Abstract
A cell line was established from whole 6–8-week-old central bearded dragon (Pogona vitticeps) embryos. Cells were mid-sized and showed an elongated and polymorphic form. The cell line grew in a monolayer and has been serially passaged for 17 passages at time of publication. This cell line has been used with samples from adenovirus polymerase chain reaction (PCR)-positive bearded dragons, and 2 virus isolates have been obtained so far. The isolates show a clear cytopathic effect in inoculated cells. Both virus isolates have been serially passaged on this cell line, and have been identified by PCR amplification and sequencing of a portion of the DNA-dependent DNA polymerase gene and show 100% nucleotide identity to the corresponding region of an agamid adenovirus. Electron microscopic examination of supernatant from infected cells demonstrated the presence of nonenveloped particles, with a diameter of approximately 80 nm in both virus isolates.
Introduction
Adenoviruses (AdVs) are nonenveloped with a diameter of 60–90 nm and a double-stranded DNA genome. They have been detected in every major class of vertebrate. In reptiles, AdV infections are very commonly detected in agamid lizards, particularly central bearded dragons (Pogona vitticeps), which are one of the most popular reptiles in the pet trade. Adenoviruses have been described in bearded dragons of different ages from neonatal to adult lizards9,13,14,18 in many different European countries, the United States, and Australia.8,11,13,14,20,22,25
The most common method used for the detection of AdVs in central bearded dragons is a polymerase chain reaction (PCR) targeting a portion of the DNA-dependent DNA polymerase gene. 25 Partial characterization of AdVs from central bearded dragons has shown that the majority of AdVs found in this species are closely related to one another and are all members of the genus Atadenovirus.20,21,25 This virus is therefore known as Agamid adenovirus 1. 25 Clinical signs associated with AdV infections in bearded dragons include weakness, anorexia, lethargy, depression, as well as central nervous signs, including circling and head tilt. 16 Adenovirus infection has also been detected in these animals without any clinical signs. 14 Histological changes described in infected bearded dragons include basophilic and eosinophilic intranuclear inclusions, which have been documented in enterocytes, hepatocytes,11,13 epithelial cells of the bile ducts, renal tubules, pancreatic acini, and oral mucous membranes.18,24
To date, no AdVs have been isolated from bearded dragons in cell culture, although infections with these viruses are common in this species. There is only 1 report available on the isolation of an AdV from a lizard in cell culture. In that report, 2 closely related AdVs were isolated from helodermatid lizards (Heloderma horridum and Heloderma suspectum) in a permanent cell line from iguanas (iguana heart [IgH2] cells). 20 From snakes, however, there have been several reports of successful isolations of AdVs in cell cultures. Snake adenovirus 1 was originally isolated on IgH2 cells from the internal organs of a red corn snake (Pantherophis guttatus) with clinical signs of pneumonia. 12 The red corn snake isolate was later randomly cloned and completely sequenced,5,6 and thus serves as a prototype for reptilian AdVs. An identical virus was isolated from a boa constrictor (Boa constrictor) with inclusion body disease. In that case, the virus was isolated in viper heart (VH2) cells from internal organs and blood cells (Marschang RE, Mischling M, Benkő M, et al.: 2003, Evidence for widespread atadenovirus infection among snakes. In: Virus persistence and evolution. Proceedings of the 6th International Congress of Veterinary Virology, ed. Jestin A, Clement G, p. 152. ZOO-POLE development–ISPAIA, Ploufragan, France). Additional AdVs have been isolated from a boa constrictor with hepatic necrosis and basophilic intranuclear inclusions in the liver and small intestine, 10 as well as from a royal python (Python regius). 19 A genetically distinct AdV was detected by PCR in colubrid snakes in the United States. 7 This virus was named Snake adenovirus 2 and was later isolated in VH2 cells from an intestine sample of a red corn snake, which died with clinical signs of dyspnea and vomitus. 1 However, none of the cell lines used previously for the isolation of adenoviruses in reptiles have been permissive for the growth of agamid AdVs. It was hypothesized that agamid AdVs could be isolated on a competent cell line from bearded dragons. The current study describes the establishment of a bearded dragon cell line and the successful isolation of Agamid adenovirus 1 on these cells.
Materials and methods
Cell culture and medium
Fertilized bearded dragon eggs were obtained from a private breeder. The eggs were incubated for 6–8 weeks at 28°C and 75–85% humidity. The cells were prepared from a pool of 12 eviscerated 6–8-week-old central bearded dragon embryos (BDEs) as described previously for chicken embryo fibroblasts.
