Abstract
During a 19-month period, 5 smooth green snakes (Opheodrys vernalis) maintained as an ex situ conservation colony presented with rapid clinical progression of locally invasive oropharyngeal squamous cell carcinoma. All 5 originated from the same wild source and were housed together or in close proximity. An infectious cause was considered likely, and nested conventional polymerase chain reaction (PCR) and in situ hybridization confirmed the presence of a novel alphaherpesvirus, Opheodrys herpesvirus 1, in the neoplastic tissue in 4 of the 5 snakes. Retrospective screening of previously submitted smooth green snakes by in situ hybridization did not detect virus in prior submissions from the colony. This report documents molecular characterization of an ophidian herpesvirus as well as colocalization of its viral nucleic acid with neoplastic transformation in snakes.
Keywords
Smooth green snakes (Opheodrys vernalis) are widely distributed in North America but currently are declining in number and geographic range. The species is threatened by habitat destruction, road and human encounters, and reduction of invertebrate prey due to drought, climatic extremes, and the use of pesticides. O. vernalis is a small snake (average weight, 15 g) with moderate reproductive output, high hatchling and juvenile mortality, and a low rate of dispersal out of occupied habitats, which makes them at further risk for population decline. A program of capture, captive breeding, and reintroduction was initiated to protect gravid females and increase hatching and juvenile survival rates, therefore ameliorating the stress placed on the local wild populations in multiple capture sites in northeastern Illinois. 16
Neoplasia is a common occurrence in reptiles, including snakes. 3,7,10,21 Squamous cell carcinoma (SCC), in particular, has been reported repeatedly in snakes. 2,3,7,10,21,22 SCC is a malignant, locally invasive neoplasm that can be quite regionally extensive and is frequently found in the oral cavity in snakes. 7 ,21 Advanced age and chronic stomatitis seem to be predisposing risk factors in this taxon. 7
An association between viruses and oncogenesis in reptiles has been described for multiple species. Perhaps the best known and characterized example is the association of chelonid herpesvirus 5 and related scutaviruses and cutaneous and visceral fibropapillomas in green (Chelonia mydas), loggerhead (Caretta caretta), olive ridley (Lepidochelys olivacea), Kemp’s ridley (Lepidochelys kempii), and leatherback (Dermochelys coriacea) sea turtles. 1,9,11 Herpesviruses and papillomaviruses have been associated with cutaneous fibropapilloma formation in green lizards (Lacerta viridis) and spiny-tailed lizards (Uromastyx acanthinura), respectively. 6,13 Although viral infections have been suggested as causes of SCC in reptilians, an association has not been documented. Papillomaviruses have been detected via PCR from tissues of a cutaneous SCC in a Dumeril’s boa (Acrantophis dumerili). 6 Herpesviruses have been associated with chronic stomatitis in plated lizards (Gerrhosaurus major and Gerrhosaurus nigrolineatus). 26 In green tree monitors (Varanus prasinus), a herpesvirus was detected in animals with proliferative stomatitis and occasionally neoplastic transformation of the oral mucosa, and the virus was localized to the lesion using in situ hybridization (ISH). 25
A population of 26 O. vernalis, both wild caught and captive born, were housed in multiple small groups at a single institution for captive breeding and eventual reintroduction. Among this group, 5 snakes (3 males, 2 females; 4–5 years of age at death) in the span of 19 months (December 2014 to July 2016) presented with rapid clinical progression of locally invasive oropharyngeal SCC. At presentation, 2 snakes had extensive oral lesions that were deemed untreatable, and these animals were euthanized. The remaining 3 snakes were initially treated for infectious stomatitis, but clinical progression was rapid despite treatment, and all were subsequently euthanized. Time from presentation to euthanasia varied from 1 day to 10 weeks. All 5 snakes had similar gross lesions of 1 or more masses arising from the maxillary and or mandibular oropharyngeal mucosa, which were pale tan, multinodular, and had adherent surface accumulations of brown friable material. Histologically, oropharyngeal mucosa and submucosa, and in some cases portions of maxillary or mandibular bone, were regionally distorted or effaced by a nonencapsulated, poorly demarcated, and invasive neoplasm (Fig. 1) composed of anastomosing trabeculae, cords, and islands of neoplastic epithelial cells with scant supporting fibrovascular stroma (Fig. 2). Cells were arranged in up to 12 disorganized layers with minimal to large regions of squamous differentiation (Fig. 3). Cells were polygonal, with abundant eosinophilic cytoplasm and distinct margins. Many cases had variably sized regions with a distinct basal layer of smaller cells with scant cytoplasm. Nuclei were oval with vesicular chromatin and 1 or 2 large nucleoli. Mitoses ranged from 20 to 33 in 10 (400×) high magnification fields. Anisocytosis and anisokaryosis were moderate. Abrupt squamous differentiation, keratin pearls, prominent intercellular bridges, mild heterophilic inflammation, and surface compact keratin layers were observed in 1 or more cases. The mucosal aspect of some masses was necrotic with thick surface accumulations of mixed necrotic cellular debris, degenerate heterophils, and macrophages. No intranuclear inclusion bodies were observed.
