Abstract
Lead toxicity due to ingestion of spent ammunition is an ongoing cause of mortality in bald eagles. While gross and histologic lesions of lead intoxication have been described in a few individuals of this species, the prevalence of lesions is underreported. A retrospective study of 93 bald eagles with severe lead intoxication was performed to describe the associated lesions and their prevalence and to compare the lesions with blood, liver, kidney, and/or bone lead concentrations. Gross lesions associated with lead toxicity were most frequent within the heart (51/93 birds) and consisted of multifocal myocardial pallor and rounding of the apex. Within the brain, gross lesions included petechiae or hemorrhagic necrosis (13/93 birds). Histologic lesions compatible with lead toxicity occurred within the heart (76/93 birds), brain (59/93 birds), and eyes (24/87 birds). Lead toxicity in bald eagles is characterized by fibrinoid necrosis of small- to medium-caliber arteries, most commonly affecting the heart, brain, and eyes. Gross and histologic lesions are consistent with ischemia caused by a primary vascular injury. A blood lead concentration of greater than 4 ppm and markedly elevated liver lead concentrations were associated with a greater likelihood of lesions in the heart. Severe lead intoxication is frequently associated with lesions that are histologically detectable in bald eagles. The presence of fibrinoid arterial necrosis and parenchymal degeneration, necrosis, and/or hemorrhage within the heart, brain, and/or eyes is suggestive of lead toxicity in bald eagles and warrants evaluation of liver or bone lead concentrations.
Keywords
Lead toxicity in bald eagles (Haliaeetus leucocephalus) is primarily associated with scavenging of carcasses or offal from animals that have been killed with lead-based ammunition. 5,22,25,26,32,36,37 Lead-intoxicated bald eagles may demonstrate neurologic, ocular, gastrointestinal, and/or respiratory clinical signs or may be found deceased. 8,10,14,20,25 A review of eagles submitted to the National Wildlife Health Center between 1975 and 2013 found trauma and poisonings to be the most frequent causes of death of bald eagles, with lead intoxication accounting for 63.5% of diagnosed poisonings. 33
While elevated lead concentrations in blood, liver, kidney, and/or bone have been frequently reported in bald eagles,* gross and histologic lesions in bald eagles with lead toxicity are mostly limited to small numbers of animals, 8,23,25,30 with only 1 previous publication of a large series. 11 Blood lead concentrations of ≥1 ppm are considered evidence of severe incurable lead poisoning in bald eagles. 36
Gross and histologic lesions in bald eagles with lead toxicity have been described in the heart, gastrointestinal tract, and infrequently within the brain, eye, and kidney, with an underlying vascular insult suspected. 6,8,23,25,30 In 5 bald eagles with experimentally induced lead toxicity, medial fibrinoid necrosis of small arterioles was occasionally observed within the ventriculus, proventriculus, liver, spleen, heart, and kidney. 30 In previous reports of lead toxicity in bald eagles, there has been sporadic documentation of areas of pallor in the heart corresponding histologically to cardiomyopathy, myocardial degeneration, and/or fibrosis. 11,23 Lesions within the brain of bald eagles with lead toxicity have only been infrequently reported and are grossly characterized by red to brown discoloration or red pinpoint foci. 6,23
The purpose of this retrospective study was to describe gross and histopathologic lesions associated with severe lead toxicity (≥1 ppm blood lead concentration), including their distribution and prevalence of lesions in a large series of bald eagles. Blood, liver, kidney, and/or bone lead concentrations were analyzed to determine if these were correlated with the presence and distribution of lesions and if blood lead concentrations were correlated with lead concentrations in the liver, kidney, or bone.
Materials and Methods
All bald eagles with blood lead concentrations of 1.0 ppm or greater on admission to The Raptor Center between December 2004 and December 2015 were selected retrospectively. All birds underwent necropsy at the University of Minnesota Veterinary Diagnostic Laboratory after they died spontaneously or were euthanized. Cases were excluded if heart or brain was not examined histologically.
