Abstract
The microsporidian pathogens Vairimorpha apis and V. ceranae are known to cause intestinal infection in honey bees and are associated with decreased colony productivity and colony loss. The widely accepted method for determining Vairimorpha colony infection level for risk assessment and antibiotic treatment is based on spore counts of 60 pooled worker bees using light microscopy. Given that honey bee colonies consist of as many as 1,000 times more individuals, the number of bees collected for Vairimorpha detection may significantly impact the estimated colony infection level, especially in the case of uneven distribution of high- and low-infected individuals within a hive. Hence, we compared the frequency and severity of Vairimorpha infection in individual bees to pooled samples of 60, 120, and 180 bees, as well as compared the Vairimorpha spp. prevalence in pooled samples of 60 and 180 bees. Overall, we did not find significant differences in spore counts in pooled samples containing incremental numbers of bees, although we observed that, in less-infected colonies, a low frequency of highly infected individuals influenced the estimated colony infection level. Moreover, Vairimorpha spp. prevalence did not differ significantly among the pooled bee samples tested. Increasing the number of pooled bees from the recommended 60 bees to 180 bees did not yield a more accurate representation of colony infection level for highly infected colonies, but the clinical importance of a low frequency of highly infected individuals in less-infected colonies needs to be addressed in future studies.
The Western honey bee (Apis mellifera) plays a crucial economic and ecologic role in providing pollination services for agriculture and global food production. 17 However, managed honey bees are faced with a large variety of biotic and abiotic stressors such as the fungal pathogens Vairimorpha apis and V. ceranae (Nosematidae; formerly Nosema apis and N. ceranae 26 ), which cause severe and sometimes fatal disease in adult bees. 13 Clinical signs of vairimorphosis (nosemosis) include dysentery with nutritional stress and malnutrition, shortened lifespan, impaired flight ability, and disorientation in individual forager bees in correlation with reduced colony productivity as a result of poor foraging performance and brood care.7,20,25 At the colony level, Vairimorpha spp. infection weakens colonies and increases the risk of overwinter mortality, accompanied by high economic losses for the beekeeping industry.3,4,10,11 The widely accepted method for estimation of Vairimorpha spp. infection at the colony level is to collect 60 forager bees from the hive entrance in spring and fall, macerate the bees to extract intestinal contents, followed by a microscopic spore count in a hemocytometer to estimate the spore load per bee.2,16,21 The total number of bees in a colony ranges from ~20,000 during early spring to ~60,000 during late summer, with 30% of the oldest bees in a colony being foragers, and reportedly having the highest spore loads in a colony.21,27 Considering that only a small portion of bees (0.1–0.3%) is used for estimation of the colony infection level, the true parasite burden and the distribution of infection levels of individual bees inside a colony may not be reliably reflected with a sample size of 60 foragers.14,16,21 Vairimorpha spp. surveillance of our research apiaries (~200 hives) in the summer of 2021 revealed highly variable spore counts and Vairimorpha spp. composition in sequential samples (containing 60 bees) from the same hive within one day and on the following days (Suppl. Table 1), which is in strong concordance with another report of the variation in Vairimorpha species prevalence after subsequential sampling. 2
To evaluate the effect of sampling methods on estimation of Vairimorpha colony infection level, we infected colonies with ~50:50 mixture of V. apis and V. ceranae in the fall and performed the following analysis during the following spring: 1) compared the average number of spores per bee determined in incremental, pooled samples of bees; 2) counted spores in individual bees to determine the frequency of severely versus mildly infected bees, and 3) compared the ratio of V. apis to V. ceranae in individual bees and in incremental, pooled samples of bees. A pooled sample can consist of a low number of highly infected bees or an equal distribution of moderately infected bees, resulting in a similar calculated spore count per bee. 16 Accordingly, we analyzed the frequency of spore levels in individual bees and correlated it to pooled samples to find a reliable number of bees to represent the colony infection level (objectives 1, 2). Moreover, single bees can be infected with one or both Vairimorpha species, 22 so we determined the species composition in pooled samples with quantitative real-time PCR (qPCR) to correlate the species ratio with sample size (objective 3).
