Abstract
Nongestational choriocarcinoma is a rare malignancy in humans with poor prognosis. Naturally occurring choriocarcinoma is also rare in laboratory mice, and no genetic mouse model accurately recapitulates the features of this cancer. Here we report development of a genetically engineered mouse (GEM) model with alterations in Brca2, Trp53, and RB that develops ovarian tumors. Most of the ovarian tumors displayed histological characteristics of nongestational choriocarcinoma of the ovary (NGCO) (47%) with abundant syncytiotrophoblasts and cytotrophoblasts, positive immunolabeling for human chorionic gonadotropin, and positive periodic acid–Schiff reaction. The rest of the ovarian tumors were serous epithelial ovarian carcinoma (SEOC) (26%) or mixed tumors consisting of NGCO and SEOC (26%). We further established syngeneic orthotopic mouse models for NGCO by in vivo passaging of GEM tumors. These metastatic models provide a platform for evaluating new treatment strategies in preclinical studies aimed at improving outcomes in choriocarcinoma patients.
Nongestational choriocarcinoma of the ovary (NGCO) in humans is an exceedingly rare germ cell malignancy occurring with very low incidence. 9,20,21 These tumors present as hemorrhagic and necrotic masses on the ovary and microscopically display the presence of syncytiotrophoblasts and cytotrophoblasts. Characteristic production of human chorionic gonadotropin (hCG) is confirmed in blood or by immunolabeling. NGCO commonly presents admixed with other types of ovarian tumors (dysgerminoma, teratoma, endodermal sinus tumor, embryonal carcinoma) rather than as a pure ovarian choriocarcinoma. 11 The prognosis is usually poor due to widespread metastases to the lungs and brain. 17 Treatment includes chemotherapy but can differ based on the major histological variant of the mixed tumor. 21
NGCO is equally rare in laboratory mice. It was reported in 2 females in the B6C3F1 strain, 5 in a single case in a genetically engineered mouse (GEM) heterozygous for loss of Hic1 and Trp53 (Hic1+/– p53+/– ), 3 and in 2 cases of GEM with loss of Trp53 targeted to the endothelial cells (Tie2-Cre, Trp53fl/fl mice). 2
Previously, we reported the development of a GEM model for the serous type of epithelial ovarian cancer (SEOC), specifically induced in ovarian surface epithelium, that harbors perturbations in the Brca1 or Brca2 genes together with Trp53 deletion and RB suppression. 19 However, a subset of these mice developed ovarian lesions other than SEOC that were suspected to be choriocarcinoma but were not explored further (unpublished data). In this study, we established a new cohort of GEMs to confirm, by pathology and biomarker analysis, that the most frequent ovarian lesion other than SEOC is choriocarcinoma. Since there is no syngeneic mouse model available for this type of rare cancer, we also sought to develop a transplantable allograft syngeneic mouse model for NGCO that can be used in preclinical studies to evaluate novel therapies for this type of cancer.
A cohort of 59 Brca2fl/fl (STOCK Brca2tm1Brn ), Trp53fl/fl (FVB; 129-Trp53tm1Brn ), and Tg(K18GT121)tg/+ FVB mice 16 was injected under the right ovarian bursa with adenovirus expressing Cre recombinase to induce the genetically engineered alleles (Brca2fl/fl , Trp53fl/fl , and Tg(K18GT121)tg/+ ) specifically in the epithelial cells of the ovarian surface, as described previously. 19 Induction of these alleles results in loss of BRCA2 and TRP53 and expression of 121 amino acids of the SV40 large T antigen that efficiently inhibit proteins of the RB family (RB, P107, and P130). All animal procedures used in this study were performed at the Frederick National Laboratory for Cancer Research (FNLCR) and were approved by the National Cancer Institute–Frederick Animal Care and Use Committee (ACUC) prior to initiation. The care and use of animals were conducted in accordance with the principles outlined in the current guidelines published by the National Institutes of Health’s Guide for the Care and Use of Laboratory Animals. All experimental procedures followed standard operating procedures previously approved by the institutional ACUC and respected the Good Laboratory Practice principles.
