Abstract
A cerebral granular cell tumor is described in a 6-year-old, shorthaired, female cat. The tumor was observed above the corpus callosum and completely infiltrating the third ventricle. Histologic examination revealed that the tumor was characterized by large cells containing densely packed intracytoplasmic granules and expressed psammoma body-like patterns and cholesterinic degeneration. immunohistochemical analysis revealed granular neoplastic cells that were diffusely and strongly vimentin-positive, while they did not express cytokeratins, lysozyme, and synaptophysin. Based on morphologic and immunohistochemical findings, the tumor under study was considered to be of meningeal origin arising directly from the meninges or from meningeal elements scattered in the tela choroidea of the third ventricle roof.
Granular-cell tumors (GCTs) of the central nervous system (CNS) have been reported to be of meningeal cell derivation in the rat, ferret, and dog. 9 In human oncology, granular cell tumors comprise a large number of neoplasms occurring within the nervous system as well as extraneurally. The former are considered to be of astrocyte or pituicyte derivation, 7 while the extraneural granular cell tumors that have been reported in several sites (e.g., skin, tongue, breast, and gastrointestinal and biliary systems) are considered to be of peripheral nerve origin. 1 Similar extraneural granular cell tumors have been reported in dogs, horses, cats, cockatiels, and laboratory rodents. Once thought to be of myoblastic origin, they are now considered most likely to be of peripheral nerve origin. 6 To date, no intracranial granular cell tumors have been reported in the cat.
This report describes the histomorphologic and immunohistochemical findings of a cerebral tumor in a 6-year-old, shorthaired breed, female cat. The animal was previously affected by tremors and seizures associated with nystagmus and opistotonus. Spontaneous death occurred after a sudden onset of lethargy, stiffness, and respiratory failure. Unfortunately, the owner did not grant permission for further neuroimaging studies of the brain.
