Abstract
Equus caballus papillomavirus 2 (EcPV2) has been proposed as an etiologic agent for genital squamous cell carcinoma (SCC), the most common malignant tumor of the horse penis. EcPV2 is commonly detected by polymerase chain reaction (PCR) on normal horse genitalia; therefore, unraveling the virus’ role in oncogenic transformation requires other methods of detection. In this study, a highly sensitive multiple-probe chromogenic in situ hybridization (ISH) technique was designed to recognize the E6/E7 oncogenes of EcPV2. ISH demonstrated abundant virus within 6 of 13 penile and preputial SCCs, whereas evidence of solar damage was found in 6 cases that were negative for EcPV2 by ISH. The ISH technique is valuable for studies of pathogenesis, since it demonstrates for the first time that the vast majority of neoplastic cells contain virus. Moreover, hybridization was present in all metastases examined, implying stability of E6/E7 expression in these clonal populations of neoplastic cells. This study contributes to the accumulating evidence for a causal role of EcPV2 in a subset of genital SCCs in horses.
In horses, squamous cell carcinoma (SCCs) is a common male genital tumor. 9 Epidemiologic, clinical, and experimental data so far support a causative role for Equus caballus papillomavirus type 2 (EcPV2). EcPV2 has been consistently identified in equine genital SCC and precursor lesions. 1,6 –8,10 –12,14 The oncogenic potential of papillomavirus is well recognized in the association between human papillomaviruses (HPVs) and cervical carcinoma, and HPVs are increasingly recognized in tumors of the penis, vulva, and vagina. 3 By polymerase chain reaction (PCR), EcPV2 can be present in the genital region of horses without related clinical lesions or disease, and therefore studies designed to evaluate pathogenesis must hold a higher burden of proof than simple detection of viral DNA. 1,6 –8,10 –12,14 In HPVs associated with cervical cancer, the role of the virus has been demonstrated by the expression of E6 and E7 proteins and their subsequent interactions with multiple host proteins involved in cell cycle regulation and apoptosis. 5 In addition to manipulation of the cell cycle, E6 and E7 have been shown to cause genetic instability. 4
In horses, oncogenic E6 transcript from EcPV2 was detected by reverse transcriptase PCR in genital SCCs, which suggests an active pathogenic role of the virus. 13,15 Nonetheless, because the virus replicates in surface epithelium, these techniques by themselves cannot demonstrate causality because they do not provide information on the distribution of expression within the tumor. Several studies have convincingly used oligomer in situ hybridization (ISH) to demonstrate that EcPV2 DNA is present within nuclei of a subset of tumor cells. 6,10,12 These reports have described viral nucleic acid within superficial cells of early lesions (eg, carcinoma in situ) or scattered cells within invasive tumors. We suggest that if oncogenic E6/E7 protein is essential for the development of SCC, at a minimum, viral nucleic acid and/or E6/E7 transcript should be detectable in the majority of tumor cells.
