Abstract
Encephalitis in hamsters, which was induced by equine herpesvirus (EHV)–9, EHV-1 strain Ab4p, and zebra-borne EHV-1, was investigated and compared to assess viral kinetics and identify the progression and severity of neuropathological findings. Hamsters were inoculated with EHV-9, EHV-1 strain Ab4p, and zebra-borne EHV-1 via the nasal route and euthanized at 24, 48, 72, 96, 120, 144, and 168 hours postinoculation (HPI). The inoculated hamsters had mild to severe neurological signs at 60 to 72, 96, and 120 HPI, and the mortality rate was 75%, 0%, and 0% for animals inoculated with EHV-9, EHV-1 strain Ab4p, and zebra-borne EHV-1 viruses, respectively. Inoculated hamsters had varying degrees of rhinitis and lymphoplasmacytic meningoencephalitis, as well as differences in the severity and distribution of cerebral lesions. Furthermore, the cellular distribution of viral antigen depended on the inoculated virus. Neuronal necrosis was widely detected in animals inoculated with EHV-9, while marked perivascular cuffs of infiltrating inflammatory cells and gliosis were detected in animals inoculated with EHV-1 strain Ab4p and zebra-borne EHV-1. In the present study, 3 viruses belonging to the herpesvirus family induced encephalitis after initial propagation in the nasal cavity. These viruses might travel to the brain via the olfactory pathway and/or trigeminal nerve, showing different distributions and severities of neuropathological changes.
Equine herpesvirus (EHV)–9 was first identified in an outbreak, in 1993, in Japan, where it caused fatal encephalitis in Thomson’s gazelles 8,42 (Gazella thomsoni). EHV-9 has been isolated from a number of species, including giraffe 19 (Giraffa camelopardalis reticulata), Grevy’s zebra (Equus grevyi), and polar bear 5,34 (Ursus maritimus). In experimental studies, EHV-9 infection results in fulminant encephalitis in a variety of species, including mice and rats, 8 hamsters, 7 goats, 38 pigs, 28 dogs and cats, 41,43 and common marmosets. 21 The reservoir host for EHV-9 remains undetermined. Active infection of Equidae with EHV-9 has never been conclusively recorded. Infected zebras seroconvert but do not develop clinical disease. Thus, it is hypothesized that equids are the natural and definitive host of EHV-9. 3,34 EHV-9 is genetically similar to EHV-1. However, the restriction enzyme cleavage profiles of EHV-9 and EHV-1 were significantly different. A major pathogen of domestic horses worldwide, EHV-1 causes significant economic losses for horse breeders and is associated with abortion, respiratory tract disease, and neurological disorders. 2,22,23,36 Equine herpesvirus myeloencephalitis (EHM) remains one of the most devastating manifestations of EHV-1 infection. Reports on EHV-1–induced encephalitis have been increasing over the past decade, particularly in Europe and the United States. 31 Similar to EHV-9, EHV-1 has been isolated from a number of nonequid species, 17,20,32 including camels, antelopes, giraffes, cattle, fallow deer, alpacas, llamas, and Thomson’s gazelle (Eudorcas thomsoni). 20
Herpesviruses with a close genetic similarity to EHV-1 have been isolated from numerous equid species, including Persian onagers (Equus hemionus onager), 10 Damara zebra (Equus quagga), Grant’s zebra (Equus quagga boehmi), Burchell’s zebra (Equus quagga burchelii), and Grevy’s zebras (E. grevyi). 10,18 A recombinant EHV-1 containing EHV-9 sequences in the Pol gene was isolated from a polar bear (U. maritimus) and Indian rhinoceros (Rhinoceros unicornis) in Germany, both of which had died of acute encephalitis. 1,13 The full genome sequences between the 3 strains of EHV-1 virus isolated from a zebra (E. grevyi), onager (E. hemionus onager), and gazelle (Eudorcas thomsonii) revealed 98% or 95% genetic identity with EHV-1 and EHV-9, respectively. 14 Based on these findings, the virus was initially identified as recombinant between EHV-1 and EHV-9 but is now classified as zebra-borne EHV-1. 1 The exact pathway through which the newly emerging zebra-borne EHV-1 virus travels from the nose to the brain is still unclear.
A difference in the virulence of EHV-9 and EHV-1 in the horse has been reported in a previous study. Intranasal inoculation with EHV-9 caused fever, interstitial pneumonia, and viremia with encephalitis. 37 Histological lesions induced by EHV-9 infection in the horse, including perivascular mononuclear cell infiltrations and glial reaction in the olfactory and limbic systems without vascular lesions, differed from EHV-1 myeloencephalitis, which is characterized by endothelial necrosis, thrombosis, perivascular lymphocytic cuffing, and hemorrhage, which results in secondary hypoxic degeneration in adjacent neural tissue. 2
The present study compares the distribution and severity of the pathological changes induced in the brain of animals inoculated with EHV-9, EHV-1 strain Ab4p, and zebra-borne EHV-1.
Materials and Methods
Virus Culture Preparation
EHV-9, EHV-1 strain Ab4p, and zebra-borne EHV-1 were propagated on Madin-Derby bovine kidney (MDBK) cells. The inocula were prepared by culturing the virus from original seed stocks of EHV-9, 9 EHV-1 strain Ab4p, and zebra-borne EHV-1 (P20, fifth passage in MDBK cells) in MDBK cells. A plaque-forming assay was performed on MDBK cells for viral titration (Laboratory of Veterinary Microbiology, Gifu University, Japan).
Animals and Inoculation of Virus
Three-week-old male Syrian hamsters (Mesocricetus auratus) were purchased from a breeder (SLC, Hamamatsu, Japan), housed in an isolated biohazard cabinet and fed basal pellets (Oriental MF, Oriental Yeast Co., Tokyo, Japan). Food and water were freely available to the hamsters during the course of the experiment. The experiment was performed according to the guidelines of the pertinent laws and related standard operating procedures on the treatment and use of laboratory animals. The experimental protocol was approved by the Animal Experiment Committee of the Faculty of Applied Biological Science at Gifu University, Japan. The animals were divided into 3 groups (16 animals each) and observed for 3 days before virus inoculation. All 3 viruses were inoculated intranasally into both nostrils (bilateral) with 25 μL (104 plaque-forming units [PFU/mL]). Three inoculated animals in each group were euthanized at 24, 48, 72, 96, and 120 hours postinoculation (PI), respectively, or when they were moribund (1 animal at 120 HPI in the EHV-9–inoculated group). Four hamsters that served as uninfected controls were inoculated, via the nasal cavity, with a sham inoculum that consisted of sterile cell culture medium and were euthanized at the end of the experiment (168 HPI). The animals were checked for clinical signs at least twice daily—namely, during the first and last 12 hours.
To follow the route of encephalitis in EHV-1 strain Ab4p and zebra-borne EHV-1, 2 additional groups, each with 6 animals, were inoculated with EHV-1 strain Ab4p and zebra-borne EHV-1, respectively. Three animals from each group were sacrificed at 144 and 168 HPI. The experimental period of the EHV-9–inoculated animals was not extended because by 120 HPI, 75% of the inoculated hamsters were dead, with the remaining animal euthanized because of the presence of exaggerated nervous signs.
