximately 10- to 530-fold in comparison with the expression level in normal bladder tissues (Fig. 1). In addition, urine CCL17 concentration in dogs with urothelial carcinoma was significantly higher than that in normal control dogs and in dogs with nonneoplastic urinary tract diseases, except for polypoid cystitis (Fig. 2). Interestingly, tumoral CCL17 expression and urine CCL17 concentration varied among cases: not all dogs with urothelial carcinoma exhibited high CCL17 expression (Figs. 1, 2).
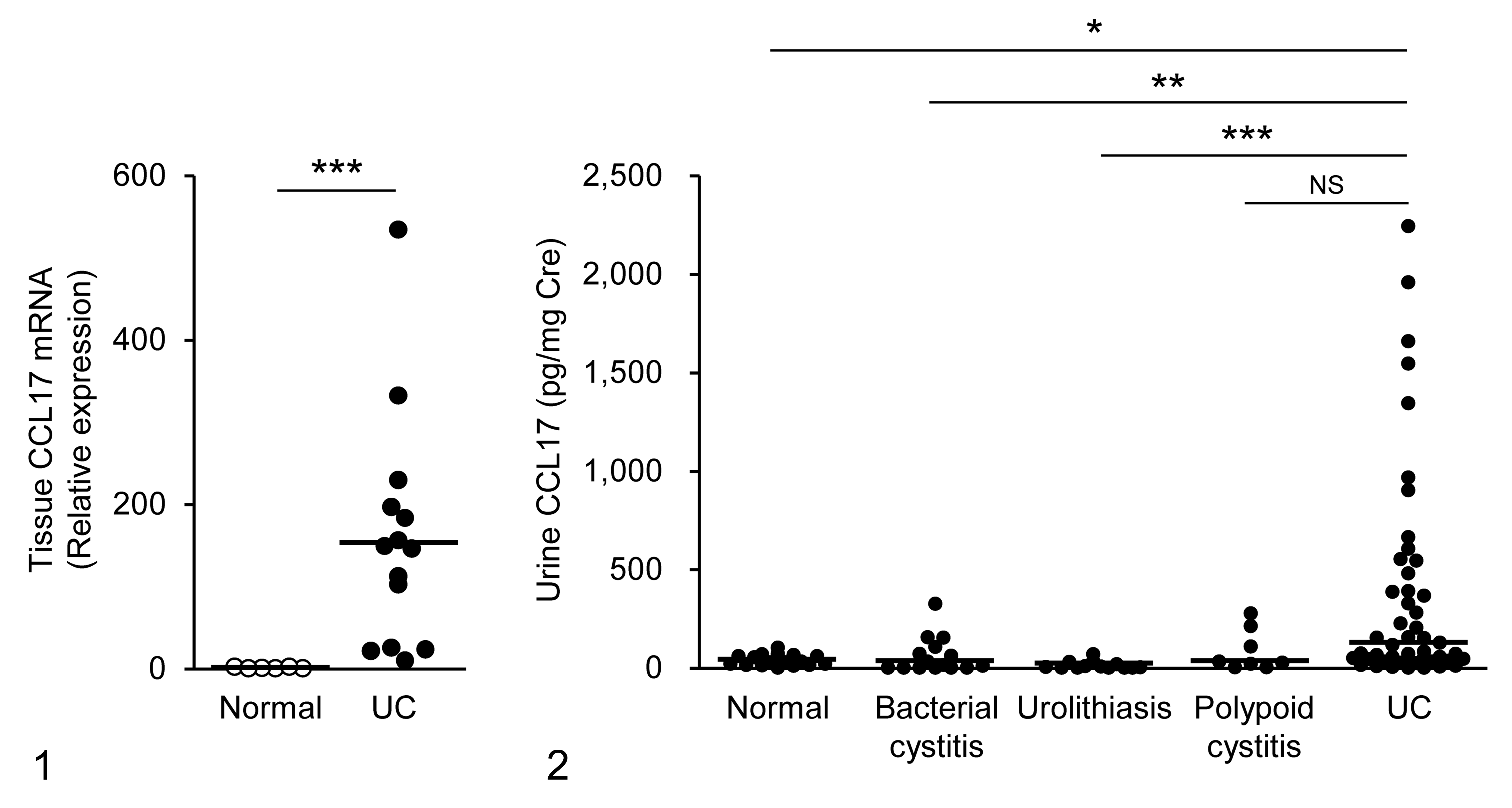
CCL17 mRNA expression in the normal bladder (
Tumor-Infiltrating Tregs Correlate With CCL17 Expression in Dogs With Urothelial Carcinoma
