Abstract
The infectivity and pathology of equine herpesvirus 9 (EHV-9), a new neurotropic equine herpesvirus isolated from gazelles, was studied in hamsters experimentally infected via nasal, ocular, oral, intravenous (IV), or peritoneal routes. Clinically, all animals inoculated by the nasal route and ~25% inoculated by the oral and peritoneal routes showed neurological signs on days 3, 6, and 9 postinoculation (PI), respectively. Neurological signs were not observed in animals administered EHV-9 by the IV and ocular routes. With the exception of animals administered EHV-9 by the IV route, all infected animals had lymphocytic meningoencephalitis. Although there were a number of differences in the severity and distribution of the lesions depending on the route of inoculation, the basic features of lymphocytic meningoencephalitis caused by EHV-9 were common. Lesions consisted of neuronal necrosis, perivascular aggregates of lymphocytes, plasma cells, and neutrophils, gliosis, intranuclear inclusion bodies, and diffuse lymphocytic infiltrates in the meninges. Viral antigen was detected in degenerated neurons in infected animals inoculated by the nasal, ocular, oral, and peritoneal routes. The distribution of EHV-9 antigen was somewhat dependent on inoculation route. There were no microscopic abnormalities or viral antigen in animals treated by the IV route. This study provides new data about experimental EHV-9 infection in hamsters through routes other than the IV route. These results suggest that in the animals infected by the oral, ocular, and peritoneal routes, EHV-9 might travel to the brain through nerves, other than by the olfactory route, after initial propagation at the site of viral entry.
Equine herpesvirus 9 (EHV-9), the newest member of the equine herpesvirus family, is a highly neurotropic herpesvirus originally isolated from the brain of Thomson’s gazelles (Gazella thomsoni) that died of fulminant encephalitis. 6 Serologically, EHV-9 is closely related to equine herpesvirus 1 (EHV-1), but its DNA fingerprint is different from that of EHV-1 and other equine herpesviruses. 6 The predicted amino acid sequence of EHV-9 showed 93 and 92% identity to EHV-1 and EHV-8, respectively. 6
Although the natural host of EHV-9 is still uncertain, some members of the Equidae family have been suspected as being a reservoir. Burchell’s zebras have a relatively high seroprevalence (60% of 45 animals) in the Serengeti National Park in Tanzania. 1 More recently, EHV-9 was detected in a polar bear with progressive meningoencephalitis, 4 and the source was traced to members of a potential equid reservoir species, Grevy’s zebras. In addition, EHV-9 was found in an aborted Persian onager in a zoo. 12
Many experimental studies on the infectivity of EHV-9 have been conducted using rodents, domestic animals, and a new-world monkey to clarify the possible emerging aspects of the virus. 5,6,8,11,13–16 Following inoculation of EHV-9 via the nasal route, mice, rats, 6 goats, 14 , pigs, 11 hamsters, 5 cats, and dogs 15,16 developed fulminant encephalitis, while horses 13 exhibited only moderate encephalitis characterized by lymphocytic perivascular aggregates and gliosis. Most of the experimental infections were conducted by the nasal route because it is hypothesized to be the most probable route of transmission in the field. However, natural transmission of EHV-9 by other routes (eg, oral and ocular) may be possible. Although there is a report on the oral transmission of EHV-9, which induced encephalitis in pigs, 11 information on the possible transmission of EHV-9 by routes other than the nasal route has been scarce to date, as has data on route-specific pathological features.
This study aimed to clarify the neurotropism of EHV-9 via different routes of inoculation, including the nasal, ocular, oral, peritoneal, and intravenous (IV) routes, and to compare the histopathology and distribution of viral antigen in the brain based on these different routes of inoculation.