We chose a sensitive multiple gene-targeted ISH method (RNAscope; Advanced Cell Diagnostics, Hayward, CA), using probes designed to hybridize to EcPV2 E6/E7, to examine a retrospective set of male equine genital SCCs. 16 RNAscope ISH uses a patented, branched DNA technique, but its name is somewhat misleading for this study, where our target is a double-stranded virus and probes targeting viral messenger RNA (mRNA) would recognize both E6/E7 mRNA and viral genomic DNA. Nonetheless, because the primary purpose of this study was to detect even a low copy number of virus or E6/E7 transcript in clinical tissue specimens, recognition of either DNA and/or mRNA was useful. Using EcPV2 Genbank reference sequence EU503122.1, 20 probe pairs within a 972-nucleotide region spanning the E6/E7 region were designed and synthesized (Advanced Cell Diagnostics). ISH was performed on 5-μm-thick formalin-fixed, paraffin-embedded tissue sections using the RNAscope 2.0 assay according to the manufacturer’s protocol. 16 Successful hybridization requires binding of side-by-side probe pairs on the targeted stretch of nucleic acid, initiating a cascade that leads to deposition of diaminobenzidine (DAB) stain. The final deposit is brown and punctate, with a direct correlation between the amount of the deposit and the number of DNA/RNA target sequences. 16
