Abstract
The New York State (NYS) beekeeping industry generated >$11M worth of honey in 2020 and >$300M in pollination services to agriculture annually. Bees are frequently exposed to pesticides through foraging and husbandry practices. Lipophilic pesticides can remain in beeswax for extended periods. We analyzed for pesticides in wax comb samples collected from NYS apiaries at the end of the growing season, comparing residue numbers and concentrations among beekeepers of different operation scales: commercial beekeepers (>300 colonies), sideliners (50–299 colonies), and hobbyists (<50 colonies). We analyzed samples collected from 72 managed honey bee colonies for 92 insecticides, herbicides, and fungicides by liquid chromatography–tandem mass spectrometry. Pesticides were detected in all samples and included 34 fungicides, 33 insecticides, and 22 herbicides. Each wax sample contained 7–35 different residues (x¯ = 17.8 residues). Wax from colonies managed by commercial beekeepers contained the most residues (x¯ = 21.9 residues), hobbyists were second (x¯ = 16.3 residues), and sideliners had the fewest (x¯ = 11.7 residues). Nearly all wax samples (98.6%) contained the pesticide synergist piperonyl butoxide, most samples (86%) contained common varroacides used to control honey bee parasites, including coumaphos and amitraz breakdown products, and 93.1% contained the fungicide difenoconazole. We detected 34 fungicides, 7 of which were found in 50% or more of the samples. We detected 22 herbicides. We found pesticide contamination of beeswax to be common, with commercial beekeepers experiencing the greatest contamination.
There are ~3,000 beekeepers in New York State (NYS), managing ~80,000 colonies of bees. 14 The western honey bee (Apis mellifera L.) is the most common domestic species used for pollination and hive products, but they are also found as feral colonies in the wild in NYS and elsewhere. 37 NYS beekeepers generated > $12.5M in income from honey in 2021 and are estimated to have an agricultural value > $300M annually from pollination services.14,41 The number of pesticides to which honey bees are exposed has been increasing over time, and NYS has high pesticide diversity and concentrations compared to the rest of the continental United States. 40 Beeswax can also contain relatively high concentrations of varroacides used to control parasitic mites in honey bees. 27
Pesticide exposure may vary among different operation scales of beekeepers (commercial, sideliner, hobbyist) for several reasons. Commercial beekeepers conduct most crop pollination, such as almonds in California, apples in NYS, and alfalfa and canola in the Great Plains states. 16 Because these crops are often grown under pesticide-intensive management practices, greater pesticide residues have been reported in bee bread and wax from commercial hives compared to samples collected from hives of sideliners and hobbyists.21,24 Similarly, hives placed in agricultural areas can have higher pesticide exposures compared to those in suburban areas, where many hobbyists are present.11,23 Because the average foraging radius of a honey bee colony can be ≥ 1 km, they are excellent landscape-scale environmental toxin scavengers, commonly bringing back pesticides to hives placed in mixed agricultural landscapes. 16 Finally, management of diseases and pests, especially for the parasitic Varroa mite, varies greatly among beekeepers, and chemical varroacides are typically one of the largest sources of beeswax contamination. 27
Although beekeepers provide wax-coated frames (foundations) for honey bees to build on, the bees themselves produce wax. The wax is heavily reused by bees; thus, the same wax can remain in the hive indefinitely. Because wax is made from hydrocarbons, it can become a lipid sink for lipophilic pesticides. 38 Indeed, beeswax is universally contaminated with agrochemicals and often contains the greatest number of residues compared to honey, pollen, and the bees themselves. 6 Because bees use wax in the brood nursery and for storage of food (honey, pollen, bee bread), there is the potential for contamination of in-hive workers and future generations of bees.6,54 Honey and beeswax are both sold for human consumption and cosmetic use; thus, residues in wax can potentially impact the human food chain and environment. 12 For these reasons, it is recommended that beekeepers remove beeswax from the hive and rotate fresh new combs every few years. 6
We designed our study to address 2 main questions: 1) What is the scope of pesticide contamination of beeswax in managed honey bee hives across NYS? 2) Does contamination vary among commercial, sideliner, and hobbyist beekeepers? We addressed these questions by sampling beeswax from 72 managed honey bee colonies and screening for 92 pesticides via a multi-residue liquid chromatography–tandem mass spectrometry (LC-MS/MS) analysis.