At necropsy, gross pathologic findings were limited to the brain. The brain was fixed in buffered 10% formalin and cut into transverse sections. A whitish-gray, firm, well-circumscribed extraparenchymal mass, approximately 1.5 cm in diameter, was observed rostrally, above the corpus callosum, symmetrically compressing the cingulate cortex. In the coronal sections from the diencephalon, this mass showed a widely calcified and irregular cut surface and completely occupied the third ventricle up to the hippocampus (Fig. 1). The mass extended to the dorsal surface of the mesencephalon between the occipital lobes.
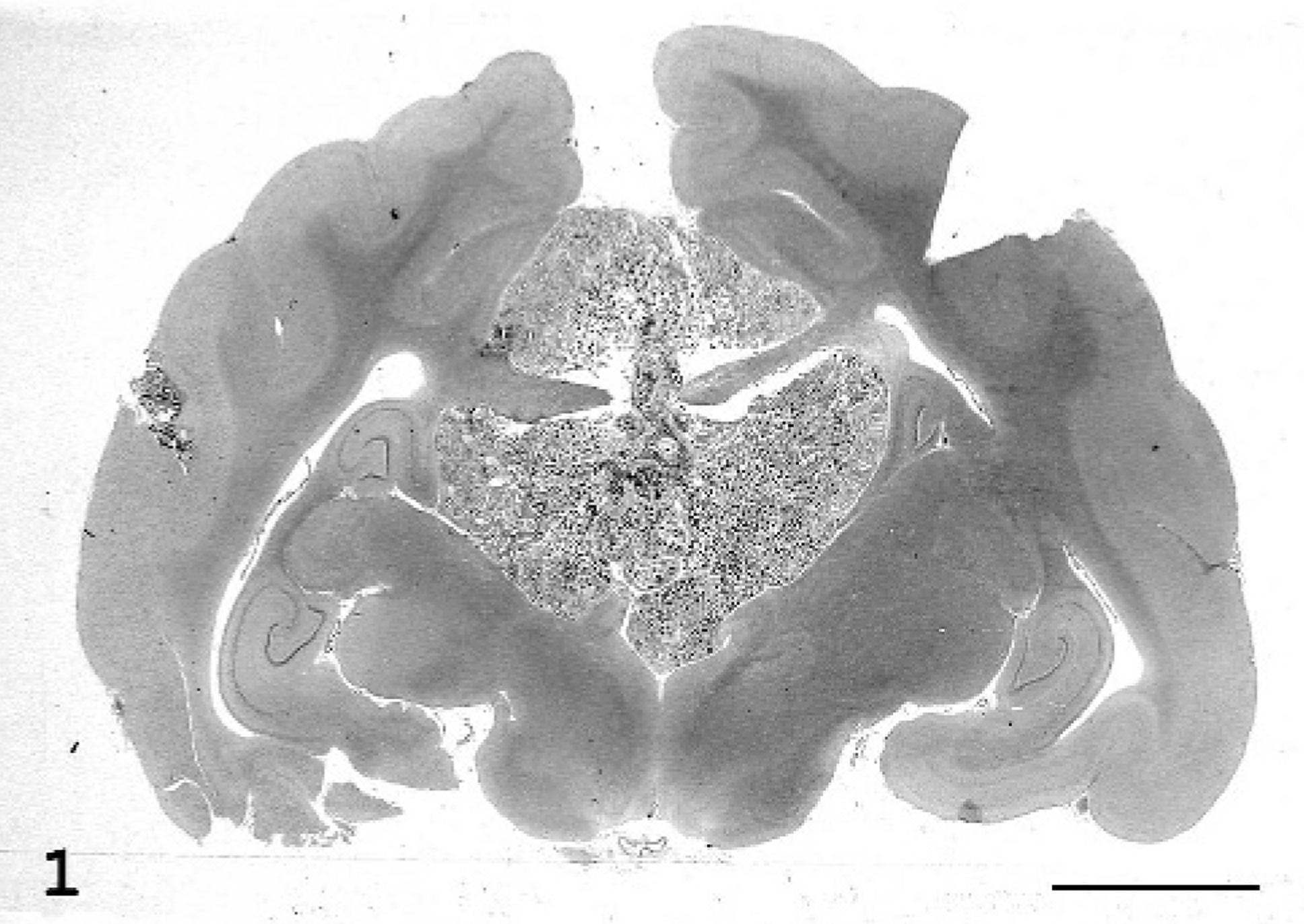
Brain; cat. The tumor extends from the cingulate cyrus to the third ventricle crossing the corpus callosum. HE stain. Bar = 6 mm.
Five-micrometer sections from paraffin-embedded coronal samples were stained with hematoxylin and eosin (HE). Histochemical stains included diastase-resistant periodic acid-Shiff (PAS) and Gabe Martoja trichromic staining for the collagen matrix. To identify the histogenesis of this tumor, immunohistochemical staining was performed using the streptavidin-biotin-peroxidase method according to the manufacturer's instructions (LSAB+ Kit, Dako, Milan, Italy). Mitotic index was investigated by Ki67. The primary antibodies used are summarized in Table 1. The chromagen was 3,3′-diaminobenzidine (Dako), and the slides were lightly counterstained with Mayer's hematoxylin. Appropriate positive and negative control tissues were included in all staining procedures.
Primary antibodies used in evaluation.
∗ GFAP = glial fibrillary acidic protein.
† NSE = neuronal specific esterase.
‡ PR88 = progesterone receptor.
On histologic examination, the newly formed intracerebral tissue appeared to be composed of large cells arranged in sheets. The cells had abundant, pale, eosinophilic cytoplasm with densely packed intracytoplasmic granules, distinct cell margins, and peripheral nuclei (Fig. 2). The granules were variably PAS-positive diastase–resistant. Numerous neoplastic cells had vacuolated cytoplasm. A number of interstitial cells with a round to oval or irregular nucleus and a slightly eosinophilic cytoplasm, sometimes without well-defined cellular borders, were observed in a fine collagen matrix. Numerous foci of calcification and peripheral cholesterol clefts were also observed. Mitotic figures were rare. A number of medium- and large-caliber vessels were identified by Gabe-Martoja staining in a poorly developed stroma, bound to the margin of the tumor and the recognizable choroid plexus. The tumor appeared to be circumscribed by a meningeal or ependymal layer and did not infiltrate the adjacent periventricular and subpial nervous tissue.