We visualized Foxp3 using immunohistochemistry and examined the localization and number of tumor-infiltrating Tregs in dogs with urothelial carcinoma. Foxp3+ Tregs were present abundantly in both the peritumoral stroma and intratumoral area (Figs. 3, 4). In comparison with the normal bladder (Fig. 5), urothelial carcinoma showed increased number of Foxp3+ Tregs (Fig. 6). The density of Foxp3+ Tregs was positively correlated with urine CCL17 concentration (Fig. 7). A tendency for the correlation between the number of Foxp3+ Tregs and tumoral CCL17 expression was observed, but there was no significant difference (
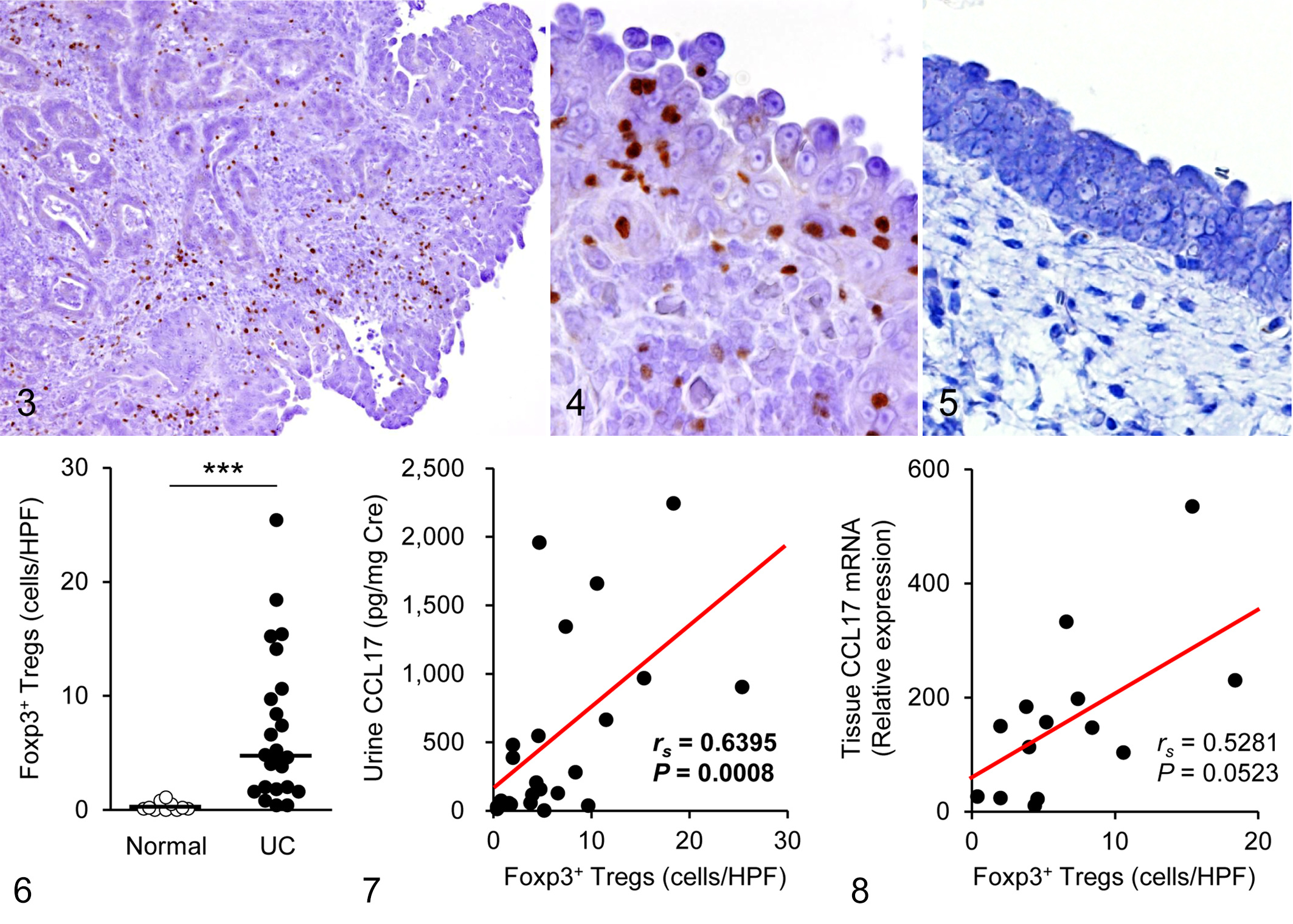
Urothelial carcinoma, urinary bladder, dogs. There is nuclear immunolabeling of Foxp3+ in mononuclear cells, indicating their identity as regulatory T cells (Tregs). Immunohistochemistry for Foxp3.