All mice were submitted for complete necropsies upon reaching the predetermined end points: presence of tumors measuring 2 cm in diameter and/or presence of hemorrhagic ascites, labored breathing, or any other clinical signs that caused distress to the animal. The following tissues were submitted for histopathological analysis: female reproductive tract, liver, lung, pancreas, spleen, and mesenteric lymph node(s). All tissues were fixed in 10% formalin for 48 hours, trimmed, routinely processed for histology, sectioned at a 5-μm thickness, and stained with hematoxylin and eosin. Selected samples were also stained with periodic acid–Schiff (PAS).
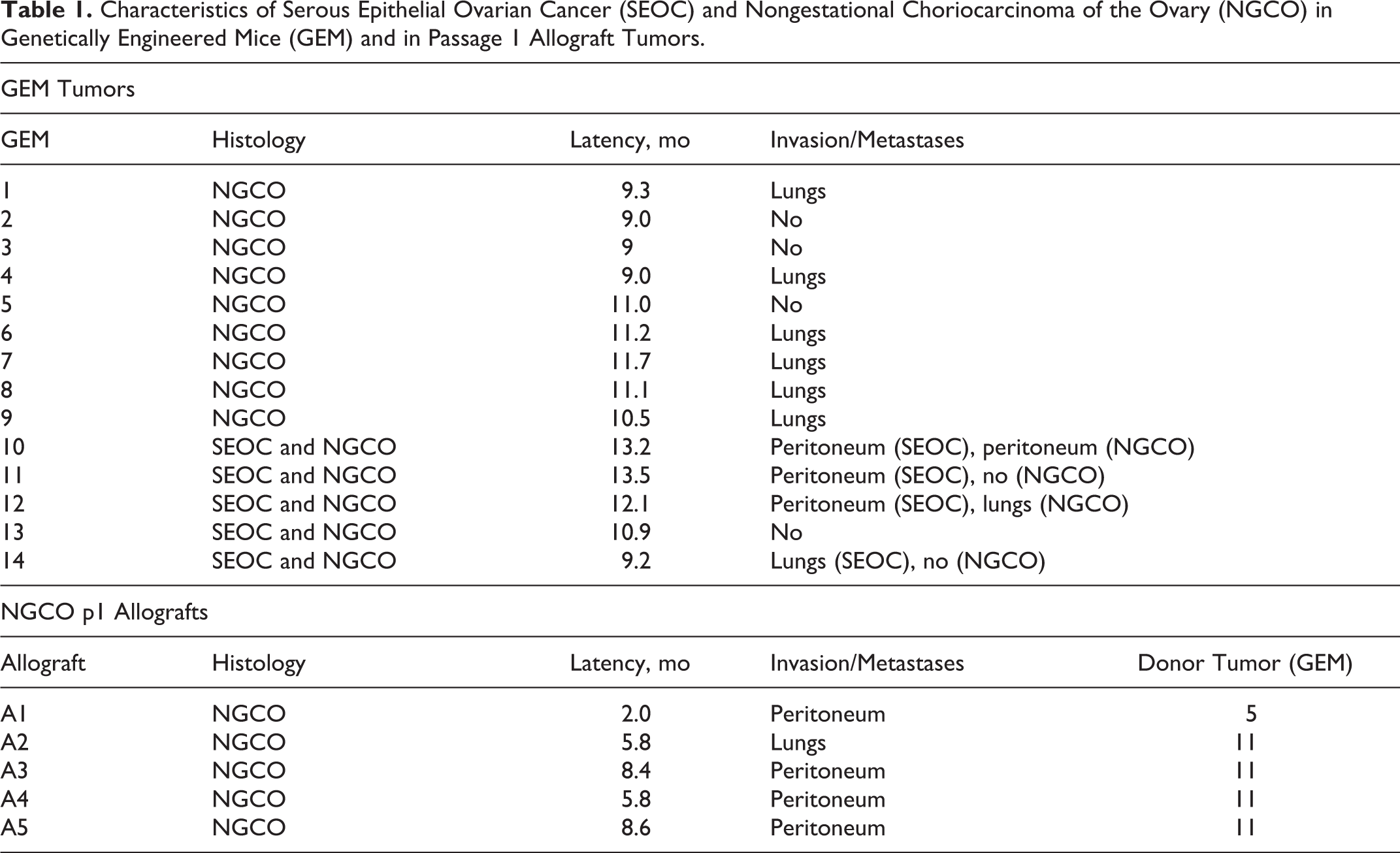
All the mice that survived beyond 9 months (19/59, 32% of the cohort) developed large ovarian tumors with latencies of 9 to 13 months post-induction. Of those, 5 of 19 (26%) were histologically characterized as SEOC (Fig. 1), 9 of 19 (47%) as pure choriocarcinoma (Fig. 2), and 5 of 19 (26%) as SEOC mixed with choriocarcinoma (Fig. 3). The remainder of the cohort was euthanized between 3 and 9 months post-induction due to development of thymic tumors (thymoma or lymphoma) (9/59, 15%), multicystic renal dysplasia (19/59, 32%), a combination of both kidney and thymus phenotypes (5/59, 8%), or other reasons (7/59, 12%). Although no ovarian tumors were observed at gross examination in the mice that developed ectopic tumors, histological examination confirmed the presence of early pathological changes, carcinoma in situ (CIS), or cellular atypia of the ovarian surface epithelium with no significant invasion into the underlying ovary and/or overlying bursa on the induced ovaries, indicating that the tumor phenotype was fully penetrant.