Materials and methods
Sample collection
Beekeepers participated voluntarily in our study. The 27 volunteers enrolled included 9 commercial beekeepers with > 300 colonies, 10 sideliner beekeepers with 50–299 colonies, and 8 hobbyists with < 50 colonies. Colonies that were sampled were spread across 9 regions and 22 counties of NYS, including the Mohawk Valley, Finger Lakes Region, Western New York, Southern Tier, Capital District, Central New York, Northern Country, Long Island, and New York City. The spatial spread and operation scale diversity of colonies sampled was meant to broadly represent the NYS beekeeping industry.
Wax samples were taken from the brood nest area (i.e., cells from the area where immature bees develop) of 72 colonies in September 2021. Samples were taken from the brood nest area given that pesticide exposure to developing bees is a particular concern, pollen and nectar are continuously brought to the area to feed developing bees, and the brood nest is where varroacides are typically applied to control the Varroa mite. The oldest areas of wax, identified by a darker color, were preferentially sampled when possible. Unfortunately, it is rare to know the exact age of combs because few beekeepers keep track of comb age. At the same time, our goal was not to relate comb age to pesticide contamination. Instead, we sought to obtain a snapshot of contamination across the NYS beekeeping industry, which was afforded by our sampling procedure. Empty cells were scraped into collection vessels using clean spatulas. Wax samples were ≥ 5 g each. Samples were stored at –80°C for 3 mo before analysis.
Sample preparation and analysis via LC-MS/MS
The samples were extracted using a modified version of the EN 15662 QuEChERS procedure 10 and screened for 92 pesticides (including some metabolites and breakdown products) by LC-MS/MS (Table 1) using methods adapted from previous studies.13,15,50
Pesticides in beeswax as determined by liquid chromatography–tandem mass spectrometry analysis.
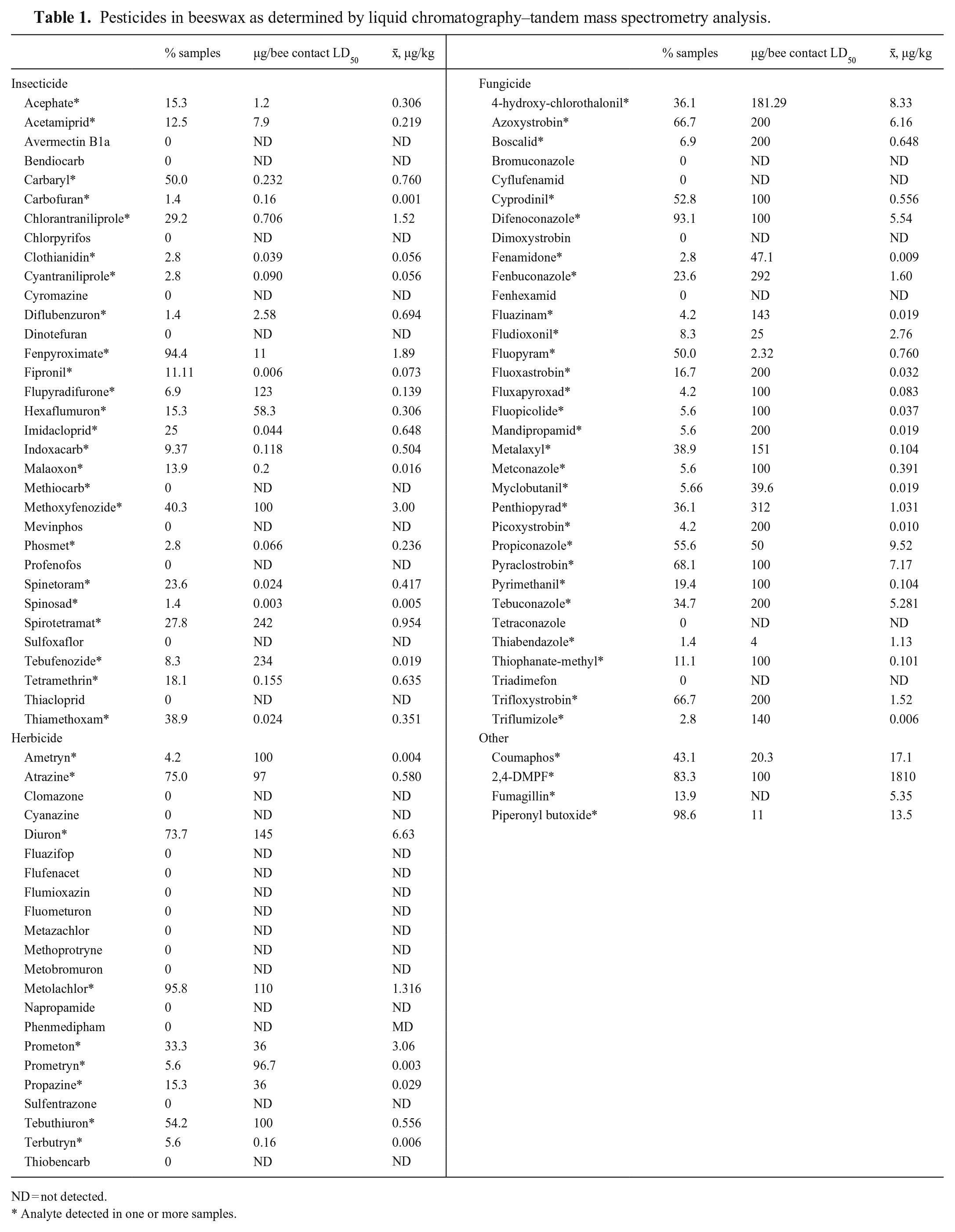
ND = not detected.
Analyte detected in one or more samples.
