Abstract
Deformed wing virus (DWV) is a single-stranded RNA virus of honey bees (Apis mellifera L.) transmitted by the parasitic mite Varroa destructor. Although DWV represents a major threat to honey bee health worldwide, the pathological basis of DWV infection is not well documented. The objective of this study was to investigate clinicopathological and histological aspects of natural DWV infection in honey bee workers. Emergence of worker honey bees was observed in 5 colonies that were clinically affected with DWV and the newly emerged bees were collected for histopathology. DWV-affected bees were 2 times slower to emerge and had 30% higher mortality compared to clinically normal bees. Hypopharyngeal glands in bees with DWV were hypoplastic, with fewer intracytoplasmic secretory vesicles; cells affected by apoptosis were observed more frequently. Mandibular glands were hypoplastic and were lined by cuboidal epithelium in severely affected bees compared to tall columnar epithelium in nonaffected bees. The DWV load was on average 1.7 × 106 times higher (P < .001) in the severely affected workers compared to aged-matched sister honey bee workers that were not affected by deformed wing disease based on gross examination. Thus, DWV infection is associated with prolonged emergence, increased mortality during emergence, and hypoplasia of hypopharyngeal and mandibular glands in newly emerged worker honey bees in addition to previously reported deformed wing abnormalities.
Recently observed declines in honey bee populations have been attributed to a variety of factors, including agricultural intensification, pesticide use, and spread of disease. 16 Varroa destructor, an ectoparasitic hemophagous mite (Suppl. Figs. S1–S4), has emerged as a major threat to honey bee health since the middle of the 20th century. 12,16 Varroa has 2 life cycle phases: (1) a reproductive phase, during which the mites reproduce and feed on prepupae and pupae (collectively referred to as capped honey bee brood), and (2) a phoretic or dispersal phase, during which the mites parasitize adult bees. To begin the reproductive phase, the mite enters the brood cell just before capping (7–8 days after egg laying for worker bees) and exits with the newly emerged bee (20–21 days after egg laying for worker bees). Varroa mites preferentially reproduce in drone brood because of longer development time required for drone brood (24 days), which generates on average 2 to 2.5 mature females per reproductive cycle compared to 1.3 to 1.45 mature female offspring in worker bee brood. 12
Varroa mites affect bees in multiple ways: they consume hemolymph of developing (pupae) and adult bees, decrease immunity, and transmit viruses. 18 Among the viruses transmitted by the Varroa mites, deformed wing virus (DWV) has been closely associated with overwinter colony losses. 16 Currently, 2 main genotypes of DWV have been described, type A and type B, which differ in virulence and distribution. 7 The morphological effects of DWV infection in honey bees have been insufficiently investigated. Previous studies have identified DWV in brain, midgut epithelium and content, queen ovaries and fat body, and drone seminal vesicles. 4,13 Clinical signs of DWV infection include pupal death and, in newly emerged bees, deformed wings and bloated, discolored, and shortened abdomens. 9 However, little is known about the histological lesions caused by DWV infection. Histopathology has not been applied systematically to study bee diseases. Therefore, the purpose of this study was to characterize microscopic morphologic changes associated with natural DWV infection in newly emerged worker honey bees.
Varroa-Infested Hives
To obtain high Varroa infestation, 5 experimental hives were left untreated with miticides during the fall of 2016 and spring of 2017. Varroa mite infestation rate was measured by dislodging and counting the mites in an alcohol-containing solution. For this purpose, approximately 300 worker bees were collected in a jar containing 200 ml automotive windshield washer fluid containing methyl alcohol (Turbo Power –40°C; Recochem, Montreal, Canada). Subsequently, the jars were shaken at 200 revolutions per minute (rpm) for 20 minutes (Orbit shaker 3520; Lab-line Melrose Park, Illinois) to dislodge the mites. The bees were then washed with tap water and the mites counted in the flow-through.