Necropsy Examination, Histopathology, and Immunohistochemistry
Complete necropsy was conducted immediately after euthanasia by intraperitoneal administration of pentobarbital sodium (100 mg/kg), and the brains were bisected on the sagittal plane. Organs, including the brain and nasal cavity, were sampled for histopathological examination. Following fixation in 10% neutralized buffered formalin, the organs were dehydrated and embedded in paraffin wax by routine methods, sectioned at a thickness of 5 μm, stained with hematoxylin and eosin, and examined using light microscopy.
Immunohistochemistry was performed as previously described 42 on all tissues examined histologically, including sections of the olfactory epithelium and brain. Rabbit polyclonal antiserum against the EHV-9 (1:800; kindly provided by Dr. H. Fukushi, made in the Veterinary Microbiology Laboratory) was used as the primary antibody for the detection of EHV-9 antigen, with the avidin-biotin-complex (ABC) method using ABC kits (Vector Laboratories, Burlingame, CA). Following the application of the secondary antibody (biotinylated anti-rabbit IgG; DAKO Cytomation, Carpinteria, California), the bound peroxidase was detected with the liquid DAB substrate chromogen system (DAKO Cytomation). Sections were then counterstained with hematoxylin. Tissue sections from hamsters with confirmed EHV-9 infection and sera from a nonimmunized rabbit and goat were used as controls. Since there is a serological cross-reaction between EHV-1 and EHV-9, 8,33 rabbit polyclonal antiserum against EHV-9 was used as a primary antibody for the detection of EHV-1 antigen on the sections obtained from animals inoculated with EHV-1 strain Ab4p and zebra-borne EHV-1.
For further confirmation, rabbit polyclonal antiserum against the EHV-1 strain HH1 (rabbit anti–EHV-1 HH1 polyclonal antibody, provided by Dr. Kirisawa, Rakuno, Gakuen University, Japan) was used as the primary antibody, at a 1:1000 dilution, on sections obtained from animals inoculated with EHV-1 strain Ab4p and zebra-borne EHV-1.
Brain sagittal sections were examined in the rostrocaudal direction, including the olfactory bulb, olfactory tubercle, cerebral cortex, hippocampus, thalamus, hypothalamus, pons, medulla oblongata, and cerebellum. Microscopic lesions were graded as –, absence of lesions; +, lesions affecting <10% of brain tissue; ++, lesions affecting 10% to 50% of brain tissue; and +++, lesions affecting >50% of brain tissue. Presence of the viral antigen was graded as –, absent; +–, very few (<3/400× field); +, low (3–5/400× field); ++, moderate (6–30/400× field); and +++, numerous (>30/400× field).
Results
Clinical Findings
EHV-9
All animals inoculated with EHV-9 showed nasal discharge, salivation, and various degrees of nervous manifestations, including restlessness, uncoordinated movements, convulsions, tremors, and lethargy, which started at 60 HPI in 6 of 10 animals and at 72 HPI affected 9 of 10 animals. By 96 HPI, all animals (7 of 7 animals) exhibited severely uncoordinated movement, convulsions, and ruffled fur. Of the 4 remaining animals, 1 died at 109 HPI, 2 died by 120 HPI, and 1 showed severe nervous manifestations and was euthanized.
EHV-1 strain Ab4p
At 96 HPI, animals inoculated with EHV-1 strain Ab4p showed mild to moderate nervous manifestations and bloody nasal discharge in 6 of 7 animals during the first 12 hours and in 4 of 4 animals during the last 12 hours. The nervous signs decreased or were absent at 144 and 168 HPI.
Zebra-borne EHV-1
Animals inoculated with zebra-borne EHV-1 showed mild to moderate nervous manifestations and nasal discharge in 1 of 4 animals at 108 HPI and in 3 of 4 animals at 120 HPI. All animals recovered at 144 to 168 HPI.
Pathologic Findings: Brain
At necropsy, gross lesions were not observed in any of the animals except for meningeal congestion. Microscopic examination consistently identified congestion of the brain and meninges, lymphoplasmacytic meningoencephalitis, and rhinitis albeit to varying degrees. Although the severity and distribution of the lesions differed depending on the virus that the animal was inoculated with, basic features of lymphoplasmacytic meningoencephalitis (Figs. 1, 2) were common. Lesions consisted of varying degrees of neuronal degeneration and necrosis (Fig. 3), indicated by the shrinkage of the perikaryon with intense eosinophilia of the cytoplasm, central chromatolysis, condensation of the nucleus, and multifocal perivascular cuffing with plasma cells, lymphocytes, and macrophages (Fig. 4), which mainly affected the gray matter and, to a lesser degree, the white matter. In animals infected with EHV-9, degenerated neurons frequently contained intranuclear eosinophilic (Fig. 5) and amphophilic inclusion bodies that filled the neuronal nuclei and caused margination of chromatin. In contrast, inclusion bodies were rare in animals infected with zebra-borne EHV-1, and amphophilic inclusion bodies that filled the nucleus were only rarely detected in animals infected with EHV-1 strain Ab4p. Glial nodules and gliosis were also observed. Parenchymal infiltrations of neutrophils and mononuclear cells occurred around necrotic neurons. Severe neuronal degeneration and necrosis with rarefaction of adjacent brain tissue were prominent features of encephalitis found in the brains of hamsters inoculated with EHV-9. Extensive perivascular cuffing and gliosis were frequently observed in the brains of animals inoculated with EHV-1 strain Ab4p and zebra-borne EHV-1.