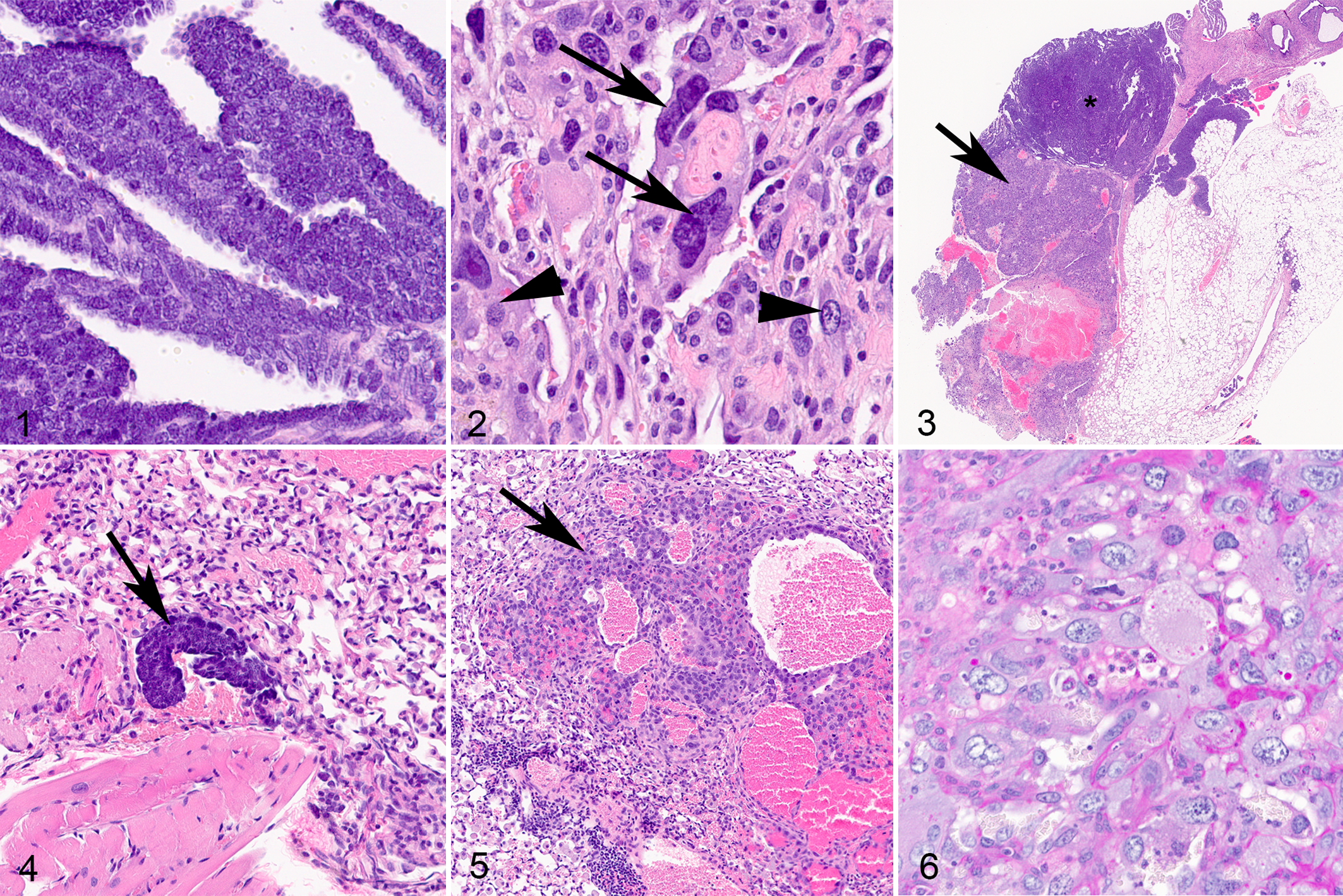
Ovarian cancer, ovary, mouse.