Synchronization of the Age and Observation of Experimental Bees
Queens in the experimental hives heavily infested with Varroa were restricted in a cage with an empty wax comb frame for 24 hours to obtain an experimental frame of eggs of approximately the same age. After 24 hours, the experimental frame was removed from the cage and incubated within the same hive for 19 to 20 days, after which it was transferred into an observation chamber (Suppl. Fig. S5). The observation chamber was a standard wooden hive box with transparent removable wall, allowing observation and sampling of emerging bees. It was placed on top of a healthy colony, separated by a wire mesh, to ensure preservation of the microclimate environment inside the observation chamber. The time of emergence was recorded from the first signs of emergence (piercing of the wax cap) to the exit of the worker bee from the brood cell. Mortality during emergence, degree of wing deformity (according to Williams et al 17 ; Table 1), and/or presence of protruded proboscis were also recorded. The newly emerged bees were collected and fixed in formalin (10% neutral buffered) or frozen on dry ice and stored at –80°C. For histological examination and polymerase chain reaction (PCR), only normal bees (degree 0) and bees with the highest degree of wing deformity (degree 5) were used (see Table 1).
Degrees of Wing Deformity in Deformed Wing Virus–Affected Bees as Proposed by Williams et al. 17

Histology
The formalin-fixed honey bee workers (degree 5, n = 10; degree 0, n = 8) were routinely processed for histopathology with the heads, thoraces, and abdomina separately embedded in paraffin blocks. The heads and thoraces were embedded in transverse orientation, while abdomina were embedded in sagittal orientation. Heads were sectioned serially every 50 μm; thorax and abdomen were sectioned every 100 μm. Sections were mounted on glass slides and stained with hematoxylin and eosin. 15 Eyes, brain, hypopharyngeal and mandibular glands, thoracic muscles, and major abdominal organs (crop, proventriculus, ventriculus, Malpighian tubules, small intestine, and fat body) were evaluated with light microscopy. The area of the hypopharyngeal and mandibular glands in each serial section was determined using Image-Pro Premier 9.1 software (Media Cybernetics, Rockville, MD) and summed to obtain the total area for each bee (number of sections per bee, mean ± SD: hypopharyngeal glands, degree 0: 26 ± 3, degree 5: 21 ± 4; mandibular glands, degree 0: 18 ± 3, degree 5: 11 ± 2).
Real-Time Quantitative PCR
RNA was extracted from 15 affected (degree 5) and 15 nonaffected (degree 0) newly emerged adult sister bees that had been fresh-frozen (RNeasy Mini Kit; Qiagen GmbH, Hilden, Germany). Real-time quantitative PCR (RT-qPCR) with previously reported primers for DWV type A, DWV type B (VDV-1), acute bee paralysis virus (ABPV), Kashmir bee virus (KBV), Israeli acute paralysis virus (IAPV), black queen cell virus (BQCV), chronic bee paralysis virus (CBPV), sac brood virus (SBV), slow bee paralysis virus (SBPV), and β-actin was performed. 8 RNA was quantified using Bio-Rad CFX96 (Bio-Rad, Hercules, CA) and SYBR Green fluorescence reporter (Perfecta SYBR Green SuperMix; Quanta-Bio Beverly, Massachusetts) according to the manufacturers’ instructions.
Statistical Analysis
Mann-Whitney U test was performed to compare time of emergence and viral load between affected and nonaffected bees; independent sample t test was used to compare total areas of hypopharyngeal and mandibular glands. Statistical analyses were performed using SPSS 23 (SPSS, Inc, an IBM Company, Chicago, IL), and graphs were created using Prism 6 (GraphPad Software, La Jolla, CA).
The mean ± standard deviation (SD) Varroa infestation rate was 9.2 ± 3.1 mites per 100 bees, which is considered enough to lead to a colony collapse. 12 A total of 215 emerging bees were observed, 11.6% of which exhibited signs of DWV infection (Figs. 1–2). All of the affected bees, but not normal bees, also demonstrated a protruded proboscis during emergence (Figs. 3–4).

Honey bee, newly emerged with normal wings.