Clinical data were included when available. Bald eagles were classified as immature (≤2 years), after second year, or adult (>4–5 years). Birds were classified as female, male, or unknown sex. Anemia was defined as a packed cell volume (PCV) of 0.35 or less. Additional information collected included whether the bird was euthanized, if and how long birds were treated with ethylenediaminetetraacetic acid (EDTA), or dimercaptosuccinic acid (DMSA) chelation therapy, and radiographically visible metal fragments (interpreted as likely lead) in the gastrointestinal tract.
Complete necropsies were performed or supervised by 1 of 2 board-certified veterinary pathologists (A.W. and A.G.A.). Body condition at necropsy was designated as overweight, good, underweight, emaciated, or not documented. Metal fragments within the gastrointestinal tract were documented when present. At a minimum, heart and brain were examined histologically for all birds included in this study. However, additional tissues were frequently examined histologically. The number of histologic sections examined for each case varied.
Blood lead analysis and tissue mineral element and heavy metal analysis were performed at the University of Minnesota Veterinary Diagnostic Laboratory (UM VDL) for samples originating from December 2004 through April 2009. All blood lead analyses after April 2009 were conducted at a commercial laboratory (Marshfield Laboratories, Marshfield, WI). Tissue mineral element and heavy metal analysis were performed at Michigan State University Veterinary Diagnostic Laboratory (MSU Veterinary Diagnostic Laboratory) for samples from May 2009 through 2015. All tissue mineral concentrations were measured by either inductively coupled plasma (ICP) atomic emission spectrometry (AES) or mass spectrometry (MS).
At the UM VDL, tissue mineral element and heavy metal analysis was conducted using a Varian Liberty 150 ICP AES (Varian, Palo Alto, CA) equipped with a Varian autosampler/diluter. Each quantitative analysis used a set of National Institute of Standards (NIST) standard reference material (SRM) for matrix-matched quality control. This included bovine liver SRM 1577b and oyster tissue SRM 1566b. Appropriate blanks and standards were used. All procedures were carried out in accordance with Varian’s operation manuals and training. The limit of quantitation (LOQ) for lead (Pb) was 500 ppb. For blood lead analysis, samples were evaluated on a Varian Spectra AA330 Atomic Absorption Spectrometer fitted with a graphite furnace and Varian auto sampler. Quality control was performed by using interlaboratory blood lead samples as supplied monthly by the University of Wisconsin. The LOQ was 10 ppb.
At Marshfield Laboratories, blood lead samples were evaluated using furnace/atomic absorption technology with a ThermoFisher M6 AA/GFS-35Z graphite furnace analyzer. The LOQ was 10 ppb.
At the MSU Veterinary Diagnostic Laboratory, tissue mineral element and heavy metal concentrations were determined on an Agilent 7500ce ICP-MS (Agilent Technologies, Santa Clara, CA) equipped with a Cetac Auto Sampler (Cetac, Omaha, NE) and MicroMist Nebulizer (Agilent). The instrument was calibrated with appropriate dilutions of Specpure 1000-μg/ml stock solutions (Alfa Aesar, Ward Hill, MA) of each element of interest according to in-house generated standard operating procedures derived from the Agilent Operator’s Manual (Agilent Technologies 2004). Each quantitative analysis used NIST bovine liver SRM 1577C and mussel tissue SRM 2976 for matrix-matched quality control. Appropriate blanks and standards were used. The LOQ for lead was 200 ppb.