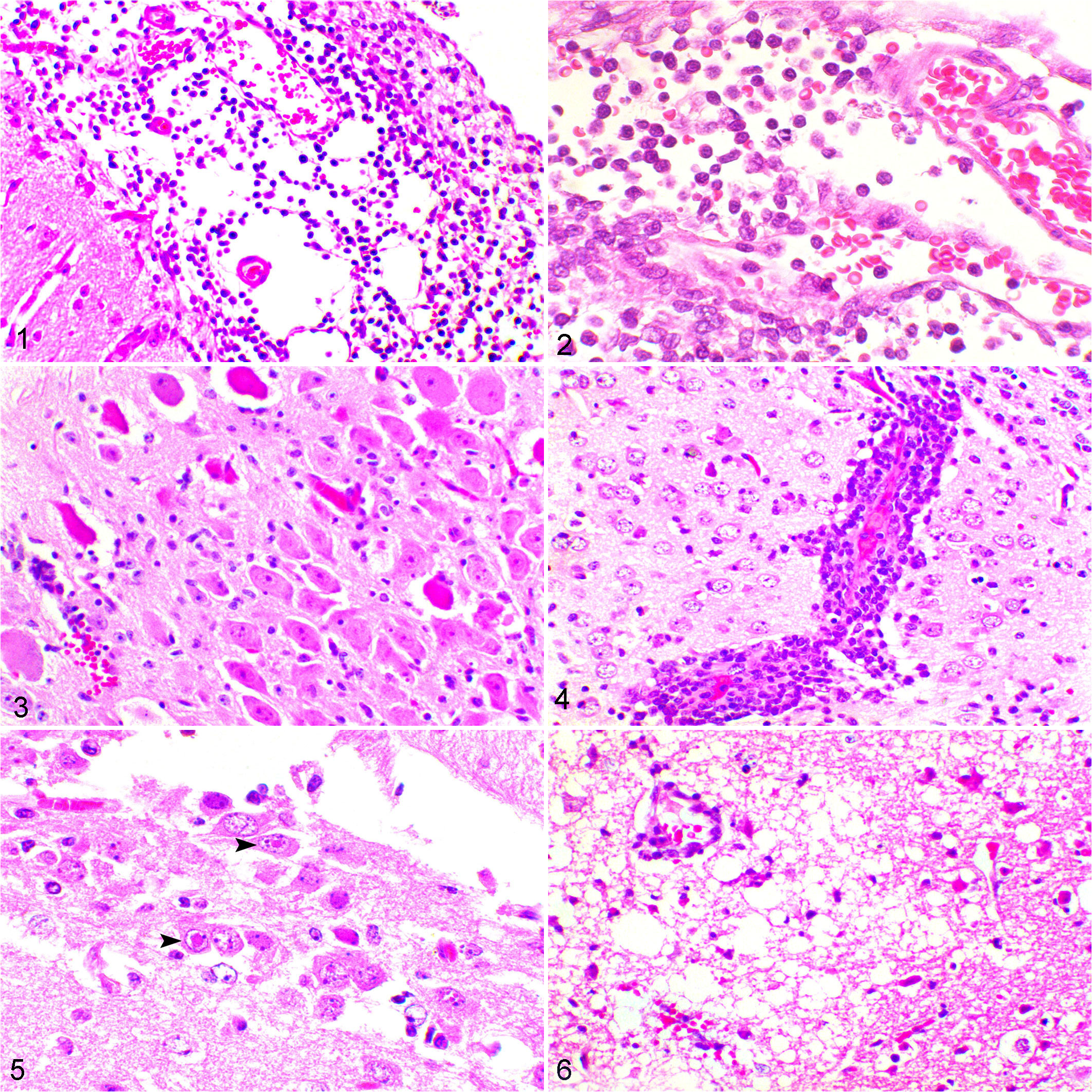
Equine herpesvirus (EHV)–9 infection, hamster, brain, 120 hours postinoculation (HPI).
Histopathological alterations in the brain in animals inoculated via the nasal route with EHV-9, EHV-1 strain Ab4p, and zebra-borne EHV-1 are summarized as follows (Tables 1 –3).
Distribution and Severity of Histologic Lesions and Viral Antigen Detection in the Brain of Hamsters Inoculated With Equine Herpesvirus–9.
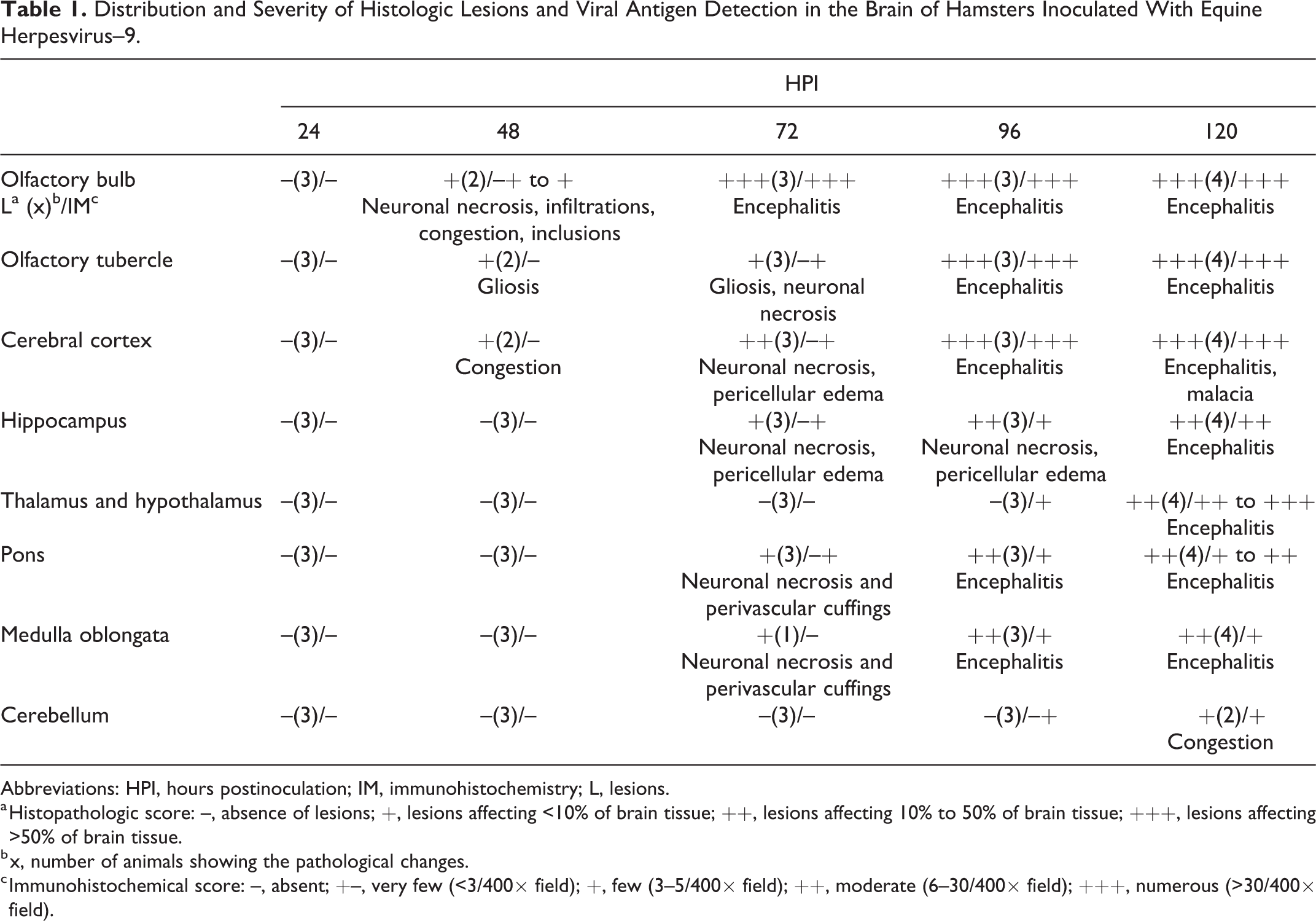
Abbreviations: HPI, hours postinoculation; IM, immunohistochemistry; L, lesions.
a Histopathologic score: –, absence of lesions; +, lesions affecting <10% of brain tissue; ++, lesions affecting 10% to 50% of brain tissue; +++, lesions affecting >50% of brain tissue.
b x, number of animals showing the pathological changes.
c Immunohistochemical score: –, absent; +–, very few (<3/400× field); +, few (3–5/400× field); ++, moderate (6–30/400× field); +++, numerous (>30/400× field).
Distribution and Severity of Histologic Lesions and Viral Antigen Detection in the Brain of Hamsters Inoculated With Equine Herpesvirus–1 Strain Ab4p.