23
As a growth medium for these cells, Dulbecco modified Eagle medium (DMEM) was used (13.4 g/l of DMEM powdered medium with 4.5 g/l of
The bearded dragon cells were passaged after reaching confluency, approximately 2 days after seeding. For subculturing, the cells were detached with 0.05% trypsin–versene (136 mM NaCl, 3 mM KCl, 1 mM KH2PO4, 6 mM Na2HPO4 × 12 H2O, 0.4 mM MgSO4 × 7 H2O, 0.9 mM CaCl2 × 2 H2O, b trypsin dry substance [1:250], d 3 mM versene, b 37,900 U/l streptomycin sulfate, a and 100,000 U/l penicillin G a dissolved in double-distilled water, adjusted to pH 7.0 with 1 N NaOH, and sterile filtered). The cells were first passaged into 25-cm2 tissue culture flasks e and later into 75-cm2 tissue culture flasks. Afterward, the cells were passaged every week, at a split ratio of 1:2. The density of cells was calculated with a hemocytometer as described previously. 15
Samples and PCR
In the first case, oral and cloacal swabs were taken from a captive-bred bearded dragon with clinical signs of egg binding. In the second case, a mixed oral and cloacal swab was taken from a captive-bred animal demonstrating central nervous system signs consisting of tremor and circling. DNA was extracted from swabs from each animal using a commercial kit f following the instructions of the manufacturer. A nested PCR for the detection of AdVs was performed as described previously. 25 All PCR products were separated on 1.5% agarose gels g containing 0.5 μg of ethidium bromide in Tris–acetate–ethylenediamine tetra-acetic acid, and visualized under 320-nm ultraviolet light. The PCR amplicons of approximately 320 bp were purified with a commercial extraction kit h and sequenced by a commercial company. i The obtained sequences were compared to the data in GenBank (http://www.ncbi.nih.gov) using the BLASTN option. The original samples from PCR products, which demonstrated 100% nucleotide identity to the Agamid adenovirus 1 (accession no. DQ077706), were inoculated onto the bearded dragon cell line for virus isolation. Swabs from both animals were also tested by PCR for the presence of invertebrate iridoviruses, 24 ranaviruses, 17 and paramyxoviruses. 2
Isolation of the viruses
The swabs were sonicated in 3 ml of DMEM supplemented with antibiotics. The samples were centrifuged at low speed (2,000 × g, 10 min) for the removal of cell debris and bacteria. Afterward, 200 µl of the sample homogenate was inoculated onto approximately 70% confluent cell monolayers in 30-mm-diameter tissue culture dishes. The dishes were then incubated for 2 hr at 28°C after which 2 ml of nutrient medium (DMEM supplemented with 2% FCS, 1% NEA, and antibiotics) was added to each dish.
Cells were examined for cytopathic effects (CPEs) every 3 days with an inverted light microscope. Extensive CPE was seen 7–10 days after inoculation, and the dishes were frozen at −80°C. Dishes showing no CPE were frozen after 2 weeks of incubation for blind passaging. Additional passages were performed after a single freeze (–80°C)-thaw cycle and low speed centrifugation.
The dishes demonstrating CPE were also tested by PCR (as described above) for the presence of AdV. For this, DNA was extracted from 200 μl of the cell culture supernatant. Both virus isolates were quantified by serial 10-fold dilution and inoculation onto BDE cells. Titers were calculated according to a previously described method. 4
Electron microscopy
Cell culture supernatants from both isolates were negatively stained with 2% potassium phosphotungstate at pH 7.3 on 3.05-mm copper grids. j Both isolates were examined with a transmission electron microscope k for the presence of viral particles.
Results
A cell line was established from the BDEs. The established cells grow well at 28ºC, are mid-sized, and have a polymorphic and elongated form (Fig. 1). The cells form a monolayer 2 days after seeding and can be subcultured at a split ratio of 1:2 every 5–7 days. Seeding density of the cells after the third passage was 2.66 × 106 cells/ml. The cells have been carried in continuous culture for 17 passages at time of publication. The morphological characteristics of the cells remained consistent in all 17 passages as well as during the 2-week incubation between subcultures.