Wax samples (4.96–5.05 g) were mixed with 7 mL of acetonitrile and 5 mL of water and then homogenized for 1 min (2.8-mm ceramic beads; Bead Ruptor 24, Omni International). After complete homogenization, 6.5 g of EN 15662 salts were added (4 g of MgSO4; 1 g of NaCl; 1 g of sodium citrate tribasic dihydrate; 0.5 g of sodium citrate dibasic sesquihydrate). Samples were then thoroughly vortexed and centrifuged at 7,300 × g for 5 min. One milliliter of supernatant was collected and transferred into a d-SPE (dispersive solid-phase extraction) tube containing 150 mg of MgSO4 and 25 mg of PSA. After the d-SPE step, 496 µL of supernatant were collected, and 4 µL of internal standard solution (d4-imidacloprid 0.07 ng/µL; d10-chlorpyrifos 0.2 ng/µL; d7-bentazon 0.1 ng/µL; d5-atrazine 0.02 ng/µL; d7-propamocarb 0.1 ng/µL) was added. The samples were then filtered (0.22-µm polytetrafluoroethylene filter) and stored at −20°C before analysis.
Sample analysis was performed with an ultra-high-performance liquid chromatography (UHPLC) system (Vanquish Flex; Dionex Softron) coupled with a TSQ Quantis mass spectrometer (Thermo Scientific). The UHPLC system was equipped with an Accucore aQ column (100 mm × 2.1 mm, 2.6-µm particle size). The mobile phase consisted of A) methanol:water (2:98, v/v) with 5 mM ammonium formate and 0.1% formic acid, and B) methanol:water (98:2, v/v) with 5 mM ammonium formate and 0.1% formic acid. The column was maintained at a temperature of 25°C throughout the run; the flow rate was set at 300 µL/min. The solvent gradient went as follows: 1.5 min equilibration (0% B) prior to injection, 0–0.5 min (0% B, isocratic), 0.5–7 min (0–70% B, linear gradient), 7–9 min (70–100% B, linear gradient), 9–12 min (100% B, column wash), 12–12.1 min (100–0% B, linear gradient), and 12.1–14.5 min (0% B, re-equilibration). The flow from the LC system was directed to the mass spectrometer through a heated electrospray probe (H-ESI). The settings of the H-ESI were: spray voltage 3,700 V for positive mode and 2,500 V for negative mode, sheath gas 40 (arbitrary unit), auxiliary gas 10 (arbitrary unit), sweep gas 1.5 (arbitrary unit), ion transfer tube temperature 325°C, and vaporizer temperature 350°C.
The MS/MS detection was carried out using the selected reaction monitoring (SRM) mode. For each compound, 2 transitions were monitored: one for quantification and the other for confirmation. The resolution of both Q1 and Q3 was set at 0.7 FWHM (full width at half maximum), the cycle time was 0.5 s, and the pressure of argon in the collision cell was set at 2 mTorr.