As reported previously, 10,19 SEOC presented as soft ovarian tumors composed of a well-delineated, nonencapsulated population of malignant epithelial cells exhibiting solid to papillary architecture (Fig. 1). Individual tumor cells were variably distinct, round to oval, with prominent anisocytosis and anisokaryosis. Nuclei were round to oval with granular cytoplasm, 1 to 3 variably indistinct nucleoli, and a high mitotic rate (4–7 per 400× high-power field).
The non-SEOC ovarian lesions presented as large hemorrhagic masses composed of well-delineated, nonencapsulated large pleomorphic cells arranged in sheets. These neoplastic cells exhibited 2 distinct populations consistent with cytotrophoblasts and syncytiotrophoblasts, indicative of choriocarcinoma (Fig. 2). Most of neoplastic cells (cytotrophoblasts) exhibited distinct borders and were polygonal to oval, up to 75 μm diameter, with abundant finely granular to vacuolated lightly basophilic cytoplasm. Nuclei were round to oval with vesicular chromatin, had indistinct to multiple nucleoli, and exhibited occasional binucleation. Anisocytosis and anisokaryosis were prominent, and bizarre mitotic figures were present. A small population of neoplastic cells (syncytiotrophoblasts) exhibited pronounced multinucleation (up to 12 nuclei). Multifocally, blood-filled cysts (up to 1 mm in size) lined by trophoblast-like cells were also noted along with both hemorrhage and necrosis.
In addition, histological examination of the hemorrhagic masses often revealed the presence of 2 distinct tumor populations that completely replaced the normal architecture of the ovary and the oviduct (Fig. 3). The 2 tumor populations were similar in overall distribution (approximately 1:1). The first population of neoplastic cells was consistent with SEOC histomorphology (Figs. 1, 3), while the second population displayed characteristics of NGCO (Figs. 2, 3). Both tumor populations displayed capabilities for local spread in the form of carcinomatosis in the peritoneal cavity and for distant metastases in lungs (Table 1). The 2 tumor populations were clearly distinguishable at invasion sites, based on their differential histology (Figs. 4, 5).
Characteristics of Serous Epithelial Ovarian Cancer (SEOC) and Nongestational Choriocarcinoma of the Ovary (NGCO) in Genetically Engineered Mice (GEM) and in Passage 1 Allograft Tumors.
To confirm the choriocarcinoma diagnosis, we evaluated tumors with PAS stain and with immunohistochemistry for hCG, as mouse ovarian choriocarcinoma is expected to exhibit intracellular accumulations of PAS-positive material and positive staining with hCG. 1 Cytotrophoblasts occasionally contained amorphous droplets of PAS-positive material (Fig. 6). In addition, immunohistochemistry was performed on paraffin-embedded tissues using heat-induced antigen retrieval with citrate at pH 6.0 (hCG, CD31) or EDTA at pH 9.0 (CD34, von Willebrand factor [vWF]) and primary antibodies for CD31 (Abcam, Cambridge, MA), CD34 (eBioscience, now part of ThermoFisher, Grand Island, NY), vWF (Proteintech, Rosemont, IL), and hCG (Origene, Rockville, MD) on Leica Biosystems’ Bond Autostainer (Buffalo Grove, IL). Internal positive and negative controls were used for optimization of CD31, CD34, and vWF immunohistochemistry, and Origene E13 placenta was used as positive and negative controls for hCG. Immunolabeling for hCG (Fig. 7) exhibited variable positive cytoplasmic immunoreactivity in cytotrophoblasts. 12 Immunolabeling was negative for endothelial markers CD31, CD34 (not shown), and vWF (Fig. 8). This finding, along with tumor morphology, effectively ruled out the possibility of a concurrent endothelial cell malignancy (angiosarcoma).
