Abstract
Susceptibility of individuals and groups to toxicants depends on complex interactions involving the host, environment, and other exposures. Apiary diagnostic investigation and honey bee health are truly population medicine: the colony is the patient. Here we provide basic information on the application of toxicology to the testing of domestic honey bees, and, in light of recent research, expand on some of the challenges of interpreting analytical chemistry findings as they pertain to hive health. The hive is an efficiently organized system of wax cells used to store brood, honey, and bee bread, and is protected by the bee-procured antimicrobial compound propolis. Toxicants can affect individual workers outside or inside the hive, with disease processes that range from acute to chronic and subclinical to lethal. Toxicants can impact brood and contaminate honey, bee bread, and structural wax. We provide an overview of important natural and synthetic toxicants to which honey bees are exposed; behavioral, husbandry, and external environmental factors influencing exposure; short- and long-term impacts of toxicant exposure on individual bee and colony health; and the convergent impacts of stress, nutrition, infectious disease, and toxicant exposures on colony health. Current and potential future toxicology testing options are included. Common contaminants in apiary products consumed or used by humans (honey, wax, pollen), their sources, and the potential need for product testing are also noted.
Keywords
Introduction and background
The field of apiary toxicology in the United States had its official start in 1881, when George Thompson of Geneva, Illinois (64 km [40 mi] west of Chicago) reported that shortly after spraying his fruit trees with the arsenical pesticide Paris green, his bee colonies were decimated. 121 The presumptive diagnosis in this case was clear, but honey bee colony morbidity and mortality events are most often multifactorial: agricultural chemicals, environmental contaminants, pharmaceuticals, and natural toxins must be considered, along with other biological factors and stressors. 18
The importance of veterinary care for honey bees was emphasized in 2017, when the U.S. Food and Drug Administration (FDA) implemented the Veterinary Feed Direc-tive Rule (https://www.fda.gov/animal-veterinary/development-approval-process/fact-sheet-veterinary-feed-directive-final-rule-and-next-steps), which classified honey bees as livestock, requiring veterinary prescriptions for antibiotics, which, in turn, required a veterinarian–client–patient relationship. 31 Interest in honey bee veterinary medicine is also growing in the European Union, but veterinary diagnosticians in the E.U. also have relatively limited experience with honey bee testing; veterinary surveillance of honey bees in China and Australia (https://www.agriculture.gov.au/agriculture-land/animal/health/laboratories/tests/routine-tests) is focused on infectious diseases; and honey bee surveillance in sub-Saharan Africa is minimal, although exposure to agricultural stressors may be lower in parts of Africa as well as Latin America.4,36,128,134,142 As veterinarians in the United States and elsewhere become more involved in honey bee health, an increased need for honey bee testing, including toxicology, can be expected. However, as with other livestock species, honey bee health is complex, and honey bees are generally exposed to multiple xenobiotics, including natural phytotoxins, pesticides, and also antimicrobial and acaricidal products applied to the hive itself, leading to the potential for additive, antagonistic, and synergistic interactions between compounds. As with other species, susceptibility to toxicants is dependent on factors such as stress, nutrition, and, particularly in honey bees, pathogen load, all of which must be considered. 133 Our purpose here is to give basic information on the application of toxicology to domestic honey bees and to expand on some of the challenges of interpreting analytical chemistry findings as they pertain to hive health in light of recent research.
Honey bees are the epitome of population medicine: evaluating the health of the hive is more useful than assessing the individual bee. 40 The honey bee colony is a “super organism,” consisting of 20,000–50,000 genetically related individuals functioning as a single unit.10,34 Domestic western honey bees (Apis mellifera L.) are integral to the human food system and produce a variety of products that are important to the human food chain and cosmetics industries, including honey, propolis, pollen, royal jelly, and beeswax.31,44,112,139 More importantly, 90% of crops producing deciduous fruit, vegetables, legumes, and oil seeds require insect pollinators, and 96% of animal-pollinated crops are pollinated by honey bees and native bees.70,118 Mixtures of agrochemicals that are applied to these crops can impact pollinators at all stages of life.18,136
The built environment of the hive is provided by the apiarist and the bees themselves, and hive structure and function are important considerations in assessing the colony. Wax provides storage of brood and food within the hive.64,109,112 Once manufactured by the bees, wax can be used for many years, making it a potential depot for lipid-soluble contaminants. 25 Pollen, collected by foragers and containing phytotoxins, agricultural field chemicals, and other contaminants, is layered into the cells and processed to encourage fermentation, producing bee bread.25,88,93,115,130 Beneficial fungi and other microbes are essential for fermentation, thus agricultural fungicides can impact bee bread production and longevity.34,133 Imbalanced microflora may contribute to mycotoxin production or spoilage in bee bread.64,108 The phenomenon of “entombed pollen,” which occurs when bee bread becomes discolored and is subsequently capped, has been associated with chlorothalonil, a common agricultural fungicide, and impacts the survival of bees and larvae. 130 Propolis is made from plant resins and contains phenolic compounds and other biologically active components, has antimicrobial properties, and is used to line the hive and seal cracks.10,64,130
Honey bees are unique among agricultural animals in that the apiarist has limited control of their nutrition, but they are not unique in requiring balanced diets containing protein, lipids, sugars, vitamins, minerals, and water in various ratios during different life stages.130,133 A diversity of food sources is required for balanced nutrition; however, monoculture cropping, habitat fragmentation, and introduced species all have had detrimental effects on honey bee nutrition.31,108,132,133 Malnutrition was implicated in bee losses between 2007 and 2015. 123 Water is collected by foragers from plant guttation fluids and waterways, and can be contaminated with agricultural chemicals, livestock drugs, and microplastics.3,35,55,63,67,71,111,118
Diagnostic investigation
As is the case with other animal health issues, diagnosing diseases caused by toxicants in honey bee colonies requires physical examination of the colony; ruling out other causes of mortality by analyzing the history and management of the colonies, including pathogen load, drug and acaricide use, location of the hive with respect to industry, agriculture, and other factors; and clinical signs. 133 There are limitations of the available toxicity data for honey bees, as described later. General health, pathogen and parasite load, and phytotoxins that are inherently part of the bee diet are likely to have specific effects on the ability of honey bees to detoxify xenobiotics and thus impact susceptibility, as we will present in more detail with xenobiotic exposure and disposition. The following recommendations for sampling and testing should consider the overall health and risk factors of the hive. Interpreting analytical results requires special considerations about the interactions of xenobiotics with various matrices associated with the honey bee colony.