Abbreviations: HPI, hours postinoculation; IM, immunohistochemistry; L, lesions.
a Histopathologic score: –, absence of lesions; +, lesions affecting <10% of brain tissue; ++, lesions affecting 10% to 50% of brain tissue; +++, lesions affecting >50% of brain tissue.
b x, number of animals showing the pathological changes.
c Immunohistochemical score: –, absent; +–, very few (<3/400× field); +, few (3–5/400× field); ++, moderate (6–30/400× field); +++, numerous (>30/400× field).
Distribution and Severity of Histologic Lesions and Viral Antigen Detection in the Brain of Hamsters Inoculated With Zebra-Borne Equine Herpesvirus–1.
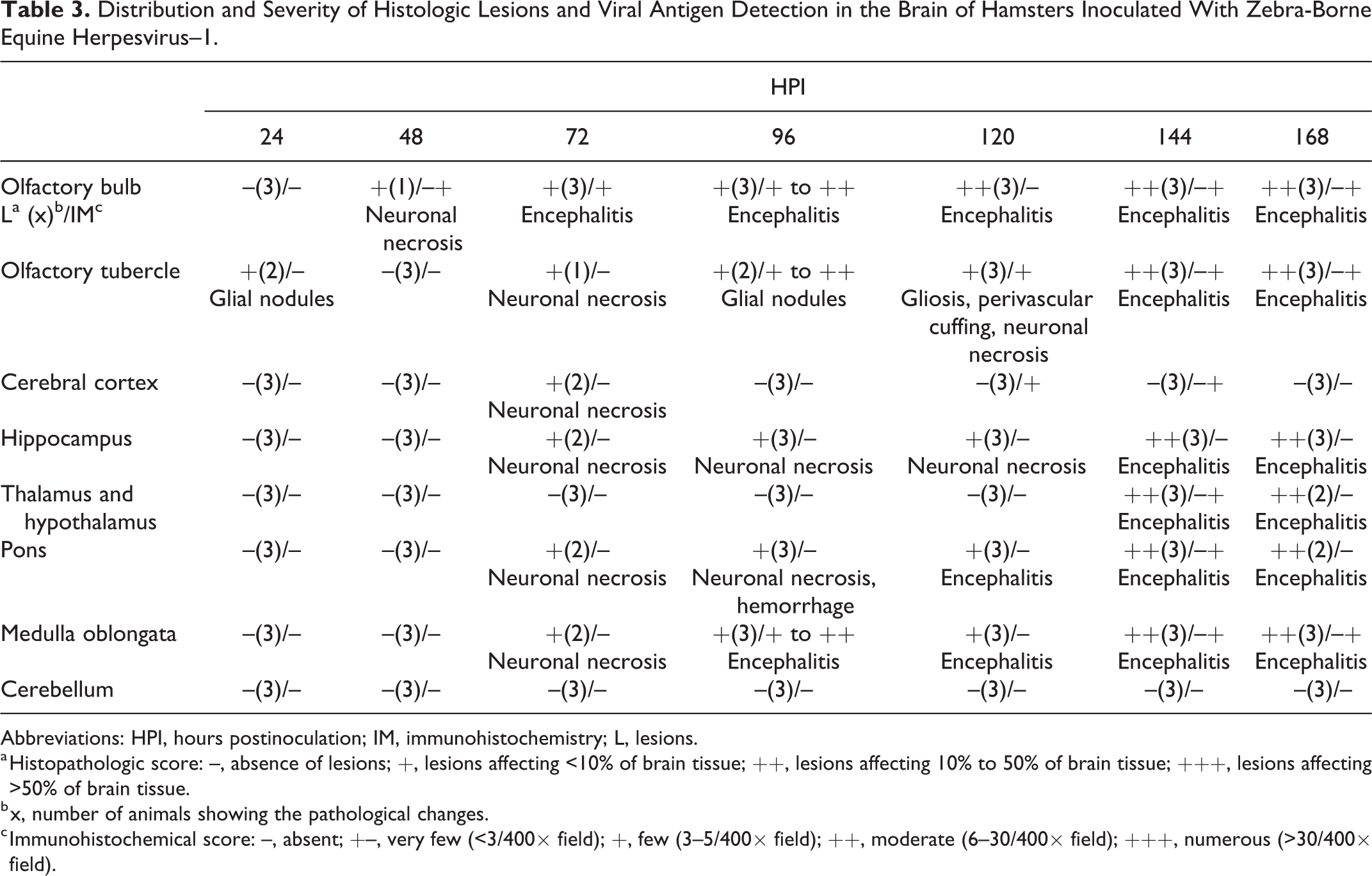
Abbreviations: HPI, hours postinoculation; IM, immunohistochemistry; L, lesions.
a Histopathologic score: –, absence of lesions; +, lesions affecting <10% of brain tissue; ++, lesions affecting 10% to 50% of brain tissue; +++, lesions affecting >50% of brain tissue.
b x, number of animals showing the pathological changes.
c Immunohistochemical score: –, absent; +–, very few (<3/400× field); +, few (3–5/400× field); ++, moderate (6–30/400× field); +++, numerous (>30/400× field).
At 48 HPI
Brain of EHV-9 inoculated animals showed mild hemorrhage, neuronal degeneration, and necrosis with a few intranuclear inclusion bodies, a few infiltrations of macrophages and lymphocytes in the glomerular and granular layers of the olfactory bulb, and gliosis of the olfactory tubercle. EHV-1 strain Ab4p-inoculated animals had glial nodules in the olfactory tubercle. Zebra-borne EHV-1–inoculated animals had mild neuronal necrosis of the granular layer of olfactory bulb.
At 72 HPI
Animals inoculated with any of the 3 viruses had lymphoplasmacytic meningoencephalitis at the level of the olfactory bulb, neuronal degeneration and necrosis, and multifocal glial nodules and/or gliosis in the olfactory tubercle, albeit to varying degrees. EHV-9–inoculated and zebra-borne EHV-1–inoculated animals had neuronal degeneration and necrosis in the cerebral cortex, hippocampus, pons, and medulla oblongata. EHV-9–inoculated animals had meningitis at the level of entrance of the sensory branch of the trigeminal nerve to the brainstem and mild perivascular cuffing in the pons and medulla oblongata.
At 96–120 HPI
In EHV-9–inoculated animals, encephalitis affecting the main olfactory bulb, accessory olfactory bulb, olfactory tubercle, cerebral cortex, hippocampus, and the brainstem at the level of the pons and medulla oblongata was observed at 96 HPI. Further extension of encephalitis to the thalamus, hypothalamus, and midbrain with appearance of areas of malacia in the cerebral cortex was observed at120 HPI. In EHV-1 strain Ab4p-inoculated animals, at 96 HPI, the meningoencephalitis was confined to the main olfactory bulb, accessory olfactory bulb, olfactory tubercle, and pons. By 120 HPI, malacia (Fig. 6) and pericellular edema appeared at the olfactory bulb and olfactory tubercle together with perivascular cuffing in the cerebral cortex, thalamus, and hypothalamus. In zebra-borne EHV-1–inoculated animals, meningoencephalitis was observed at 96 HPI in the olfactory bulb and medulla oblongata along with glial nodules in the olfactory tubercle and neuronal necrosis in the hippocampus, in the pons, and at the entrance of the trigeminal nerve to the brainstem. In addition to meningoencephalitis of the olfactory bulb, pons, and medulla oblongata, especially at the entrance of the trigeminal nerve, perivascular cuffing and gliosis were detected with mild neuronal necrosis in the olfactory tubercle at 120 HPI.