The median (and 25th–75th percentile) emergence time for normal sister bees (wing deformity degree 0) was 41 (31–60.5) minutes; for affected bees (wing deformity degree 1 to 5), it was 83 (45.5–182.5) minutes (Fig. 5). Thus, the emergence time for the bees with deformed wings was approximately 2 times longer (U = 1149.5, P < .05) than for nonaffected bees. Approximately 30% (30 of 93) of the affected bees died during the emergence process, during which the head emerged out of the cell but the thorax and abdomen remained inside the cell.
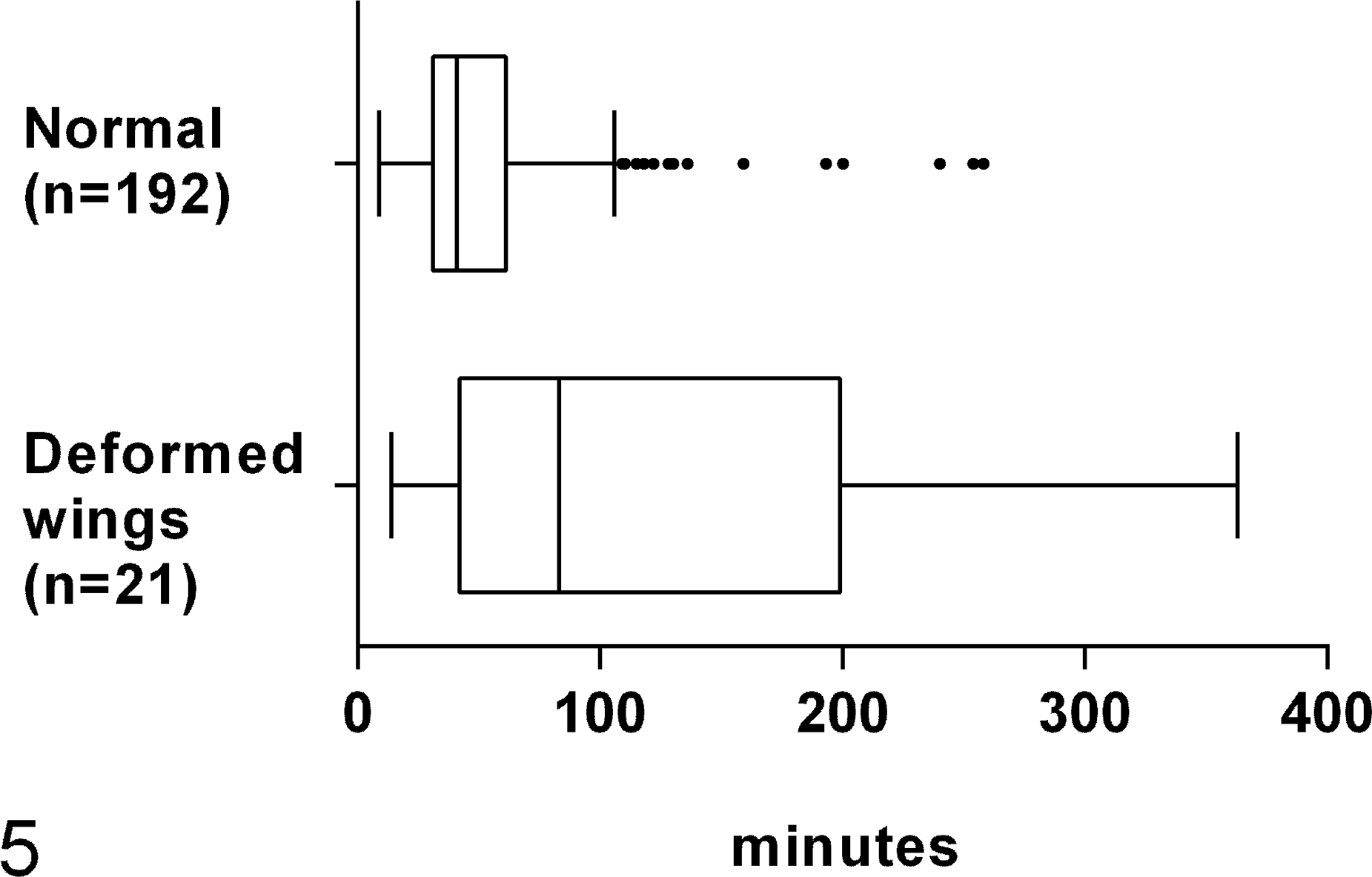
Emergence time of worker honey bees. Deformed wing virus–affected bees took approximately 2 times longer to emerge compared to normal bees (U = 1149.5, P < .05). The line represents the median, the box represents interquartile range (IQR), the whiskers represent values inside 1.5 IQR, and the dots represent values outside IQR.