Sample collection
Samples for analysis should be collected as soon as poisoning is suspected. 133 We have illustrated important sampling considerations (Fig. 1); other publications have provided more in-depth information on hive husbandry and pathogen assessment.19,133,140 Samples for analytical chemistry should be ≥ 50 g, and include bees collected alive (~500 bees), pollen (~50 cells), bee bread, honey (50 cells scraped out with a metal or wooden spatula), and wax from empty cells. 140 Given that many xenobiotics are ubiquitous in colonies, it is best to collect these samples from unaffected as well as affected hives for comparison. 140
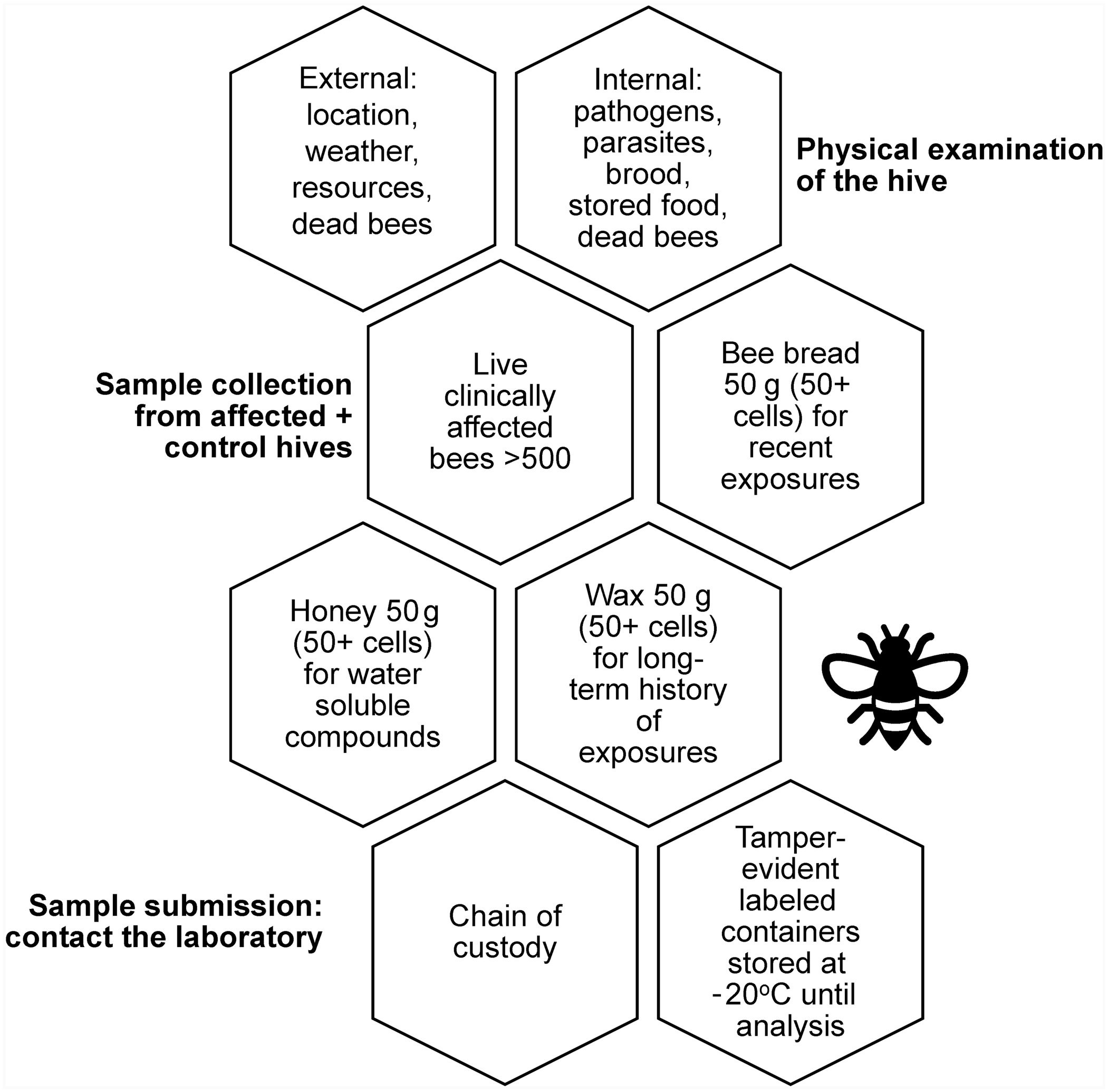
Hive examination and sample collection for honey bee toxicology testing.
Collection of live bees that are clinically affected (dying or twitching) is critical because bees undergo rapid autolysis, and pesticide concentrations decrease within hours of death but are more stable in live and frozen bees.69,118 Samples should be collected in labeled plastic bags or 50-mL centrifuge tubes, both of which can be wrapped in foil to prevent exposure to sunlight. 140 Live bees, and other samples, should be frozen at −20°C and kept frozen during shipment and until analysis.65,140 Sample containers must be properly labeled with the collector name, owner name, date and time of collection, colony number, and sample type, and a description of the history, including onset of the problem, colony condition, and toxicants that may be involved. 140 Because of the potential for legal action, it is wise to include chain of custody forms and use tamper-evident packaging. 140
Unfortunately, diagnosis of the cause of morbidity and mortality is never guaranteed. Because of rapid autolysis and biodegradation of pesticides, false-negative pesticide results are possible, and analyzing for metabolites can be useful. 133 Additionally, data on concentrations of xenobiotics in bees and bee products that are associated with mortality or sublethal effects are usually lacking, making comparison to unaffected hives useful. 133 Pollen samples can be used if a recent toxicant exposure is suspected and dying bees are not observed; bee bread is useful in determining past exposures. 140 Wax gives a longer history of pesticide exposure, given that lipid-soluble xenobiotics remain dissolved in wax for years or even decades, although little is known about the bioavailability of contaminants in wax.65,140
Analytical testing
The variety of organic substances and environmental matrices to which honey bees may be exposed presents an analytical challenge. Modifications of the quick, easy, cheap, effective, rugged, and safe (QuEChERS) extraction protocol, developed for the determination of pesticide residues in fruits and vegetables, are commonly used for the analysis of whole bees, larvae, honey, beeswax, bee bread, and other hive products, coupled with liquid or gas chromatography–tandem mass spectroscopy (LC-MS/MS or GC-MS/MS) for efficient, simultaneous screening for a large number of acaricides, fungicides, pesticides, and metabolites.1,6,29,69,75,97 Modified QuEChERS extraction in tandem with LC-MS/MS has also been applied to the analysis of wild mason bee nest contents such as feces, leaf capsules, and mud. 96 Direct surface analysis utilizing desorption atmospheric pressure photoionization–MS for determining neonicotinoid insecticide residues has been reported in plant material but has yet to be proven for analysis of whole bees or hive products. 127 Analysis for biomarkers is also amenable to veterinary diagnostic laboratory methods, including peptide and protein analysis, and PCR to look at gene upregulation, as we describe in the metabolism section. Triple quadrupole and quadrupole time-of-flight mass spectrometers, coupled to LC, have also enabled targeted and untargeted screening of neuropeptides and proteins to elucidate the mechanisms by which toxic exposures may exert sublethal effects.47,58 Although biomarker identification methods are currently utilized in the research context only, data from these investigations may assist in the interpretation of the significance of residual toxicant concentrations.
The complexity of the matrices investigated generally requires matrix-matching of standards for accurate quantitation because many analytes are susceptible to matrix effects, including ion suppression and/or enhancement. 69 Simultaneous analysis of multiple matrices appears to be required for an accurate picture of pesticide contamination load given that pesticide partitioning is driven by chemical interactions between pesticides and hive components (e.g., lipophilicity of beeswax and hydrophilicity of honey).25,112 Octanol:water partition coefficients for various agricultural chemicals and the matrices in which they have been detected are available for many, but not all, compounds (Table 1).
Agrochemicals that may impact bees, based on classification, mechanism of action, use, and the log of the octanol:water partition coefficient (log Kow) and the matrices in which the chemical has been detected. The higher the log Kow, the more likely the compound is to dissolve in lipophilic matrices such as wax; the lower the log Kow, the more likely the compound is to dissolve in hydrophilic matrices such as honey.