We analyzed samples for 33 insecticides, including τ-fluvalinate, which is also used as a varroacide in bee colonies; 33 fungicides, including chlorothalonil metabolite 4-hydroxy chlorothalonil; and 22 herbicides (Table 1). In addition, 2 varroacides used against Varroa mites, amitraz metabolite 2,4-dimethylphenyl formamide (2,4-DMPF) and coumaphos, the antimicrobial fumagillin, and the insecticide potentiator piperonyl butoxide were included in our analysis (Table 1). This 92-pesticide multi-residue analysis is available to the public (https://blogs.cornell.edu/ccecf/). Our analysis includes the most common agrochemicals and varroacides used in NYS and other parts of North America that can be quantified via LC-MS/MS. Values are reported in µg/kg.
Statistical analysis
Data analysis was conducted using R v.4.2.3 (https://www.r-project.org/) with the glmmTMB package. 3 We constructed a generalized linear-mixed model (GLMM) that evaluated whether the number of pesticides detected in wax varied among beekeeper operation scales (commercial, sideliner, hobbyist), including beekeeper ID as a random effect. To determine significance, we employed a likelihood ratio test and compared the model to one with the same response, distribution, and random-effect structure, but that excluded operation scale as a predictor. Post-hoc independent contrasts were evaluated via the R emmeans package.
Results
Pesticide residues were detected in all 72 of the wax samples analyzed; the number of unique pesticide residues per colony ranged from 7 to 35 (x¯ = 17.8; Table 1). All but one sample contained the pesticide synergist piperonyl butoxide. The herbicides metolachlor, atrazine, and diuron were found in more than half of the colonies, respectively, and the insecticide fenpyroximate was found in most colonies. Amitraz metabolite 2,4-DMPF was found in most samples, and at concentrations sometimes > 4,000 µg/kg. Fungicides difenoconazole, pyraclostrobin, azoxystrobin, and trifloxystrobin were each found in more than half of the samples.