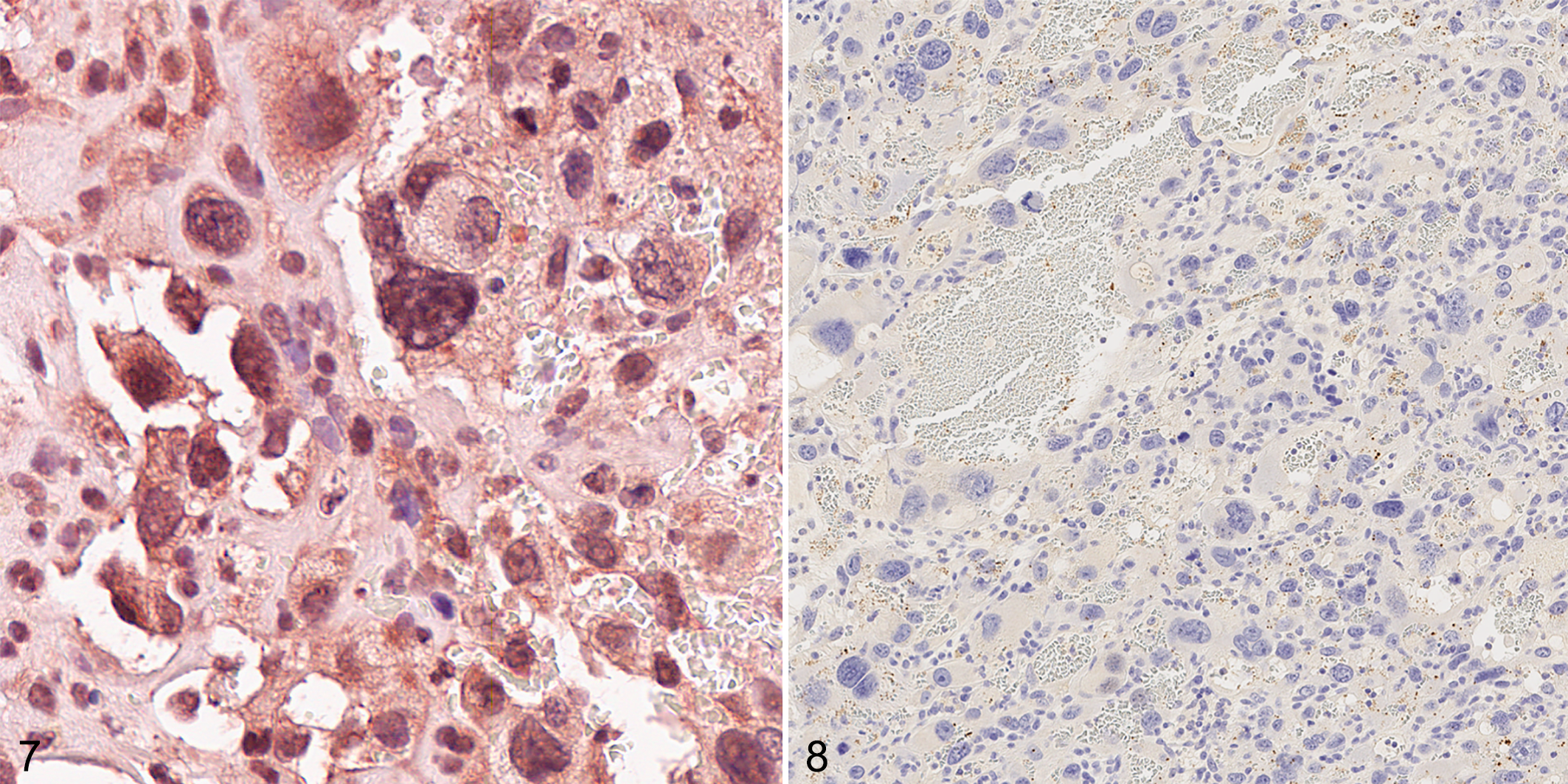
Nongestational choriocarcinoma of the ovary (NGCO), mouse. Positive cytoplasmic cytotrophoblast immunolabeling for human chorionic gonadotropin (hCG) (Figure 7) and absence of immunolabeling for von Willebrand factor (Figure 8) confirm NGCO pathology.