We performed further analyses to examine whether tumor-infiltrating Tregs, urine CCL17 concentration, and tumoral CCL17 mRNA increased along with the histologic type (WHO classification), tumor grade, and lymph node involvement. The number of Tregs and CCL17 expression tended to be increased in dogs with infiltrating and high-grade tumor; however, no significant difference was evident between histologic type/grade and Tregs/CCL17 expression (Supplemental Figs. S2–S4). Lymph node involvement was not related to tumor-infiltrating Tregs, CCL17 concentration in urine, or tumoral CCL17 mRNA (data not shown).
BRAFV595E Mutation Correlates With Tumor-Infiltrating Tregs and CCL17 Expression in Dogs With Urothelial Carcinoma
We further investigated whether BRAFV595E mutation influences tumor-infiltrating Tregs and CCL17 expression. As shown in Figures 9 to 11, Foxp3+ Tregs were increased in cases with BRAFV595E mutation in comparison with cases of wild-type BRAF. Moreover, tumoral CCL17 mRNA expression and urine CCL17 concentration in cases with the BRAFV595E mutation were higher than those in cases with wild-type BRAF (Figs. 12, 13).
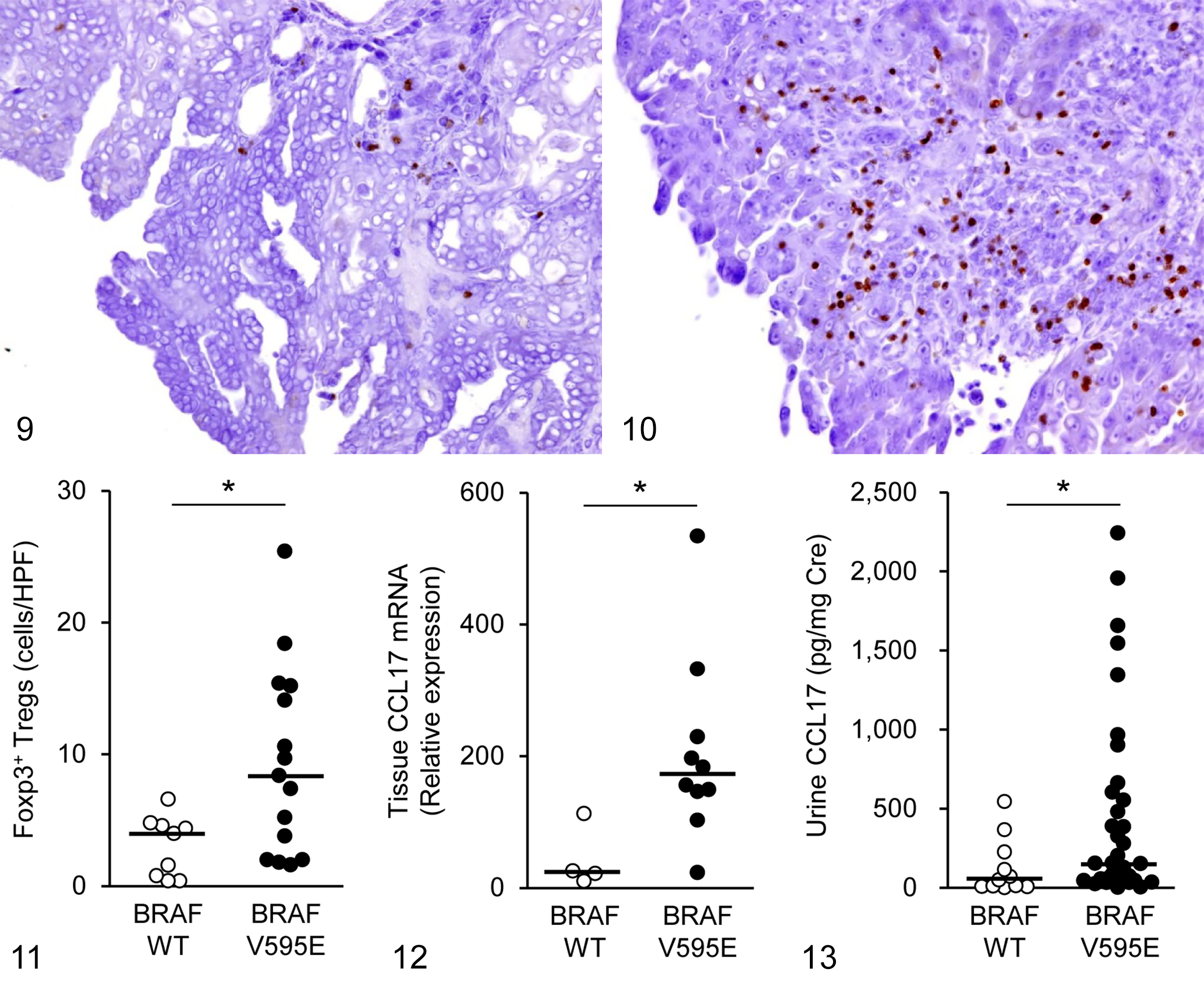
Urothelial carcinoma, urinary bladder, dogs. The number of tumor-infiltrating Foxp3+ regulatory T cells (Tregs) was relatively low in cases with wild-type (WT) BRAF (Fig. 9) and relatively high in cases with the BRAFV595E mutation (Fig. 10). Immunohistochemistry for Foxp3.
Survival Analysis
During follow-up, 14 of 24 dogs with total cystectomy died and 10 dogs were alive at the end of the study period. The median overall survival was 298 days (range = 10–1333 days). Based on the median number, we classified each case as having a high or low number of tumor-infiltrating Tregs or urine CCL17, then assessed the correlation between Tregs/CCL17 expression and prognosis. The overall survival for cases with number of high tumor-infiltrating Tregs was shorter than that for cases with low number of tumor-infiltrating Tregs (Fig. 14). Furthermore, high CCL17 concentration in urine correlated with shorter overall survival (Fig. 15). However, BRAFV595E mutation was not significantly related to the prognosis (Fig. 16).