Wax contamination differed significantly among beekeeping operation scales (χ21 = 15.9, p < 0.001). Wax from colonies managed by commercial beekeepers contained the most residues (x¯ = 21.9), hobbyists were second (x¯ = 16.3 residues), and sideliners had the fewest (x¯ = 11.7 residues; Fig. 1). Pesticides that were not detected in any samples included avermectin B1A, bendiocarb, bromuconazole, chlorpyrifos, clomazone, cyanazine, cyflufenamid, cyromazine, dimoxystrobin, dinotefuran, fenhexamid, fluazifop, flufenacet, flumioxazin, fluometuron, metazachlor, methiocarb, methoprotryne, metobromuron, mevinphos, napropamide, phenmedipham, profenofos, sulfentrazone, sulfoxaflor, tetraconazole, thiacloprid, thiobencarb, and triadimefon. Lack of these detections could be the result of lack of exposure, degradation, or poor solubility in the wax substrate.
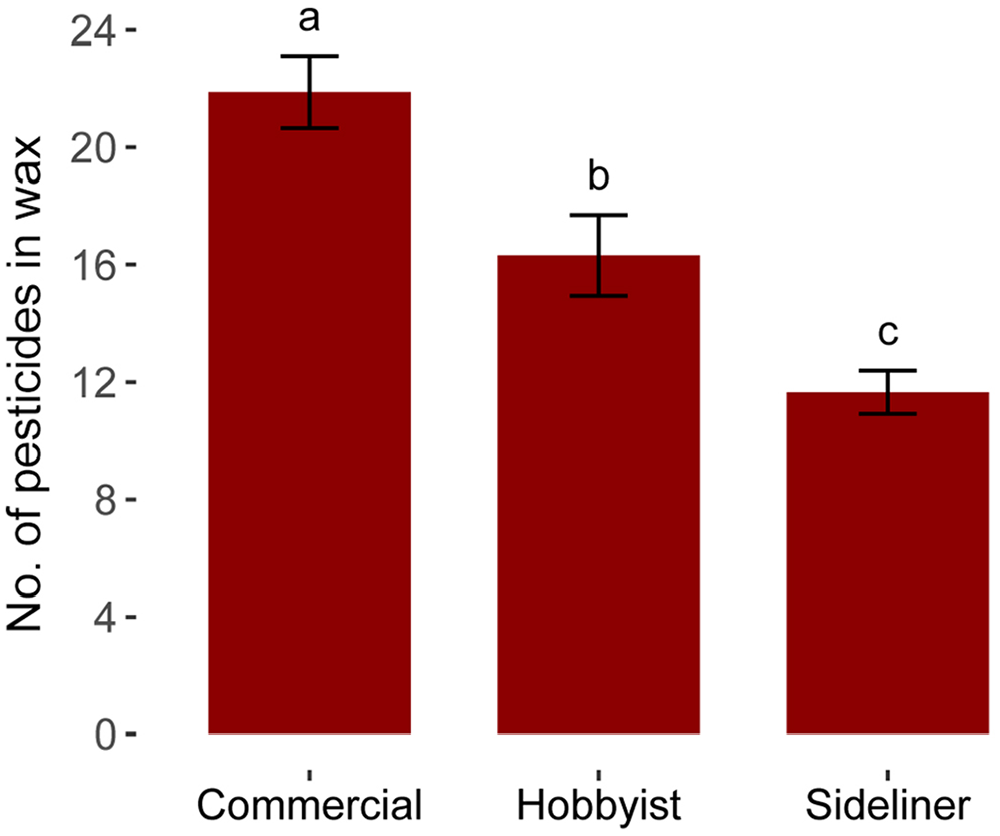
Number of pesticide residues in beeswax sampled from commercial beekeepers (> 300 colonies), sideliners (50–299 colonies), and hobbyists (< 50 colonies). Different letters indicate significance (p < 0.05) via emmeans post-hoc contrasts. x¯ ±SE shown.
Total varroacide concentrations in wax samples were 14.2–7,460 µg/kg with most being coumaphos and 2,4-DMPF. Fungicides were the second most common pesticides (x¯ = 57.7 µg/kg), followed by piperonyl butoxide, insecticides (12.8 µg/kg), and herbicides (13.3 µg/kg).
Discussion