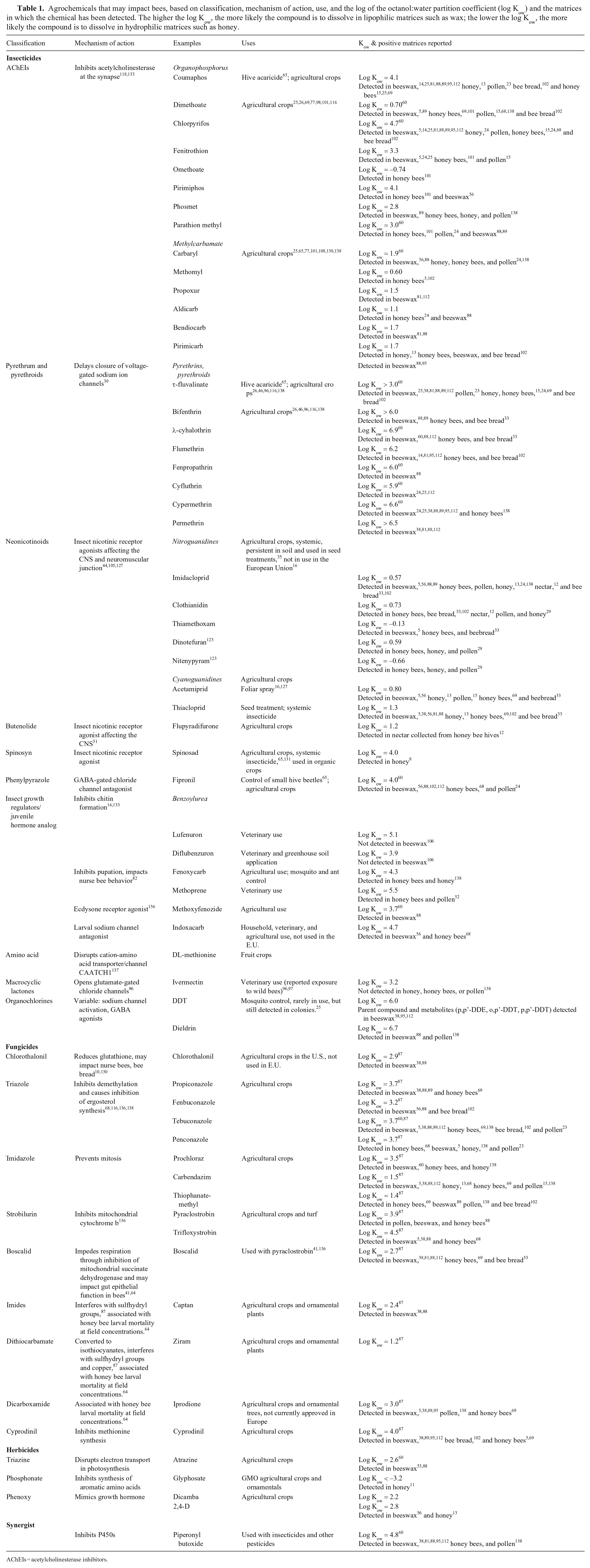
AChEIs = acetylcholinesterase inhibitors.
Mineral and metal analyses of honey bees, honey, pollen, and other hive materials have been accomplished using inductively coupled plasma–optical emission spectroscopy (ICP-OES), ICP-MS, and emission scanning electron microscopy, the latter of which has enabled the investigation of metal nanoparticles in bees.2,37,144 In ICP-based methods, sample preparation typically involves microwave-assisted digestion of material into a mixture of acid(s) and hydrogen peroxide. Toxic elements appear to be more concentrated in honey bees than in honey, indicating that whole bees may be a more informative specimen than hive products in identifying toxicoses from these analytes. 37 Both ICP-OES and ICP-MS are well-suited to multi-element analyses for a comprehensive picture of elemental toxin load.
Treatment
Once poisoning incidents have been diagnosed, treatment options are limited. Removing the source of exposure, or moving colonies away from the source of exposure, is essential. Either situation may require replacement of old frames with new frames to remove residual contamination, plus supplemental feeding to replace the loss of honey and bee bread. 133 Very often, severely affected colonies are destroyed. 133
Xenobiotic exposure and disposition in honey bees
Exposure and absorption
Honey bee colonies are exposed to xenobiotics through food and environmental material collected by foragers and through pharmaceuticals and pesticides applied directly to the colonies for control of pathogenic microbes, parasites, and other pests.34,43 Foragers are the end-stage of the worker caste and have the most exposure to environmental contaminants through direct contact and resource collection.34,118,136 The cuticle of the bee has a surface composed of hydrocarbons that can be crossed by many lipophilic compounds. 34 The wings are intensely vascularized and account for about a third of the surface area of the bee; thus, they are a potential route of exposure and absorption, particularly to volatilized pesticides. 100 Respiratory exposure and absorption are also likely when foragers are exposed to volatilized pesticides. 136
Exposure is likely to occur when foragers are exposed to agrchemicals being applied to crops, or after, when residues remain on surfaces, in plant tissues, guttation droplets, or other water sources.30,133 There are prohibitions against the use of certain insecticides during the daytime, when bees are foraging, and on blooming crops that attract bees.65,118 However, these prohibitions generally do not apply to agrichemicals other than insecticides, and spayed chemicals on non-blooming crops can drift onto wild flowering plants, thus exposing foragers. 83 Some pesticides, such as many of the neonicotinoids, spinosads, and fipronil, are systemic pesticides; similar to the spot-on parasiticide products used in dogs and cats, once applied to plants, these insecticides remain in the tissues for a prolonged period.30,133 Foragers can ingest xenobiotics in the field through nectar or water, and share contaminants through allogrooming or trophallaxis.34,136 When foragers are exposed to acutely toxic compounds at sufficient concentrations, rapid death has a protective function for the colony by preventing these foragers from bringing contaminated material back to the hive. 118 Chronic exposure to low concentrations of some chemicals can lead to accumulation in the bee’s fat bodies and potentially to chronic morbidity. 34
Contaminants that foragers bring back to the hive can expose the queen, drones, brood, and younger adult workers.64,93,115,118 Lipophilic pesticides can accumulate in the wax, which can be reused by bees for years, or even decades, potentially exposing stored products and larvae, thus history should include when the wax frames were introduced to the hive, if possible.25,34 The color of wax darkens over time in the hive, and older, likely more contaminated wax, will be darker than new wax. The wax and pollen loads from foragers typically contain the highest concentrations of pesticides compared to brood and honey, and thus are appropriate samples to determine chronic (wax) and recent (pollen) exposures. 25 Many of the pesticides detected in wax have been acaricides used by beekeepers, although field pesticides are also detected routinely.88,93 Additionally, wax is used in food and cosmetics, indicating potential for minor human exposure. 139 Managed colony frames should be replaced every few years to minimize residues in wax. 25
Because many pesticides are lipophilic, honey samples generally have the lowest pesticide concentrations compared to pollen, bees, larvae, and wax. However, honey can accumulate some hydrophilic compounds and is also a potential source of minor exposure for humans to these contaminants. 25 As noted, pollen collected by foragers is a significant source of xenobiotics to the hive, where it is stored in wax as bee bread to be consumed by nurse bees and young workers. 112 Neonicotinoid insecticides, which tend to be water-soluble and are found in pollen from treated plants, have been detected in bee bread.35,40,96 The herbicide glyphosate has been found at relatively high concentrations, and fungicides have also been detected in bee bread, potentially impacting fermentation.39,133 Royal jelly, bee bread, and honey are mildly acidic, which can decrease ionization and increase gastrointestinal absorption of weakly acidic compounds such as glyphosate, whereas more basic chemicals, such as the sulfonylurea herbicides, will be ionized and therefore have decreased gastrointestinal absorption. 34
Metabolism and elimination pathways
Metabolism of xenobiotics in insects, similar to vertebrates, utilizes phase I–III enzyme systems.34,55 Metabolic enzymes in bees are located in the cuticle, fat body, Malpighian tubules of the excretory system, midgut, nervous system, and within mitochondria.34,51,135 The fat body, in particular, serves a detoxification role similar to the liver. The fat body is the target of Varroa destructor mites, which may have implications for xenobiotic metabolism. 104 The unique complement of metabolic enzymes in honey bees and related species, the potential for interactions caused by simultaneous exposure to multiple xenobiotics in the field, and the recently discovered potential for damage to fat bodies caused by the common parasite V. destructor, necessitates further research, development, and adoption of clinical assessment tools for enzyme activity in honey bees. Such tools will enhance the ability of the toxicologist to assess honey bee susceptibility to toxicants on a case-by-case basis and inform the diagnosis of toxicoses.
