Abstract
Introduction:
Ovarian cancer is still a major health problem in Indonesia. development of breast cancer gene-related personalized medicine to increase the survival outcome of epithelial ovarian cancer patients in Indonesia is expected to be achieved. This research aims to evaluate the impact of pathogenic breast cancer gene 1 and breast cancer gene 2 tumor mutation on high-grade serous epithelial ovarian cancer survival outcome.
Methods:
This study is an observational analytic study, using a historical cohort study design. A total of 68 from 144 patients diagnosed with International Federation of Gynecology and Obstetrics 2014 stage IIB-IV high-grade serous epithelial ovarian cancer between January 1st, 2015 until March 31st, 2021, at three centers in Jakarta. Next-generation sequencing tumor breast cancer gene 1 and breast cancer gene 2 testing and were included in this cohort historical study. We compared patient’s overall survival outcomes, according to pathogenic breast cancer gene 1 and breast cancer gene 2 tumor mutational status. Clinicopathological characteristic factors that might affect patient’s survival outcomes were also investigated.
Results:
In the group of individuals with pathogenic breast cancer gene 1 and breast cancer gene 2 tumour mutations, the risk of death was significantly lower by 86% (adjusted RR 0.149; 95% CI: 0.046–0.475; p-value = 0.001), and the median survival time was significantly better (median 46 months; 95% CI: 34.009–57.991; p-value = 0.001) compared to the group without pathogenic breast cancer gene 1 and breast cancer gene 2 tumor mutations (median 23 months; 95% CI: 15.657–30.343; p-value = 0.001). The multivariate analysis revealed that the presence of a pathogenic breast cancer gene 1 and breast cancer gene 2 tumor mutation is an independent and positive prognostic factor for survival outcome. The adjusted relative risk was 0.149, with a 95% CI of 0.046–0.475, p-value = 0.001.
Conclusions:
In high-grade serous ovarian cancer patients, the pathogenic breast cancer gene 1 and breast cancer gene 2 tumor mutations group have a better prognosis with longer survival outcomes than those without pathogenic breast cancer gene 1 and breast cancer gene 2 tumor mutations.
Keywords
Introduction
Ovarian cancer ranks as the third most prevalent gynecological cancer globally, following cervical and uterine cancer. It also has the second highest fatality rate, surpassed only by cervical cancer. 1 There are still no effective tools for general population screening. This also reflected economically and cost-effective strategies for early detection and prevention of ovarian cancer have been investigated over the last decade. The cost of treatment per patient with ovarian cancer remains the highest among all cancer types. According to a study conducted by Yue et al., 2 ovarian cancer had the greatest yearly cost per person ($13,566), followed by uterine cancer ($6852), and cervical cancer ($2312). The primary constituents of medical expenses were of hospital inpatient stays, accounting for 53% ($2.03 billion), followed by office-based visits at 15% ($559 million), and outpatient visits at 13% ($487 million). 2 In Indonesia, ovarian carcinoma economic burden was depends on chemotherapy regimen and length of stay. 3
The Globocan 2020 study reported an increase in the number of new cases and mortality cases of ovarian cancer. Specifically, there were 313,959 new cases and 207,252 mortality cases in 2020, compared to 295,414 new cases and 184,799 mortality cases in 2018. 4 The current anticipated global 5-year prevalence of ovarian cancer is 823,315 cases, which represent an increase compared to the 2018 figure of 600,000 cases. Asia has the greatest rates of incidence, mortality, and 5-year prevalence among all continents. Specifically, there are 170,759 (54.4%) instances of incidence, 112,936 (54.5%) cases of mortality, and 435,574 (52.9%) cases of 5-year prevalence in Asia.5,6 Ovarian cancer in Indonesia has the second highest incidence, mortality, and 5-year prevalence rate among all cancers, behind cervical cancer. There are 14,896 cases, 9581 deaths, and 37,533 cases that have lasted for at least 5 years. 7 This makes Indonesia the third highest country in Asia in terms of ovarian cancer cases, after China and India.7,8
CA125 and HE4 are the sole authorized biomarkers for application in epithelial ovarian cancer; yet, they are inadequate for early identification. Multivariate index (MVI) assays have been created to address the restrictions of using individual blood indicators in the assessment of epithelial ovarian cancer, particularly when evaluating adnexal masses before surgery. 9 The Risk of Malignancy Algorithm (ROMA) utilizes menopausal state, CA125, and HE4 levels to accurately diagnose women with a pelvic tumor. miRNAs possess significant promise in multiple facets of predicting epithelial ovarian cancer. However, additional research is required to fully characterize it as a biomarker. Specifically, it is necessary to establish consistent procedures for processing samples and improve the accuracy of platforms used to detect miRNA in tumors and blood. 10