Materials and methods
Experimental colony preparation and Vairimorpha infection
In September 2021, ~80 honey bee colonies at 2 research apiaries near Saskatoon, SK, Canada were examined for Vairimorpha infection by microscopy and qPCR (60 bees per colony), and 21 colonies were identified as not infected. All colonies were treated with label dosages of oxytetracycline and amitraz, for control of American foulbrood and Varroa mites, respectively, per our region’s standard beekeeping practices. In October 2021, after colonies were wrapped for outdoor overwintering, we randomly assigned the 21 Vairimorpha-free colonies into 3 experimental groups (control, low-dose, high-dose), with 7 colonies per group. Colonies assigned to the low- and high-dose groups were inoculated with 90 × 106 and 900 × 106 Vairimorpha spores, respectively (90 mL of homogenate containing 106 or 107 spores/mL); colonies in the control group were administered 90 mL of a sucrose solution. Vairimorpha spores for the inoculum were obtained from ~1,000 bees from a donor colony that was determined by qPCR of 60 bees to be infected with 50% of both Vairimorpha species. 27 To prepare the inoculum, we crushed the bees in a blender with NH4Cl to avoid spore germination and mixed the spores with 2:1 (w/v) sucrose syrup to the final concentration, having 1:1 (w/v) sucrose syrup in the final suspension. 12 We applied the spore suspension in the inter-frame spaces directly on the bee cluster of the top super (box) with a syringe (Fig. 1). The colonies that we used had never been treated with fumagillin, either in advance of or during our experiment.

Experimental inoculation of a honey bee colony with Vairimorpha suspension.
Colony strength, and Varroa infestation assessment
As an estimate of colony strength, the cluster size of the top super was analyzed from images taken with a digital camera (16.2 megapixel Nikon D7000; Minato) in October 2021, before inoculation, when colonies were already wrapped for outdoor overwintering, and again in March 2022, coinciding with the first visual overwinter survival assessment. 23 We estimated the cluster size by counting the number of inter-frame spaces in the top super occupied by adult bees to the nearest 0.25. 23 Varroa mite infestation per colony was analyzed in March 2022 using the alcohol wash technique.6,18
Colony Vairimorpha assessment
We collected ~300 bees with a vacuum from the lid of the top super in March, April, and May 2022 from all live colonies. Bees were divided into subsamples of 3 × 60 bees and 30 individual bees. Single bees were each macerated in 500 μL of sterile dH2O, and the 3 subsamples containing 60 bees were each homogenized with 60 mL of sterile dH2O. To generate pooled samples of 120 and 180 bees, we combined 400 μL of two 60-bee subsamples (= 120 pooled bees) and 400 μL of three 60-bee samples (= 180 pooled bees). Vairimorpha spore loads per bee were determined by evaluation of a 10-μL aliquot of each individual or 60-, 120-, or 180-bee sample using a hemocytometer and light microscope. 12 Additionally, genomic DNA was extracted from the 60- and 180-bee samples (Blood and tissue kit; Qiagen), and Vairimorpha species composition was determined by qPCR with species-specific primer–probe combinations based on the small ribosomal subunit rDNA. 27 The Ct values from qPCR were transformed to spore numbers using the slope function from duplicate standard curves prepared from 10-fold diluted spores (107–10 spores) of each species.
Statistical analysis
All statistical tests were conducted with R v.4.2.2 (https://www.r-project.org/) and an α = 0.05 significance level. Normality was assessed with the Shapiro–Wilk test. The number of surviving colonies was compared by treatment group and sampling time using the Pearson χ2 test. Vairimorpha spore counts and species prevalence were compared among pooled samples using repeated measures one-way ANOVA with a post hoc paired t-test and Wilcoxon matched-pairs signed rank test, respectively. The effect of treatment and sample size on species prevalence was determined by 2-way ANOVA. One-way ANOVA or the Kruskal–Wallis test were used to analyze the interaction effect of sampling time and treatment on cluster size and Varroa infestation. The correlation between colony infection level and frequency of infection severity or intensity (non-infected to highly infected) or Vairimorpha spp. prevalence was assessed with the Pearson correlation.
Results
Colony overwinter survival, strength, and Varroa infestation level
Of 21 colonies, 10 survived until March 2022, with a statistically nonsignificant dose-response in colony survival (Fig. 2; χ2 = 6.5243, df = 6, p = 0.3671). Surviving colonies in all groups had significantly decreased colony strength from October 2021 to March 2022 (F(1,36) = 113.247, p < 0.0001), but the colony strength did not differ significantly among the control and infected groups at each time (F(2,36) = 1.071, p = 0.353; Table 1). In March 2022, the Varroa infestation did not differ significantly among treatment groups (F(2,7) = 1.938, p = 0.214; Table 1).