Bees have fewer than half of the genes coding for detoxification enzymes that are found in other insects.34,55 This may be a result of the mutualistic relationship between pollinators and angiosperm plants.10,78 Whereas bees have a symbiotic relationship with plants, taking nectar and pollen in exchange for pollination services, other herbivorous insects ingest vegetative tissue of plants, in which phytotoxins have evolved as a defense mechanism. 10 Metabolic enzymes in bees have evolved to metabolize flavonoids and other chemicals specifically found in nectar and pollen. 34 Phase I and phase II enzymes are inducible in both larval and mature bees, and upregulation in larvae may be associated with contaminants in food or wax. 105
The primary class of phase I metabolic enzymes in bees is the cytochrome P450 (CYP) monooxygenase group, although bees have fewer isotypes than other insect species. 34 There are 46 CYP genes in honey bees (Table 2).10,34 Bees have reduced CYP3 and CYP4 enzyme activity compared to other insects.10,34 The CYP9 enzymes are inhibited by demethylation-inhibiting (DMI) fungicides and the pyrethroid insecticide bifenthrin. 40 Interestingly, the P450 enzymes in honey bees are not induced by phenobarbital, as they are in insects of the orders Lepidoptera and Diptera, and in Drosophila melanogaster. 66 Honey induces P450 enzyme production in both bees and humans, most likely in response to the flavonoids present. 66
An incomplete list of important phase I cytochrome P450 (CYP) monooxygenase enzymes in honey bees and their known functions.

Methods for determining CYP gene expression in honey bees using RNA extraction and sequence analysis, and PCR, have been developed. 78 Messenger RNA expression for P450s was measured by isolating RNA via automated gel electrophoresis and using an mRNA sequence. 42 Similarly, mRNA was isolated using electrophoresis and synthesized cDNA primers for PCR. 51 An assay was developed for CYP9Q1 activity based on the addition of quercetin dissolved in dimethyl sulfoxide to midgut homogenate, then analyzing an ethyl acetate extract via LC for quercetin. 78 Although CYP6As also metabolizes quercetin, the CYP9Q1 has higher activity. 78
The other major phase I enzyme group includes the carboxylesterases, which are relatively deficient in bees compared to other insects. 34 Honey bees have 24 genes for carboxylesterases, but some mosquitoes have twice as many genes. 34 Expression varies with age and caste, and is highest in foragers. 34 Carboxylesterases are upregulated by some insecticides including neonicotinoids, pyrethroids, spinosyns, and fipronil, and inhibited by the organophosphorus compound SSS-tributyl phosphorotrithionate. 18
Phase II enzymes are involved in conjugation reactions for elimination of xenobiotics. The delta and epsilon classes of glutathione and glutathione S-transferases (GST), specific to insects, are of prominent importance in honey bees for detoxifying reactive oxygen and nitrogen species and electrophiles.34,117 The pyrethroid insecticides and coumaphos induce GST; diethyl maleate, used in malathion production, inhibits GST. 117 A method has been developed to directly measure GST activity in honey bee tissues using glutathione and 5,5′-dithiobis(2-nitrobenzoic acid) as substrates and measuring the UV spectrum at 320 nm. 70 Messenger RNA expression was measured for GST by isolating mRNA and comparing it to the sequence library. 42
Phase III proteins are transporters used to move metabolites across cell membranes and promote excretion through Malpighian tubules. 34 These are found mostly in the fat body, Malpighian tubules, hindgut, and cuticle. 34 Important among them are the multi-drug-resistance (MDR) protein transporter group, and in particular the ABC multidrug transporters, which require ATP to move metabolites across cell membranes. 50 There are 41 genes for ABC transporters in bees, including 3 subfamilies, and they are induced in larvae exposed to nicotine.34,105 Inhibitors of ABC transporters include azole fungicides, oxytetracycline, fumagillin, and quercetin, and substrates include several neonicotinoid insecticides and macrocyclic lactone pesticides.34,50
Metabolism of toxicants is associated with significant oxidative stress in insects, necessitating antioxidant enzymes. 117 These include superoxide dismutase (SOD) and catalase (CAT). Also included with the antioxidant enzymes is GST. Honey contains CAT, potentially to reduce hydrogen peroxide concentrations. 117 Both fipronil and spinosads induce CAT activity in honey bees. 22 SOD and CAT expression was measured using mRNA isolation and sequencing. 42 An assay for CAT activity was adapted based on incubating tissue extracts with hydrogen peroxide in a phosphate buffer and measuring absorbance at 240 nm. 22
Elimination of metabolic waste is accomplished through the Malpighian tubules. There are 4 tubules in larvae and 64 in adults, but pupae lack Malpighian tubules, which may impact their ability to eliminate xenobiotics. 34
Limitations of data from honey bee toxicity studies
The U.S. Environmental Protection Agency (EPA) regulates pesticide use in the United States. The EPA is responsible for and has published guidelines on pesticide use and risk assessment for pollinators, divided into 3 tiers (Table 3).124–126 Tier 1 involves estimates of exposure based on established estimated environmental concentrations (EECs) and acute and chronic toxicity testing data.
U.S. EPA pesticide risk assessment tiers for honey bees based on life stage, duration of exposure, and endpoints, which include the no-adverse-effect level (NOAEL).