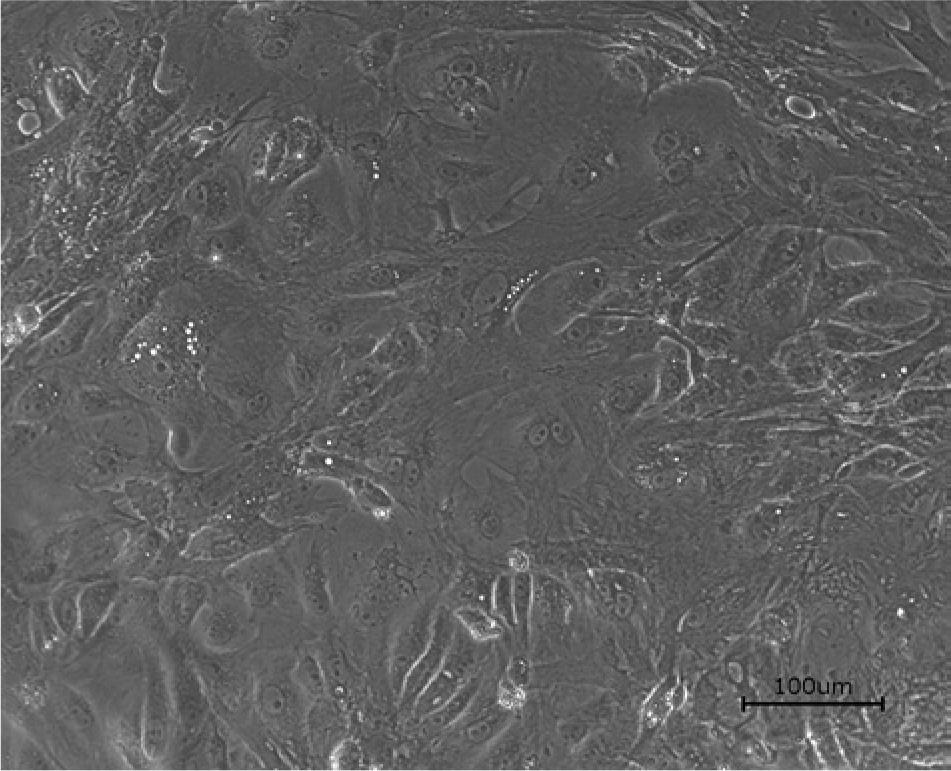
Noninfected bearded dragon embryo cells, second passage. Bar = 100 μm.
The nested PCR for the detection of AdVs gave positive results with the swab samples from both animals. In the first case, AdV was detected in the oral swab, in the second case in the mixed oral and cloacal swab. These samples were therefore inoculated onto the BDE cells. The PCR assays for the detection of invertebrate iridoviruses, ranaviruses, and paramyxoviruses in these samples were negative. Both isolated viruses demonstrate a CPE with rounding and detachment of cells (Fig. 2). The isolates were identified as AdVs by PCR and sequencing. The sequencing results were identical before and after isolation. The partial DNA-dependent DNA polymerase gene sequences obtained from both isolates were 272 nucleotides long and were identical to one another and to the corresponding part of the Agamid adenovirus 1 genome (DQ077706). Both isolates have been serially passaged on the established BDE cells. Seven additional passages of each virus isolate have been performed to date, following freeze–thaw cycles and low speed centrifugation. No difference was observed in adenovirus replication between different cell passages. DNA from the seventh passage of each isolate was also prepared, and PCR and sequencing carried out as described. Sequencing results remained identical to previous results. The virus titer of the highest passages was 104 and 103.5 mean tissue culture infectious dosage (TCID50). Both isolates were also identified as AdVs by electron microscopic examination of the cell culture supernatants. Nonenveloped particles, with a diameter of approximately 80 nm, were detected by negative staining electron microscopy in both cases (Fig. 3).