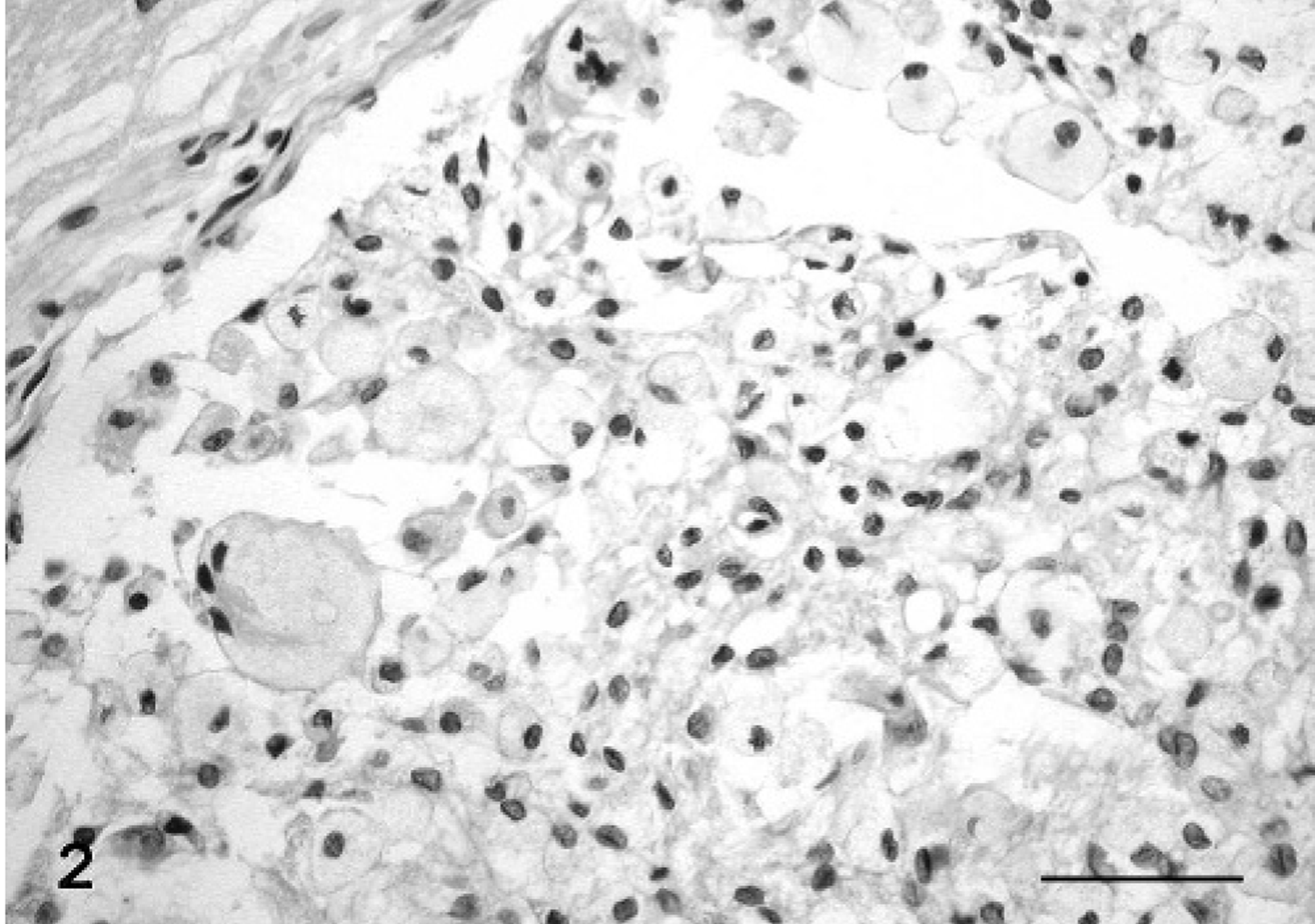
Brain tumor; cat. The neoplastic cells, some multinucleated, are arranged in sheets, and they appear to be distended by a finely granular material. HE stain. Bar = 50 μm.
The only structure involved in the expanding tumor was the corpus callosum above the third ventricle.
By immunohistochemistry, granular neoplastic cells were diffusely and strongly vimentin-positive (Fig. 3), but they did not express cytokeratins or synaptophysin. The granules of neoplastic cells appeared inconsistently S-100 positive, and occasionally neoplastic cells expressed progesterone (PR)-receptors. A marked immunoreactivity was observed in the nongranular interstitial cell population using anti-S-100 protein, anti-neuron-specific enolase (NSE), and antilysozyme antibodies (Fig. 4). The ependymal layer and scattered interstitial cells involved in the tumor stained positive for glial fibrillary acidic protein (GFAP. Laminin immunolabeling was confined to blood vessel basal membranes. The mitotic index, using a Mib-1 marker, was low (Table 2).