Tissue samples were chosen from archived biopsies and/or necropsies selected from the William R. Pritchard Veterinary Medical Teaching Hospital Anatomic Pathology archives at the University of California, Davis. Thirteen genital SCCs were selected, representing ∼30% of the cases over the previous 10-year period, all from geldings. Cases were selected for quality (no autolysis and <48 hours in formalin) and the presence of intact bordering mucosa or skin. The age range at the time of diagnoses was 6 to 33 years. Anatomic locations were determined from clinical or pathology case records with 5 cases localized to penis, 2 to both penis and prepuce, and 6 to prepuce alone (cases 1–13, Table 1). A routine workup in 4 cases included papillomavirus immunohistochemistry (IHC, rb-anti-BPV1; B0580; Dako, Carpinteria, CA), all of which were negative. Twelve non-SCC cases, including normal penile skin, hyperplastic squamous epidermal skin plaques, genital and nongenital papillomas, and mucous membranes from various locations, were tested in parallel (cases 14–25, Suppl. Table S1). Two bovine papillomas were included to ensure probe specificity (cases 24–25, Suppl. Table S1). A board-certified dermatopathologist (V.K.A.) assessed epidermal pigmentation and the presence of solar damage for samples that contained peritumoral skin or mucosa. Evidence of solar damage, specifically the presence of dermal elastosis and vascular wall hyalinization, was present in 6 of 13 SCC cases in nonpigmented skin but none of the 5 SCC cases that occurred in pigmented skin (Table 1). In the remaining 2 cases, the presence or absence of pigment and/or of solar damage was masked by ulceration and/or inflammation.