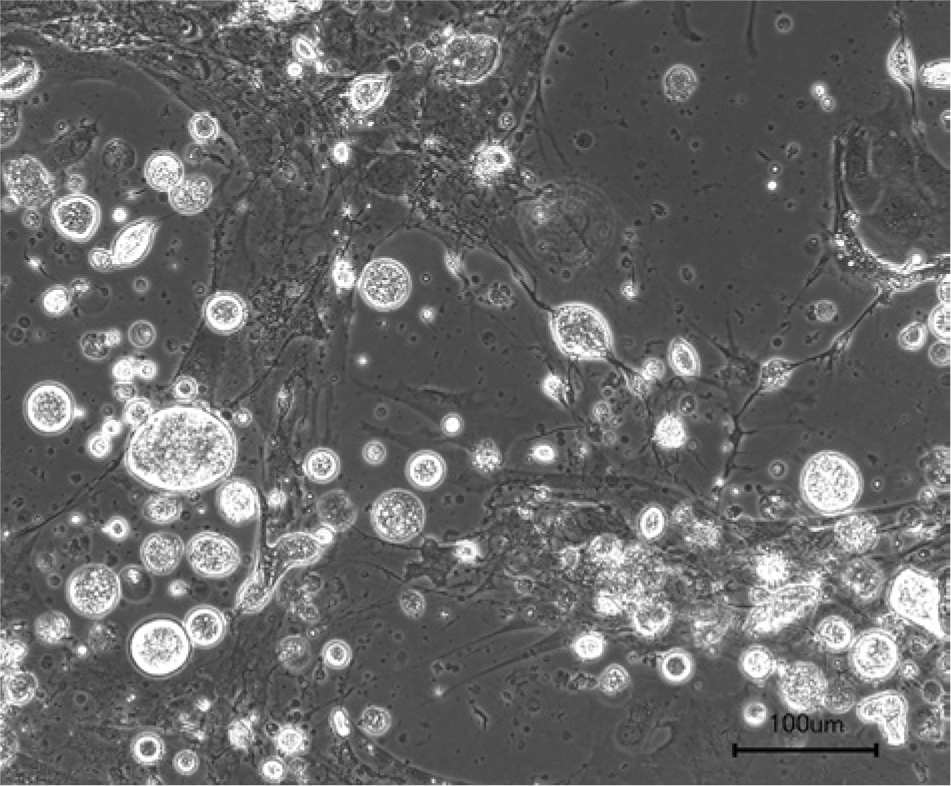
Adenovirus isolate from a central bearded dragon (Pogona vitticeps). Cytopathic effect in bearded dragon embryo cells, 7 days after inoculation, sixth passage of an isolate. Bar = 100 μm.
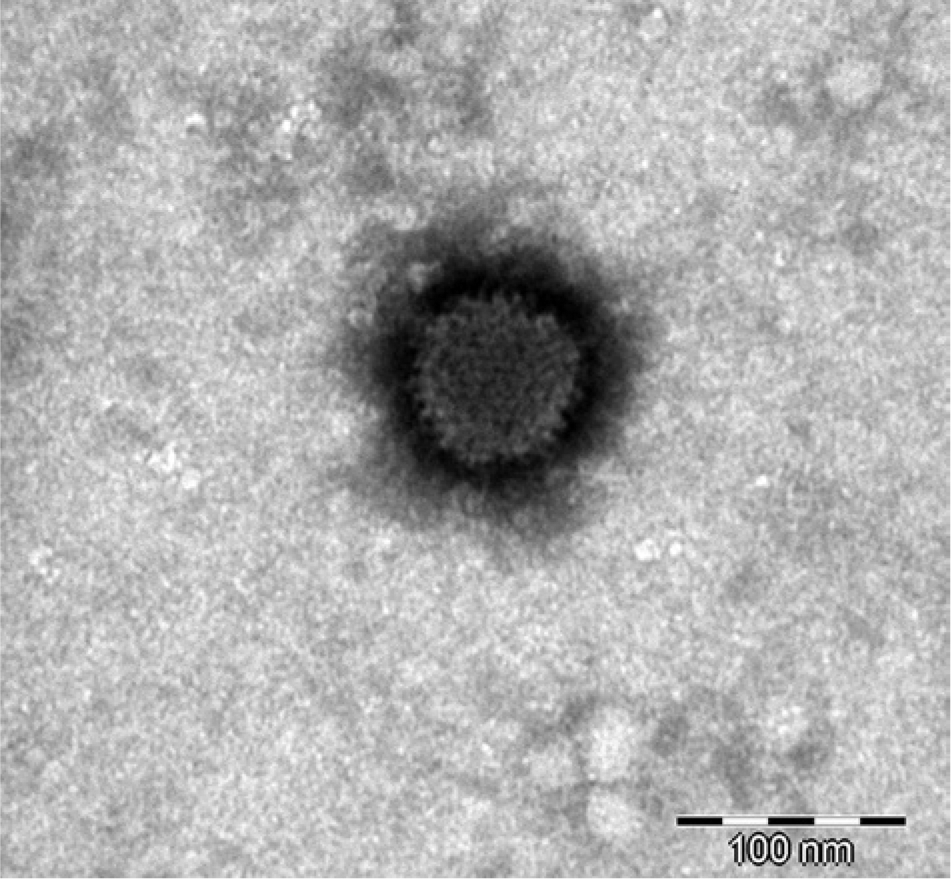
Electron micrograph of icosahedral nonenveloped virus particles in cell culture supernatant from one of the central bearded dragon (Pogona vitticeps) isolates (fifth passage on bearded dragon embryo cells). Bar = 100 nm.