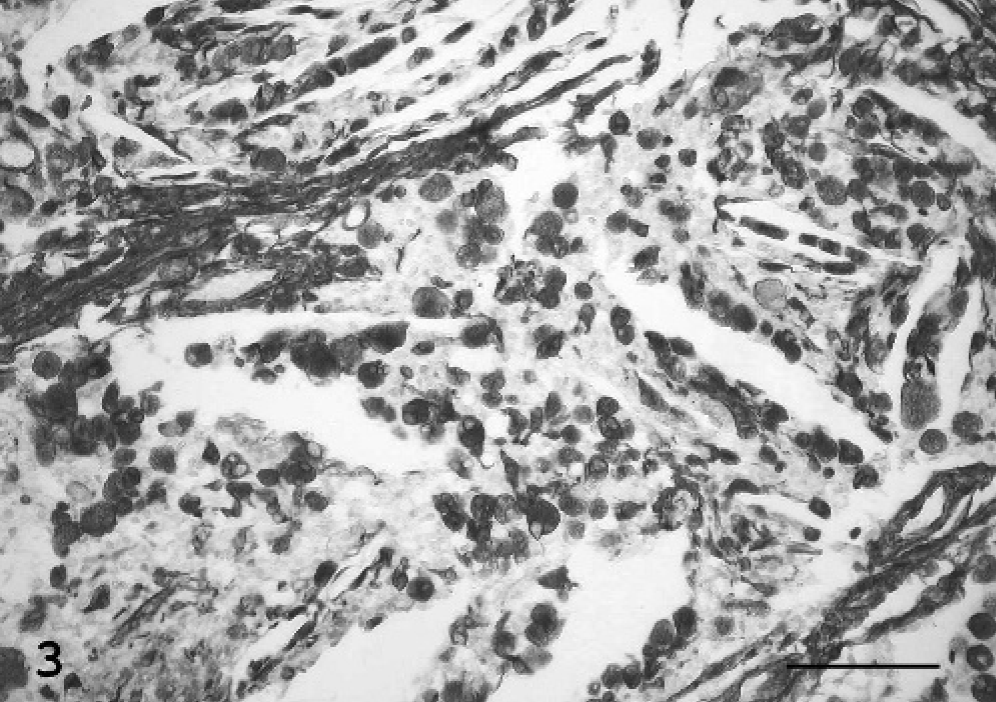
Brain tumor; cat. The granular cytoplasm of neoplastic cells stains strongly positive for vimentin along with mesenchymal stroma. Avidin-biotin peroxidase complex method. Mayer's hematoxylin counterstain. Bar = 88 μm.
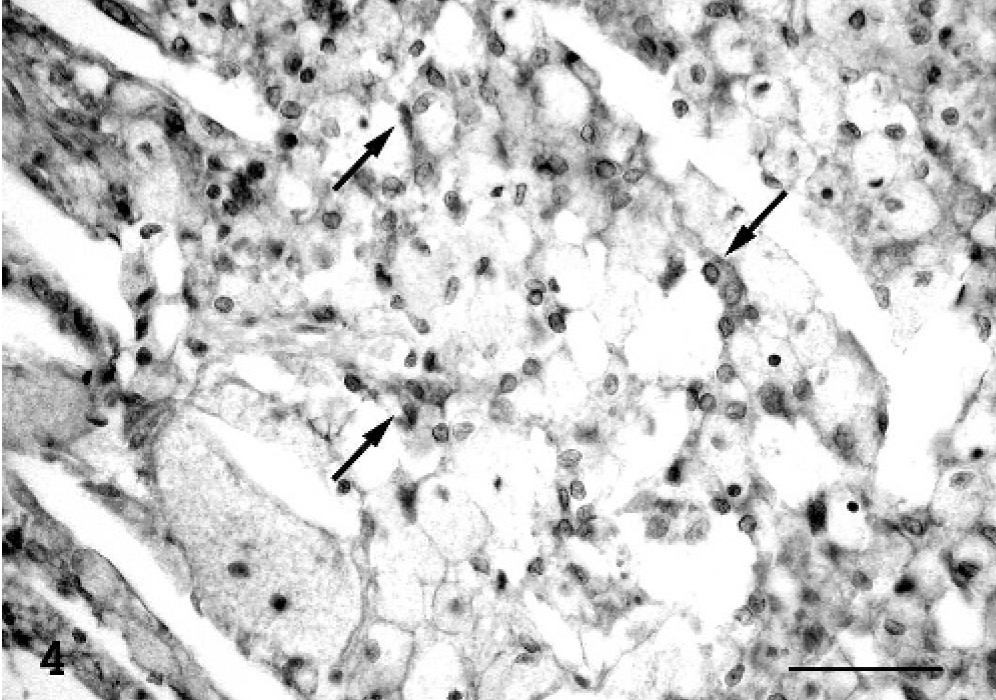
Brain tumor; cat. A number of interstitial cells react to lysozyme (arrow), while the neoplastic granular cells do not express this histiocytic phenotype marker. Avidin-biotin peroxidase complex method. Mayer's hematoxylin counterstain. Bar = 50 μm.