All demographic, clinical, and laboratory data were entered into a spreadsheet (Excel; Microsoft, Redmond, WA). Data cleaning and recoding of variables were done in the spreadsheet. For the purpose of statistical analysis, blood lead concentrations greater than 4 ppm were recoded as “1” while those below (1 to <4 ppm) were recoded as “0.” Data analysis was done with a statistical software package (Statistix 10.0). This included summarizing descriptive continuous data by mean, median, and range and categorical data as frequencies. A 2-sample t test was done to compare mean lead tissue (liver, kidney, and bone) concentrations by impact of chelation (chelation therapy = yes or no). To examine the relationship of blood and tissue (ie, kidney, liver, and bone) lead concentrations, Pearson correlation coefficients were calculated. To further examine the linear relationship between these lead concentrations, linear regression and adjusted correlation coefficients (R2) were derived. Lead concentrations in blood and tissues (liver, kidney, and bone) were compared to the presence or absence of histologic lesions in the brain, heart, and eye using the χ2 tests. To determine the strongest predictor of histologic lesions, logistic regression was performed using the following predictor variables: liver, kidney, bone, and blood lead concentrations. In a separate analysis, chelation therapy was factored in.
Results
Ninety-three bald eagles were included in the study (Table 1). Chelation treatment was attempted for 43 of 93 birds (46.2%). Chelation attempts ranged from 1 to 15 days (mean of 3.9 days and median of 2 days). Of the 93 cases, 79 were euthanized by intravenous or in very exceptional cases by intracardiac injection of barbiturates under anesthesia, in accordance with the euthanasia guidelines published by the American Veterinary Medical Association. 24 Most of the birds were euthanized “in extremis” due to clinical signs associated with lead toxicity such as lethargy and dyspnea (open-mouth breathing). All birds treated with chelation either died or were euthanized due to deterioration of clinical signs or a continued lack of response to treatment.
Demographics and Clinical Findings of Bald Eagles With Lead Toxicity.
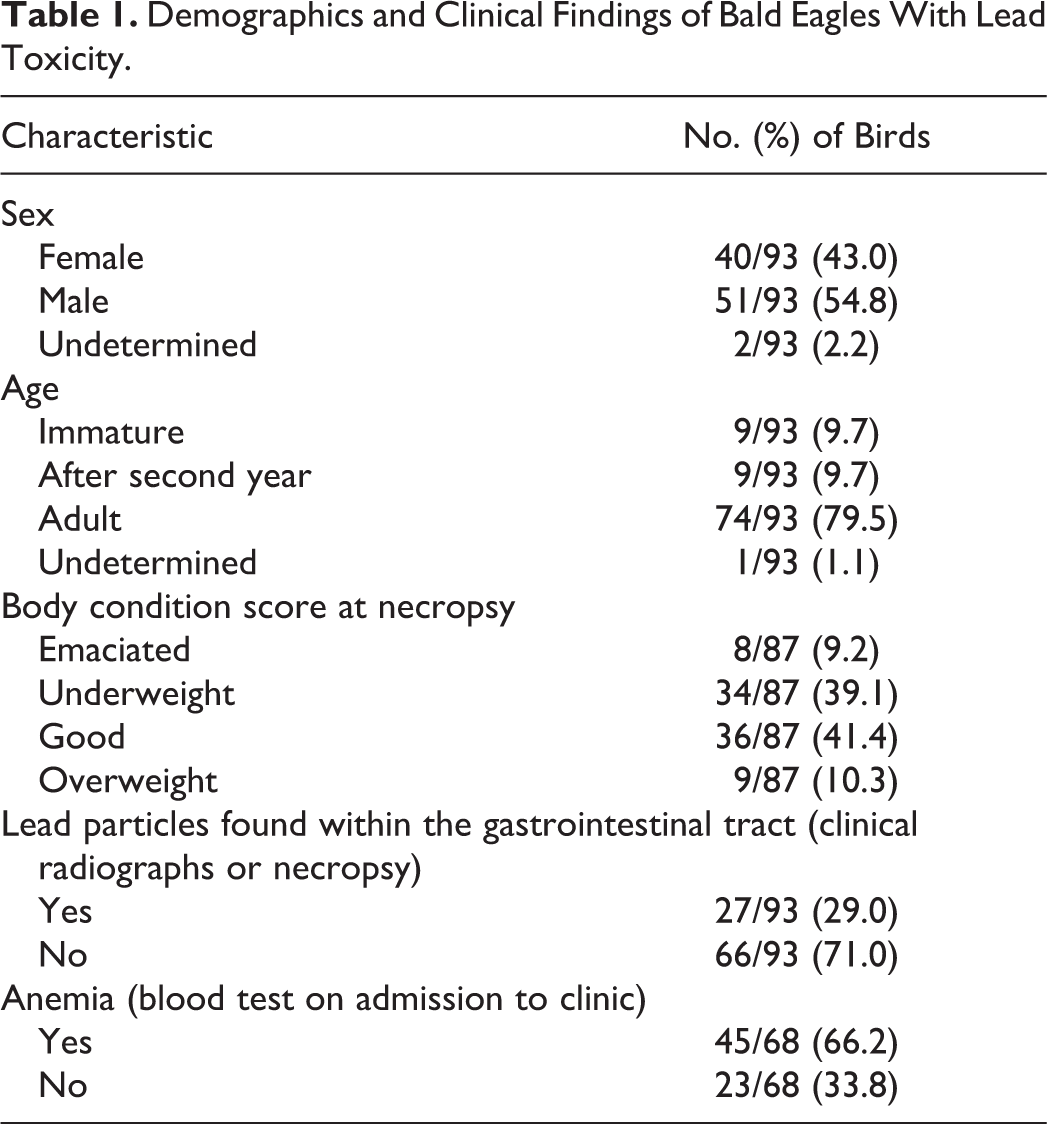
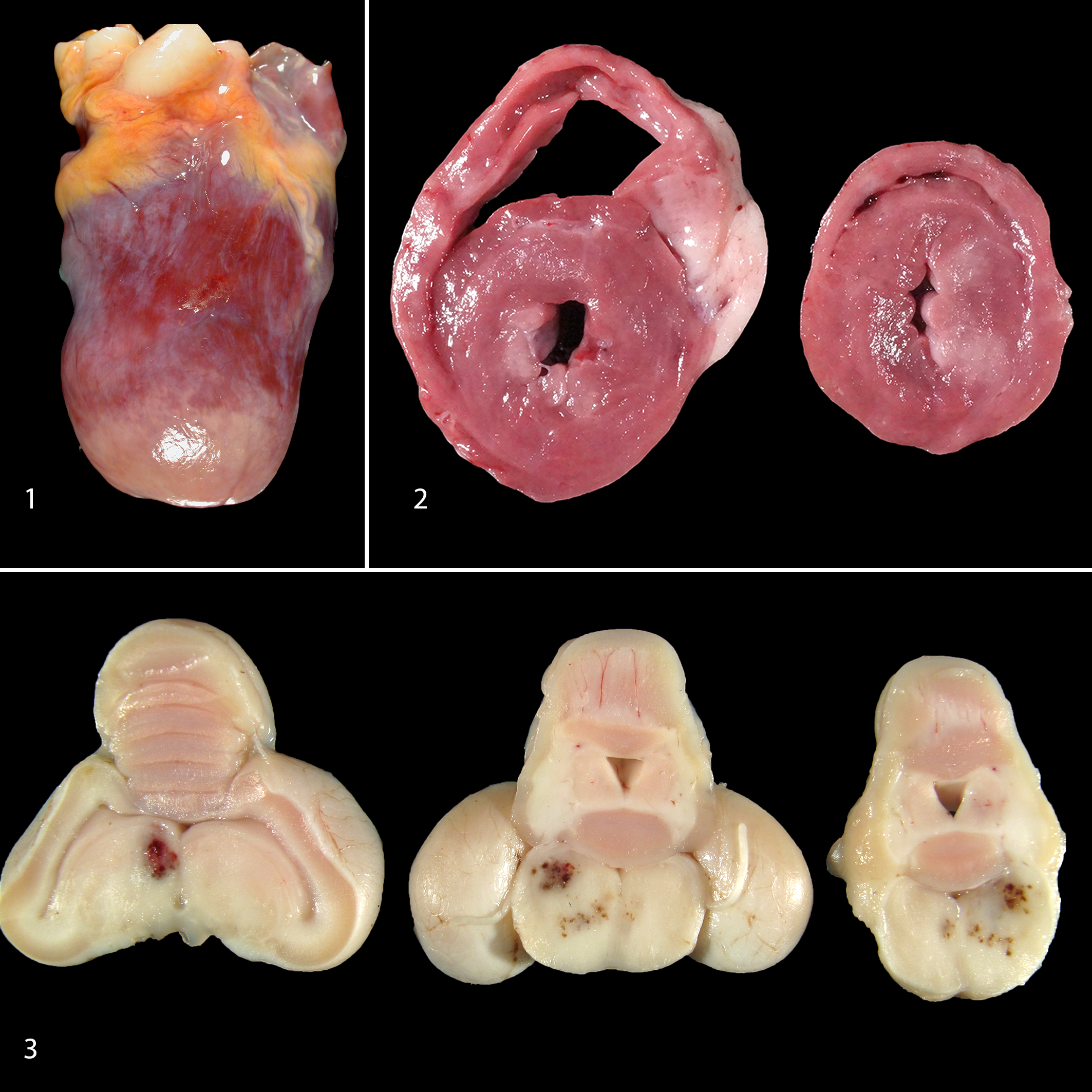
Gross lesions compatible with lead toxicity were present in 59 of 93 birds. Gross lesions were most frequently observed within the heart (Table 2). The most common cardiac gross abnormality was multifocal, pale tan areas within the myocardium (Fig. 1). Often visible from the epicardial surface, areas of pallor extended into the underlying myocardium on cut surface (Fig. 2). Affected areas often included the papillary muscles but were otherwise randomly distributed throughout the