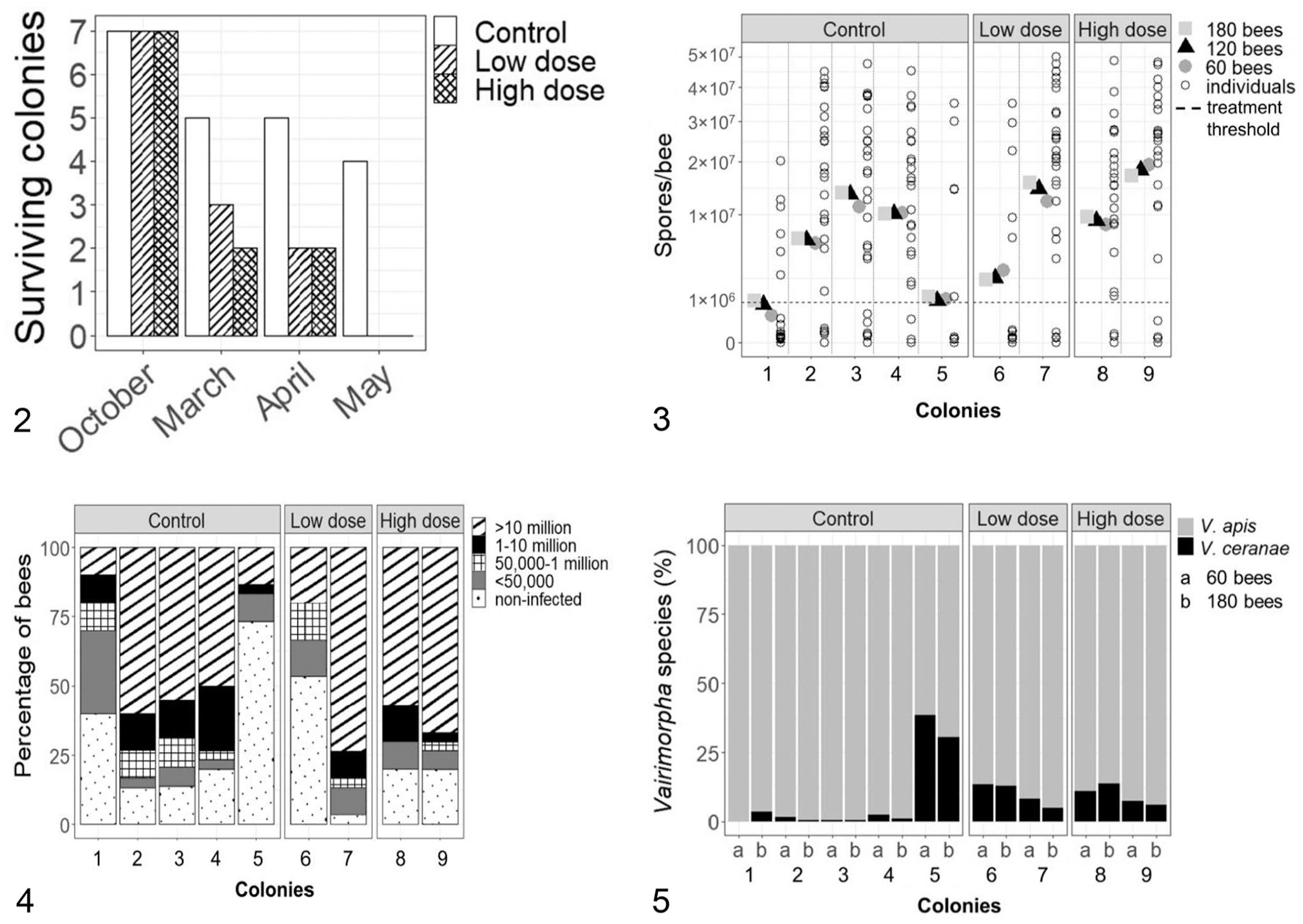
Colony overwinter survival of control and Vairimorpha spp.–infected colonies over time.
Cluster size and percent Varroa infestation in surviving control and Vairimorpha spp.–infected colonies.