Immunohistochemical results in the granular cell tumor.∗
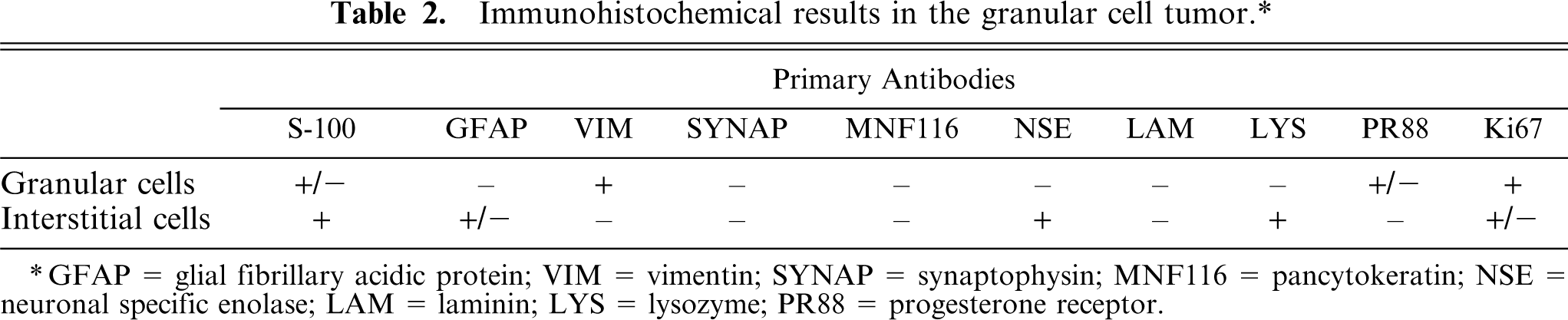
∗ GFAP = glial fibrillary acidic protein; VIM = vimentin; SYNAP = synaptophysin; MNF116 = pancytokeratin; NSE = neuronal specific enolase; LAM = laminin; LYS = lysozyme; PR88 = progesterone receptor.
Feline granular round-cell tumors of the small intestine and mesentery have been referred to as being of globule leukocyte and large granular lymphocyte origin. 5 Immunocytochemical studies on feline granular round-cell tumors occurring in the tongue, tonsil, vulva, and digit suggested that granular-cell tumors have different cellular origins despite their similar morphologic features. 3 While GCTs are well documented in the CNS of humans and rats, they are rarely reported in dogs and have never been reported in the cat. In humans, cerebral GCTs occurring in the infundibulum of neurohypophysis are suspected to be of specialized astrocyte origin. 8 In some cases, human intracranial GCTs, like extraneural GCTs reported in the skin, tongue, breast, and biliary and gastrointestinal systems, are considered to be of Schwann cell origin. 1 Canine cerebral GCTs appeared to be of meningeal cell origin 2 or mesenchymal and neural nature. 3
The tumor under study appeared to develop outside the neural parenchyma and histologic examination showed features commonly observed in meningeal tumors, such as psammoma body-like patterns and areas of cholesterol degeneration. Based on the negative staining for GFAP expression, the tumor was not considered to be of astrocytic derivation. Neoplastic granular cells did not stain for GFAP and cytokeratin thus excluding their origin from the ependyma and choroid plexus, respectively. Moreover, the uniformly positive vimentin-immunolabeling, along with the focally positive reaction to S-100, supports a meningeal origin, while negative staining for lysozyme excluded a monocyte/histiocyte phenotype of these cells. The granular cell expression of progesterone receptors suggests a meningeal origin. 4 The complete loss of synaptophysin immunolabeling in the granular neoplastic cells would exclude a neural or epithelial neuroendocrine origin. GFAP, S-100, lysozyme, and NSE interstitial immunoreactivity suggested the presence of a mixed reactive cell population including ependymal, histiocytoid, and glial cells, instead of precursor cells, transitioning toward large granular cells. 2 Therefore, the present study suggests this feline GCT originated from the meninges of the cingulate cyrus and expanded through the corpus callosum to the third ventricle or, conversely, from the meningeal elements scattered in the choroidea tela of the third ventricle roof to the cingulate cyrus. The latter hypothesis is well documented in the cat, where intraventricular meningiomas, sometimes with a multifocal pattern, have been reported. To our knowledge, this is the first report of a cerebral granular cell tumor of probable meningeal origin in the cat.