The low survival rates, and high mortality rates in advanced ovarian cancer patients, are associated with low optimal debulking rates. Based on the Cochrane database review report, optimal debulking surgery increased overall survival and progression-free survival (PFS) significantly.11,12 Indonesia has a low optimal debulking rate, based on a cross-sectional descriptive study at Dr. Cipto Mangunkusumo National Central Referral Hospital, in 2012–2016, which was 46%, compared to developed countries, 60%–70%. 13 These factors contribute to the high ovarian cancer case-fatality rate in the developing countries, such as Indonesia, which reached 59.2%–63.8%, while developed countries only reached 54.8%. The case fatality rate of ovarian cancer in Indonesia ranks 5th highest in Asia, after India, China, Japan, and Pakistan.14–16
Although the majority of epithelial ovarian cancers represent sporadic disease, associated with TP53 mutations (>95% type II), approximately 15%–23% are known to represent a hereditary group, associated with mutations in the BRCA1/2 cancer susceptibility gene (±20% to 29% of ovarian cancers). The prevalence rate of mutations varies based on histological subtypes, the highest being high-grade serous epithelial ovarian cancer (HGSOC), 20%–27% of epithelial ovarian cancer.17–19 The BRCA1/2 gene contributes to the process of DNA repair, cell-cycle checkpoint control, protein ubiquitylation, and chromatin remodeling. In the DNA-repair process, BRCA1/2 is involved in the repair of DNA damage, binds to RAD51, initiates homologous recombination, and repairs double-strand breaks DNA. In the process of chromatin remodeling, BRCA1 plays a role in DNA repair, forming multimeric complexes with chromatin-remodeling complexes (SW1 and SNF), and histone deacetylase complexes. Mutations in this gene will interfere with the process of chromatin remodeling in DNA damage. All of these mechanisms, which play a role in the carcinogenesis of HGSOC, are related to the BRCA1/2 mutation.20,21
According to a meta-analysis, BRCA1/2 mutations respond better to Platinum chemotherapy, have higher complete response rates, lower partial responses, and longer PFS, compared to the wild-type group. This is associated with inhibition of DNA repair pathways, making tumor cells more sensitive to the DNA-damaging effects of chemotherapy. The pathogenic BRCA1/2 mutations are also associated with the development of targeted therapy for poly (ADP-ribose) polymerase (PARP) inhibitors, as personalized medicine. In the state of homologous recombination deficiency due to BRCA1/2 mutations, cancer cells are highly dependent on the PARP-mediated base excision repair (BER) mechanism of DNA single-strand breaks to repair DNA damage spontaneously. PARP inhibitors exert a significant antitumor effect, related to synthetic lethality in HGSOC patients with pathogenic BRCA1/2 mutations. 22
Six main routes of DNA damage repair (DDR) have been identified. These processes are utilized to repair double-strand DNA breaks (DSB) and single-strand DNA breaks (SSB) caused by various causes of injury. Homologous recombination (HR) and nonhomologous end joining (NHEJ) are the primary mechanisms involved in mending double-strand breaks (DSBs). The HR pathways are activated during the S/G2 phase as a result of the presence of a sister chromatid, while NHEJ repairs DSBs during all phases of the cell cycle except the M phase. Nonhomologous end joining (NHEJ) is a more rapid process compared to homologous recombination (HR) and predominantly takes place during the G1 phase of the cell cycle. In addition to the well-established proteins Ku70/80, DNA-PKcs, Artemis, DNA pol λ/μ, DNA ligase IV-XRCC4, and XLF, several novel proteins participate in the nonhomologous end joining (NHEJ) process. These include PAXX, MRI/CYREN, TARDBP of TDP-43, IFFO1, ERCC6L2, and RNase H2. MRI/CYREN has a dual function, promoting NHEJ during the G1 phase of the cell cycle and suppressing the pathway during the S and G2 phases. 23
Based on the description above, ovarian cancer is still a major health problem in Indonesia. An effort to detect risk factors for hereditary ovarian cancer susceptibility genes, and determine targeted treatment, intending to increase survival rates, is closely related to the pathogenic BRCA1/2 mutations analysis. The Dr. Cipto Mangunkusumo Hospital, as a center of a national referral hospital, Persahabatan Hospital, and MRCCC Hospital Siloam, as the largest cancer referral center in Jakarta, need to collect accurate data regarding the effect of pathogenic BRCA1/2 tumor mutations, on the HGSOC survival outcome. Briefly, the development of BRCA-related personalized medicine to increase the survival outcome of epithelial ovarian cancer patients in Indonesia is expected to be achieved. This research aims to evaluate the impact of pathogenic BRCA1/2 tumor mutation on HGSOC survival outcome at Dr. Cipto Mangunkusumo National Central Referral Hospital, Persahabatan Central Referral Hospital, and MRCCC Central Referral Hospital Siloam Jakarta.
Patients and methods
This study is an observational analytic study, using a historical cohort study design, aiming to determine the effect of pathogenic BRCA1/2 tumor mutations on high-grade serous epithelial ovarian cancer patient’s overall survival, at Dr. Cipto Mangunkusumo National Central Referral Hospital, Persahabatan Central Referral Hospital, and MRCCC Central Referral Hospital Siloam Jakarta.
The research was carried out after obtaining an Ethical Review from Komisi Etik Penelitian Kesehatan (KEPK) Medical Faculty of Indonesia University/Dr. Cipto Mangunkusumo National Central Referral Hospital, No.: KET-198/UN2.F1/ETIK/PPM.00.02/2020, February 24th, 2020; and No. ND-291/UN2.F1/ETIK/PPM.00.02/2021; Ethical Review of KEPK Persahabatan Central Referral Hospital, No. 106/KEPK-RSUPP/10/2020, October 1st, 2020; and Research Approval Letter of MRCCC Central Referral Hospital Siloam Jakarta, No. 735/SS/Dir/V/2021.