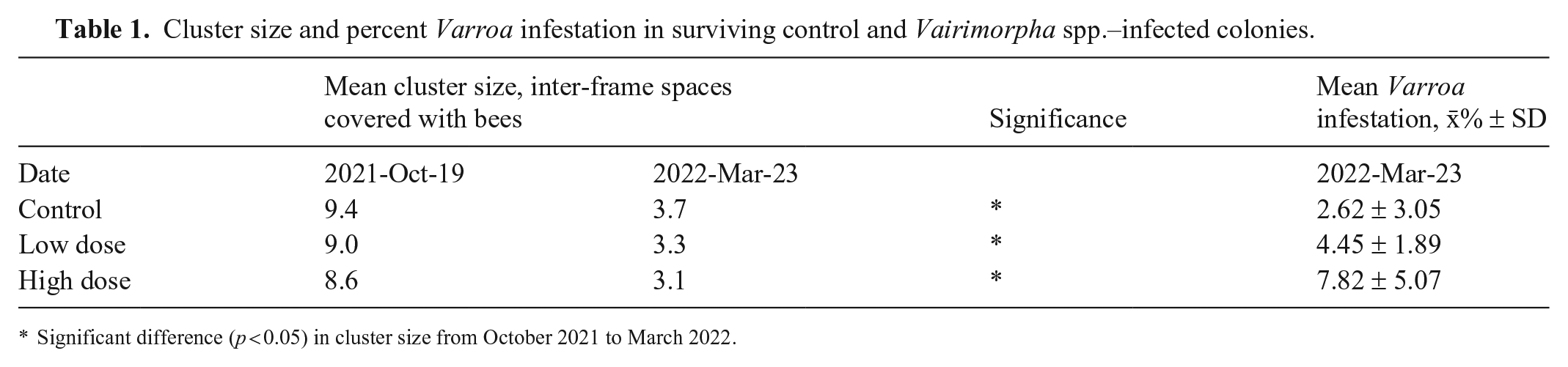
Significant difference (p < 0.05) in cluster size from October 2021 to March 2022.
Comparison of Vairimorpha spore counts in pooled vs. individual samples
There was a significant (F(2,33) = 3.754, p = 0.034) dose-response in Vairimorpha infection level in April 2022, with mean spore counts in control, low-dose, and high-dose groups of 7.3 ± 5.7 × 106, 9.7 ± 9.2 × 106, and 14.2 ± 5.9 × 106 spores/bee, respectively, based on the mean spore counts in 60, 120, and 180 bees. The mean spore count per bee in 30 individually analyzed bees, and 60, 120, and 180 pooled bees was not statistically different within each treatment group (control F(1.1,4.4) = 5.505, p = 0.0717; low-dose F(1,1) = 2.019, p = 0.3904; high-dose F(1,1) = 1.324, p = 0.4555), nor among the treatment groups (F(3,24) = 0.846, p = 0.4823), and the interaction of treatment and sampling method (pooled sampling) was nonsignificant (F(6,24) = 0.033, p = 0.9998; Fig. 3).
Comparison of Vairimorpha spore counts from pooled bee samples with the frequency of Vairimorpha infection intensity in individual bees
An increase in mean spores per bee based on pooled sampling (Fig. 3) was correlated significantly with a decrease in the percentage of non-infected individual bees (r = –0.7975, p = 0.01; Fig. 4), and also with an increase in the percentage of highly infected individual bees (r = 0.904, p = 0.0008). Colonies with similar mean Vairimorpha spore loads per bee based on a pooled sample also had a comparable distribution of individual bees with different infection intensities (Figs. 3, 4). For example, colonies 1, 5, and 6 had the lowest mean spores per bee based on pooled samples (Fig. 3) and, similarly, showed the lowest frequency (< 20%) of highly infected individuals (Fig. 4). However, in colony 5 and 6, most individual bees were non-infected (53–75%; Fig. 4) and yet the mean spores per bee based on pooled sampling (Fig. 3) was above the treatment threshold of 1 × 106 spores/bee.
Comparison of Vairimorpha species prevalence in pooled 60- and 180-bee samples
The species prevalence did not differ significantly between samples of 60 or 180 bees from each colony (W = 0.8798, p = 0.426), nor among the treatment groups (F(2,12) = 0.046, p = 0.955) or the combination of treatment and number of bees per pooled sample (F(2,12) = 0.014, p = 0.986; Fig. 5). The Vairimorpha spp. ratio was 61.9–99.9% V. apis to 0.01–38.1% V. ceranae; the trend toward increased V. apis prevalence with increased mean spores per bee based on pooled samples was nonsignificant (r = 0.3835, p = 0.0781). Moreover, an increasing percentage of non-infected bees per colony (Fig. 4) was correlated significantly with decreasing V. apis prevalence (r = –0.8044, p = 0.0089).