Histologic appearance of hypopharyngeal and mandibular glands is presented for a normal bee (Fig. 6) and a severely affected bee (degree 5) (Fig. 7). In clinically normal bees, acini of hypopharyngeal glands consisted of cells with inconspicuous nuclei obscured by numerous intracytoplasmic secretory vesicles (Fig. 6, upper inset). In contrast, in severely affected bees (degree 5), the acinar cells had deeply basophilic cytoplasm with large coarsely granular nuclei and few vesicles, which often contained eosinophilic homogeneous material. Occasionally, the acinar cells were shrunken with condensed fragmented nuclear material (interpreted as apoptotic cells) (Fig. 7, upper inset). The total area of hypopharyngeal glands of the severely affected bees was 3.2 times smaller compared to clinically nonaffected bees (mean ± SD: 7.98 ± 2.71 mm2 in apparently normal bees, 2.50 ± 1.51 mm2 in affected bees; t 16 = 5.5, P < .001; Fig. 8).
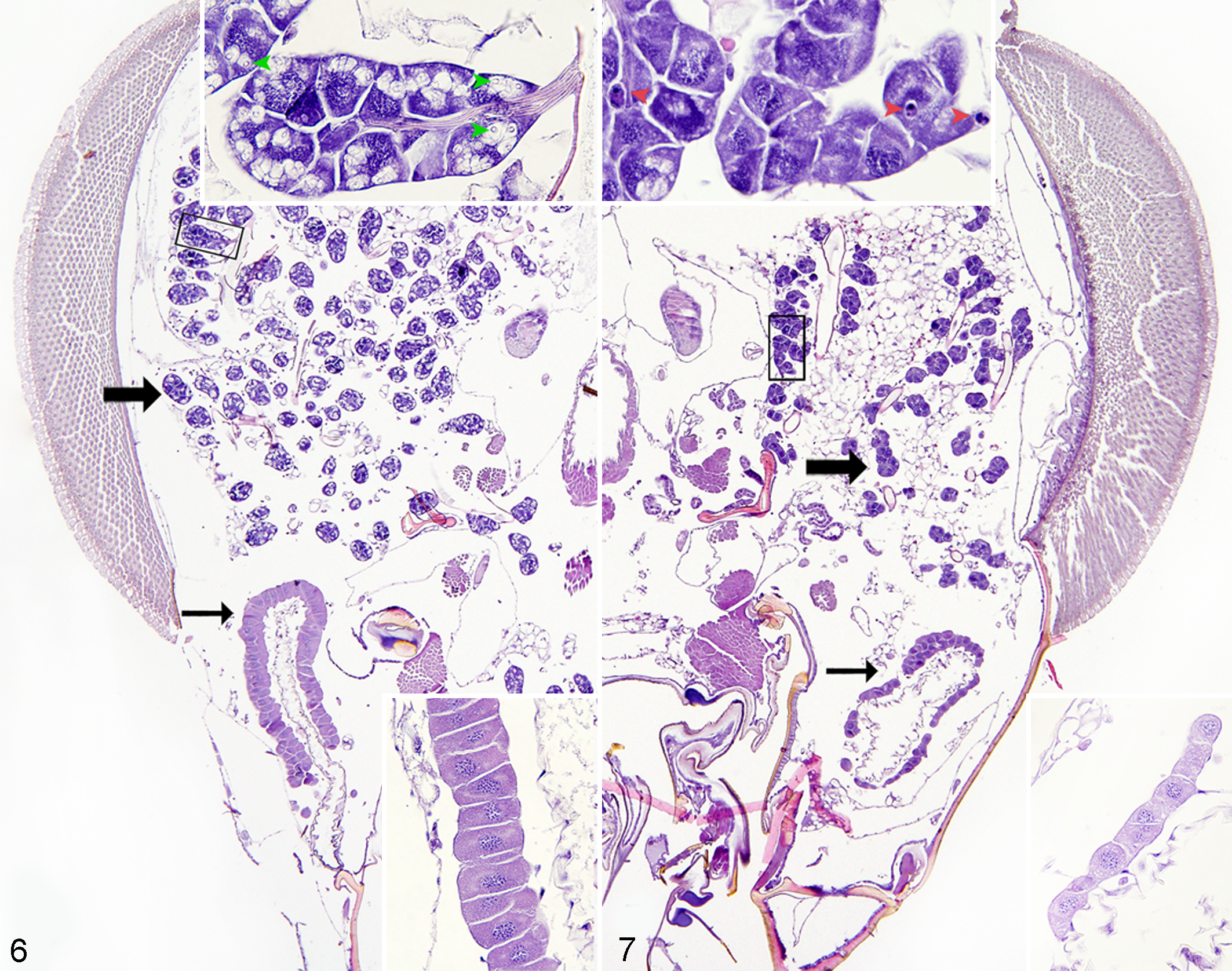
Normal worker honey bee. Hypopharyngeal (thick arrow) and mandibular (thin arrow) glands. Top inset: higher magnification of the boxed area of hypopharyngeal gland acini. Note secretory vesicles (green arrowheads). Bottom inset: higher magnification of mandibular gland showing simple columnar epithelium. Hematoxylin and eosin (HE).
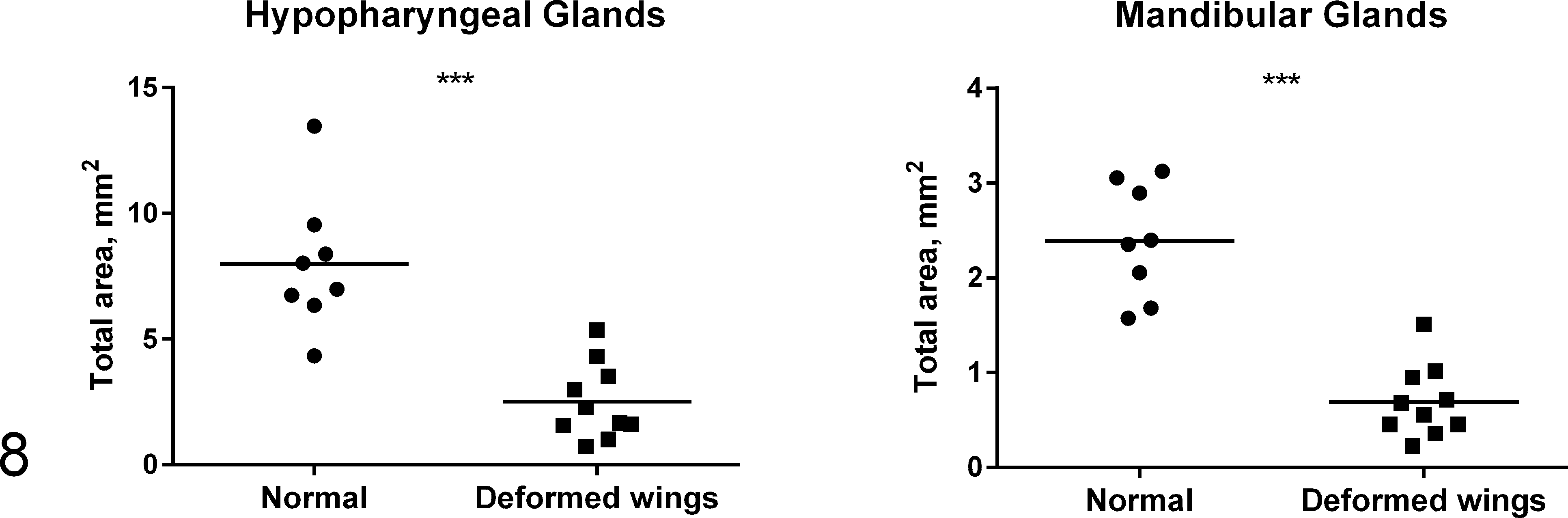
Total area of the hypopharyngeal and mandibular glands in individual affected (▪) and normal bees (•). The average total area of hypopharyngeal and mandibular glands was 3.2 and 3.5 times smaller, respectively, in affected bees compared to normal bees (***P < .001).