The subjects were selected by consecutive sampling from archival data and medical records of Anatomic Pathology Department—Medical Faculty of Indonesia University/Dr. Cipto Mangunkusumo National Central Referral Hospital, Persahabatan Central Referral Hospital, and MRCCC Central Referral Hospital Siloam Jakarta, with a diagnosis of high-grade serous epithelial ovarian cancer, evidenced by the results of histopathological examination, Formalin-Fixed and Paraffin-Embedded (FFPE) tumor tissue blocks and HE slide staining, from January 1st, 2015 until March 31st, 2021 period.
Inclusion criteria of this study were patients with advanced stage high-grade serous epithelial ovarian cancer in the Gynecologic Oncology Subdivision, as confirmed by histopathological examination results from the pathology anatomy department, patients who have undergone optimal debulking surgery and adjuvant chemotherapy (paclitaxel-carboplatin), patients with FFPE samples that meet quality control criteria. Exclusion criteria in this study were unwilling to participate in the study, specimens that do not meet analysis requirements, incomplete medical record data.
Patient selection was carried out based on the inclusion and exclusion criteria of the study, reviewed the HE slide by a single expert pathologist, confirmed the histopathological type of high-grade serous epithelial ovarian cancer, and selected the best FFPE block with the highest tumor cell content. The data collection of pathogenic BRCA1/2 tumor mutation sequencing analysis was performed at MedGenome Labs. Ltd., India, and KALGen Innolab Indonesia Clinical Laboratory.
The study aims to determine the impact of pathogenic BRCA1/2 tumor mutations and clinical characteristics on the survival of patients with advanced stage high-grade serous epithelial ovarian cancer. The estimated sample size was calculated using the formula for the Test of Difference in Proportions for Two Independent Samples with 80% power analysis. Estimated total sample size for pathogenic mutation of BRCA1/2 tumor was 52 sample with unexposed outcome of 96%, exposed outcome 60%, OR 0.07 and prevalence ratio 0.62:
The laboratory analysis begins with the Quality Control selection, at least 10% of the viable neoplastic cells—tumor content (>150 tumor cells/HPF), is considered as an acceptable criteria to continue the pathogenic BRCA1/2 tumor mutation analysis. The DNA extraction, library preparation, targeted enrichment, and sequencing: DNA extraction from FFPE tumor tissue block, is used to perform targeted gene capture, using a custom capture kit for the complete coding region of the BRCA1 and BRCA2 genes. The DNA captured library was sequenced on the Illumina HiSeq series to produce 2 × 150 bp sequence reads at 80–100× at the target sequencing depth.
Statistical analysis
The next-generation sequencing (NGS) data analysis and clinical reports: clinically associated mutations, annotated using variants published in the literature and disease database tools—ClinVar, OMIM, GWAS, HGMD, SwissVar, cBioPortal, OncoMD (MedGenome’s lab curated somatic database, including TCGA and COSMIC). The variants were most frequently filtered by minor allele frequency (MAF) on 1000 Genome phase 3, ExAC, gnomAD, dbSNP141, 1000 Japanese Genome, and the internal Indian population database. The biological effects of nonsynonymous variants were calculated using multiple prediction algorithms such as PolyPhen, SIFT, Mutation Taster2, and LRT. The reportable mutations, prioritized and reported, according to the AMP-ASCO-CAP guidelines. 24 Only nonsynonymous and splice site variants were found in the coding regions of the BRCA1 and BRCA2 genes, which will be used for clinical interpretation. Acceptance of BRCA1/2 tumor mutation analysis results from the MedGenome Laboratory and KALGen Innolab Indonesia Clinical Laboratory. The subjects’ monitoring was done until the time subjects experienced a death event, or were declared alive (sensor) until the completion of the research observation period. The subject data is then processed and analyzed using IBM SPSS Statistics version 29.0.1.0 (IBM Corp. Released 2023. IBM SPSS Statistics for Windows, Version 29.0. Armonk, NY, USA: IBM Corp.).
According to the data presented in Figure 1, a total of 144 out of the 226 sample lists from three different hospitals (Dr. Cipto Mangunkusumo National Central Referral Hospital with 106 samples, Persahabatan Central Referral Hospital with 24 samples, and MRCCC Central Referral Hospital Siloam Jakarta with 14 samples) met the criteria for inclusion. These samples were diagnosed as Stage IIB-IV high-grade serous epithelial ovarian cancer and underwent primary treatment involving laparotomy debulking and adjuvant chemotherapy. Complete data were available for histopathology slides and FFPE block preparations. Due to factors such as FIGO stage IIB (early stage), inadequate HE slide, and/or FFPE block data, the study could not include the remaining 82 samples. Out of the 144 available samples, 87 met the research inclusion criteria set by the pathologist. These criteria included reassessment of histopathological subtype, degree of differentiation, stage IIB-IV, and tumor cell content greater than 20%. On the other hand, 57 samples did not meet the reassessment criteria for inclusion in the research.

Sample selection of the study.