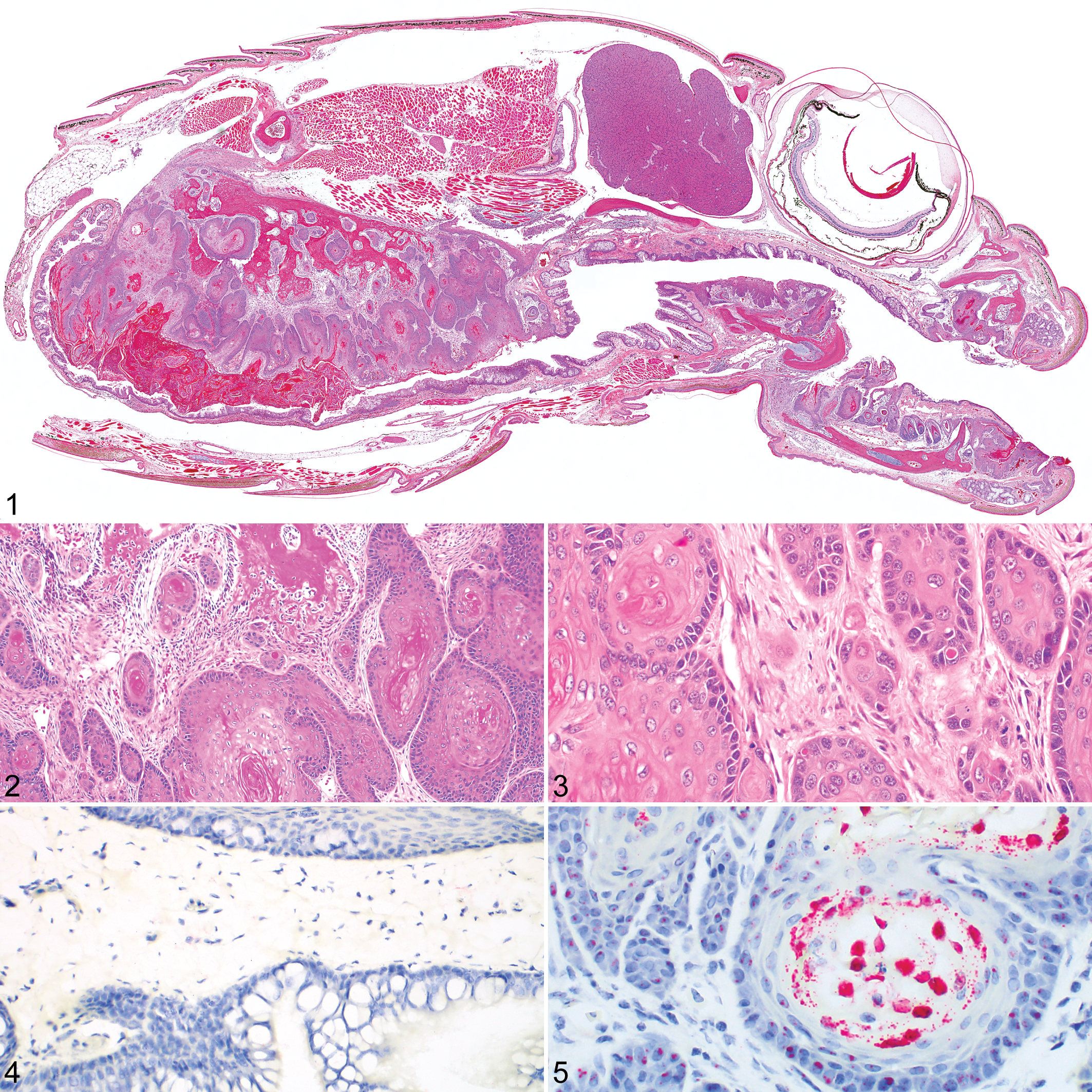
Oropharyngeal squamous cell carcinoma (SCC), smooth green snake.