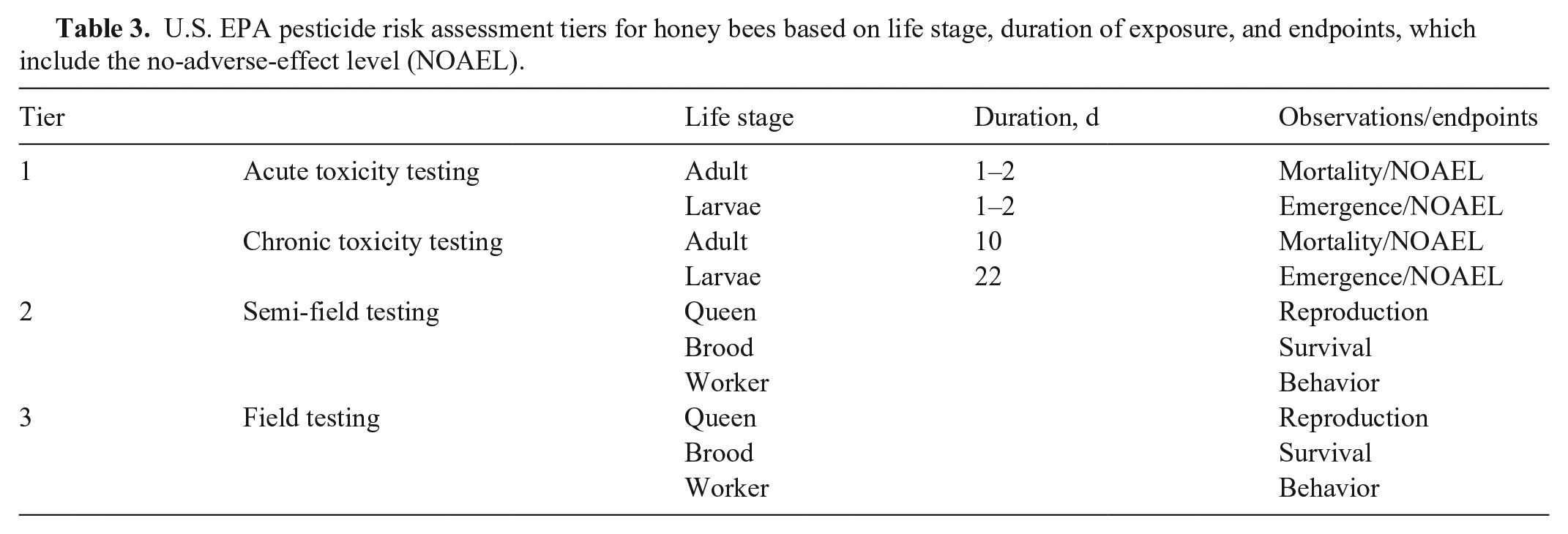
Toxicity data generated for the tier 1 EPA guidelines rate pesticides by categories (Table 4). 126 There are several drawbacks to the use of this type of toxicity testing. First, these bioassays were designed to determine effectiveness on killing target species of pest insects. 119 These assays were not developed for eusocial insects; thus, they consider the impact of the pesticide on the individual forager bee and do not consider colony-level effects.10,119 Forager bees can transfer the xenobiotic to others in the colony through retrieved resources, impacting other individuals, even generations, within the hive, and potentially impacting the long-term survival of the colony.10,120,122 Underlying heath issues commonly seen in honey bees, such as poor nutritional status and pathogen load, are not considered in toxicity testing. 119 Also not considered is the importance of mixture toxicology: honey bees in the real world are exposed to multiple pesticides, phytotoxins, surfactants, potentiators, mycotoxins, and other xenobiotics on a daily basis.30,119,136
Toxicity ratings for honey bees based on tier 1 acute toxicity testing and given in µg/bee. The LD50 is the calculated lethal dose for 50% of the population.

EPA tier 2 requires semi-field and field testing (Table 3) and can involve studies on small captive colonies in tunnel enclosures, or on small free-ranging colonies fed known concentrations of the pesticide in question.65,83,124 Tier 3 studies are intended to best represent field conditions; a colony is released near a bee-attractive blooming crop that has been treated with the pesticide under study.65,124
Other methodologies for assessing the effects of pesticides on honey bee colonies are in development, including cell culture techniques, which have the potential to improve knowledge of mechanistic toxicology. 46 Modeling applications to look at the big picture have been designed (Table 5). The Bee Informed Partnership (BIP) is particularly suited for the superorganism that is the honey bee colony (https://beeinformed.org/). BIP uses tools developed by public health practitioners, such as the socio-ecologic system framework, and approaches risk factors through epidemiologic investigation to inform management practices. 119 Through these mechanisms, improvements to understanding of apiary toxicology are likely on the horizon.
Modeling applications that have been designed to assess xenobiotic exposure and impacts on domestic honey bee colonies.

Impacts of poisoning on bee colonies
Bee colonies are exposed to a number of different toxicants by several different routes. 18 Pesticides, often in mixtures with other pesticides, solvents, and potentiators, have been inadvertently sprayed directly on hives, but more commonly are brought home by workers who have foraged on contaminated cultivated or wild plants.1,83,131 Agricultural chemicals have a wide range of toxicities and mechanisms of action, but generally, insecticides are more toxic to bees than fungicides or herbicides. 65 Other organic and elemental contaminants (such as heavy metals) can accumulate in urban and industrial areas where bees forage. These anthropogenic toxicants can remain present in stored food or within the wax and propolis of the hive itself and become an ongoing source of exposure for larvae, young workers, and the other castes that live in the colony. Plants themselves produce a number of alkaloids, flavones, terpenoids, and other phytotoxins that can impact foragers and stored food. Toxigenic fungi can grow on food stores in the colony. Beekeepers and veterinarians treat hives directly with parasiticides and antimicrobials to protect their colonies from disease, and these can also remain in the wax for a long time. Effects of these multiple xenobiotics can be additive, synergistic, or antagonistic, depending on the combinations.25,65 Clinical effects are dependent not only on chemical exposures, but also on environmental, health, management, and genetic factors. 64 Effects can run the gamut from sublethal or subclinical changes to outright mortality. 30
Clinical signs with acutely toxic insecticides
With the exception of the rare cases in which hives are sprayed directly, rapidly lethal chemicals used in the field can have somewhat protective actions by preventing contaminated foragers from bringing the chemicals back to the colony. 90 It is unlikely that dead bees will be observed, given that they die in the field and away from the hive. 90 When observed bee kills do occur, often more than one colony is affected. 65 Bees are found dead or dying around the entrance to the hive or on the bottom board.65,118 Poisoned bees are likely to be nurse bees who have ingested contaminated pollen or nectar, which impacts the brood and queen that rely on nurse bees for food. 65
Clinical signs with sublethal and chronic toxic pesticide exposures
The most common impacts of xenobiotics on colony health are sublethal: pesticides brought to the hive by foragers impact the colony, but no mortality is observed directly.30,67,133 Contaminants can accumulate in the fat body of the bee over time. 34 Although there is no initial impact observed, sublethal exposures can decrease the colony’s potential for growth and long-term survival.30,133 Behavioral effects are considered a more sensitive indicator of toxicant exposure than overt mortality. 116 Altered foraging behavior has been reported, as have declines in learning and cognitive function in workers, with potential long-term impacts on colony growth and viability.30,64,65 Increased food intake for nurse bees is reported with some pesticides, which increases exposure to the nurse bees and transmission to the queen and brood. 10 This can impact queen fecundity and behavior, as well as brood development and survival, both of which have implications for future generations of the hive. 10 Effects have also been reported on immunocompetence, including social effects (through grooming and other sanitary behaviors), as well as cellular and humoral immunity. 30
Chemical and metabolite residues in the hive can cause chronic toxicant exposure. 133 Agrochemicals that have been reported in honey, pollen, and wax samples from hives have included pyrethroids, organophosphorus and methyl carbamate acetylcholinesterase inhibitors (AChEIs), neonicotinoid insecticides, insect growth regulators (IGRs) organochlorine insecticides, synergist compounds (such as piperonyl butoxide), a variety of fungicides, herbicides, acaricides, and other products used in colony management.25,68,69,88,112,133,138
Important toxicants in honey bee health
Pharmaceuticals and acaricides
The most common pesticide compounds found in wax and other samples from honey bee colonies are acaricides used to treat Varroa mites (Table 6), parasites that were introduced to European honey bees and that, uncontrolled, can cause significant colony losses.25,88,119 The most common miticides found include the pyrethroid τ-fluvalinate, the organophosphorus compound coumaphos (both were present in ≥ 98% of wax samples collected in North America 88 ), and metabolites of amitraz. 93 Parasite control is critical, but involves trade-offs because acaricides are also toxic to bees; for example, the use of amitraz or τ-fluvalinate and coumaphos did not impact growth or winter survival of bees, but Varroa mite infestations of > 3 per 100 bees does impact winter survival of new colonies. 93 Coumaphos, a cholinesterase-inhibiting acaricide, has been associated with decreased larval survival.54,64 Coumaphos can persist in the hive for > 1 y. 133 The pyrethroid τ-fluvalinate is also used on field crops and has been detected in bees, pollen, wax, and honey.25,138 Amitraz is used in polyethylene strips or combustible tablets for fumigation. 99 Amitraz is not usually found in wax or honey because of rapid degradation, but breakdown products that can be detected in bees and honey, especially after fumigant use, include 2,4-dimethylphenyl formamide, N-2,4-demethylphenyl formamide, and 2,4-dimethyl aniline.69,99,138
Acaricides used in apiaries, their pesticide classifications, and some examples of known interactions with other xenobiotics.