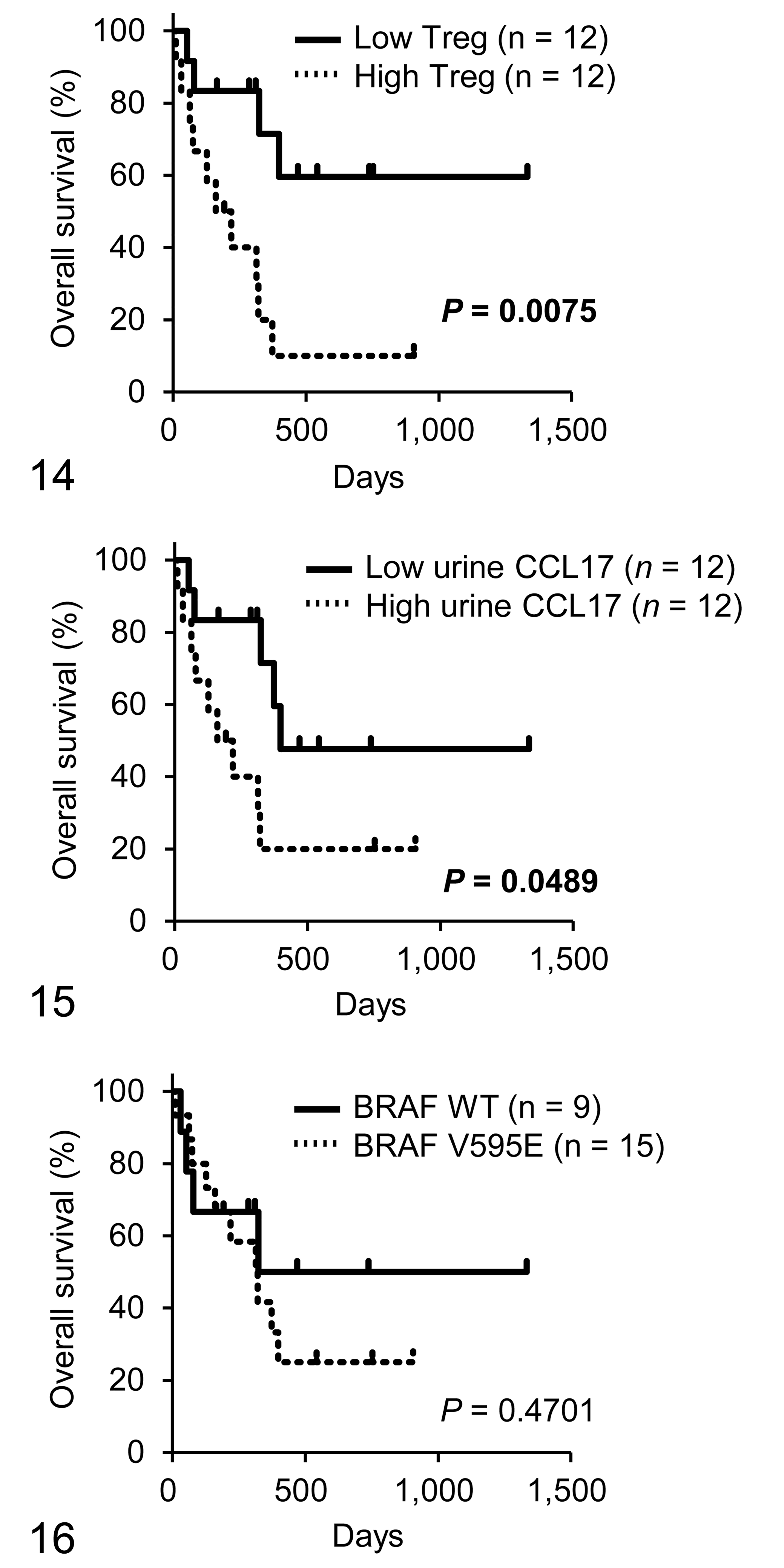
Kaplan-Meier analyses of overall survival of dogs with urothelial carcinoma according to the number of regulatory T cells (Tregs; Fig. 14), urine CCL17 concentration (Fig. 15), and BRAFV595E mutation (Fig. 16).
BRAF Inhibitor Decreases CCL17 Production in Canine Urothelial Carcinoma Cells With the BRAFV595E Mutation
We used 2 canine urothelial carcinoma cell lines, named, Love and K9TCC-Sh, for in vitro BRAF inhibition experiments. A digital PCR assay confirmed that the BRAFV595E mutation was observed in Love cells, whereas K9TCC-Sh cells had wild-type BRAF (data not shown). These cell lines were treated with the BRAF inhibitor dabrafenib to examine the effect of BRAF inhibition against canine urothelial carcinoma. A cell proliferation assay showed that Love and K9TCC-Sh cells were relatively resistant to dabrafenib (Fig. 17). In K9TCC-Sh (cells with wild-type BRAF), dabrafenib showed no antitumor effect even at the highest dose (100 μM). Western blot analysis revealed that dabrafenib completely inhibited phosphorylation of ERK, the downstream signaling pathway of BRAF, in Love cells but not in K9TCC-Sh cells (Fig. 18). Higher CCL17 production was detected in Love cells in comparison with K9TCC-Sh cells (Fig. 19). Dabrafenib showed dose-dependent inhibition of CCL17 production in Love cells but not in K9TCC-Sh cells (Fig. 19).