To establish syngeneic allograft models for further studies, 10 of the harvested tumors were orthotopically transplanted under the ovarian bursa of strain-matched mice, as described previously. 18 Of the 10 donor tumors, 2 were choriocarcinomas, 4 were mixed tumors of SEOC and NGCO origin, and 4 were SEOC. Transplanted tumors grew with much shorter latency periods than tumors in GEM models (Table 1). In general, allografts recapitulated the histology of donor tumors, with the exception of mixed SEOC/NGCO tumors, which mostly retained the SEOC histology after transplantation. This might have resulted from the technical difficulty of implanting choriocarcinoma tissue that appears hemorrhagic, rather than its potential to establish tumors after transplantation. Nevertheless, after a single passage, we established 5 NGCOs that grew with a latency of 2 to 8.6 months and retained their capacity to spread and metastasize (Table 1). These allograft models are available to be continuously passaged in syngeneic immunocompetent mice and to be used in preclinical studies evaluating potential therapeutics for choriocarcinoma.
Currently, most of available choriocarcinoma models use JEG-3 and BeWo human choriocarcinoma cell lines that were established decades ago 8,14 from human choriocarcinoma serially transplanted through hamster and rat. The exact subtype of the original human choriocarcinoma was not described. A similar cell line, JAR, was derived from gestational choriocarcinoma that developed after pregnancy. 15 Another model of gestational choriocarcinoma was established in nude mice by subcutaneous or intraperitoneal injection of immortalized human primary normal trophoblast cells that were retrovirally transduced with activated H-RAS. 7 Due to their human origin, all of the above cell lines must be used in immunodeficient mice to achieve in vivo growth. Here, we described GEM models with defined genetic aberrations that develop nongestational choriocarcinoma of the ovary, with the capacity to spread throughout the peritoneum and distantly to the lungs. Moreover, we have established a transplantable allograft model that retains the NGCO histology and metastatic capacity when passaged orthotopically in syngeneic mice (Table 1). The benefits of the immunocompetent transplant model include the capacity to increase the cohort size as needed for preclinical evaluation and optimization of treatments, as well as the ability to evaluate immunotherapeutics in a more natural tumor microenvironment. The tumors also have defined genetic aberrations: loss of Trp53, inhibition of RB tumor suppression, and loss of Brca2.
Pathogenesis of choriocarcinoma is poorly understood, and data for the mutation profile in these tumors are extremely limited. A study examining copy number alternations (CNAs) and microsatellite markers of a set of human gestational choriocarcinoma (GC) and nongestational choriocarcinoma (NGC) samples identified a total of 200 CNAs affecting all chromosomes. 13 The changes for NGCs included about the same proportions of losses and gains, with gain of 21p11.1 being the only significant rare CNA. However, the functional analysis of CNAs was performed in silico, and thus the exact mutational gene profile was not discerned. BRCA2 germline mutations confer an increased risk of developing ovarian cancer, but germ cell tumors are not correlated with BRCA1 or 2 loss. 6 However, TRP53 mutation may be correlated with risk of germ cell choriocarcinoma. 4 While we cannot determine whether choriocarcinoma arose in this model as a result of Tp53 loss only or other perturbations, the presence of mixed SEOC/choriocarcinoma tumors in some of the mice indicates that the viral injection may induce germ cells as well as target epithelial cells. Regardless of the origin, we have shown that the NGCO can be reliably passaged in recipient mice, thereby generating cohorts that can be further used to evaluate novel therapies for this disease.
Footnotes
Authors’ Contribution
The authors contributed in the following way: LS, ZWO: conception and design; LS: data analysis and interpretation, writing of the manuscript, figure generation; MG, LL: technical assistance and data acquisition; BK, NP: pathological analysis and data interpretation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project has been funded in whole or in part with federal funds from the National Cancer Institute, National Institutes of Health, under contract HHSN261200800001E. The content of this publication does not necessarily reflect the views or policies of the Department of Health and Human Services, nor does mention of trade names, commercial products, or organizations imply endorsement by the US government.