Materials and Methods
Animals
Twenty-five 5-week-old male Syrian hamsters (Mesocricetus auratus) were obtained from a commercial breeder (SLC Inc., Hamamatsu, Japan). After receipt, animals were randomly assigned to 1 of 6 groups (4 to 5 animals per group) and were group housed in stainless-steel wire-bottom cages and kept in an isolated biohazard cabinet for approximately 1 week acclimation. Animals were provided a basal pellet diet (Oriental MF; Oriental Yeast Co., Tokyo, Japan) and bottled water ad libitum. This animal experiment was conducted in accordance with pertinent laws and related standard operating procedures on the treatment and use of laboratory animals. The experimental protocol was approved by the Animal Experiment Committee of the faculty of Applied Biological Science at Gifu University.
Inoculation of Virus
Twenty of the 25 hamsters were administered a single dose of 50 μl (2 × 106 PFU) of EHV-9 virus solution via the nasal, ocular, oral, IV, or peritoneal routes (Table 1). The 5 remaining animals were used as a control group. The animals were examined for clinical signs 3 times per day.
Experimental Design

a NA, not applicable; animals were not administered dosing vehicle.
Histopathology and Immunohistochemistry
All animals found moribund, and those remaining at the termination of the experiment (day 15) were euthanatized by exsanguination under ether anesthesia and a complete necropsy was performed. The nasal cavity, brain, heart, lungs, liver, spleen, kidneys, stomach, small intestine, eyes, pancreas, stomach and intestine, adrenals, and lymph nodes were collected and fixed in 10% buffered formalin. After fixation, the formalin-fixed paraffin-embedded (FFPE) tissues were processed for routine staining by hematoxylin and eosin and examined by light microscopy.
In addition, FFPE sections of the brain, nasal mucosa, and lungs were immunolabeled with EHV-9 rabbit antiserum by the avidin–biotin–complex (ABC) immunoperoxidase method (ABC Kits; Vector Laboratories, Burlingame, California) as described previously. 16 The primary antibody was prepared by immunizing rabbits with purified EHV-9 virions, and the specificity of the antiserum was confirmed by indirect immunofluorescence assay by using EHV-9–infected cultured cells and Western blotting. The EHV-9 antiserum (1:800; Veterinary Microbiology Laboratory at Gifu University) followed by application of a secondary antibody (biotinylated anti-rabbit IgG; DAKO Cytomation, Carpinteria, California) with the Liquid DAB Substrate Chromogen System (DAKO Cytomation) used as the chromogen and hematoxylin counterstain.
Results
Clinical Findings
All 4 animals (100%) inoculated via the nasal route, 1 of the 4 (25%) inoculated via the oral route, and 1 of the 4 (25%) inoculated via the peritoneal route showed clinical signs compatible with EHV-9 infection, which consisted of body weight loss, crouching posture, nasal and lacrimal discharges, and salivation on days 3, 6, and 9 postinoculation (PI). By the second day from the onset of the clinical signs, clinically affected animals often became hyperreactive to stimuli, with some having tremors and uncoordinated movements of the limbs and some progressing to convulsions and a severe moribund state. In addition, the orally inoculated animals demonstrated rapid, progressive weight loss and neurological signs from the onset of the signs on day 6. In 3 of the 4 animals from the ocular route, body weight remained stable or decreased slightly over a period of several days, after which it returned to normal. No other obvious signs were observed in any of the animals inoculated via the ocular route, nor were there any obvious clinical signs in the animals administered EHV-9 via the IV route.
Pathologic Findings
At necropsy, no gross abnormalities were observed in any organs or tissues in any of the inoculated animals. The distribution and severity of the histopathological findings in animals administered EHV-9 via the nasal, oral, peritoneal, ocular, and IV routes are summarized in Table 2. Depending on the route of administration and subanatomical region of brain examined, there were differences in degree of inflammation observed. In the cerebrum, the histopathologic score ranged from slight to severe in animals inoculated with EHV-9 via the nasal, oral, and peritoneal routes and slight to moderate in the animals exposed via the ocular route (Table 2). Although there were a number of differences in the severity and distribution of the lesions depending on the route of inoculation, the basic features of lymphocytic meningoencephalitis caused by EHV-9 were common. The lesions consisted of neuronal degeneration and loss; varying degrees of gliosis; perivascular aggregates of lymphocytes, plasma cells, and neutrophils (Fig. 1 ); and slight focal or diffuse lymphocytic infiltrates in the meninges. These findings were similar to previously reported cases in hamsters, dogs, and cats of EHV-9 inoculated via the nasal route. 5,14–16 There were frequent shrunken acidophilic neuronal cells scattered in the affected areas. The affected neuronal cells showed severe chromatolysis or karyorrhexis. The degenerating neuronal cells frequently possessed Cowdry A-type and/or full-type intranuclear inclusion bodies (Fig. 2 ), particularly in the olfactory bulb and rhinencephalon.