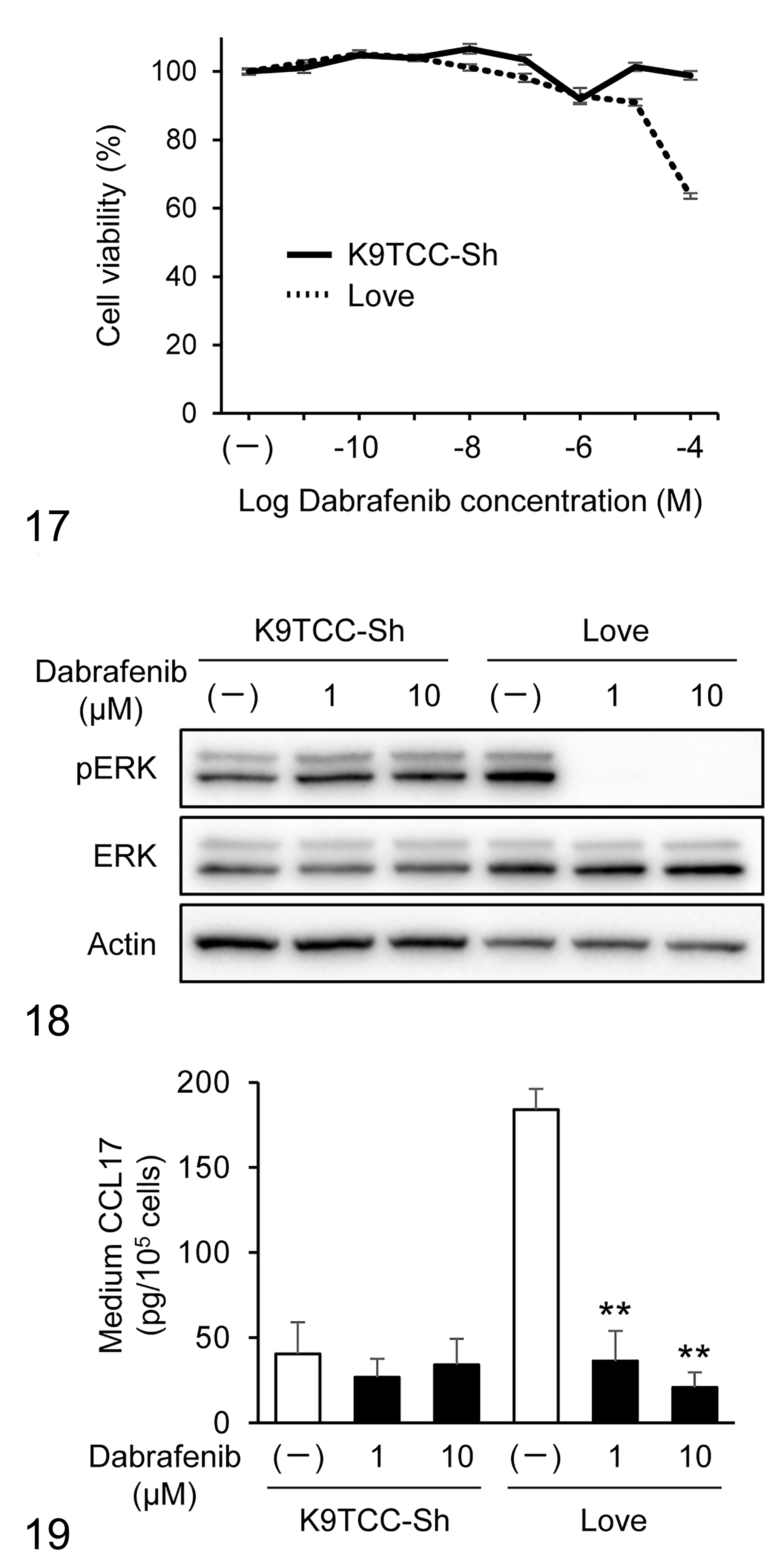
Cell viability of canine urothelial carcinoma cell lines after treatment with dabrafenib. K9TCC-Sh (with wild-type BRAF) and Love (with BRAFV595E mutation) cells were treated with 0.01 nM to 100 μM dabrafenib. After 24 hours, cell viability was determined. Data are presented as mean ± SEM.