In Situ Hybridization for E6/E7 Viral Nucleic Acid and Evidence of Solar Damage in Horses With Genital Squamous Cell Carcinoma.
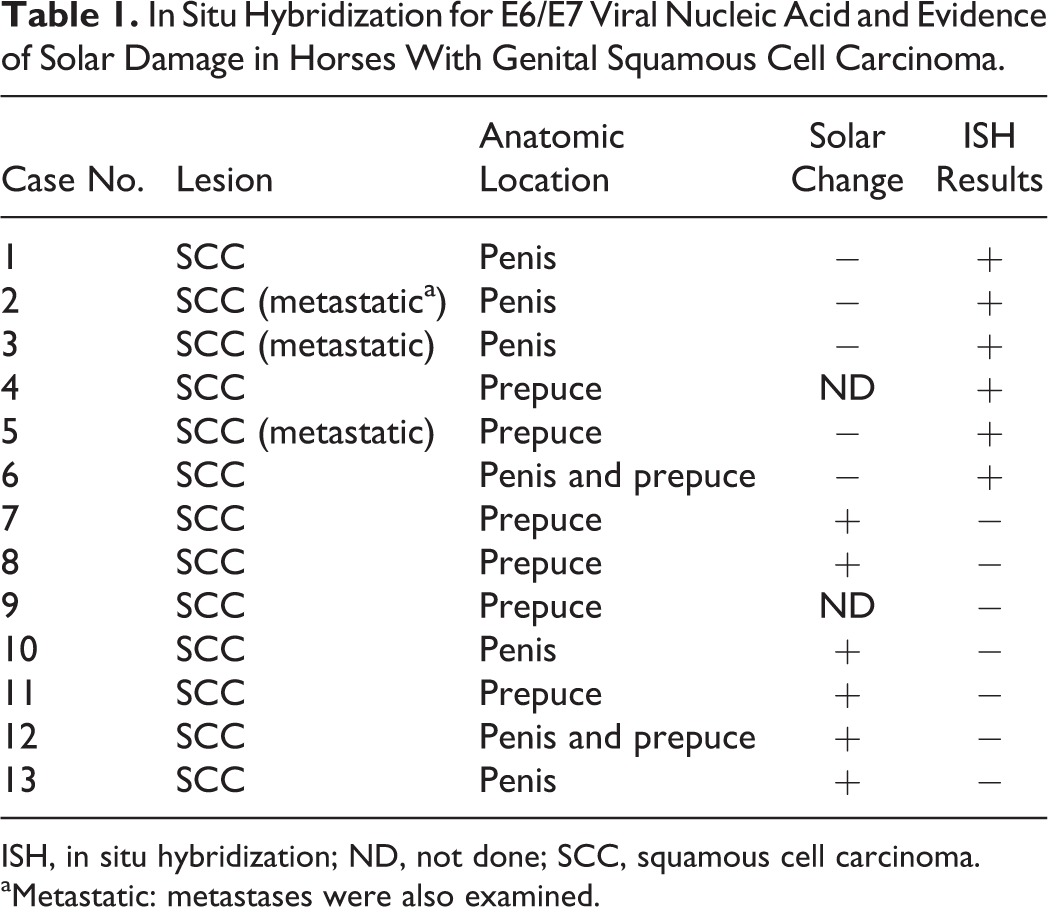
ISH, in situ hybridization; ND, not done; SCC, squamous cell carcinoma.
aMetastatic: metastases were also examined.
The pattern of E6/E7 probe hybridization in benign (nonprogressive) genital papillomas recapitulated the known natural history of productive papillomavirus infection in a stratified squamous epithelium (Fig. 1). Probe hybridization within the basal layer was present but light, often within a few adjacent basilar cells each containing a single dot (deposit) (Fig. 1c). In contrast, probe hybridization was strong in the nuclei of cells within the upper strata, where viral copy number is expected to increase (Fig. 1c).
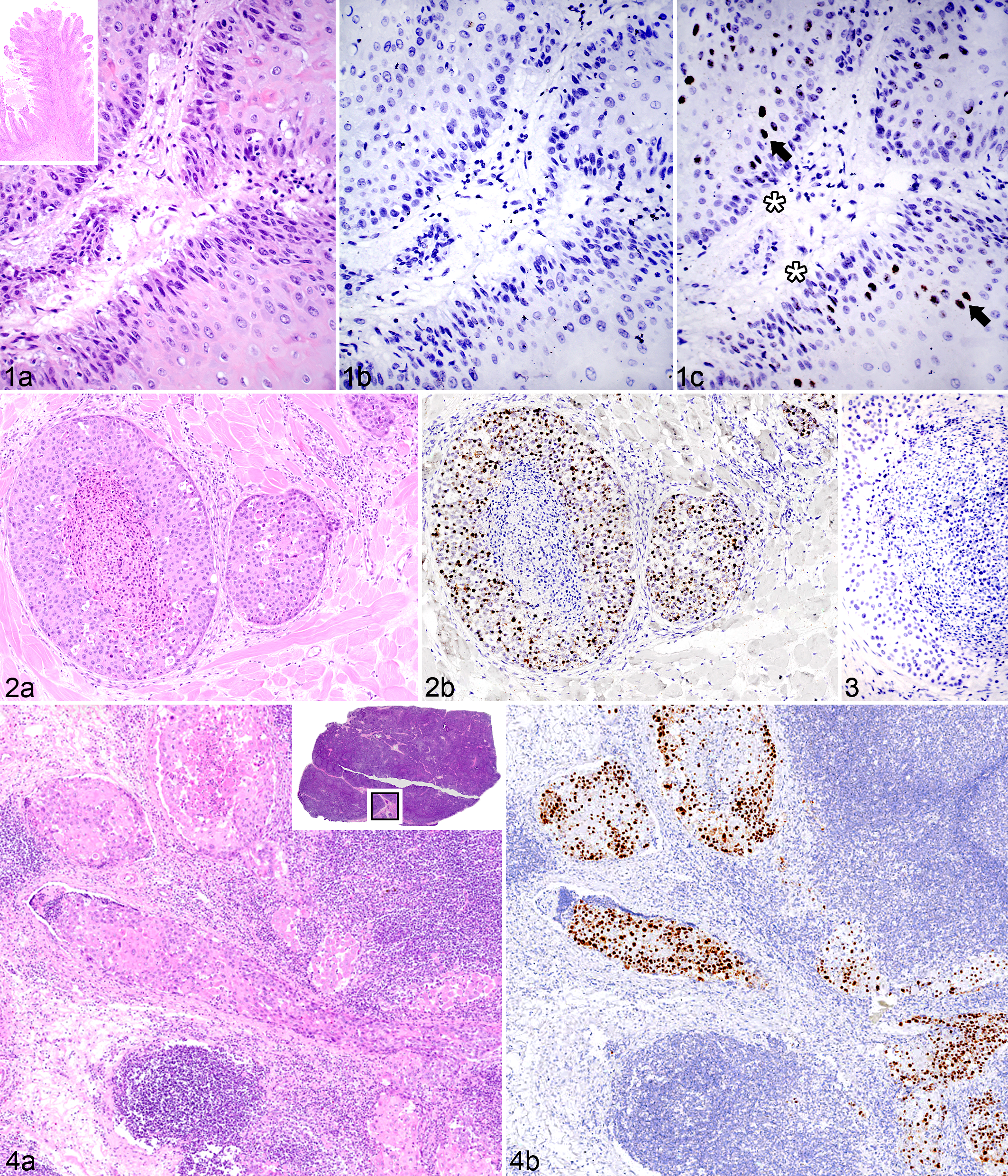
Papilloma, preputial sheath; horse No. 16. (a) Basal epithelium palisades along the fibrovascular core. Inset: Fronds of acanthotic, hyperkeratotic epidermis radiate from a central fibrovascular stalk. Hematoxylin and eosin (HE). (b) No hybridization is present using a probe for bovine papillomavirus. In situ hybridization (ISH), BPV1/2 probe. (c) EcPV2 E6/E7 probe hybridization is present in small but detectable amounts within basilar epithelial nuclei (asterisks) and more robust within nuclei of differentiating cells of the upper strata (arrows). ISH, EcPV2 E6/E7 probe.
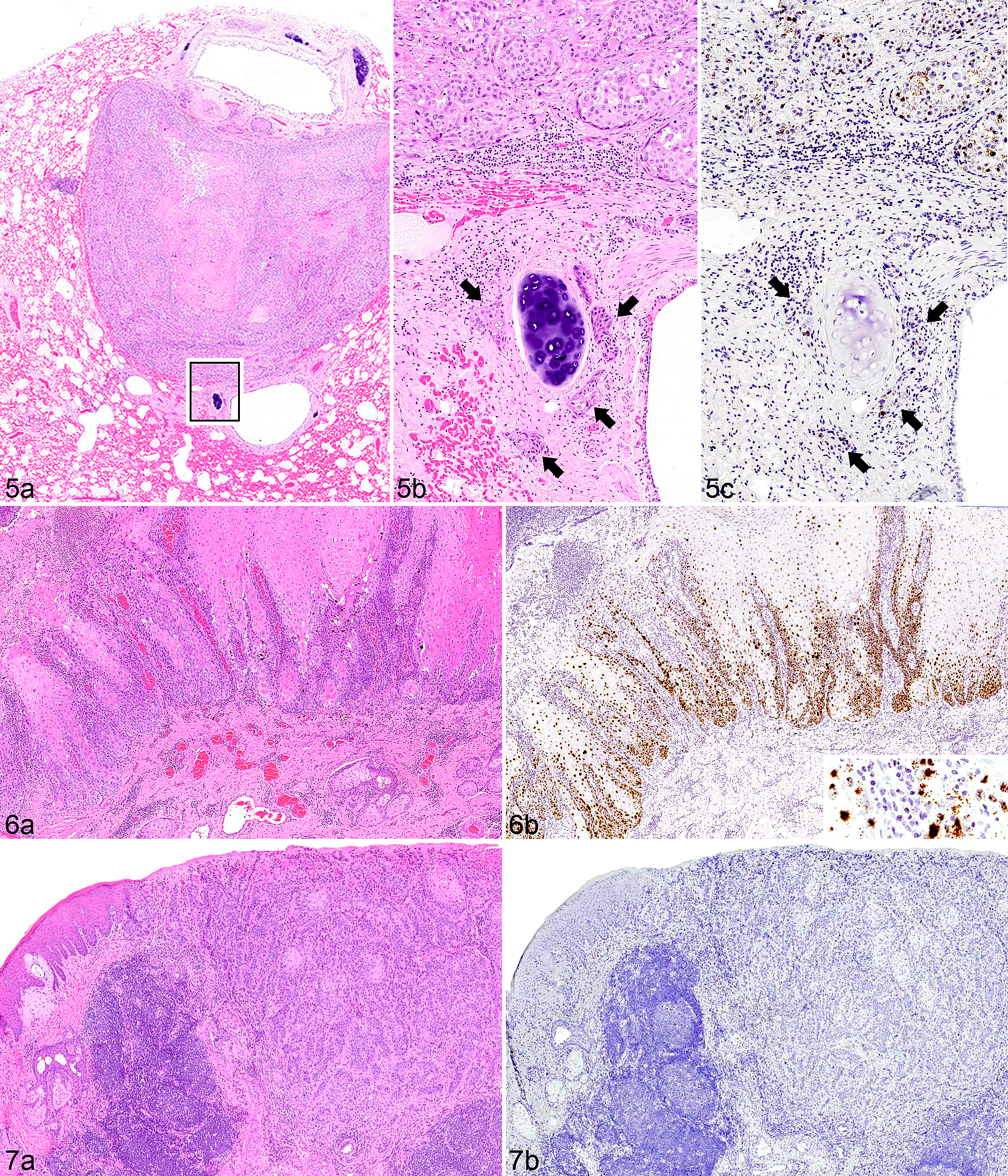