Distribution and Histopathologic Severity Score of Lesions in Clinically Affected Animals Inoculated With Equine Herpesvirus 9 by Nasal, Oral, Peritoneal, Ocular, and Intravenous Routes

a Histopathologic score: –, none; +, slight; ++, moderate; +++, severe.
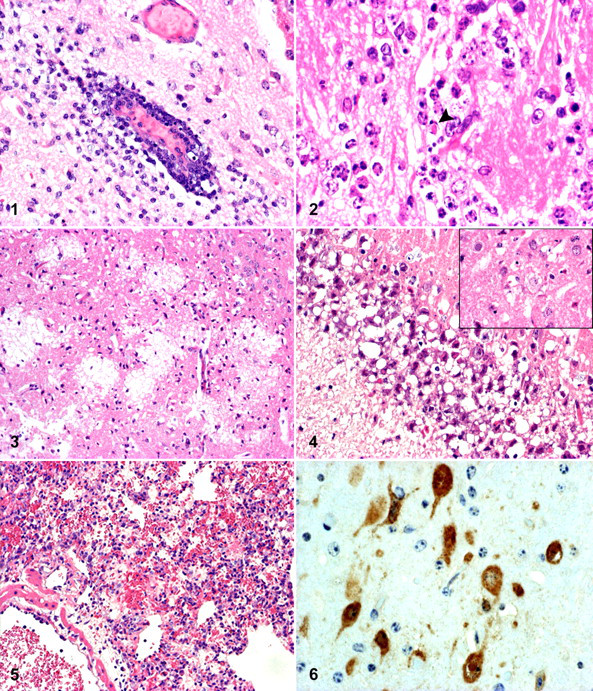
In the clinically affected animals inoculated via the nasal route, there was marked diffuse neuronal loss and intranuclear inclusion bodies in the olfactory bulb as well as severe inflammation in all layers of the olfactory bulb (eg, glomerular, mitral, and granular layers), while animals inoculated via the oral or peritoneal route had varying degrees of inflammation depending on the olfactory bulb layer (Table 2). There was slight inflammation in the glomerular layer, a moderate degree of inflammation in the mitral layer, and severe inflammation in the cells of the granular cell layer.
In addition to the above-described microscopic changes that were commonly observed in animals administered EHV-9 via the nasal, oral, peritoneal, and ocular routes, findings specific to the route of administration included the following: (1) orally inoculated animals had malacia and hemorrhage in the glomerular layer (Fig. 3 ) and marked neuronal vacuolization (Fig. 4 ); (2) animals inoculated via the ocular route (3 of 4) had varying degrees of gliosis, perivascular cellular aggregates, focal to diffuse meningitis, and mild neuronal necrosis; and (3) animals inoculated via the oral or peritoneal routes had neuronal necrosis and mild gliosis in the cerebellum. No abnormalities were observed in the cerebellum in the animals infected via the nasal and ocular routes.
In the nasal cavity of all 4 animals infected via the nasal route, rhinitis was observed. Lesions consisted of multiple foci of necrosis of the olfactory epithelial cells with intralesional intranuclear inclusion bodies and inflammatory cell infiltrates within the mucosa and admixed with the desquamated epithelial cells. In animals infected via all other routes, the olfactory epithelium was intact with no pathological features of rhinitis.