Discussion
In this study, we have shown that the BRAFV595E mutation is associated with CCL17 expression and Foxp3+ Treg infiltration in dogs with urothelial carcinoma. Furthermore, BRAF inhibitors attenuated downstream ERK phosphorylation and CCL17 production in canine urothelial carcinoma cells with the BRAFV595E mutation. These results suggest that the high CCL17-mediated Treg recruitment in canine urothelial carcinoma is caused, at least in part, by BRAFV595E mutation-driven oncogenic MAPK signaling. BRAF inhibitors may exert antitumor activity by affecting antitumor immunity rather than directly inhibiting tumor cell growth.
Evidence suggests that tumor-infiltrating Foxp3+ Tregs correlate with a poor prognosis in certain human and canine tumors. 1,2,10,13 –15,17,18,23,33,34,45 In this study, we showed that tumor-infiltrating Tregs and urine CCL17 concentration correlated with shorter overall survival in dogs with urothelial carcinoma, suggesting that tumor-produced CCL17-mediated Treg recruitment is associated with adverse outcomes. Contrary to our expectation that BRAFV595E mutation would be a negative prognostic factor in canine urothelial carcinoma, we failed to identify a significant correlation between BRAFV595E mutation and overall survival. The possible explanations for the lack of correlation are as follows: (1) the sample size might be too small to detect a significant correlation or (2) BRAFV595E mutation might not directly correlate with prognosis because mechanisms other than BRAFV595E mutation might influence CCL17 expression. Additional studies are needed to clarify the association between BRAFV595E mutation and prognosis.
Prostaglandin E2 (PGE2) is a major arachidonic acid-derived lipid mediator that is abundantly produced by cyclooxygenase-2 (COX-2) upon inflammation. 41 The biological effects of PGE2 are mediated through 4 types of receptors, EP1–4. 44 In human dendritic cells, CCL17 expression is upregulated by PGE2 through the EP2 and EP4 receptors. 4,25 In dogs with urothelial carcinoma, COX-2 overexpression and subsequent PGE2 overproduction are observed. 5,16,31,52 In addition, our group, using RNA sequencing, showed that the EP2 receptor was the most activated upstream regulator in canine urothelial carcinoma, 24 indicating that COX-2-derived PGE2/EP2 signaling plays a role in the pathogenesis of canine urothelial carcinoma. A recent study demonstrated that BRAFV595E mutation promotes COX-2 expression and PGE2 production in canine urothelial carcinoma cell lines. 51 This evidence collectively suggest that a BRAFV595E mutation-induced COX-2/PGE2/EP2 pathway is involved in CCL17 expression and Treg infiltration in dogs with urothelial carcinoma. Mechanisms linking BRAFV595E mutation and upregulated CCL17 expression in canine urothelial carcinoma will require further investigation.
Although the numbers of tumor-infiltrating Tregs and CCL17 expression were increased in urothelial carcinoma cases with BRAFV595E mutation, some cases with wild-type BRAF showed increased Treg infiltration and CCL17 expression. This phenomenon may be due to other mechanisms leading to the activation of the MAPK pathway and subsequent CCL17 expression. Previous studies have shown that dogs with urothelial carcinoma overexpress several receptor tyrosine kinases, namely, HER2, EGFR, and PDGFR-β. 12,24,46,49 These receptor tyrosine kinases can activate downstream signaling cascades such as the MAPK pathway. Thus, additional comprehensive studies are needed to evaluate BRAF genotype, expression status of receptor tyrosine kinases, activation of the MAPK pathway, CCL17 expression, and Treg infiltration simultaneously in dogs with urothelial carcinoma.