At 144–168 HPI
In animals inoculated with EHV-1 strain Ab4p, meningoencephalitis severely affected the main olfactory bulb, accessory olfactory bulb, olfactory tubercle, and the brainstem at the level of the pons and medulla oblongata. In animals inoculated with zebra-borne EHV-1, the main olfactory bulb, accessory olfactory bulb, olfactory nerve, olfactory tubercle, hippocampus, hypothalamus, ventral part of the midbrain, pons, and medulla oblongata were moderately affected.
Fig. 7 outlines the distribution of histopathological changes over time in brains of hamsters that were infected with EHV-9, EHV-1 strain Ab4p, and zebra-borne EHV-1.

Distribution of histopathological changes in sagittal sections of brains of hamsters infected by intranasal inoculation with equine herpesvirus (EHV)–9 (left column), EHV-1 strain Ab4p (middle column), and zebra-borne EHV-1 (right column). The lesions present at different times after infection are represented by the following:  , perivascular cuff;
, perivascular cuff;  , neuronal necrosis; ○, parenchymal infiltration;
, neuronal necrosis; ○, parenchymal infiltration;  , gliosis and/or glial nodules;
, gliosis and/or glial nodules;  , malacia. The lesions are severe and widely distributed. Neuronal necrosis is marked after infection with EHV-9. Perivascular cuffing and gliosis are the predominant changes after infection with EHV-1 strain Ab4p and zebra-borne EHV-1.
, malacia. The lesions are severe and widely distributed. Neuronal necrosis is marked after infection with EHV-9. Perivascular cuffing and gliosis are the predominant changes after infection with EHV-1 strain Ab4p and zebra-borne EHV-1.
Pathologic Findings: Olfactory Epithelium
The olfactory epithelium showed varying degrees of rhinitis in animals inoculated with any of the 3 viruses: severe rhinitis with EHV-1 strain Ab4p, mild to moderate with EHV-9, and very mild to mild with zebra-borne EHV-1. At 24 HPI, the surface contour of the olfactory epithelium became irregular, with frequent necrosis of the olfactory epithelial cells. Hemorrhage was observed in the lumen of the nasal cavity of animals inoculated with EHV-1 strain Ab4p (Figs. 8, 9). The olfactory mucosa of the nasal cavity showed frequent clusters of neutrophils, some of which desquamated into the lumen and infiltrated the lamina propria. Severe and widespread erosion with prominent neutrophil infiltration was also observed in some areas of the olfactory epithelium in animals inoculated with EHV-1 strain Ab4p. The degree of inflammation decreased or completely resolved by 5 days postinoculation (DPI), 5 DPI, and 6–7 DPI in animals infected with EHV-9, EHV-1 strain Ab4p, and zebra-borne EHV-1, respectively.
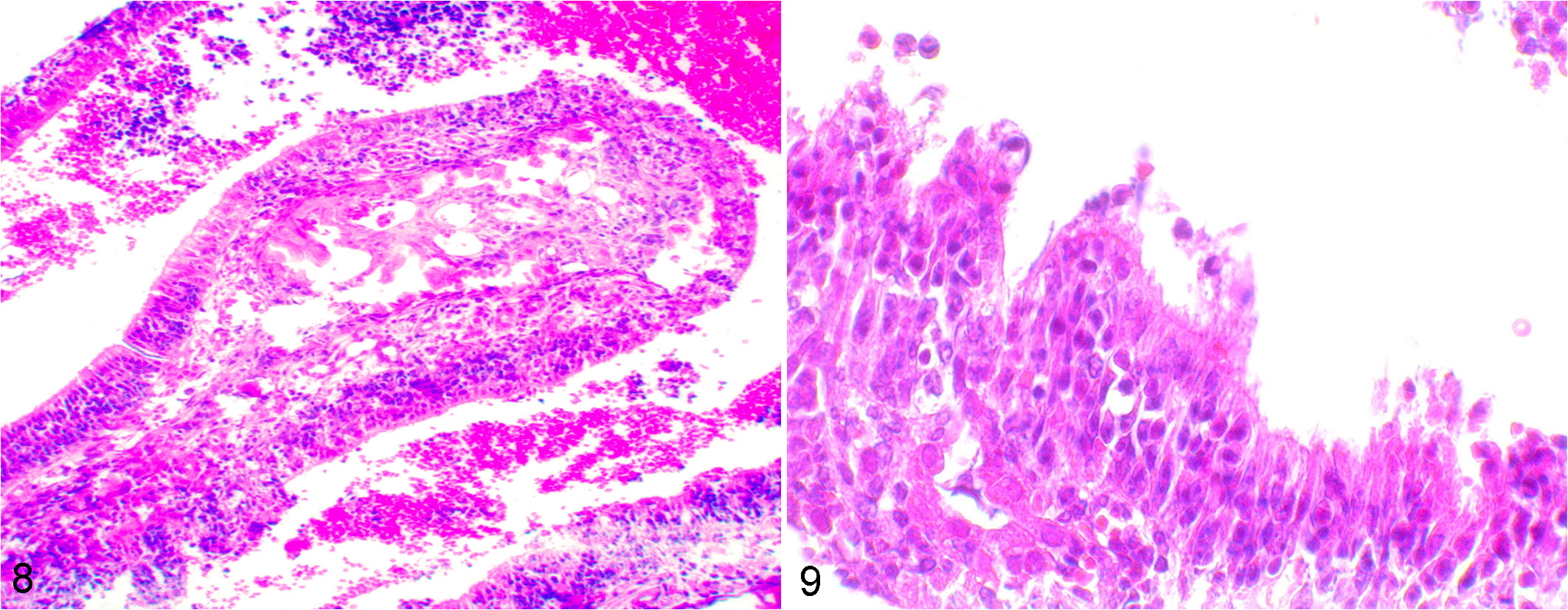
Equine herpesvirus (EHV)–1 strain Ab4p infection, hamster, olfactory epithelium, 72 hours postinoculation. Figure 8. There is severe necrosis of the nasal epithelium with erosion/ulceration and inflammation. Inflammatory exudate and hemorrhage are present in the lumen. Hematoxylin and eosin (HE). Figure 9. There are degenerative changes and necrosis. HE.
No microscopic abnormalities were observed in the 4 nontreated control animals.
Immunohistochemistry: Brain
Immunohistochemical staining with EHV-9 immunoreactivity was used to determine viral kinetics of transmission from the nose to the brain, as well as the distribution of immunopositive cells in the brain (Tables 1 –3).
At 24–48 HPI
The brain showed no immunoreactivity after inoculation with any of the 3 viruses at 24 HPI. In animals inoculated with EHV-9, immunolabeling was present at 48 HPI in a few neurons of the glomerular and granular layers of the olfactory bulb, as well as in inflammatory cells (predominantly macrophages) and very few neurons of the mitral layer. In animals inoculated with EHV-1 strain Ab4p, immunoreactivity was observed only in a few neurons in the glomerular layer and in very few neurons in the mitral and granular layers. One of the 3 animals inoculated with zebra-borne EHV-1 had immunolabeling in very few neurons of the glomerular layer.