There were no microscopic abnormalities noted in the 4 animals inoculated via the IV route or in the 5 nontreated control animals.
No abnormalities were found in other organs, except for moderate interstitial pneumonia in the animals administered EHV-9 by the nasal route (Fig. 5 ). The pulmonary changes consisted of diffusely thickened alveolar septa with varying numbers of macrophages and neutrophils. Also, bronchitis/bronchiolitis was observed in 2 of these animals characterized by multifocal mucosal necrosis, degeneration, and desquamation of the epithelial cells with abundant inflammatory cells infiltrates within the mucosa and in airway lumen.
Immunohistochemistry
In all of the clinically affected animals, EHV-9 antigen was detected in the cytoplasm and intranuclear inclusions in the degenerating neuronal cells (Fig. 6 ). Distribution of EHV-9 antigen in the clinically affected animals inoculated with EHV-9 by the nasal, oral, peritoneal, ocular, and IV routes are summarized in Table 3.
Distribution and Presence of EHV-9 Antigen in the Clinically Affected Animals Inoculated With Equine Herpesvirus 9 by Nasal, Oral, Peritoneal, Ocular, and Intravenous Routes
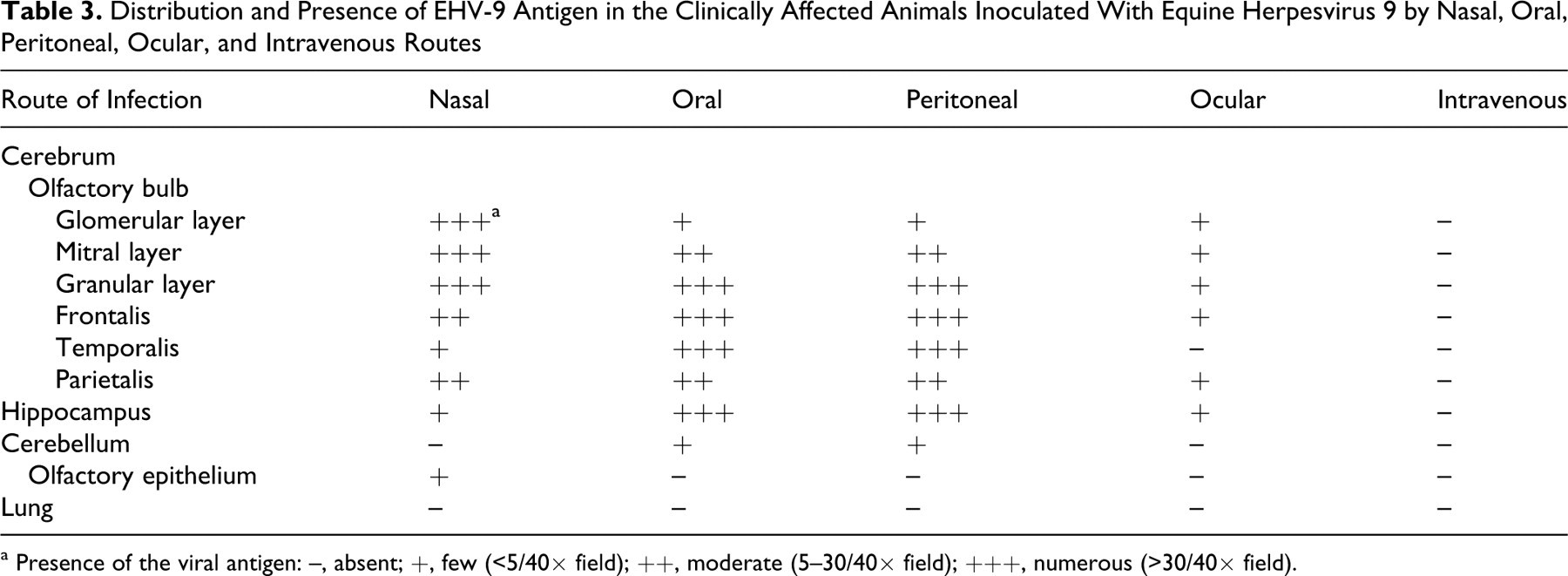
a Presence of the viral antigen: –, absent; +, few (<5/40× field); ++, moderate (5–30/40× field); +++, numerous (>30/40× field).