Robust E6/E7 probe hybridization was detected in 6 of 13 genital SCCs (3 penile, 2 preputial, and 1 involving both tissue types; Table 1, Figs. 2, 4, and 5). Probe hybridization was similar, strong, and primarily nuclear in all 6 cases, occurring in 80% to 100% of the neoplastic cells, often with punctate staining of the entire nucleus. Probe hybridization occurred in islands of neoplastic cells in the deep subcutis (Fig. 2b), in vessels of regional lymph nodes (Fig. 4), and in metastases (Fig. 5). There was a distinct pattern within hyperplastic mucosa adjacent to regions of invasion in which hybridization was most intense within a broad band of basilar cells and rarely within individual cells of the upper epithelial strata (Fig. 6). Hybridization was lightly scattered within the cytoplasm in some regions (Fig. 6b). Intracellular localization of the signal can be imprecise because tissue sections are boiled during the ISH procedure, which can compromise cellular microarchitecture.
Penile squamous cell carcinoma metastatic to lung; horse No. 2. (a) A nodule of neoplastic tissue between 2 bronchioles. The boxed region is shown at higher magnification in Fig. 5b, where tumor is present within peribronchiolar vessels (arrows) and replacing alveolar parenchyma (top half of image). Hematoxylin and eosin (HE). (c) E6/E7 probe hybridization is detected within the nuclei of metastatic neoplastic cells. Arrows indicate the tumor in peribronchiolar vessels. In situ hybridization (ISH), EcPV2 E6/E7 probe.