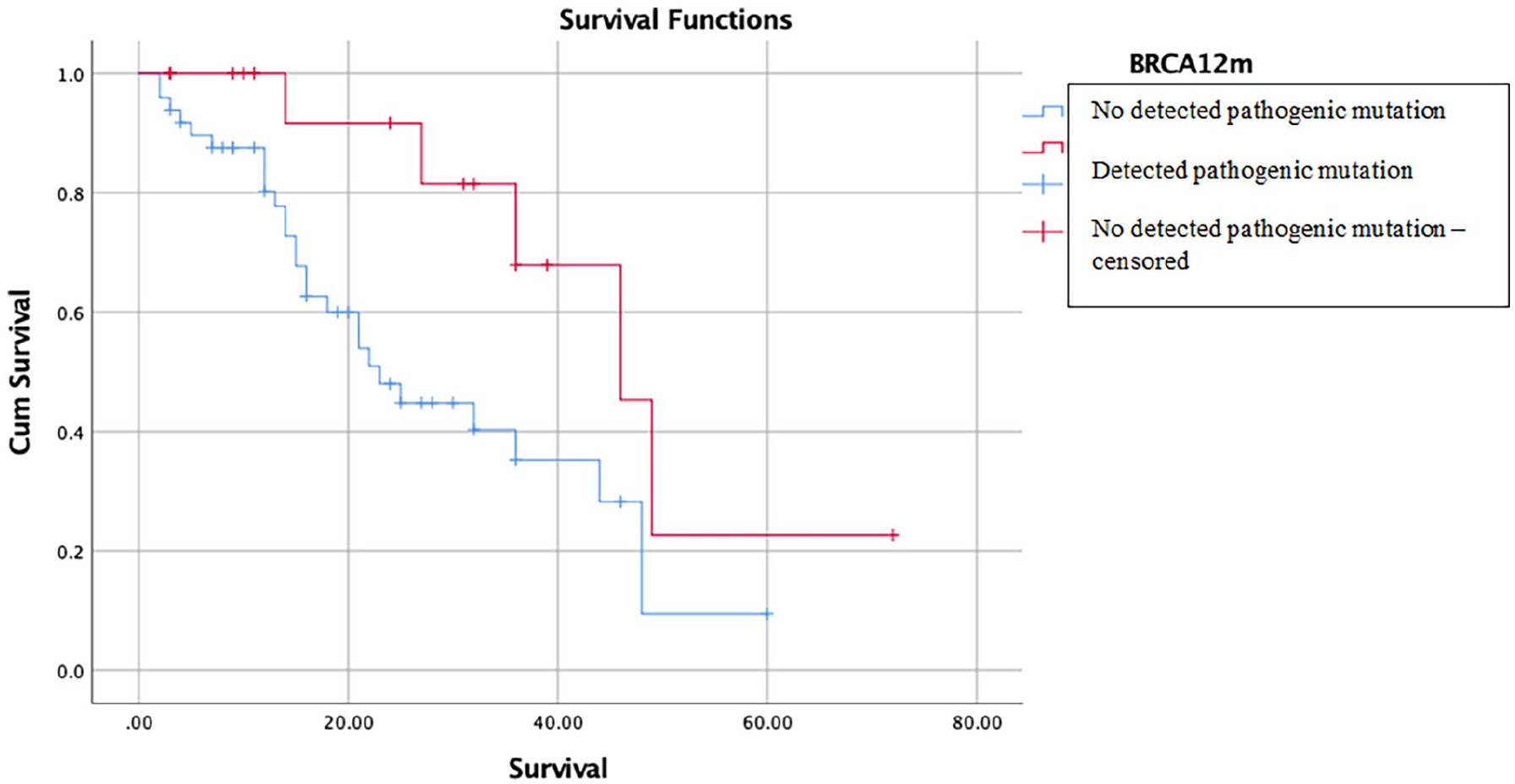
Kaplan–Meier Curve effect of pathogenic BRCA1/2 tumor mutation on survival of high-grade serous epithelial ovarian cancer patients.
All of the 87 samples mentioned above were examined for pathogenic BRCA1/2 gene tumor mutations analysis. A total of 68 FFPE samples were declared to meet the quality control (QC) criteria, followed by DNA isolation, library preparation, template preparation, and DNA sequencing. The remaining 19 FFPE samples were declared not fulfill quality control criteria, due to the low DNA Integrity Index (DIN), due to high DNA fragmentation (<120–150 bp), in suboptimal FFPE quality.
Results
Based on univariate analysis, most of the samples were 50 years old, that is, 76.47% (52/68); only 23.53% (16/68) of the sample was <50 years old. A total of 72.05% (49/68) of the sample was in the menopausal age group, and 11.76% (8/68) of the sample was nulliparous. A total of 20.58% (14/68) of the sample had a family history of breast cancer and/or ovarian cancer (HBOC). A total of 0.03% (2/68) of the sample had a previous history of breast cancer. Most of the patients were diagnosed at FIGO 2014 stage III, that is, 67.64% (46/68), the rest were FIGO IIB stage at 22.05% (15/68), and FIGO IV stage at 10.29% (7/68). A total of 60.29% (41/68) samples had preoperative CA-125 levels ⩾500 mIU/mL, and 54.41% (37/68) samples with intraoperative ascites volume ⩾500 mL. A total of 27.94% (19/68) of the sample had a cytoreductive residual lesion ⩾1 cm (sub-optimal debulking). A total of 11.76% (8/68) of the sample received neoadjuvant chemotherapy. Adjuvant chemotherapy was administered to 88.23% (60/68) of the sample, and 47.05% (32/68) of the sample was recorded to have died in this study.