Mandibular glands in normal bees (degree 0) were lined by simple high columnar epithelium with amphophilic cytoplasm (Fig. 6, lower inset). In contrast, mandibular glands in severely affected bees (degree 5) were lined by cuboidal epithelium with cytoplasmic microvesiculation (Fig. 7, lower inset). The total area of mandibular glands of the severely affected bees (degree 5) was 3.5 times smaller compared to clinically nonaffected bees (mean ± SD: 2.39 ± 0.60 mm2 in normal bees, 0.69 ± 0.38 mm2 in affected bees; t 16 = 7.3, P < .001; Fig. 8). No obvious light microscopic differences between affected and nonaffected bees were found in thoracic and abdominal organs; therefore, quantitative morphometric analysis of these organs was not performed.
Affected bees had a significantly higher viral load of DWV compared to nonaffected bees with fold difference of 1.7 × 106 (U = 225, P < .001), with dCt coefficient of variation 33% and 30% for affected and nonaffected bees, respectively. PCR efficiency was 90.6% (slope –3.57) for DWV primers and 87.9% (slope –3.65) for β-actin. No DWV type 2 (VDV-1) RNA was detected. Ten of 15 affected bees and 14 of 15 nonaffected bees were also positive for BQCV, and the viral load was 44 times higher in the BQCV-positive affected bees compared to BQCV-positive nonaffected bees (U = 140, P < .001). PCR for ABPV, KBV, IAPV, CBPV, SBV, and SBPV was negative for both DWV-affected and nonaffected bees.
To our knowledge, this was the first characterization of DWV infection with histopathology. We have found that a protruded proboscis, prolonged emergence time, increased mortality during emergence, and hypoplastic hypopharyngeal and mandibular glands were associated with DWV infection.
The proboscis is a composite organ forming a tube that functions as a pump for delivering liquids to the mouth. When not in use, the retractor muscles of the labium contract to fold the proboscis behind the head. The pathogenesis of protruded proboscis and prolonged emergence time was not determined in this study. We have not been able to identify histopathologic changes in muscle or nervous tissue to explain these clinical observations; however, functional or ultrastructural changes cannot be ruled out. Alternatively, a protruded proboscis and prolonged emergence could be associated with generalized energy depletion of the affected bee caused by combined effects of the mite feeding and viral proliferation. This could also explain, at least in part, why some bees were not able to emerge at all and eventually died within the comb cells.
Based on our PCR results, we have found that DWV was the most abundant virus present in the affected as well as clinically normal bees; however, affected bees had 1.7 million times higher DWV load compared to clinically normal bees. In addition to DWV, BQCV was also identified in 67% affected and 93% clinically normal bees. There was a mild increase (44 fold) in BQCV load in deformed bees compared to clinically normal bees; however, it was much lower compared to the increase in DWV. None of the other common bee viruses were found. These results suggest that DWV infection is associated with prolonged emergence, increased mortality, protruded proboscis, and hypoplastic hypopharyngeal and mandibular glands, all of which may have negative effects on colony performance. The association of BQCV with the aforementioned pathological changes cannot be ruled out. However, since BQCV was detected only in two-thirds of the affected bees and its load was substantially lower than DWV load, it may be less likely to have the same impact as DWV.