All of the SCCs that had positive hybridization of the E6/E7 probe occurred in pigmented skin or mucosa, and half of these cases were metastatic (Table 1). In the remaining 7 SCCs, no virus was detected by EcPV2 ISH probe hybridization (Fig. 7). Six of the virus-negative tumors occurred within regions that had detectable solar damage. Horses with virus-associated tumors and those with evidence of solar damage in peritumoral tissue were of similar age (average, 20 and 15 years of age, respectively) (Suppl. Table S1).
Controls included both nucleotide-matched scrambled probes (Fig. 3) and BPV probes (Fig. 1B). No control probe hybridization was detected in non-SCC lesions, normal penile skin, and preputial skin (Suppl. Table S1). BPV probe hybridization was positive in bovine papilloma and negative in all SCC tissues tested.
We performed EcPV2-specific PCR and sequencing of amplicons from SCC cases only. Total nucleic acid was isolated from scrolls of formalin-fixed, paraffin-embedded tissue using a Qiagen DNeasy Blood and Tissue Kit (Qiagen, Valencia, CA). PCR primers for EcPV2 evaluation were designed to amplify a 224-bp product of E1 between nucleotides 2107 and 2330 (Genbank reference sequence EU503122.1; EcPV2-1F: ATTACCGCAGAGCGGAGATG, EcPV2-1 R: GCTGGACTTGCCAGTGTTTG). The 25-μl reactions contained 1× buffer, 200 μM of each dNTP, 1 μM each of the primers, 1U Taq Polymerase (Qiagen), and 4 μL template. PCR conditions were as follows: 94°C for 3 minutes and 40 cycles of 94°C for 1 minute, 55.5°C for 30 seconds, and 72°C for 1 minute, followed by 72°C for 5 minutes. Amplicons were sequenced using a BigDye Terminator v3.1 Cycle Sequencing Kit (Applied Biosystems, Foster City, CA). EcPV2 DNA was amplified from all 13 equine genital SCC samples included in the study. This result is within the range of other reports, consistent with the 43% to 100% prevalence of PCR-positive cases identified in other studies. 10,11 In this study, EcPV2 DNA was detected by PCR in 7 SCCs that were negative by ISH. The reasons for this disparity are not clear, but there are several potential explanations. This result could reflect virus on the surface of the skin in the genital region. It is also possible that PCR is more sensitive than this ISH method and able to detect fewer infected cells. Last, while appropriate buffer and single primer controls were all negative, it is difficult in a retrospective study such as this to control for contamination at the level of collection and/or tissue processing.
These data show that in a subset of genital SCCs, the vast majority of neoplastic cells contain E6/E7 nucleic acid of EcPV2. There are additional key observations from this study that contribute to our understanding of the biology of EcPV2-associated tumors. First, solar damage and viral nucleic acid were detected in genital SCC in horses, but they were never detected in the same case. It is unlikely that these mechanisms are exclusive; indeed, solar change and viruses are jointly present in some SCCs of humans. 2 Second, all 3 of the metastatic tumors were associated with EcPV2. This is the first time EcPV2 has been detected in metastases using ISH, and the fact that E6/E7 segregates with a clonal population is minimal correlative evidence that continued expression could contribute to stable replication. In our records spanning the proximate 10 years, 16% of genital SCCs were metastatic. There could be different behaviors (and potentially different prognoses) for virus-associated SCCs compared with nonviral SCCs, but verification would require additional studies.
Footnotes
Acknowledgements
We thank the UC Davis VMTH Histopathology Laboratories for their technical expertise and Kevin Keel for his assistance with obtaining the images.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is funded by grants from the Bernice Barbour Foundation and Boerhinger-Ingleheim Vetmedica to the Pesavento Laboratory.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