Based on Table 1 above, the proportion of pathogenic BRCA1/2 tumor mutation in this study was 27.94%. The pathogenic BRCA1 tumor mutation was identified in 10 samples (14.7%), pathogenic BRCA2 tumor mutation in 10 samples (14.7%), with pathogenic tumor mutations to BRCA1 and BRCA2 at the same sample known in 1 sample (1.47%). The incidence of BRCA1/2 variants of uncertain significance (VUS) in this study was 29.41% (20/68), with 25% (5/20) of them, known to have both BRCA1/2 and VUS pathogenic mutations. The incidence of the wild-type BRCA1/2 gene in this study was 50% (34/68).
The pathogenic BRCA1/2 tumor mutation, VUS, and wild type distribution in high-grade serous epithelial ovarian cancer.

Ca-125: cancer antigen 125.
Based on Table 2, the total incidence of death in this study was 47.05% (32/68). The highest incidence of death was seen in the age group <50 years (56.3%), parity ⩽1 (53.3%), BMI <25 kg/m2 (54.5%), no previous history of breast cancer (48.5%), no HBOC family history (50.0%), FIGO 2014 stage ⩾III (52.8%), preoperative CA 125 level ⩾500 IU/mL, and intraoperative ascites volume ⩾500 mL (63.4% and 62.2%, respectively), postdebulking laparotomy positive residual lesions (73.5%), administration of neoadjuvant chemotherapy (87.5%), no adjuvant chemotherapy (75.0%), and no pathogenic BRCA1/2 tumor mutation (55.1%).
Survival analysis of high-grade serous epithelial ovarian cancer.

BMI: body mass index; R0: a complete resection of the tumor (R0 resection) was achieved; Ca-125: cancer antigen 125.
The multivariate analysis using logistic regression revealed that among the five variables examined in Table 3, which was body mass index (BMI kg/m2), family history of cancer related to HBOC, preoperative CA 125 levels (U/mL), intraoperative ascites volume (mL), and residual lesions after debulking laparotomy, a family history of HBOC-related cancer significantly influences the tumor’s BRCA1/2 pathogenic mutation (adjusted RR 5.212; 95% CI: 1.495–18.167; p-value = 0.010). The analysis of Tables 3 and 4 indicates that factors such as age, parity, BMI, history of breast cancer, FIGO 2014 stage, preoperative CA 125 level (U/mL), intraoperative ascitic fluid volume (mL), lesion size residual, administration of NACT, and administration of adjuvant chemotherapy did not influence the presence of BRCA1/2 tumor pathogenic mutations (p-value > 0.05).
Key factors affecting mortality rate on high-grade serous epithelial ovarian cancer patients mortality rate.

Logistic regression analysis by using ENTER method to variables with p-value of <0.2 acquired through bivariate analysis.
FIGO: international federation of gynecology and obstetrics; Ca-125: cancer antigen 125.
Most influential variables on advanced stage high-grade serous epithelial ovarian cancer mortality rate.

Multivariate analysis result from multivariate variable with p < 0.2.
No R0: a complete resection of the tumor (R0 resection) was not achieved; Ca-125: cancer antigen 125.
Multivariate analysis was continued on the variables with p-value < 0.2 by Cox regression analysis. In this study, five main variables were considered to have the most influence on survival, which presented on Table 4, that is, pathogenic BRCA1/2 tumor mutation, parity >1, preoperative CA-125 levels ⩾500 IU/mL, positive residual tumor lesion, and no adjuvant chemotherapy. In the group of individuals with the pathogenic BRCA1/2 tumor mutation, the risk of death was significantly reduced by 86% (adjusted RR 0.149; 95% CI: 0.046–0.475; p-value = 0.001), compared to those without the pathogenic BRCA1/2 tumor mutation. In the group with a parity more than 1, the probability of death is likewise reduced by 73% (adjusted relative risk 0.371; 95% CI: 0.168–0.823; p-value = 0.015), compared to the group with a parity of 1 or less. In the group with a preoperative CA 125 level of ⩾500 mIU/mL, the risk of death was 3.2 times greater (adjusted RR 3.189; 95%CI: 1.091–9.324; p-value = 0.034) compared to the group with a preoperative CA 125 level of <500 mIU/mL. In the group of patients with positive residual tumor lesions, the risk of death was 6.98 times higher (adjusted relative risk 6.989; 95% CI: 2.523–19.357; p-value < 0.001) compared to the R0 group (no visible residual tumor). In the group of study participants who did not get adjuvant chemotherapy, the risk of death was found to be 46.94 times higher (adjusted RR 46.949; 95% CI: 11.114–198.340; p-value < 0.001) compared to the group that received adjuvant chemotherapy.