In beeswax collected from locations across North America between 2007 and 2008 and analyzed for 200 different chemicals, an average of 6.5 pesticides was found per sample, but some wax samples contained up to 39 pesticide residues. 27 We only analyzed samples for 92 compounds, but found more than twice that mean number of residues (17.8); the lowest residue count was 7. We did not assess the most common contaminant in the previous study because it was not amenable to the LC-MS/MS analytical method that we used. The varroacide τ-fluvalinate was detected in 98.1% of 259 samples. 27 Several studies in Spain found amitraz metabolites in wax (Table 2). The remaining studies detected an amitraz metabolite (not 2,4-DMPF) in 46.5% and 70–91.7% of wax samples at concentrations up to 3,530 µg/kg and 1,490 µg/kg, respectively.4,5
Residues found in >50% of beeswax samples from New York State, compared to previous studies that have assessed pesticide contamination of beeswax.
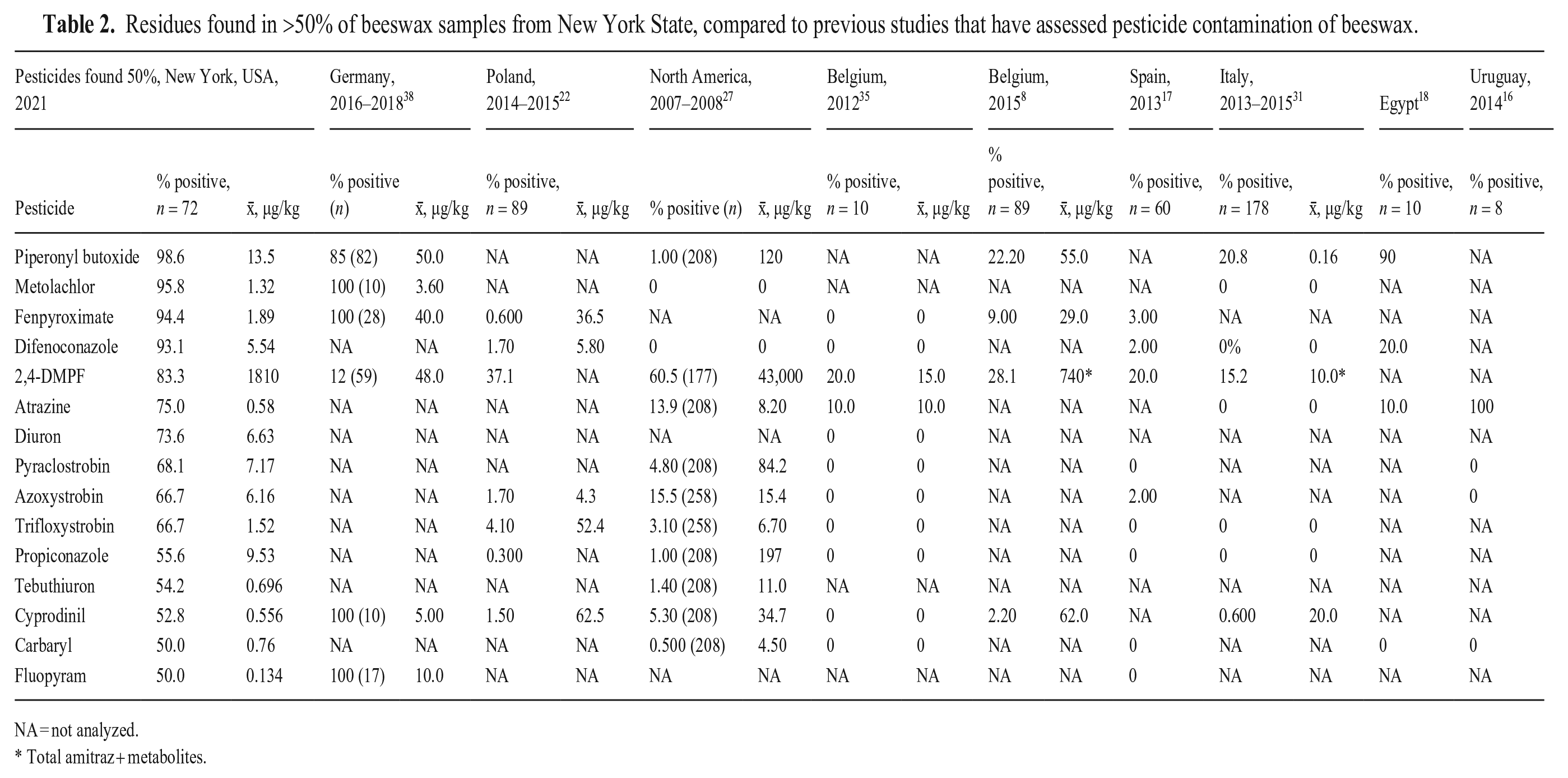
NA = not analyzed.
Total amitraz + metabolites.
The varroacides τ-fluvalinate, amitraz, and coumaphos are routinely used for the control of Varroa mites in honey bees and are frequent residues in beeswax. 27 Coumaphos, in particular, is commonly detected in beeswax in Europe and North America, frequently at relatively high concentrations (Table 3). These were the 3 most common pesticides (and metabolites) found in commercial beeswax collected from Swiss apiaries.21,25 The detection of coumaphos-resistant Varroa mites in the eastern United States has led to reduced use of coumaphos, thus the reduced amount found in NYS wax was expected, although coumaphos residues are known to persist in wax.6,19,32,53
Coumaphos residues in our and previous studies assessing contamination of beeswax.
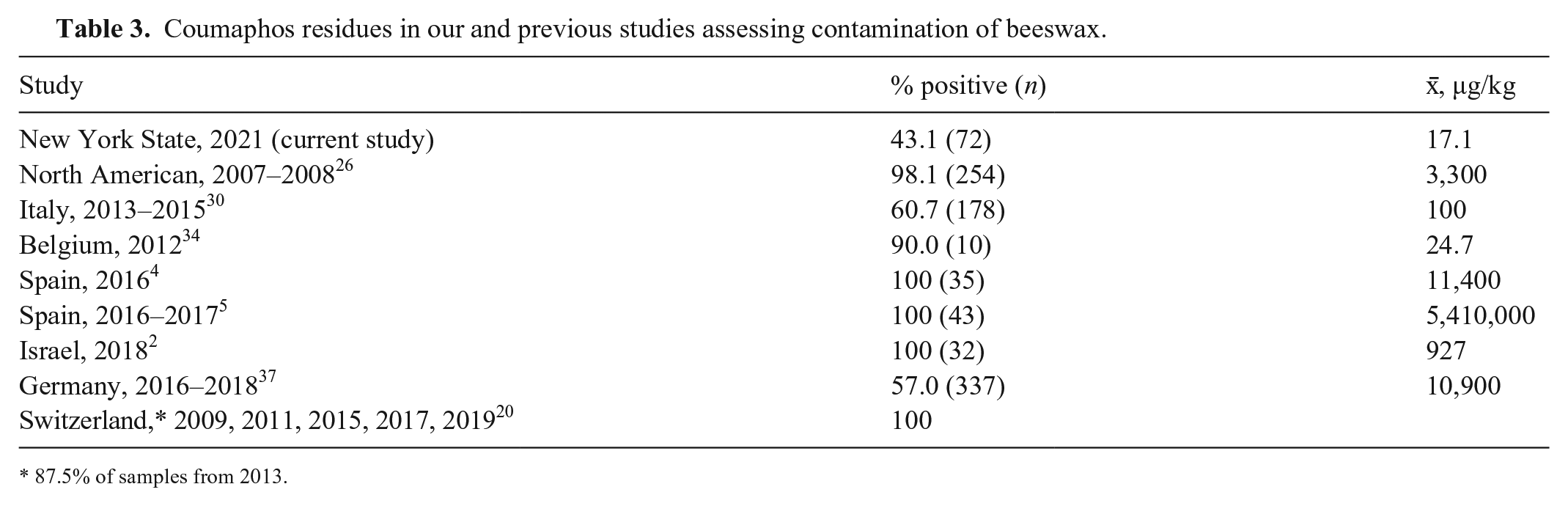
87.5% of samples from 2013.