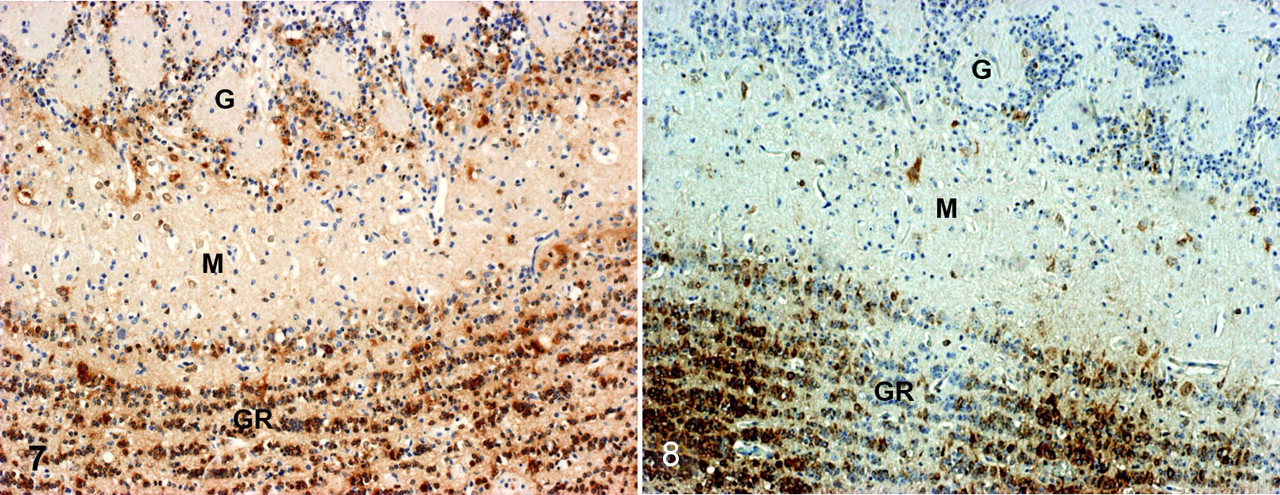
The distribution of EHV-9 antigen was somewhat dependent on route of inoculation. In the clinically affected animals that were inoculated via the nasal route, EHV-9 antigen was detected in numerous neuronal cells in all 3 cell layers of the olfactory bulb (Fig. 7 ), with few to moderate numbers of antigen-positive cells in the hippocampus and frontal, parietal, and temporal lobes. The converse pattern of antigen expression was observed in the clinically affected animals inoculated via the oral and peritoneal routes with more numerous antigen-positive cells detected in the hippocampus and frontal, parietal, and temporal lobes than in the olfactory bulb (Table 3, Fig. 8 ). In addition, EHV-9 antigen was detected in areas showing neuronal vacuolization in the clinically affected animals inoculated via the oral route. EHV-9 antigen was not detected in the cerebellum in any of the infected animals, except for few antigen-positive cells detected in animals from the oral and peritoneal inoculation groups. A weak EHV-9 antigen expression (<5 positive cells/40× field) was detected in neurons in the olfactory bulb, hippocampus, parietal, and frontal lobes in the microscopically affected animals inoculated via the ocular route (Table 3). EHV-9 antigen was also detected in the cytoplasm of degenerating epithelial cells in the nasal cavity in clinically affected animals inoculated with EHV-9 via the nasal route. No immunolabeling was detected in the lung.