Despite the strong association of DWV with the characteristic signs such as wing deformities, its etiological role and the pathogenesis of DWV infection are still not well understood. 9 A number of studies demonstrated a link between DWV infection and anomalous development of wings and other signs. Thus, DWV replication was demonstrated in various tissues of honey bees, including wings, head, thorax, legs, hemolymph, and gut. 3 In bees with crippled wings, viral replication was shown to take place in the head, thorax, and abdomen, while in nonaffected infected bees, the replication occurred in the thorax and/or abdomen but not in the head. 19 DWV RNA was identified within the brain of honey bees using in situ hybridization. 13 The development of wing deformity was also found to correlate with virus replication in Varroa mites. The mites that could cause wing deformities contained 102 to 104 times higher DWV load compared to mites that did not cause overt DWV infection. 5 Also, injection of DWV in adult forager bees was reported to result in learning deficits and a decrease in motor activity, 9 shortened foraging period, and decreased survivial. 2 Protruded proboscis has anecdotally been associated with Varroa infestation; however, the mechanism or importance of this lesion remains unknown. In this study, we found that protruded proboscis and prolonged emergence were associated with deformed wings. Taking into account the established effect of DWV on motor and neural activity of honey bees, it is possible that prolonged emergence and protruded proboscis may be associated with Varroa-mediated DWV infection of the nervous and/or muscular systems. 9
Hypopharyngeal and mandibular glands are particularly developed in young nurse bees. 20 Hypopharyngeal and mandibular glands produce a wide array of enzymes and a protein-rich secretion called royal jelly, which is used by nursing bees to feed larvae and the queen. Mandibular glands also produce pheromones that regulate reproductive and foraging behavior. These structures undergo physiological atrophy in older worker bees, as they begin performing other tasks, such as foraging. 20
A number of studies have demonstrated that the size of hypopharyngeal glands depends on a number of factors, including age, presence of queen in the colony, 14 pollen quality, 10 and insecticide toxicity. 6,11,20 The size of hypopharyngeal and mandibular glands decreased in bees that were heavily parasitized with Varroa mites during development. 1 The results of our study suggest that hypoplasia of hypopharyngeal and mandibular glands is also associated with DWV infection, which is vectored by Varroa mites. As hypopharyngeal and mandibular glands are crucial for brood development and colony organization, hypoplasia and dysfunction of these glands in individual bees might affect colony-level performance and could potentially contribute to honey bee population declines.
One limitation of this study is that direct visualization of the viral RNA and/or proteins in the honey bee tissues, especially hypopharyngeal and mandibular glands, was not performed. The results of our study add to the body of knowledge on the effects of DWV infection and can be valuable in guiding future studies on viral pathogenesis in honey bees.
Supplemental Material
Supplemental Material, Combined_supplemental_materials-Koziy_et_al - Deformed Wing Virus Infection in Honey Bees (Apis mellifera L.)
Supplemental Material, Combined_supplemental_materials-Koziy_et_al for Deformed Wing Virus Infection in Honey Bees (Apis mellifera L.) by Roman V. Koziy, Sarah C. Wood, Ivanna V. Kozii, Claire Janse van Rensburg, Igor Moshynskyy, Ihor Dvylyuk and Elemir Simko in Veterinary Pathology
Footnotes
Acknowledgements
We thank Jess Morris for the invaluable assistance in fieldwork with honey bees and to Dr. Carlos Castillo for his molecular virology expertise.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Western Grains Research Foundation (ADF 20150125, 2016), Saskatchewan Canola Development Commission (ADF 20150125, 2016), Saskatchewan Agricultural Development Fund (20160157, 2017), North American Pollinator Protection Campaign (2015), Canadian Bee Research Fund (2015), Saskatchewan Beekeepers Development Commission (2015), and WCVM Wildlife Health Research Fund (2016), Mitacs (IT09712, FR22300, SK–ISED), Project Apis m. Costco Canada. The authors also acknowledge financial support provided to RVK by the University of Saskatchewan Graduate Scholarship and Western College of Veterinary Medicine Graduate Enhancement Fund.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