At 72 HPI
In animals inoculated with EHV-9, there was frequent and more intense immunoreactivity in neurons of the glomerular and granular layers of the main olfactory bulb and accessory olfactory bulb, with moderate reaction in the mitral layer. In addition, very few scattered neurons in the olfactory tubercle, frontal and parietal lobes of the cerebral cortex, hippocampus, and pons showed immunoreactivity. In animals inoculated with EHV-1 strain Ab4p, neuronal immunoreactivity was limited to a moderate reaction in the glomerular and granular layers, as well as a mild reaction in the mitral layer. In animals inoculated with zebra-borne EHV-1, immunolabeling was confined to a mild reaction in the glomerular layer.
At 96–120 HPI
In animals inoculated with EHV-9 at 96 HPI, neurons of the main olfactory bulb, accessory olfactory bulb, olfactory tubercle (Fig. 10), and cerebral cortex of the frontal and parietal lobes showed strong immunoreactivity. Macrophages and neutrophils were particularly found around blood vessels, and degenerated neurons infiltrated the olfactory bulb. In addition, at 120 HPI, few to moderate numbers of neurons in the hippocampus, thalamus, hypothalamus, midbrain, pons, medulla oblongata, and fibrous and granular layers of the cerebellum (Fig. 11) were immunopositive. In animals inoculated with EHV-1 strain Ab4p, at 96 HPI, neurons in the olfactory bulb, accessory olfactory bulb, and olfactory tubercle showed mild to moderate immunoreactivity. By 120 HPI, a decrease in immunoreactivity in the olfactory bulb, accessory olfactory bulb, and olfactory tubercle was observed. A few scattered neurons were detected in the midbrain and pons, particularly at the entrance of the trigeminal nerve into the brainstem. In zebra-borne EHV-1–inoculated animals at 96 HPI, few to moderate immunoreactive neurons and macrophages were observed in the glomerular and granular layers of the olfactory bulb, olfactory nerve, olfactory tubercle, and medulla oblongata. In 1 of the 3 animals inoculated with zebra-borne EHV-1, just 3 immunoreactive neurons were observed in the frontal cortex and olfactory tubercle at 120 HPI.
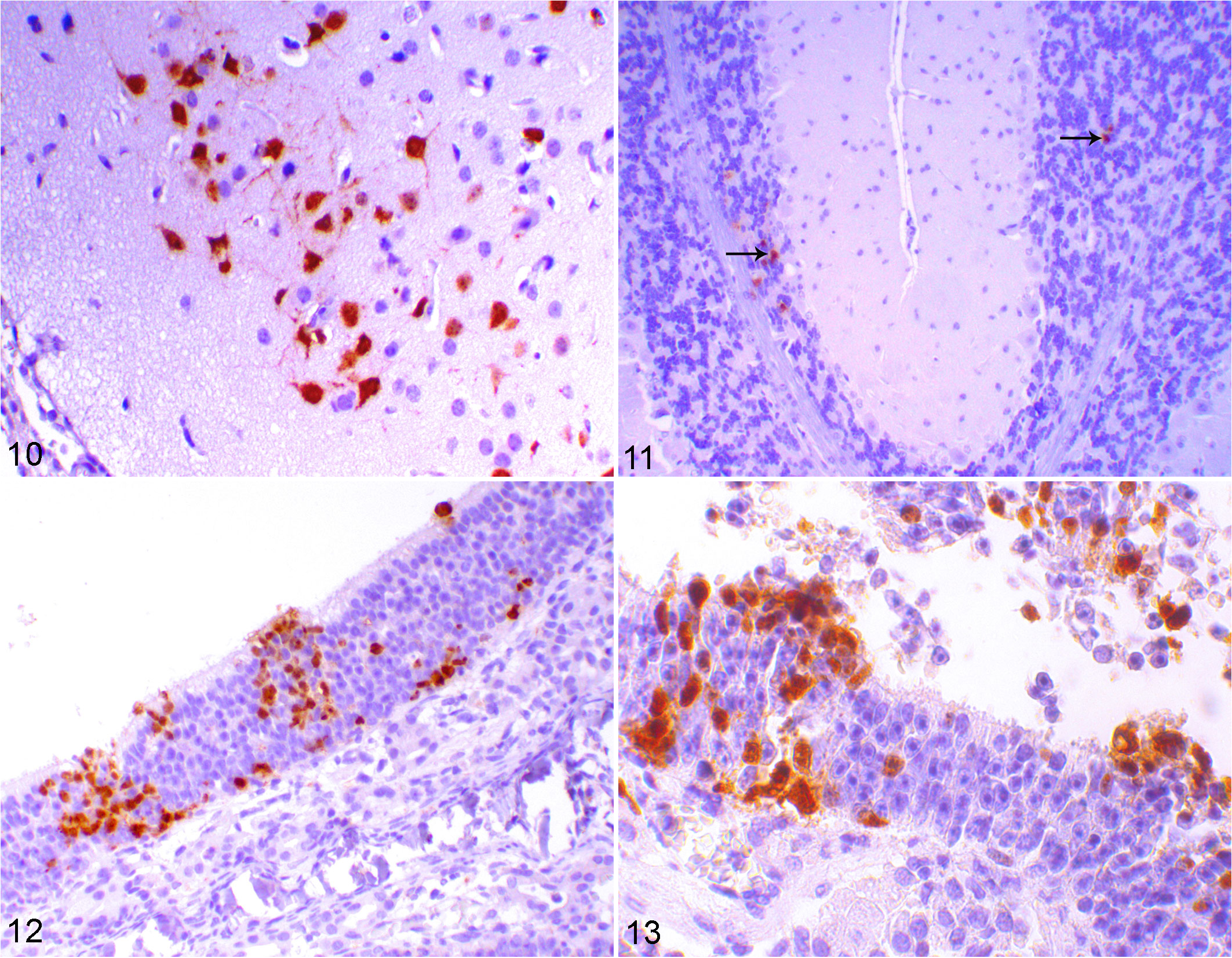
Equine herpesvirus (EHV)–9 infection, hamster, brain. Immunohistochemistry (IHC) for EHV-9 antibody. Figure 10. Immunolabeling for EHV-9 in the cytoplasm, dendrites, and nuclei of neurons of the olfactory tubercle, at 96 hours postinoculation (HPI). Figure 11. Immunolabeling for EHV-9 antibody in the granular layer of the cerebellum (arrows) at 120 HPI. Figures 12–13. EHV-1 strain Ab4p infection, hamster, olfactory epithelium, 48 HPI. Immunolabeling for EHV-9 in the olfactory neurons as well as desquamated epithelial cells and inflammatory cells in the lumen of the nasal cavity. IHC for EHV-9 antibody.