Discussion
The pathogenicity of EHV-9, the newest member of the equine herpesvirus group, has been investigated extensively in various species of domestic laboratory animals and a new-world monkey. 8 EHV-9 caused encephalitis when administered via the nasal route; however, there is a paucity of information regarding infection by routes other than the nasal route. Hence, this study aimed to clarify the neurotropism of EHV-9 via different routes of inoculation, including the nasal, ocular, oral, peritoneal, and IV routes, and to compare the histopathology and distribution of viral antigen in the brain based on the different routes of inoculation.
Clinically, animals inoculated via the nasal, oral, and peritoneal routes showed neurological manifestations on days 3, 6, and 9 PI, respectively, whereas animals inoculated via the IV route failed to exhibit any infection or clinical abnormalities after prolonged incubation (15 days). There may be discrepancies in EHV-9 infection of the brain based on the route of inoculation when animals are inoculated with the same quantity of virus. For the neurotropic herpesvirus, several routes of viral entry into the central nervous system (CNS) have been considered, which include the neural, olfactory, and hematogenous routes. 7 Other neurotropic herpes viruses, including herpes simplex virus-1 and porcine herpesvirus-1, may enter the CNS via the IV, intramuscular, and intraperitoneal routes. 7
Based on previous and present experimental studies in hamsters and pigs inoculated via the nasal route, the olfactory pathway (ie, through the olfactory nerves) was thought to be the major route of transmission of EHV-9 into the CNS. 5,11 However, this study clearly indicates that virus can enter the CNS after administration of EHV-9 via the oral, peritoneal, and ocular routes and that there are differences in the distribution of antigen-positive cells and in the location and severity of the cerebral lesions. Thus, EHV-9 may gain access to the CNS through a nonolfactory route, as these animals inoculated via these nonnasal routes did not exhibit EHV-9–induced rhinitis and the olfactory bulbs showed milder lesions and fewer viral antigen-positive cells than observed in the animals infected via the nasal route. Specifically, animals infected via the oral and peritoneal routes had milder lesions and fewer viral antigen-positive cells in the glomerular and mitral cell layers of the olfactory bulb than animals infected via the nasal route. This observation suggests that the virus most likely does not enter the brain through the olfactory nerve but possibly through other nerves. Moreover, there were differences in the quality of the lesions when animals were administered EHV-9 via nonnasal routes. One animal infected via the oral route exhibited malacia and hemorrhage in the brain, but the pathogenesis of these lesions is uncertain at present. Further investigations are needed to determine exactly how EHV-9 reaches the brain in routes other than the nasal route as well as the pathogenesis of EHV-9–induced encephalitis by these alternative routes of administration.
An interesting observation was that animals infected via the ocular route had mild and localized lesions in the rhinencephalon, which indicated that the virus had traveled to the CNS through the optic nerve. Fatal infections by Cercopithecine herpes virus 1 (B virus) in humans via ocular exposure from biological fluid from macaque monkeys have been reported. 2 Similar to B virus, the ocular route may also be important in the natural transmission of EHV-9 in some sensitive animals such as goats, cats, dogs, and gazelles.
The differences in the incubation period and paths of travel to the CNS among the various routes in the present study resulting in a variety of clinical signs and histopathological features suggests a dependency on the replication of the virus at the site of entry and its opportunity to access regional nerves to travel to the brain. A similar hypothesis was proposed in the case of pigs infected orally by EHV-9, 11 where the virus was thought to travel centripetally in the nerve fibers from the oral mucosa to the trigeminal ganglion, eventually entering the olfactory lobes. 3,9,10
This study provides new data about experimental EHV-9 infection in hamsters through routes other than the IV route. Since EHV-9 antigen was not detected by immunohistochemistry in the olfactory epithelium and internal organs of the clinically affected animals inoculated via the oral, peritoneal, and ocular routes, these results suggest that in the animals infected by these routes, EHV-9 might travel to the brain through regional nerves (nonolfactory route), after initial propagation at the site of viral entry.
Footnotes
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
This study was supported, in part, by a Grant-in-Aid for Scientific Research from the Ministry of Health, Labor and Welfare of Japan.