Retrospective review of pathology reports for this species from August 2012 to December 2016 identified 10 additional snakes with oral lesions. All were under 3 years of age, and all had histories of chronic stomatitis. Squamous metaplasia was noted in 3 individuals, and neoplastic transformation of the chronically inflamed mucosa was identified in 2 additional snakes.
Because of the abrupt change in frequency and presentation of oral SCC in multiple individuals, a viral cause was suspected. Nucleic acids were extracted and purified from frozen neoplastic oral tissue and liver that was available from 2 snakes using a commercial kit (QIAamp DNA mini kit, Qiagen, Valencia, CA) according to the manufacturer’s instructions. Nested PCR for a fragment of the herpesviral DNA-dependent DNA polymerase was performed using previously reported methods. 15,23 Emydid herpesvirus 1 and a water extraction control were used as positive and negative controls, respectively. An amplicon of approximately 500 bp was visualized with gel electrophoresis from neoplastic oral tissue of both snakes; no bands of appropriate size were visualized from liver samples. Positive samples were treated with a commercial enzymatic cleanup reagent (ExoSAP-IT, Affymetrix, Cleveland, OH) and submitted for Sanger sequencing (University of Chicago Comprehensive Cancer Center DNA Sequencing Facility). Sequences were edited, trimmed, and assembled using commercial bioinformatics software (Geneious version 10.1.2, BioMatters, Auckland, New Zealand) into a 479-bp fragment (GenBank accession no. MH299816). Basic local alignment search tool analysis of the nucleotide sequence of the fragment revealed low sequence identity to short fragments of other known herpesviruses. Basic local alignment search tool protein analysis of the predicted amino acid sequence of the herpesviral DNA polymerase fragment revealed ∼47%–59% sequence identity to many alphaherpesviruses, including but not limited to Tursiops truncatus alphaherpesvirus 1 (59%; AAU13922.1), Glyptemys herpesvirus 2 (55%; AIY27716.1), Crocodyline herpesvirus 3 (55%; ALI97594.1), and human alphaherpesvirus 1 (53%; AKE48609.1).
To confirm the presence of the virus, hereafter referred to as Opheodrys herpesvirus 1, within neoplastic cells, RNAScope ISH (Advanced Cell Diagnostics, Newark, CA) 24 was performed using a Leica Bond RX on all 5 snakes with oropharyngeal SCC, the 10 additional O. vernalis with histories of oral inflammation, and an unaffected (control) O. vernalis from the same collection. For all snakes, RNAScope ISH was performed on tissue sections of the head, including the oropharyngeal mucosa. RNAScope ISH was also performed on a full set of tissues from 1 snake with oropharyngeal SCC and the unaffected control. No positive staining was noted in the oral mucosa of the unaffected snake (Fig. 4) or in any of the 10 snakes with histories of oral inflammation; additionally, no positive staining was noted in any of the other tissues examined from the unaffected snake. In the neoplastic oropharyngeal epithelium of 4 of 5 of the snakes (80%) with SCC, strong, positive intracellular staining was noted (Fig. 5). Within the neoplastic mucosa and islands of neoplastic epithelial cells, basal cells demonstrated punctate intranuclear positive ISH staining. In keratinizing epithelial cells along the mucosal surface or at the center of keratin pearls, positive ISH staining was present throughout the cytoplasm of the cells. Positive ISH staining was also noted within the nucleus of small numbers of exocrine pancreatic cells and mononuclear cells in the spleen in the additional tissues examined from 1 snake with SCC (data not shown).