= additive; ++ = synergistic.
Naphthalene, which has been detected in wax and honey, has been used to control wax moths, but this use is not legal in the United States. 67 Other compounds that can be used legally to control moths include 1,4-dichlorobenzine and 1,2-dibromomethane, which can both be detected in wax and honey after treatment. 67 One more is the Bacillus thuringiensis (Bt) Cry toxin, which is a pore-forming δ-endotoxin from B. thuringiensis var. aizawai. Commonly found in genetically modified organism (GMO) crops, Bt Cry toxins can produce osmotic shock as a result of disruption of the midgut epithelium.64,133
Antimicrobials include lincomycin, used inappropriately for American foulbrood, oxytetracycline, used to treat European foulbrood, and tylosin.31,34 Adverse effects have been reported with tetracyclines, including damage to midgut epithelium from oxytetracycline, and slowed larval growth and changes in larval pigmentation from chlortetracycline. 64 Use of antibiotics requires proper dosing and withdrawal times to prevent residues in apiary products. 66 High-dose application of oxytetracycline has been associated with the presence of honey residues for 3 y. 68 Chloramphenicol has been detected in bee products imported from China and India, but is not legal for use on honey bee colonies in the United States. 67 Fumagillin-B is not FDA-approved and may cause residues in honey, but has been used to treat Nosema spp. protozoal parasites.31,66
Other possible sources of antibiotic residues in bee products include runoff from treated livestock and use on crops. 67 Sulfonamides and streptomycin are used on citrus crops, and sulfonamide residues in particular are very stable in honey. 67
Agricultural chemicals
The major classes of insecticides used in agriculture have included the organophosphorus and methyl carbamate AChEIs, pyrethroids, neonicotinoids, and IGRs (Table 1). 30
Pyrethroids are very common insecticidal products used in agriculture, and residues are commonly detected in samples from colonies.88,96 Because pyrethroids are rapidly neurotoxic, foragers will die in the field or lose the ability to fly, making it less likely that they will return to the colony.65,133 Permethrin concentrations of 20 ppm were found in brood and adult bees during a permethrin-caused bee kill. 88
Organophosphorus and methyl carbamate insecticides represent the AChEIs, and have been involved in bee kills.69,88,101 The use of AChEIs has been declining; these insecticides are replaced by pyrethroids and neonicotinoids, but they are still common and often detected in pollen, nectar, and honey.1,35 Sublethal effects of AChEIs include increased AChE gene transcription, increased grooming behavior, and cessation of egg laying in queens, although colony impacts may not be evident.1,107,118 Parathion methyl deserves special consideration; a microcapsule formulation was developed for delayed release and to prevent drift. 67 Unfortunately, these microcapsules are similar in size to pollen and attach to the surface of the bee, to be brought to the hive and stored, which has resulted in thousands of colonies lost in Italy and Uruguay.67,90,101
Several systemic insecticides are placed on seed coatings and circulate in the tissues of the plant as it grows, and these include the neonicotinoid class of insecticides and fipronil. 114 Experimental studies and mathematical modeling have not consistently shown increased mortality in worker bees ingesting pollen of plants treated with systemic insecticides, and the impacts on the hive are not yet understood.54,114 The neonicotinoid class of insecticides was introduced in the 1990s and, in 2010, accounted for approximately one-third of the pesticide use in agriculture.29,35,40,96 Neonicotinoids are used in 120 countries on > 140 types of crops; virtually all corn that is grown in North America has been treated with these insecticides.71,75,96 Bee kills have been associated with corn, cotton, and sunflower fields in Germany, France, Slovenia, the United States, and Canada.67,75 Most bee kills occur at the time of planting, when dust from the seed coating is exhausted into the environment and drifts over areas between fields, contaminating wild food sources such as dandelions. 71 Increased worker bee losses were recorded in Italy during corn planting time over 3 y of observation; these episodes have declined after the use of neonicotinoid insecticides in seed coats was banned in that country. 75 A similar use ban has been proposed in the state of New York. 45
There are 2 classes of neonicotinoid insecticides: the nitroguanidines and the cyanoguanidines, the latter of which are approximately one-third as toxic to bees. 40 The most common of the nitroguanidines is imidacloprid, which is rapidly metabolized in bees to 6-chloronicotinic acid, imidacloprid urea, imidacloprid guanidine, and imidacloprid olefin.25,44,69 Lesions in larvae exposed to imidacloprid include increased cell death in the hypopharyngeal gland, and, in queens, epithelial cell death in the midgut and degeneration and resorption of the ovariole have been documented.10,85 Clothianidin is another nitroguanidine neonicotinoid, and the third most common pesticide detected in dead honey bees.69,123,127 Thiamethoxam is rapidly metabolized to clothianidin.91,122 Acetamiprid, a common cyanoguanidine, is metabolized to acetamiprid-N-desmethyl and 6-chloronicotinic acid.25,44 Larvae are more sensitive to acetamiprid than mature bees, and synergism with fungicides is possible. 16
Another systemic insecticide of interest is fipronil, which is used in veterinary medicine as a systemic parasiticide, and agricultural use has been associated with bee kills. 88 Fipronil and its metabolites fipronil sulfone and disulfonyl fipronil have been detected in field-exposed bees.25,68,130 Fipronil is known to reduce drone fertility and cause histologic changes in the hypopharyngeal glands of nurse bees.65,68
The IGR class of insecticides is a broad group of compounds with endocrine effects (Table 1). They are usually more toxic to larvae than adults. 64 Methoxyfenozide is known to decrease worker survival at field-relevant concentrations.68,136
The most commonly used agricultural pesticides are the fungicides.30,66 Consequently, they are frequently detected in honey bee colonies. 64 Because they are not intended to kill insects, fungicides can be applied to crops during blooms and when bees are present.66,88 Although not likely to directly impact foraging bees, fungicides have the potential to impact beneficial fungal microflora of the hive, including those used in bee bread production.34,133 Interactions of fungicides with other agrochemicals may be relevant, for example, prochloraz inhibits P450 metabolism of pyrethroid insecticides by honey bees. 66