Survival analysis was conducted to compare the median survival rates between two groups: the group with pathogenic BRCA1/2 tumor mutations and the group without these mutations. Using the Kaplan–Meier–Log Rank Test, it was found that the group with pathogenic mutations had a significantly better median survival of 46 months (95% CI: 34.009–57.991; p-value = 0.001) compared to the group without these mutations, which had a median survival of 23 months (95% CI: 15.657–30.343; p-value = 0.001). This information is presented in Supplemental Table 1 and Figure 2.
Discussion
This pathogenic BRCA1/2 tumor mutation detection, based on the NGS study, both proportion, and evaluation of the pathogenic BRCA1/2 tumor mutation impact, on the survival outcome of epithelial ovarian cancer patients, is the first study in Indonesia.
In several developed countries, the BRCA1/2 mutations in ovarian cancer research are supported by the National Health Insurance System, which has approved and covered the cost of the BRCA1/2 gene detection in all epithelial ovarian cancer patients. In these countries, germline BRCA1/2 gene assay has been recommended for all epithelial ovarian cancer patients. In Indonesia, the detection of these genes, not yet included in the insurance financing system, has not become a routine detection in epithelial ovarian cancer patients, including at Dr. Cipto Mangunkusumo National Central Referral Hospital, Persahabatan Central Referral Hospital, and MRCCC Central Referral Hospital Siloam Jakarta. The expensive examination fees, cultural factors, social stigma, and guilty feelings toward other family members, allow patients to avoid this genetic testing. Therefore, we carried out this study to determine the proportion of pathogenic BRCA1/2 tumor mutation, and the impact of pathogenic BRCA1/2 tumor mutation on the survival outcome of HGSOC patients, at the three national center referral hospitals, as well as to socialize the importance of pathogenic BRCA1/2 tumor mutation’s detections on the risk factor detection, management, and survival of epithelial ovarian cancer patients in Indonesia.
The correlation of FIGO 2014 staging, preoperative CA 125 level, intraoperative ascites volume, and positive residual tumor lesion with mortality incidence
In the Cox regression multivariate analysis Table 3, the residual tumor lesion is one of the variables that considered to have a significant effect on survival outcomes. In the group of patients with positive residual tumor lesions, the risk of death was 6.98 times higher (adjusted RR 6.989; 95% CI: 2.523–19.357; p-value < 0.001) compared to the R0 group (no visible residual tumor). The correlation between the FIGO 2014 staging variables, preoperative CA 125 levels, and intraoperative ascites volume was not seen (p-value > 0.05, respectively).
In the previous effect of BRCA1/2 mutational status on cytoreductive surgery residual tumor lesion’s study, among 69 BRCA1/2 mutation patients, compared with 298 wild-type HGSOC patients (FIGO stage IIIC-IV), multivariate analysis showed that BRCA1/2 mutation status is not related to the residual tumor volume. Briefly, the better survival outcome of the pathogenic BRCA1/2 tumor mutation’s group was not associated with cytoreductive residual tumor lesion. 25
The correlation of the neoadjuvant chemotherapy and adjuvant chemotherapy with mortality incidence
Regarding the administration of neoadjuvant chemotherapy (NACT) variable, in a previous study report, by comparing the survival rates of 49 patients who received NACT, based on the BRCA1/2 mutation status, the NACT (IDS) group had poor OS and PFS, compared to the without NACT (PDS) group, p = 0.003 and p < 0.001, respectively. There was no OS difference in the BRCA1/2 mutation, and without BRCA mutation groups (median 67.2 and 47.8 months; p-value = 0.231). The BRCA1/2 germline mutation group has a better PFS (median 17.2 and 14.2 months; p = 0.014). Based on multivariate analysis, the BRCA1/2 mutation status was a good prognostic factor, increasing PFS, in accordance with multicenter studies. 20
Several other studies, also compared survival rates, in the primary management outcome of without pathogenic BRCA1/2 tumor mutation’s group.26–28 Although there were no significant differences in FIGO stage characteristics, and residual tumor lesion, the NACT group had a significantly poor PFS than the PDS group (median 14.2 vs 16.9 months; p = 0.003). Similar results were also reported in the retrospective multicenter study of Petrillo et al. 29 In the BRCA1/2 mutations group, although FIGO stage IV cases were more common in the NACT group, PFS did not differ between the NACT-IDS and PDS groups (p = 0.082). However, the NACT group showed poor OS than the PFS group (5-year survival rates; 57.9% vs. 82.8%; p = 0.040). 29
In this study, the highest incidence of mortality was seen in the neoadjuvant chemotherapy group, which was 87.5% (7/8). According to Table 2, 85.71% (6/7) of the incidence of death in this group, occurred in the group without pathogenic mutations, compared to 14.28% (1/7) in the pathogenic BRCA1/2 tumor mutation group. In the neoadjuvant chemotherapy group, based on the bivariate Chi-square/Fisher’s Exact analysis, the likelihood of death was 3.79 times greater (crude RR 3.795; 95% CI; 1.56–9.234; p-value = 0.003), than the group without neoadjuvant chemotherapy. Based on multivariate analysis, Cox regression analysis, however, NACT did not significantly affect survival outcome (Table 3).