myocardium of the left ventricle and less frequently the right ventricle. Less commonly, the heart was enlarged, with rounding of the apex associated with left ventricular dilation. A small number of eagles had an increased amount of watery, brown to red tinged, translucent fluid within the pericardial sac (hydropericardium).
Gross Lesions of Bald Eagles With Lead Toxicity.

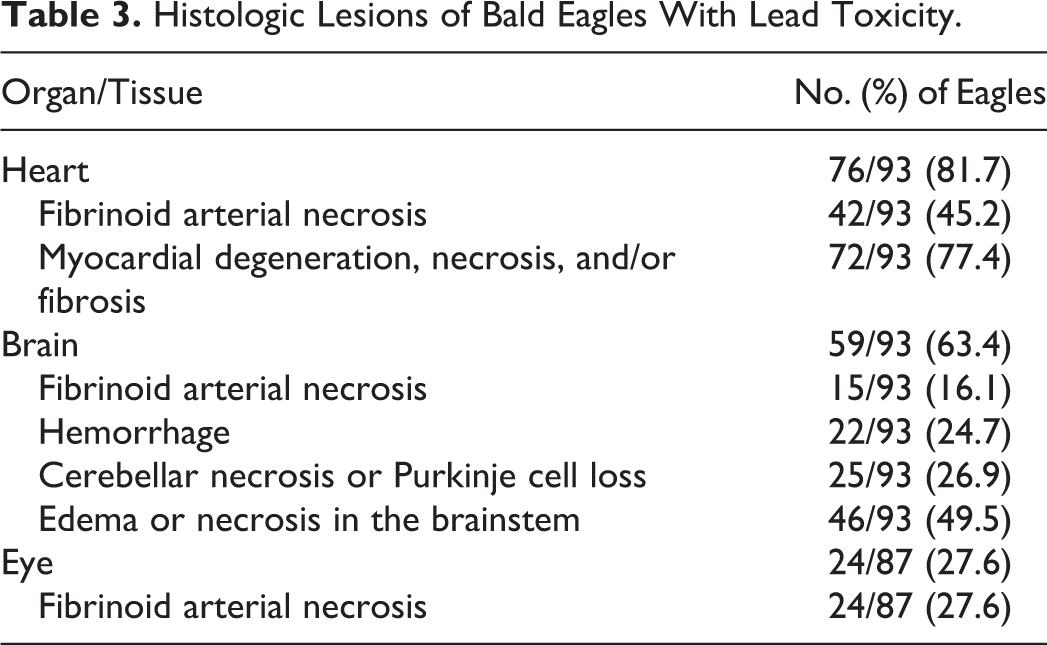
Gross lesions compatible with lead toxicity were less frequent within the brain (Table 2). The most common gross abnormalities within the brain were multifocal to extensive, variably sized, unilateral or bilaterally asymmetric dark red foci consistent with petechiae or hemorrhagic necrosis, predominately within the brainstem and cerebellum but also in the cerebrum (Fig. 3). One eagle had a focal yellow area within the brainstem.
Most bald eagles had histologic changes consistent with lead toxicity (86/93 birds; 92.5%) (Table 3). Histologic lesions, including vascular changes, occurred most frequently within the heart, brain, and eyes and sporadically in other tissues. Vascular lesions were consistent with fibrinoid arterial necrosis, characterized by loss of architectural detail of the walls of small- to medium-caliber arteries with disruption of tunica media, progressing to cell loss within the vessel wall (Fig. 4). These lesions frequently affected the arteries circumferentially. Variable