Discussion
We aimed to determine the effect of different sampling methods on the estimation of Vairimorpha spp. spore loads and species ratio at the colony level. The mean Vairimorpha spp. spore count in a sample of 60 pooled bees did not differ significantly compared to samples containing 120 and 180 pooled bees, indicating that increasing the number of pooled bees by 2–3-fold will not yield a more accurate estimate of colony infection. Interestingly, 2 colonies with mean spore counts per bee in excess of the treatment threshold of 1 × 106 spores/bee consisted of a majority of non-infected or low-infected bees, which confirms our hypothesis that a low number of high-infected bees in a pool of healthy bees can lead to misinterpretation of the colony infection level and hence to antibiotic treatment of mainly uninfected bees. It is unclear if these low numbers of heavily infected nestmates would altruistically leave or would be forced out of the colony by healthy nestmates to maintain the colony health, as shown in previous studies.1,5,24
The number of pooled bees does not appear to influence the Vairimorpha spp. composition, as evidenced by the similar species prevalence in sample sizes of 60 or 180 pooled bees. Although we inoculated the colonies with a 50% mix of each Vairimorpha spp., surprisingly all colonies were predominantly V. apis–infected in spring (90% V. apis in 7 of 9 colonies). Recent literature suggested a replacement of V. apis by V. ceranae, which is contradictory to our findings.8,15,22 One explanation for this discrepancy is that V. ceranae spores in our experiment may have lost viability during the inoculum preparation, resulting in a higher V. apis prevalence in spring. Hence, our results are not in concordance with the occurrence of V. ceranae spring peaks shown for overwintering colonies in Ontario, Canada. 9 Interestingly, colonies 5 and 6, which had mainly uninfected bees, revealed a higher V. ceranae proportion (~32%) than the other colonies (< 10%).
Our study highlights the difficulties of conducting experimental, colony-level infection with Vairimorpha spp., including high colony mortality and contamination of control colonies. Given the high overwinter mortality of our experimental colonies, we could sample only 10 of 21 colonies in spring 2022. Furthermore, spring Vairimorpha spp. infection levels in the control colonies were similar to the low- and high-dose colonies. Given the lack of protocols and studies using experimental Vairimorpha spp. infection at the colony level, we may have used exceedingly high spore doses for inoculation, ultimately resulting in high colony mortality. Also, the proximity of control colonies to Vairimorpha spp.–inoculated colonies in the same apiary may have resulted in Vairimorpha spp. transmission to control colonies through robbing of contaminated honey stores in dead or weak low- and high-dose colonies. 19
Hence, we need further colony-level studies with higher sample sizes and non-contaminated controls to investigate the clinical importance of the frequency of Vairimorpha spp. infection intensity in individual bees. Our data will subsequently contribute to evaluation of antibiotic treatment strategies and the effect of treatment in colonies with a low frequency of highly infected individuals.
Supplemental Material
sj-xlsx-1-jvd-10.1177_10406387231194620 – Supplemental material for Comparison of individual and pooled sampling methods for estimation of Vairimorpha (Nosema) spp. levels in experimentally infected honey bee colonies
Supplemental material, sj-xlsx-1-jvd-10.1177_10406387231194620 for Comparison of individual and pooled sampling methods for estimation of Vairimorpha (Nosema) spp. levels in experimentally infected honey bee colonies by Sarah Biganski, Tessa Lester, Oleksii Obshta, Midhun S. Jose, Jenna M. Thebeau, Fatima Masood, Marina C. B. Silva, Marcelo P. Camilli, Muhammad F. Raza, Michael W. Zabrodski, Ivanna Kozii, Roman Koziy, Igor Moshynskyy, Elemir Simko and Sarah C. Wood in Journal of Veterinary Diagnostic Investigation
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We received financial support from the Canadian Honey Council’s Canadian Bee Research Fund, the Saskatchewan Beekeepers Development Commission (SBDC), MITACS Accelerate, and the University of Saskatchewan.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