Regimens of chemotherapy, nonsteroidal anti-inflammatory drugs, and a combination of these are considered the mainstay treatment in dogs with urothelial carcinoma. Several studies have shown the efficacy of these regimens for dogs with urothelial carcinoma; however, the median overall survival with medical treatment is less than 1 year. 11,19 We have recently shown that anti-CCR4 treatment exerts an antitumor effect in dogs with urothelial carcinoma by blocking Treg infiltration with the CCL17/CCR4 pathway. 21 The objective response rate of anti-CCR4 treatment was 71%, and the median overall survival was 474 days. Further analyses revealed that the BRAFV595E mutation was associated with clinical response to anti-CCR4 treatment in a cohort of canine urothelial carcinoma cases (Supplemental Fig. S5), indicating the involvement of BRAFV595E in Treg infiltration via the CCL17/CCR4 pathway. It will be of great interest to investigate whether BRAF inhibitors can enhance the clinical efficacy of anti-CCR4 therapy or other treatment regimens.
The present study showed that CCL17 concentration in urine of dogs with urothelial carcinoma was significantly higher than that in healthy control dogs and dogs with nonneoplastic urinary tract diseases, suggesting the diagnostic potential of urine CCL17 for urothelial carcinoma. The cutoff value for CCL17 of 100 pg/mg creatinine could differentiate urothelial carcinoma from healthy or nonneoplastic urinary tract diseases with sensitivity of 51% and specificity of 87% (Supplemental Table S2). A previous study has shown that urine CCL2 concentration as a diagnostic biomarker for urothelial carcinoma shows a sensitivity of 95% and specificity of 38%. 40 The simultaneous analysis of urine CCL17, which has a relatively high specificity, and CCL2, which has high sensitivity, may serve as a more accurate biomarker for urothelial carcinoma.
We failed to identify a significant difference in urine CCL17 concentrations between dogs with urothelial carcinoma and polypoid cystitis. This result could be attributed to the small sample size or the elevated CCL17 expression as a result of nonspecific inflammation. Indeed, urine CCL17 concentrations were slightly increased in some cases with bacterial cystitis as well as polypoid cystitis. More extensive studies will be necessary to evaluate the utility of urine CCL17 for diagnosis of urothelial carcinoma.
The limitation of this study is that only one section was used for immunohistochemistry in each case. Although Tregs were counted in multiple fields, it is possible that we could not accurately evaluate the uneven distribution of Tregs across other sections. Another limitation is that healthy control dogs were not well-matched to the clinical cases by breed. Since it was impossible to obtain normal bladder tissues from various breeds matched to the clinical cases, all healthy control dogs used in immunohistochemical assessments and quantitative RT-PCR were of the same breed (beagle). This limitation could lead to erroneous results regarding Treg number and gene expression, which might differ across breeds. Further research is needed to investigate the breed differences in Treg infiltration and CCL17 expression in the bladder.
In conclusion, this study illustrates a link between the BRAFV595E mutation and Treg recruitment via CCL17 production in dogs with urothelial carcinoma. Furthermore, a BRAF inhibitor dabrafenib reduced CCL17 production in BRAFV595E urothelial carcinoma, and may thereby attenuate Treg infiltration. These findings pave the way for clinical trials of BRAF blockade immunotherapy for canine urothelial carcinoma.
Supplemental Material
Supplemental Material, sj-pdf-1-vet-10.1177_0300985820967449 - BRAFV595E Mutation Associates CCL17 Expression and Regulatory T Cell Recruitment in Urothelial Carcinoma of Dogs
Supplemental Material, sj-pdf-1-vet-10.1177_0300985820967449 for BRAFV595E Mutation Associates CCL17 Expression and Regulatory T Cell Recruitment in Urothelial Carcinoma of Dogs by Shingo Maeda, Ryohei Yoshitake, James K. Chambers, Kazuyuki Uchida, Shotaro Eto, Namiko Ikeda, Takayuki Nakagawa, Ryohei Nishimura, Yuko Goto-Koshino, Tomohiro Yonezawa and Yasuyuki Momoi in Veterinary Pathology
Footnotes
Acknowledgements
We thank Dr Deborah W. Knapp, Department of Veterinary Clinical Sciences, Purdue University, for kindly providing K9TCC-Sh cells.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the JSPS KAKENHI Grant-in-Aid for Young Scientists (A) (Grant No. JP16H06208), the Grant-in-Aid for Scientific Research (A) (Grant No. JP19H00968), and the Anicom Capital Research Grant (EVOLVE).
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