Piperonyl butoxide was the chemical that we found most commonly. Piperonyl butoxide is a common ingredient in many pesticide products, including pyrethroid, methylcarbamate, and pyrazole insecticides, and the herbicide metolachlor, because it acts to inhibit the activity of cytochrome P-450 enzymes found in plants, insects, and vertebrates, thereby preventing metabolism of insecticides and potentiating their function. 51 The widespread use of piperonyl butoxide across several pesticide products may partially explain its high prevalence of contamination in beeswax. Other compounds that we found commonly include the herbicides metolachlor, atrazine, and diuron, greenhouse insecticide fenpyroximate, and the fungicides difenoconazole, pyraclostrobin, azoxystrobin, and trifloxystrobin.
Systemic pesticides (Table 4), which are incorporated into plant tissues, including pollen and nectar, were detected in 39% of the samples in our study. These pesticides included the neonicotinoids acetamiprid, clothianidin, imidacloprid, and thiamethoxam, as well as fipronil and spinosad; dinotefuran and thiacloprid were not detected. Imidacloprid and thiacloprid were not detected in wax samples in Israel, but they were detected in bee bread. 2 None of the following were detected in samples of wax collected from the brood nest in apiaries in Belgium: acetamiprid, clothianidin, imidacloprid, thiacloprid, thiamethoxam, or spinosad.8,35 The European Union placed a moratorium on the use of clothianidin, imidacloprid, and thiamethoxam in 2013 to protect pollinators, which has been expanded since that time, and likely impacted the residues in beeswax in the later studies. 39 Additionally, neonicotinoids are hydrophilic, and thus likely to partition to other hydrophilic matrices, such as honey and bee bread, rather than wax, which is hydrophobic.
Systemic insecticide residues detected in beeswax in our (New York State) and previous studies.