This case series documents a novel alphaherpesvirus, Opheodrys herpesvirus 1, in oropharyngeal SCC of smooth green snakes (O. vernalis). Virus was detected by ISH in neoplastic tissue of 4 of 5 affected snakes and in no snakes with histories of chronic stomatitis. No frozen tissues were available for the single ISH-negative snake with uncomplicated oropharyngeal SCC. The neoplasm in that snake may represent unrelated spontaneous transformation, or it may be a result of viral infection below the detection limit of the ISH assay. Although a herpesvirus has been previously associated with stomatitis and oral SCC in lizards, this is the first documentation of a potential relationship between SCC and herpesviruses in ophidians. Moreover, although herpesvirus-like particles have been previously observed ultrastructurally in hepatocytes of 2 boa constrictors (Boa constrictor), endothelial cells of 16 captive horned vipers (Vipera ammodytes ammodytes), and Siamese cobra (Naja naja kaouthia) venom, 4,8,19 Opheodrys herpesvirus 1 is the first ophidian herpesvirus with partial molecular characterization. The amplified portion of the DNA-dependent DNA polymerase of Opheodrys herpesvirus 1 had highest amino acid similarity to other alphaherpesviruses. The predicted amino acid sequence was aligned to the homologous sequences of 69 other herpesviruses with multiple alignment using fast Fourier transform. 12 Maximum likelihood (gamma distributed rate variation with estimate of proportion of invariable sites with 1000 resamplings) and Bayesian (Mr. Bayes 3.2.6 with mixed amino acid substitution models, gamma distributed rate variation, 4 chains of 2 × 106 generations with 25% burn-in) were performed on the CIPRES server. 14,17,20 The results of the Bayesian (with posterior probabilities; Fig. 6) and maximum likelihood (data not shown) analyses placed Opheodrys herpesvirus 1 as the most basal alphaherpesvirus, lying between β and γ herpesviruses and other alphaherpesviruses previously identified in lizards. The identification of Opheodrys herpesvirus 1 may be important for increased understanding of the evolution of alphaherpesviruses.
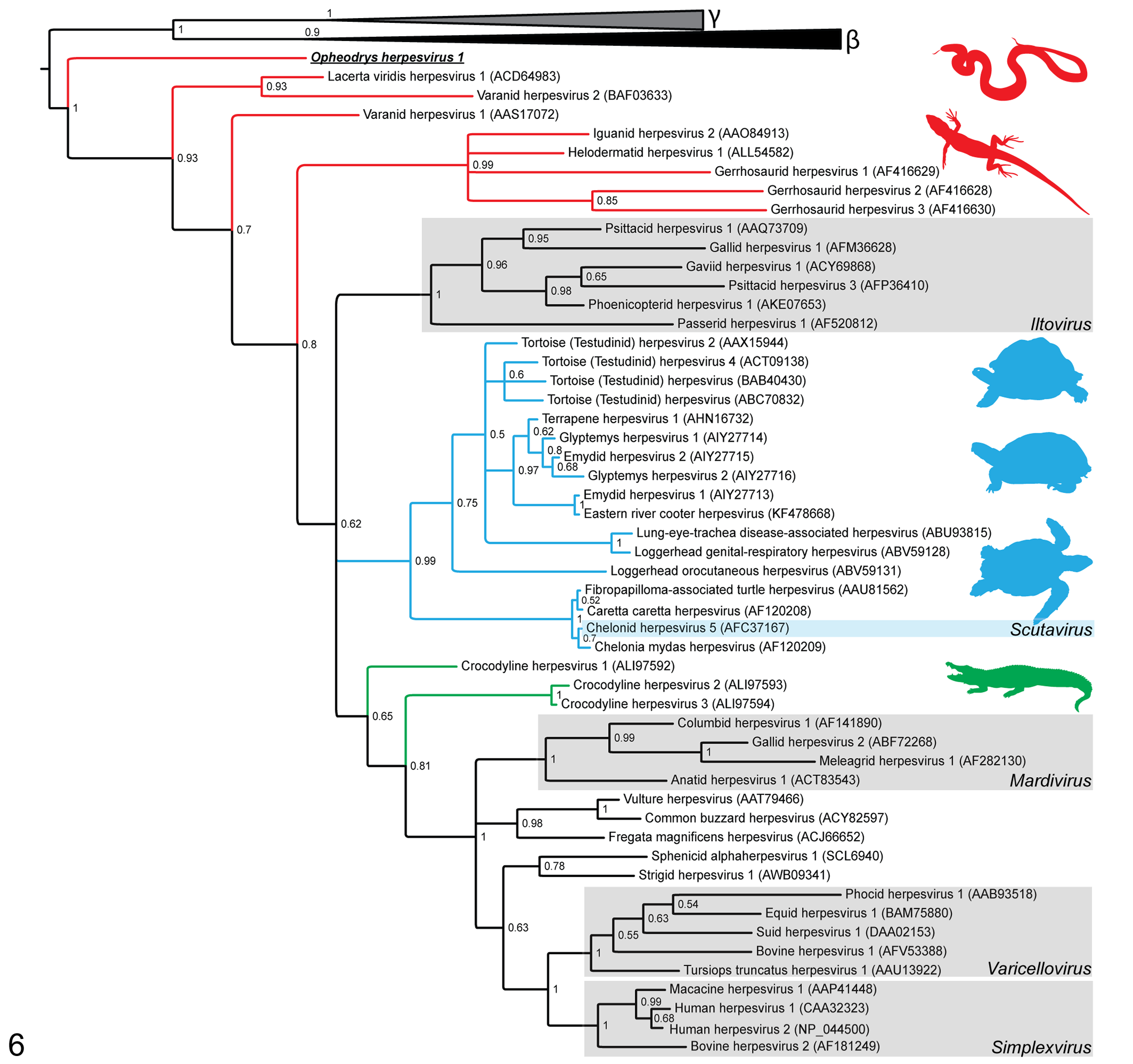
A midpoint rooted Bayesian phylogenetic tree showing the relationship of Opheodrys herpesvirus 1 (boldface and underlined) to other herpesviruses. Posterior probabilities are shown at branch points. Herpesviruses of squamates (snakes and lizards) are highlighted with red branches, chelonians (turtles and tortoises) with blue branches, and crocodilians with green branches. The recognized alphaherpesvirus genera are indicated by shaded boxes. The phylogenetic relationships of analyzed gammaherpesviruses (γ; grey triangle including AAK95475, AF319782, ADZ99583, AFM56565, NP_047983, NP_065512, and YP_438136) and betaherpesviruses (β; black triangle including ADK70825, AEW46233, AFL03576, AY728086, CCE57390, NP_042931, and YP_073778) are collapsed.