Discussion
Although AdV infections are commonly described in bearded dragons,11,13,18,20,25 previous attempts to isolate the virus in cell culture have been unsuccessful. 20 Agamid adenovirus 1 seems to be very species specific because the virus has only been detected in bearded dragons to date. 21 Based on this fact, it seemed likely that the virus might grow on bearded dragon–specific cells, although it did not grow on other reptilian cell lines tried previously. According to this hypothesis, BDEs were used to create a bearded dragon fibroblast cell line. This cell line provides a tool for the isolation of bearded dragon–specific viruses and was successfully used to isolate AdVs from 2 animals. The isolation of these viruses is important for the study of the biological properties of the virus. Because AdV infection has been described in central bearded dragons with 16 and without clinical signs, 14 the primary pathogenic role of Agamid adenovirus 1 has been questioned in many cases. Isolation of this virus opens up the possibility to carry out experimental transmission studies to help understand how this virus affects its host and what factors might influence the course of infection.
Virus isolation also opens up possibilities for the study of ultrastructural properties of this virus. To date, studies have shown clear differences in the number of fibers present on the surfaces of different atadenoviruses (Pénzes J, Menéndez-Conejero R, Ball I, et al.: The first genome sequencing and analysis of novel lizard adenovirus, unpublished data). The fiber, hexon, and penton proteins that make up the icosahedral capsids of AdVs form the main antigenic components of these viruses. 3 Only partial sequence information from the DNA-dependent DNA polymerase gene and the hexon genes are currently available from Agamid adenovirus 1 (http://www.vmri.hu/~harrach/ADENOSEQ.HTM). Further sequence data is necessary to understand the structural differences between various atadenoviruses, and ultrastructural and biological studies will help elucidate the importance of these differences for AdVs.
The isolated viruses can also be used for the development of serological tests. Neutralization tests have previously been used for the detection of AdV neutralizing antibodies in serum of infected snakes (Funk RS, Marschang RE, Romanova I, et al.: 2011, Survey of native Arizona rattlesnakes for viral infections, p. 31. In: Proceedings of the Biology of the Rattlesnakes symposium, July 20–23, 2012, Tucson, Arizona; Marschang RE, Mischling M, Benkő M, et al.: 2003, Evidence for widespread atadenovirus infection among snakes), but studies on antibodies against AdVs in lizards have been hindered by the lack of isolates available. The isolation of Agamid adenovirus 1 will allow the performance of serological tests, which are necessary to understand how common Agamid adenovirus infections are among both captive and wild bearded dragons and whether this virus might be able to spread to other reptile species. The isolates can also be used to study serological cross-reactivity between AdV isolates from various reptiles.
Footnotes
Acknowledgements
The authors would like to thank Silvia Speck for her excellent technical assistance, and Anke C. Stöhr and Mike Bauer for providing the fertilized bearded dragon eggs.
a.
Biochrom AG, Berlin, Germany.
b.
Titriplex III, Merck KGaA, Darmstadt, Germany.
c.
Cellstar, Greiner Bio-One GmbH, Frickenhausen, Germany.
d.
Serva Electrophoresis GmbH, Heidelberg, Germany.
e.
BD Biosciences, San Jose, CA.
f.
DNeasy, Qiagen GmbH, Hilden, Germany.
g.
Bioenzym, Oldendorf, Germany.
h.
peqGOLD, PEQLAB Biotechnology GmbH, Erlangen, Germany.
i.
Eurofins MWG GmbH, Ebersberg, Germany.
j.
Plano GmbH, Wetzlar, Germany.
k.
JEM-1011, JEOL Ltd, Tokyo, Japan.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by a grant from the Morris Animal Foundation.