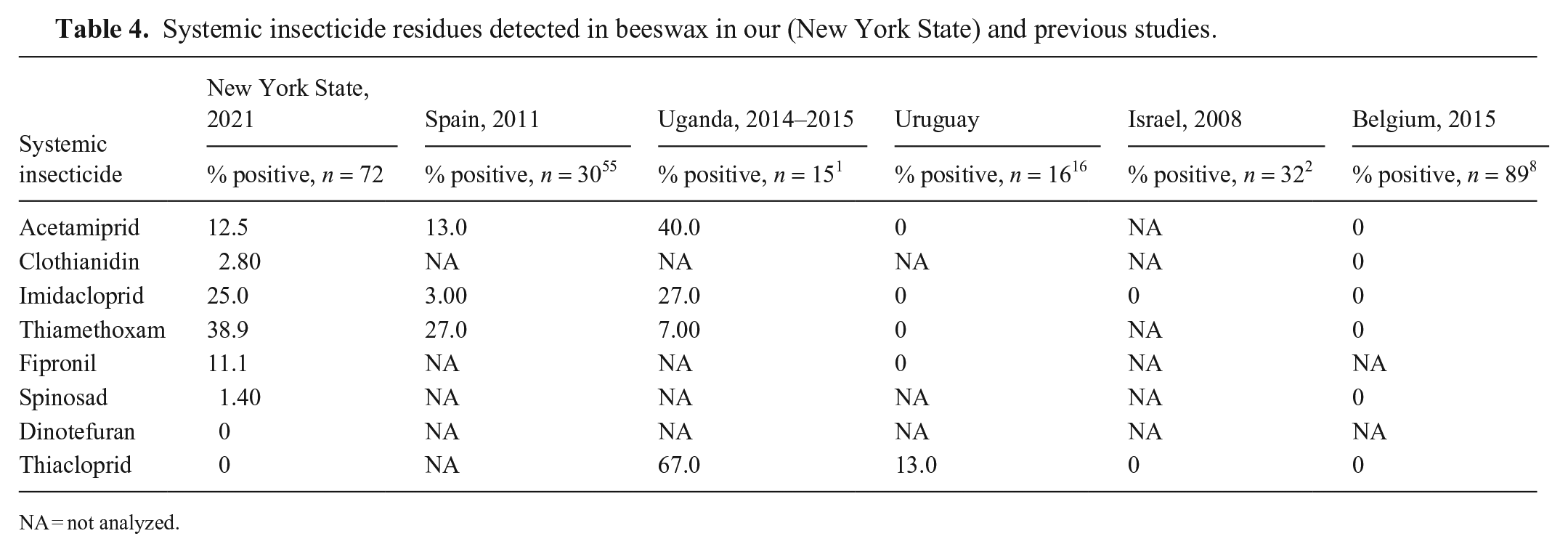
NA = not analyzed.
Pesticides that were not detected in any samples include avermectin B1A, a component of abamectin. Avermectin B1A is lipid soluble and locates predominantly in the leafy parenchyma of the plant, whereas surface contamination undergoes rapid environmental degradation, which may limit its accumulation in wax. 36 Several insecticides (bendiocarb, mevinphos, methiocarb) and herbicides (methoprotryne) are not currently in use in the United States, and the insecticide chlorpyrifos is no longer in use in NYS.25,30,34,42,45 Bromuconazole is a fungicide used primarily on ornamental plants, which may limit its exposure to colonies in our study. 49 Use of the fungicides triadimefon and tetraconazole on food crops has been restricted in the United States, and tetraconazole is further restricted from use in NYS.46,48 Several herbicides are in restricted use in the United States (cyanazine, thiobencarb) or NYS (clomazone, fluazifop, flufenacet, flumioxazin, metobromuron, sulfentrazone). 29 Other herbicides are usually used pre-emergence (cyanazine, clomazone, metazachlor, metobromuron, napropamide, thiobencarb); thus, high concentrations may not be present at the time of pollination.24,28,43,44,47,52 Some herbicides and insecticides are used on crops that are not grown in NYS, including phenmedipham (sugar beet herbicide), fluometuron (cotton herbicide), and profenofos (cotton insecticide). Fenhexamid is a fungicide that has been found occasionally in beeswax samples; it is used to control fungus on fruit after pollination.26,33 Dimoxystrobin was detected in wax samples from Germany, where it has been used on oilseed and sunflower crops. 38 Thiacloprid has been found in live bee samples but was not detected in honey or pollen. 20 Thiacloprid and sulfoxaflor are neonicotinoid insecticides and are highly water soluble, making them less likely to accumulate in lipid substrates.7,9
Overall, we found that pesticide contamination of beeswax is common in NYS and that commercial beekeepers experience the greatest levels of contamination. The average number of pesticides detected in beeswax samples in our study was generally higher than has been detected in previous studies, but the concentrations of pesticides were similar to those found previously. Our results provide a useful baseline for future longitudinal studies that seek to assess how pesticide contamination of beeswax may or may not change over time. In addition, our data will be useful for designing bioassays that assess whether the contamination levels found in our study have any negative sublethal impacts on bees.
Footnotes
Acknowledgements
We thank Conner Hinsley and Emma Walters for their assistance in sample collection. We also thank Jennifer Moiseff for her editorial assistance.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Our research was funded by the New York State Environmental Protection Fund and the U.S. Department of Agriculture (grants 2018-08603, NYC-139850). The content is solely the responsibility of the authors and does not necessarily represent the official views of the funding agencies.