At 144–168 HPI
In animals inoculated with EHV-1 strain Ab4p, neurons and macrophages in the olfactory bulb, accessory olfactory bulb, olfactory tubercle, midbrain, and pons showed mild to very mild positive reactivity at 144 HPI. Only very few neurons in the medulla oblongata showed immunoreactivity at 168 HPI, with other brain regions showing no immunoreactivity even though encephalitis was present. In animals inoculated with zebra-borne EHV-1, few scattered neurons and inflammatory cells (predominantly macrophages) showed immunoreactivity in the olfactory bulb, olfactory tubercle, frontal cortex, thalamus, hypothalamus, pons, and medulla oblongata. Furthermore, only very few neurons in the olfactory bulb, accessory olfactory bulb, olfactory tubercle, and medulla oblongata showed immunoreactivity at 168 HPI.
Immunohistochemistry: Olfactory Epithelium
Immunohistochemistry revealed viral antigen in olfactory receptor neurons (Figs. 12, 13), as well as desquamated epithelial cells and inflammatory cells in the lumen of the nasal cavity at 24 to 48 HPI. Viral antigen immunoreactivity had disappeared in 50% of the animals inoculated with EHV-9 by 5 DPI. In contrast, it was not detected in any animals inoculated with EHV-1 strain Ab4p or zebra-borne EHV-1 from 5 DPI until the end of the experiment, indicating the possibility of viral clearance in these animals.
Fig. 14 shows the distribution of EHV-9 antigen immunoreactivity in the sagittal sections of the brains of hamsters infected with EHV-9, EHV-1 strain Ab4p, and zebra-borne EHV-1, via intranasal inoculation, at 48, 72, 96, 120, 144, and 168 HPI.
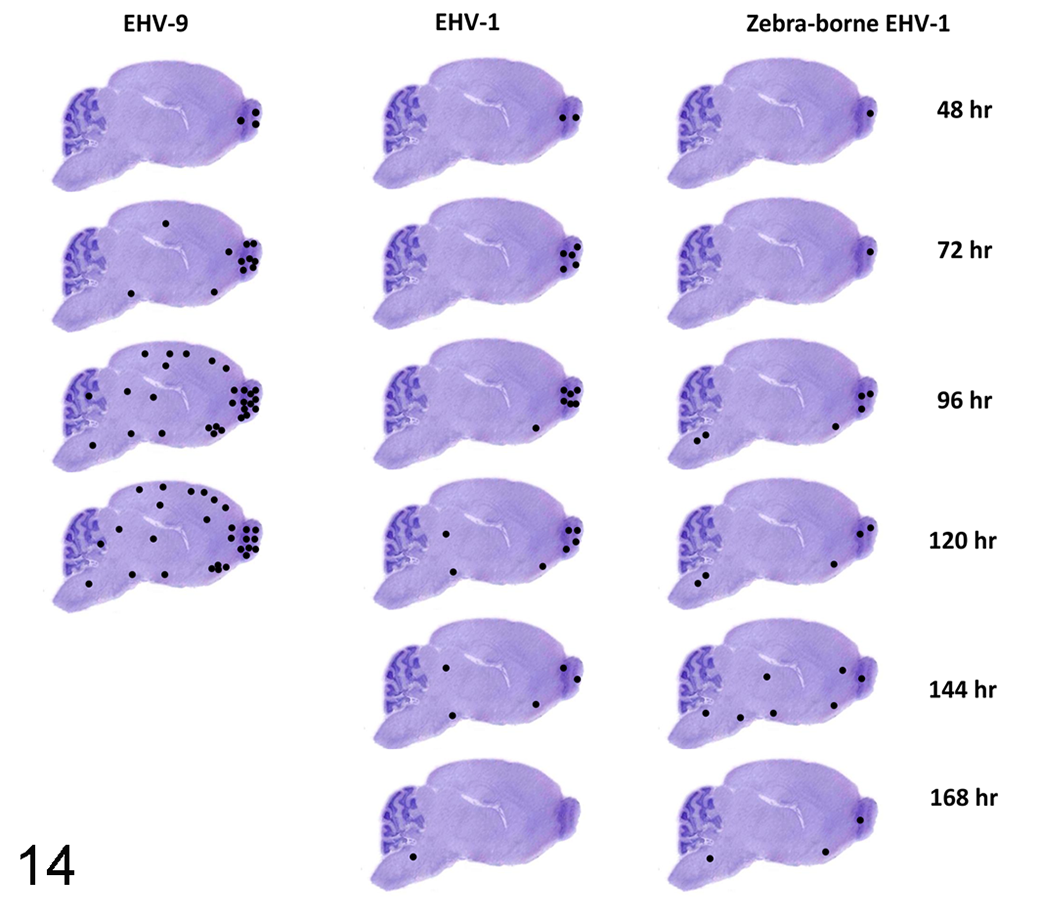
Distribution of equine herpesvirus (EHV)–9 antigen (•) in sagittal sections of the brains of hamsters infected with EHV-9 (left column), EHV-1 strain Ab4p (middle column), and zebra-borne EHV-1 (right column). Hamsters infected with EHV-9 (left column) did not survive beyond 120 hours. The brains infected with EHV-9 showed numerous widely distributed immunoreactive neurons in contrast to EHV-1 strain Ab4p and zebra-borne EHV-1.
There was no difference in the distribution of immunoreactive neurons with the 2 antibodies that were used.
Comparison of the 3 Viral Infections
Although the basic features of rhinitis and lymphoplasmacytic meningoencephalitis were common among hamsters infected with EHV-9, EHV-1 strain Ab4p, and zebra-borne EHV-1, findings that were specific for each inoculated virus included the following. First, the onset of neurological signs following infection with EHV-9, EHV-1 strain Ab4p, and zebra-borne EHV-1 occurred at 2 to 3, 4, and 5 DPI, respectively. Second, the mortality rates of animals inoculated with EHV-9, EHV-1 strain Ab4p, and zebra-borne EHV-1 viruses were 75%, 0%, and 0%, respectively. Third, although the progression of encephalitis was rapid and widespread after infection with EHV-9, it was slow and restricted to particular areas in the brain infected with EHV-1 strain Ab4p and zebra-borne EHV-1. Fourth, although EHV-9–induced encephalitis rapidly progressed and continued until the animal died, that caused by EHV-1 strain Ab4p and zebra-borne EHV-1 progressed slowly until 5 DPI, before viral antigen detection and neurological signs decreased or completely resolved at 6 to 7 DPI, which could be explained by viral clearance. Fifth, the most prominent feature of the induced encephalitis in EHV-9 was neuronal necrosis, rather than gliosis and perivascular cuffs observed in the EHV-1 strain Ab4p and zebra-borne EHV-1 infections. Sixth, encephalitis affected the olfactory bulb, olfactory tubercle, cerebral cortex, hippocampus, thalamus, hypothalamus, pons, and medulla oblongata in animals inoculated with EHV-9. However, the olfactory bulb, olfactory tubercle, hippocampal neurons, cerebral cortex, and scattered neurons in the medulla oblongata were infected in zebra-borne EHV-1, indicating a difference in susceptibility of neurons to these viruses. EHV-1 only affected the olfactory bulb, olfactory tubercle, pons, and medulla oblongata and did not induce any pathological changes in the cerebral cortex and hippocampal neurons.