Similar to fungicides, herbicides are not intended to impact insects and thus do not have use restrictions related to honey bees. 64 Herbicides are used to kill weeds, some of which are the flowering plants that bees use as forage.54,118,133 The restricted-use herbicide paraquat is known to decrease the lifespan of bees.34,64 Phenoxy-herbicides may impact thermoregulation through inhibition of ATP production. 54 Glyphosate use has increased since the 1990s given its broad spectrum and usefulness with GMO crops and no-till agriculture. 132 Effects of glyphosate on bees include disruption of the gut microbiome; impaired learning, short-term memory, and foraging behavior; antioxidant depletion; immunocompromise; and delayed larval development.39,54,132 Modulation of P450s, including downregulation of CYP4G11, by glyphosate has been documented.10,132
Phytotoxins and mycotoxins
Not all weedy plants are beneficial to honey bee health or production. Pollen contains phenolic compounds and nectar often contains other phytotoxins. 64 Flavonoids are produced by plants to avoid herbivory, but they are also important in imparting flavor to honey. 64 The flavonoid quercetin, a major component of pollen that imparts the yellow color, is also found in propolis and nectar. 80 Quercetin is an important substrate for the CYP6 and CYP9 enzymes, which are inhibited by triazole fungicides.18,80 Quercetin inhibits mitochondrial ATP synthase, compromising energy production and flight in foragers and thermoregulation of the colony. 80 It is also reported to cause ovarian activity in mature workers, potentially destabilizing the social structure of the colony. 80
Specific phytotoxins impacting bee health have been documented. A unique toxic plant problem occasionally encountered is purple brood, which is caused by foraging on the plant Cyrilla racemiflora, a tropical plant from South America that grows in parts of the United States and causes discoloration and death of larvae (https://beeinformed.org/). The saponins of Aesculus californica, the Pacific horse chestnut (syn. California buckeye) found in the northwestern United States, can cause fatal paralysis in bees. 133 The polyphenol compounds from coriander (Coriandrum sativum), an Old World plant and international culinary herb, have been associated with mortality in honey bee larvae. 72 Extensive losses of Brazilian bees have been caused by pollen from the Stryphnodendron polyphyllum tree, the cause of Brazilian sacbrood-like disease (BSBD).20,21 Many plants, including but not limited to those in the Prunus family (almonds and stone fruit), can have cyanogenic glycosides, such as amygdalin, in nectar and pollen. 64 Cyanogenic glycosides can release cyanide, which inhibits mitochondrial oxidative phosphorylation. 86 Although bees are relatively resistant, cyanide exposure can cause abdomen dragging and dorsal recumbency. 86 Plants produce a number of alkaloids that can be found in various concentrations in pollen and nectar. 64 A few have been associated with mortality in bees, including the steroidal alkaloids, which open voltage-gated calcium channels, from Veratrum spp. and Zigadenus spp., which grow in temperate climates of the northern hemisphere for the former, and is localized in the southeastern United States for the latter. 59
Other phytochemicals have variable impact on bee health and development. Pyrrolizidine alkaloids are found in pollen from many plants, including those in tribe Senecioneae, genus Crotalaria, and families Boraginaceae and Asteraceae, and bees do not detoxify them.64,94,133 High doses of pyrrolizidine alkaloids can cause mortality in honey bees, but these genotoxic compounds, which also cause pulmonary hypertension in people, are frequently detected in honey.64,94 The methylxanthines—caffeine, theophylline, and theobromine—can be detected in honey from bees that have foraged on coffee, citrus, and tea plants.64,120 Caffeine benefits olfactory learning, which influences pollen-collection behavior, in foraging honey bees. 7 Nicotine is found in pollen and nectar of tobacco and related plants and can be detected in honey.105,120 Nicotine causes upregulation of the pentose phosphate pathway in bees, leading to glycolysis, and promotes larval growth. 105 Larvae are able to metabolize nicotine to cotinine, cotinine N-oxide, and inactive 4-hydroxy-4-(3-pyriidyl) butanoic acid. 105
Occasionally, phytotoxin contamination of honey has impacted consumers, sometimes with fatal consequences.49,113,146 This was demonstrated by early experimental toxicologist and king of Pontus, Mithridates VI Eupator, who used honey from bees that foraged on plants in the family Ericaceae to poison the soldiers of Pompey the Great, circa 70 BC. 129 Contamination of bee products may increase as small-scale and amateur beekeeping continues to flourish internationally.9,143 The Ericaceae, which are found in extensive areas of Asia, but also in North America, Europe, and Australia, include rhododendrons (Rhododendron spp.), pieris (Pieris spp.), and mountain laurels (Kalmia spp.), and produce grayanotoxins, which activate voltage-gated sodium ion channels leading to hyperpolarization of the neuron. 49 Since the time of Mithridates, grayanotoxin-containing honey has been sought as a novelty and herbal remedy, and life-threatening cardiac dysrhythmias have been reported in consumers. 49 Other phytotoxin-related poisonings have been reported from ingestion of honey from Southeast Asia as a result of contamination with phytotoxins, including triptolide, a toxic diterpenoid from the tree Tripterygium wilfordii, and, in separate incidents, indole alkaloids from Gelsemium elegans. 146 Severe poisonings characterized by vomiting and seizures were reported in New Zealand and associated with honey containing tutin, a picrotoxin-like phytotoxin, after bees foraged on honeydew extracted from the plant Coriaria arborea. 9
Because toxigenic Aspergillus spp. of fungus can grow in honey, the mycotoxins aflatoxin B1 and ochratoxins are often detected in honey.64,120 Bees are able to inactivate these mycotoxins, but aflatoxin B1 may impact the life expectancy of workers. 64
Minerals, heavy metals, and miscellaneous
Elemental contaminants are environmentally stable and often have slow elimination kinetics, thus they can be associated with chronic accumulation and morbidity.57,101 Important heavy metal contaminants in the environment include lead, mercury, arsenic, and cadmium, but lithium, zinc, and aluminum will also be considered.27,37,57,101,103,110,144 Lead has been detected in samples of bees, honey, and wax in different parts of the world.37,74,103,120 Concentrations in honey are relatively low, but up to 3 ppm lead has been detected in wax. 144 Lead at clinically relevant doses (0.28 ng/bee) impaired learning and memory in worker bees. 84 Similarly, mercury has been detected in bees, honey, and wax, with wax concentrations > 1 ppm reported.41,103,144 Cadmium has been associated with decreases in honey production and population losses in exposed colonies, and there is no safe limit of exposure for bees.57,64,116 Cadmium has been detected in bees and honey.37,103,144