Based on Supplemental Table 2, the highest mortality incidence was also seen in the group without adjuvant chemotherapy, which was 75% (6/8). In that group, the likelihood of death was 14.02 times greater, significant statistically (crude RR 14.021; 95% CI: 4.612–42.626; p-value < 0.001), than receiving adjuvant chemotherapy group. Based on Cox-regression multivariate analysis, Table 4, the absence of adjuvant chemotherapy significantly increased the risk of death (adjusted RR 46.949; 95% CI: 11.114–198.340; p-value < 0.001).
The effect of pathogenic BRCA1/2 tumor mutation on survival outcome of high-grade serous epithelial ovarian cancer patients
The BRCA mutation status may have different survival effects, after undergoing different primary treatments, due to differences in disease patterns or clinical characteristics of HGSOC and differences in chemotherapy response. 30 The methods of proteomics, such as mass spectrometry and protein array analysis, have significantly improved the understanding of the molecular signaling processes and the characterization of ovarian cancer at the proteome level. An examination of the proteins in ovarian cancer and how they respond to treatment can reveal novel treatment options that may decrease the development of drug resistance and potentially enhance patient outcomes. 31
At the time of diagnosis, HGSOC patients with the BRCA1/2 mutation were reported to have a higher peritoneal tumor load, and increased proportion of lymph node enlargement significantly, compared with the wild-type BRCA gene group. 32 The retrospective study reported that nodular peritoneal disease was closely associated with BRCA mutation status, whereas mesenteric involvement and supra-diaphragmatic lymphadenopathy, were significantly associated with the wild-type BRCA gene. 33 The high response rate to platinum-based chemotherapy in the BRCA1/2 mutation group, likely to have a similar effect on the NACT-IDS and PDS groups, led to no difference in survival outcome, as shown in the results of this study.
Based on the survival outcome analysis in Table 3 and Supplemental Table 2, the pathogenic BRCA1/2 tumor mutation, significantly reduced the likelihood of death, up to 86% lower (adjusted RR 0.149; 95% CI: 0.046–0.475; p-value = 0.001), than without the pathogenic BRCA1/2 tumor mutation group. Based on multivariate analysis, between the pathogenic BRCA1/2 tumor mutations group, and without pathogenic BRCA1/2 tumor mutation, using Kaplan–Meier-Log Rank Test analysis, the pathogenic mutation group had a better median survival (median 46 months; 95% CI: 34.009–57.991; p-value = 0.001), when compared to the group without pathogenic mutations (median 23 months; 95% CI: 15.657–30.343; p-value = 0.001). Administration of NACT did not affect survival in this study.
In this study, in addition to the pathogenic BRCA1/2 tumor mutation status, another variable that influenced overall survival outcome was the parity group >1 (adjusted RR 0.371; 95% CI: 0.168–0.823; p-value = 0.015), the preoperative CA 125 level group ⩾500 mIU/mL (adjusted RR 3.189; 95% CI: 1.091–9.324; p-value = 0.034) the positive residual tumor lesion group (adjusted RR 6.989; 95% CI: 2.523–19.357; p-value < 0.001), no adjuvant chemotherapy was given (adjusted RR 46.949; 95% CI: 11.114–198.340; p-value < 0.001).
The role of BRCA1/2 gene structure and function on ovarian carcinogenesis
Currently, several BRCA1 and BRCA2 interactor proteins have been identified. RAD51 plays a role in the process of improving DSBs, the most important part of the HR process. Its function is critical to the completeness of the protein encoded by the two genes BRCA1/2. 34 Some studies have successfully described the role of BRCA2 in the regulation of intracellular transport, enzymatic activity, and function of RAD51. 35 BRCA1 exhibits physical interactions with RAD51, forming the complex responsible for resection of single-stranded DNA, at the site of the double-strand breakage. DNA damage will be followed by extensive phosphorylation of histone H2AX, and form a focus at the site of damage. BRCA1 was recruited into the foci, before involving other factors, such as RAD51. This explains that H2AX and BRCA1 initiate the DNA repair mechanism, by modifying the local chromatin structure, allowing DNA repair proteins to reach the site of damage. BRCA1 and BRCA2 also function as transcriptional co-regulators and chromatin remodeling functions. BRCA1 has the ability to co-activate p53-dependent endogenous p21 stimulation. 36
Platinum containing chemotherapy as promising therapeutic response for BRCA1/2 mutation’s ovarian cancer group
Although BRCA mutation-associated ovarian cancers appeared to be more aggressive, compared to sporadic ovarian cancers, the group showed higher sensitivity to platinum agent and other DNA-damaging regimens. 37 The majority of women with ovarian cancer are typically discovered at advanced stages, and to recurrency is common after initial platinum-based chemotherapy. This recurrence often leads to an incurable condition with limited therapeutic options. 38 Stronach et al. 39 discovered that DNA-PKcs selectively phosphorylated nuclear AKT at Ser473 in platinum-resistant OCCC and HGSOC cells, while not affecting platinum-sensitive cells.