There were also some similarities among 3 investigated viruses. The entry and propagation of all 3 viruses occurred in the olfactory epithelium. Transmission of the 3 viruses to the brain occurred via the olfactory nerve.
Discussion
Encephalitis is a hallmark of EHV-9, EHV-1, and zebra-borne EHV-1 infection, as well as the main cause of death or euthanasia of infected animals. The present study was conducted to compare the pathology of EHV-9, EHV-1 strain Ab4p, and zebra-borne EHV-1 in adult hamsters, with respect to the viral entry, spread, distribution, and severity of brain lesions following intranasal inoculation. To assess the viral kinetics, time course, and severity of neuropathological alterations, sagittal brain sections of all inoculated animals were examined histologically.
In the olfactory neuroepithelium, varying degrees of rhinitis were observed, as well as olfactory neurons that tested positive for the 3 investigated viruses. Immunohistochemistry demonstrated the presence of viral antigen as early as 48 HPI, particularly in neurons of the olfactory bulb. Considering the results of our previous experiment, 6 with intranasal inoculation of EHV-9 in suckling hamsters, the olfactory neuroepithelium is likely to represent the portal of virus entry to the brain. After initial propagation in the olfactory receptor neurons, the virus is transported to the olfactory bulb along the axons of the olfactory neuroepithelial cells, as demonstrated by the presence of virus antigen in the axons of the olfactory neurons as well as the olfactory nerve. A similar virus transmission route along the olfactory pathway following intranasal inoculation has also been reported with other viruses like Bornavirus, rabies virus, 12 Nipah virus, 26 and EHV-9 in pigs. 27 A previous report on a polar bear that was naturally infected with EHV-9 demonstrated that the olfactory bulb was the most severely affected area of the brain. Following infection of the olfactory bulb, the virus disseminates to the olfactory tubercle, from where it spreads transsynaptically via connections to the hippocampus and cerebral cortex. 7 A recent study showed that in mice that were inoculated intranasally, EHV-1 accesses the brain through the olfactory neuroepithelium along the olfactory nerve and spreads transsynaptically in a rostrocaudal direction, using olfactory and limbic neuronal networks 11 for further spread throughout the brain. In the present study, the olfactory tubercle was the second area in the brain to be affected in all 3 experimental groups. Histologically, axons forming the lateral olfactory tract and tufted cells located at the ventral portion of the olfactory bulb project from the olfactory bulb to the olfactory tubercle. 39 Therefore, it is not surprising that the olfactory tubercle region was the second area of the central nervous system (CNS) to become infected as early as 72 to 96 HPI, following infection of the olfactory bulb at 48 HPI. Thus, these results demonstrated that there was a close similarity among the 3 investigated viruses, with respect to their transmission from the nasal cavity to the brain through the olfactory nerve.
Alternatively, it has been previously hypothesized that there is a possible route of EHV-9 infection from the nasal mucosa through the trigeminal nerve, 38 following infection of the terminal arborizations of the maxillary branch of the trigeminal nerve, which lie in submucosal connective tissue. Submucosal terminal sensory axons of the trigeminal nerve can only be infected with a virus if the epithelial surface is damaged, leaving the axons exposed to the virus. Therefore, the possible role of the trigeminal nerve in transmission of viruses to the pons and medulla oblongata should be considered.
Although variable degrees of rhinitis were observed in the nasal cavity with the 3 investigated viruses, the most severe cases were observed after EHV-1 strain Ab4p infection. By using immunohistochemistry, the antigen of the 3 viruses was detected in the olfactory bulb at 48 HPI. However, the spread of EHV-9 through the brain was faster than that of the other 2 viruses. Thus, although disease onset and lesion development were faster with EHV-9 than with the other 2 viruses, this could be attributed to the high neuronal tropism of EHV-9, which mainly affected neurons, compared to that of the other 2 viruses.
Inoculated hamsters had varying degrees of encephalitis, as well as discrepancies in the severity and distribution of the lesions induced in the brain. Furthermore, the distribution of viral antigen also differed. Encephalitis affecting widespread areas of the brain as well as more widely distributed antigen-positive neurons were observed in animals inoculated with EHV-9 in contrast to animals inoculated with zebra-borne strain and EHV-1 strain Ab4p, indicating a difference in the neurotropism of these neurons to the 3 viruses. In contrast to EHV-1 strain Ab4p and zebra- borne EHV-1, EHV-9–induced encephalitis revealed severe progression. Severe neuronal necrosis may explain the rapid onset of clinical signs and high mortality observed in the animals inoculated with this strain.
In a recent study, zebra-derived EHV-1 isolates were assigned to an intermediate group according to their neurological virulence. 18 In addition, the brains of the hamsters infected with zebra-derived isolates 18 showed a degree and distribution of pathological changes that was similar to those in the present study.
CNS lesions induced by EHV-1 strain Ab4p in the present study differed from those of EHV-1 encephalomyelitis in horses. Two alternative modes of EHV-1 dissemination in the nervous system were previously reported, including neural spread and hematogenous spread. In naturally infected horses, hematogenous spread is predominant. Vasculitis, thrombosis, and ischemic damage of the neuroparenchyma are the key features following EHV-1 infection of endothelial cells. 4 In horses, direct infection of neurons has been reported only in exceptional cases. 30,35,40 This suggests that the virus is endotheliotropic, rather than neurotropic, in its natural host. Conversely, previous reports on mice inoculated with EHV-1 by different routes showed apparent neuronal and glial infections resulting in cell death and tissue injury. Moreover, no viral antigen was detected in endothelial cells in the murine CNS. This finding corroborated that of previous studies in which suckling mice were inoculated with EHV-1 intracerebrally. 16 Vasculitis, a characteristic lesion of equine encephalomyelitis, was absent in the CNS of mice infected with either NHH1 (neuroadapted HH1) or HH1 strain of EHV-1, 15 and viral antigen was detected in neurons, glial, and ependymal cells but not in the vascular endothelium, which was similar to the present findings with EHV-1 and zebra-borne EHV-1. These findings support previous results 29 that meningoencephalitis of mice clearly differs from that of horses, due to the lack of endothelial infection, vasculitis, and thrombosis. Similar reports regarding the lack of endothelial cell positivity for EHV-1 in the brain of infected animals have also been recorded. 17,24 It is noteworthy that vascular lesions were not evident in the brain of animals naturally infected with EHV-9 5,42 and zebra-borne EHV-1. 33
Since pathogenicity studies in the horse are expensive, several animal models have been developed using mice and Syrian hamsters (M. auratus). EHV-1 caused severe neurological disease in Syrian hamsters in a recent study, thus making this species an interesting model for studying EHV-1 infection in the CNS. 25 Since there is a wide range of susceptible hosts and the actual reservoir for EHV-9 is unclear, a hamster model has been developed and proven for its validity in various studies concerning EHV-9 pathogenesis. 6,7 Moreover, emerging infection in captive animals and the increasing host range of zebra-borne EHV-1 infection requires evaluation of a hamster model to study the pathogenesis of this EHV-1 strain.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by a grant from the Ministry of Education, Culture, Sports, Science and Technology, Japan, for the Joint Research Program of the Research Center for Zoonosis Control, Hokkaido University, and a grant from Ono Pharmaceutical Co., Ltd.