Arsenic, which is a metalloid, is of particular interest because it has historically been used in agricultural pesticides and wood preservatives, and thus there are circumstances in which honey bees have a high potential for exposure. 64 Aside from the mishap with Paris green in 1881, arsenical products used to preserve wood in hives have resulted in increased winter mortality.64,121 Arsenic has been detected in bees, wax, pollen, honey, and larvae. 144 Arsenic exposure is documented to impair honey bee learning and memory when fed at clinically relevant doses (0.15 ng/bee), and effects are additive with lead and copper. 84
Tin has been associated with winter mortality when used as a wood preservative in hives, and has been detected in wax at concentrations > 3 ppm.64,144 Aluminum has been associated with shortened lifespan in European honey bees, which are more sensitive than the Mediterranean subspecies, and has been detected in honey (6 ppm), larvae (11 ppm), bees (50 ppm), and pollen (60 ppm).27,144 Lithium has been considered for use in the control of Varroa mites, and although lithium formate is relatively safe, other forms were found to reduce bee lifespans. 110
Metal oxide nanoparticles, which are widely used in some industries, have been reported to impact bees.2,32 These particles are small enough to adhere to the bodies of bees and be inhaled into the spiracles, but they may also be ingested in nectar, pollen, or water, where they can cause direct epithelial damage in the midgut.2,32 Nanoparticles have been detected in lysosomes of phagocytic cells and are reported to upregulate catalase, superoxide dismutase, and CYP4G11.2,32
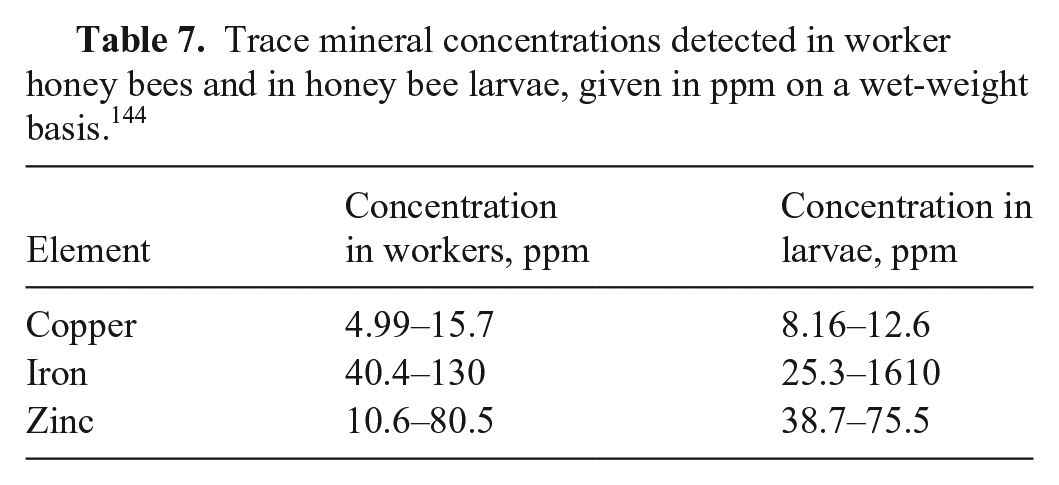
Nutritional trace minerals include copper, iron, selenium, and zinc. Copper naphthenate has been used in wood preservatives used in hives and was associated with decreased survival of the brood and queen, with a copper concentration of 21 ppm in affected bees. 64 Trace mineral concentrations have been reported in mature worker bees and larvae (Table 7). 144 Selenium concentrations in soils in the United States range remarkably across the country, and some plants are known to accumulate selenium and produce pollen concentrations up to 2,830 ppm. 64 Selenium-containing amino acids are less toxic to bees than inorganic selenium, and larvae are much more sensitive to selenium compared to adults. 64 Reported selenium concentrations in bees range from below the limit of detection to 0.76 ppm on a dry matter basis. 37
Trace mineral concentrations detected in worker honey bees and in honey bee larvae, given in ppm on a wet-weight basis. 144
Microplastics and nanoplastics are environmentally ubiquitous and mostly come from degradation of other plastics, including textiles, tires, agricultural plastics, artificial turfs, road paints, single-use plastics, and construction. 141 Microplastics are < 5 mm in diameter, and nanoplastics are small enough to cross biological membranes. 62 Microplastics and nanoplastics may act as a lipid sink for other contaminants or a vector for pathogens. 3 Ingestion of plastic particles by bees has been associated with decreased microbiome diversity, increased oxidative stress, and increased sensitivity to tetracyclines. 62 Microplastics have been found in bees and bee products in South America and Europe, and some potentially came from garments of beekeepers. 3
Conclusions and future directions
The FDA implemented the Veterinary Feed Directive Rule in 2017, which classified honey bees as livestock requiring veterinary care. 31 Part of the role of veterinarians in honey bee health is the diagnosis of honey bee diseases, including toxicoses, but few veterinary diagnostic toxicologists in North America currently have experience with honey bees, and although interest in apiary diseases is growing internationally, there is limited veterinary toxicology expertise.4,36,128,134,142 Interpreting honey bee test results requires understanding that the hive is a super-organism, familiarity with sources of xenobiotic exposure and interactions, and a basic understanding of the complex interactions between honey bees, their pathogens, and their environment. Veterinary toxicology relies heavily on analytical chemistry; proper sample collection, critical to the detection of toxicant exposures, relies upon an understanding of the exposure routes for bees of all life stages and castes, xenobiotic interactions, metabolism, and degradation, and solubility of different compounds within the various matrices of the hive. Given that honey bees are production animals, the analytic toxicology laboratory also has a role in residue avoidance in bee products such as honey, wax, and pollen. 143
Most of the current toxicity data for honey bees is based on LD50 values from individual insect studies; however, this information does not translate well to the super-organism, which can withstand the loss of a large number of individual workers, nor does it account for the complex mixtures of xenobiotics to which honey bees are exposed throughout their life cycle. 10 Chronic and sublethal exposures are likely to impact queen health and brood survival, and therefore sustainability of the colony.30,133 Additionally, historic LD50 data did not take into consideration the recently discovered impacts of Varroa spp. mites on the fat body, a major organ involved in xenobiotic metabolism in the honey bee. 104 Methods have been developed to determine CYP gene regulation and expression in honey bees, including PCR methods, enzyme function assays, and chromatography, electrophoresis, and spectroscopy techniques that are amenable to techniques already in use in veterinary diagnostic laboratories.22,42,51,70,80
Finally, honey bees are potential sentinels for environmental contamination. Precipitous declines in insect populations have been documented in areas of the world where they have been studied. 48 Many species of wild insect pollinators, including bees, are in decline. The International Union of Conservation of Nature lists 9 species of bees as critically endangered, 18 species as endangered, and 10 as threatened (https://www.iucnredlist.org/en). Although pesticides and other xenobiotics impact wild bees, there is currently limited information on these effects; examples include the effects of neonicotinoids on western bumble bees (Bombus occidentalis), 61 eastern bumble bees (Bombus impatiens), 16 and orchard mason bees (Osmia lignaria), 96 and diflubenzuron impacts on eastern bumble bees. 17 The limited comparative toxicology studies that generated data from honey bees and wild bee species have had mixed results.55,96,116 However, residue data from honey bee colonies can be used in estimating exposure for wild bee populations, and DEBtox modeling has been used to predict differences in xenobiotic impacts on different bee species.55,116 More generally, honey bees have been used as sentinels for environmental contaminants that impact public health.13,28,73,74,76,89,145
Footnotes
Acknowledgements
We thank Dr. Scott McArt for his help in reviewing and revising this manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