The PI3K pathway is commonly upregulated in epithelial ovarian tumors and has a significant impact on chemoresistance and the maintenance of genomic integrity. This pathway is involved in various aspects of DNA replication and cell cycle regulation. The inhibition of the PI3K may lead to genomic instability and mitotic catastrophe through a decrease of the activity of the spindle assembly checkpoint protein Aurora kinase B and consequently increase of the occurrence of lagging chromosomes during prometaphase. 40
Platinum agent intervention occurs in the DNA cross-links process, causing double-stranded DNA helical damage, which cannot be repaired due to disruption of HR repair mechanisms. Several studies have shown an increased long-term survival rate in women with BRCA mutation-associated ovarian cancer, with platinum agent chemotherapy, compared to the sporadic group. 41 Intraperitoneal Cisplatin chemotherapy has shown good long-term outcomes, in ovarian cancer associated with the BRCA1/2 mutation. 42
Research limitations
This study has several limitations. First, selection bias and other issues can arise regarding the design of historical cohort studies. Second, the monitoring period was short due to the limited research period and limited resource. Third, in this study, there was no assessment of therapeutic response, and evaluation of the primary disease-free interval, either in the BRCA1/2 tumor pathogenic mutation group, or in the no mutation group, due to the lack of complete postadjuvant monitoring data (periodic clinical, imaging, and tumor marker CA-125 level monitoring). Based on this study, evaluation of the completeness of medical records is an important factor for the sustainability of the research, especially in teaching hospitals. Fourth, the number of samples of the pathogenic BRCA1/2 tumor mutation group was limited, because not the entire list of target populations could be included as the study sample. A total of 39.58% of the target population did not meet the re-selection of the inclusion criteria by pathologists, due to a mismatch of histopathological types, and low tumor cell content. Suboptimal FFPE sample quality, low DNA integrity index (DIN), was recorded in 21.84% of research sample candidates, due to high DNA fragmentation (<120–150 bp), so it did not pass the NGS quality control. Both of these caused the limited number of samples in several variables of clinical characteristics (history of breast cancer, neoadjuvant chemotherapy administration, BRCA gene domain mutation), correlation analysis could not be assessed.
Strength of the study
In addition to the limitations of the study, the strengths of this study include a more specific study population, only involving high-grade serous epithelial ovarian cancer’s patients. The selection of inclusion criteria for all samples was carried out directly by single expert pathologist so that interobserver variations could be avoided. The examination of pathogenic BRCA1/2 tumor mutations was carried out in an EMQN (European Molecular Genetics Quality Network) certified laboratory. The control of survival bias was carried out by ensuring that the factors thought to influence survival were included in the statistical analysis. Research methodologies are clearly defined, produce highly useful accurate data, applicable in clinical practice.
Conclusion
This study demonstrates that HGSOC patients with pathogenic BRCA1/2 tumor mutations have significantly better survival outcomes compared to those without these mutations. Patients carrying BRCA1/2 mutations exhibited a significantly lower risk of death and a longer median survival time. Multivariate analysis confirmed that the presence of BRCA1/2 mutations serves as an independent positive prognostic factor for overall survival. These findings suggest that identifying BRCA1/2 mutations in HGSOC patients can guide more personalized treatment strategies and improve survival outcomes.
Supplemental Material
sj-docx-1-smo-10.1177_20503121241299849 – Supplemental material for The impact of pathogenic BRCA1/2 tumor mutation status on high grade serous epithelial ovarian cancer survival outcome: A multicenter study from Indonesia
Supplemental material, sj-docx-1-smo-10.1177_20503121241299849 for The impact of pathogenic BRCA1/2 tumor mutation status on high grade serous epithelial ovarian cancer survival outcome: A multicenter study from Indonesia by Sutrisno Sutrisno, Dina Marlina, Kevin Dominique Tjandraprawira and Putri Nadhira Adinda Adriansyah in SAGE Open Medicine
Footnotes
Acknowledgements
None.
Author contributions
SS was involved in the conception, design, analysis, interpretation of the data, and drafting of the article. DM and KDT collected and analyzed the data and assisted in drafting the article. SS and DM collected and analyzed the data. PNAA and KDT developed the conception design, analyzed and interpreted the data, and revised the article critically for intellectual content.
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request. The data are not publicly available due to privacy or ethical restrictions
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
The research was carried out after obtaining an Ethical Review from Komisi Etik Penelitian Kesehatan (KEPK) Medical Faculty of Indonesia University/Dr. Cipto Mangunkusumo National Central Referral Hospital, No.: KET-198/UN2.F1/ETIK/PPM.00.02/2020, February 24th, 2020; and No. ND-291/UN2.F1/ETIK/PPM.00.02/2021; Ethical Review of KEPK Persahabatan Central Referral Hospital, No. 106/KEPK-RSUPP/10/2020, October 1st, 2020; and Research Approval Letter of MRCCC Central Referral Hospital Siloam Jakarta, No. 735/SS/Dir/V/2021.
Informed consent
Written informed consent was obtained from the patient for publication and any accompanying images. All patients have signed informed consent to publish their medical information and/or images in the manuscript.
Trial registration
Not applicable.
Preprint disclaimer
This article is a preprint and has not been peer-reviewed. The authors haven’t submitted this article on any preprint servers. It reports new research that has yet to be evaluated by the scientific community and should not be regarded as conclusive or used to guide clinical practice. The findings and conclusions may be updated or refined based on further review. The published version from SAGE Open Medicine will available upon acceptance of the journal.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