The source of Opheodrys herpesvirus 1 in this population is unclear. Affected snakes were 4 to 5 years of age at the time of death and had been housed for years prior to presentation communally with both affected and unaffected animals. All but 1 was hatched in captivity from eggs laid by dams that were gravid at time of capture; the fifth snake was wild caught. Only a single dam of the affected snakes had been maintained in captivity, which later died with esophagitis due to a penetrating foreign body. There is no information about the eventual morbidity or mortality of the other dams. Further complicating this matter is the ability of herpesviruses to become latent in some species, making detection difficult and shedding intermittent. Three of the SCC snakes were hatched in captivity and had no direct contact with their dams. It is postulated that if the herpesvirus infection was acquired from the dam, it would have been vertically passed into the developing yolk mass. Vertical transmission of herpesvirus is reported in placental animals, 5,18 but limited information is available for vertical spread in oviparous species. All dams, and the 1 wild-caught snake, came from the same capture site in the same county; no snakes from other geographically distant capture sites were diagnosed with SCC.
The impact of this disease for the free-ranging population is unknown. The multiple cases of SCC were used as justification to those involved in the reintroduction project to prevent further release of snakes from captivity into the wild population, even before an infectious etiology was confirmed. However, as all snakes with SCC were of advanced age (mean, 4–6 years; average life span in captive population, 5–6 years), it was not clear whether SCC significantly affected the life span of the captive population. Comorbidities are not elucidated at this time, but none of the snakes that presented with SCC had been clinically ill previously. Because the rate and mode of transmission are not known, it is unclear how many of the remaining snakes might be affected, were infected at release, or were infected originally in the wild. As noted clinically, overt external evidence of the oropharyngeal neoplasms is difficult to appreciate. There would be difficulties observing wild individuals without capture and detailed examination, an action which would undoubtedly adversely affect a threatened population; therefore, no monitoring in the wild is occurring. At this time, snakes from this refugia population are no longer being released.
Footnotes
Acknowledgements
We would like to thank the histology staff at the Veterinary Diagnostic Laboratory at the University of Illinois Urbana-Champaign for slide preparation, Melissa Brown of the histology laboratory at the University of Florida’s College of Veterinary Medicine for ISH slide processing, and the Zoological Pathology Program faculty members and residents who performed the necropsies and histopathologic evaluations included in this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