amounts of eosinophilic, amorphous to hyaline material expanded the vessel wall and/or surrounded the vessel. In severe cases, there was almost complete obliteration of the vessel wall. Vessels were occasionally filled and occluded with similar eosinophilic fibrillary material (interpreted as peracute thrombi). Within the heart, fibrinoid arterial necrosis typically affected only a few arteries per histological section while most myocardial arteries were unaffected. Fibrinoid arterial and arteriolar necrosis was less common within the brain and predominately affected meningeal vessels and vessels within the parenchyma of the cerebellum, mesencephalon, and/or brainstem. Fibrinoid arteriolar necrosis of the choriocapillaris was present in 24 of the 87 birds in which the eyes were examined.
Histologic Lesions of Bald Eagles With Lead Toxicity.
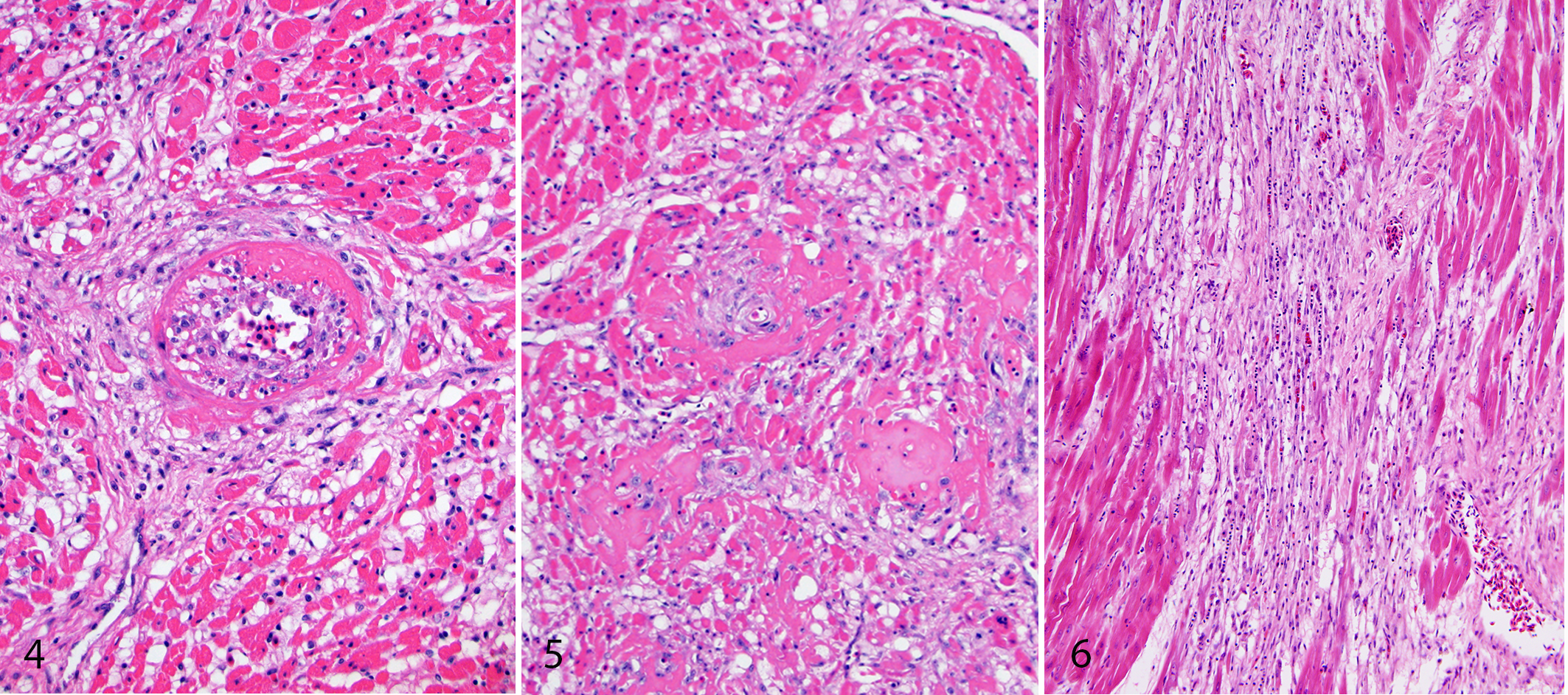
Additional histologic lesions consistent with lead toxicity were predominantly characterized by multifocal parenchymal degeneration and necrosis, likely resulting from ischemia. The heart of all eagles with hydropericardium had myocardial degeneration and necrosis. Within the heart, concurrent histologic lesions included small to large areas of degeneration and/or necrosis within the myocardium (Fig. 5). Cardiomyocytes in affected areas were swollen and hypereosinophilic or small and shrunken. Globular to floccular fragmentation of the sarcoplasm was occasionally present. This intermittently progressed to cardiomyocyte loss, proliferation of plump spindle cells with oval nuclei (fibroplasia), and, in some cases, deposition of dense collagen (fibrosis) (Fig. 6). For those cases in which special stains were applied, collagen deposition in the heart stained positively with Masson’s trichrome stain and Picrosirius red, the latter indicating deposition of type I or type III collagen. These changes were considered a continuing spectrum over time. Some birds had only acute cardiomyocyte degeneration and necrosis. While a small number of eagles had only fibrosis within the myocardium, it was more common for myocardial fibrosis to be accompanied by ongoing myocardial degeneration and/or necrosis.
Gross and histologic lesions in the heart occurred slightly more frequently in birds receiving chelation treatment (58.1% and 86.0% of birds) compared to birds that were not chelated (52.0% and 78.0% of birds).
Within the brain, histologic lesions were substantially more frequent (59/93; 63.4%) than gross lesions. Lesions considered compatible with lead toxicity within the brain comprised fibrinoid necrosis of capillaries and small-caliber arteries in the leptomeninges and neuropil, hemorrhage, necrosis, and/or parenchymal edema (Figs. 7–10). Necrosis was characterized by areas of rarefaction, pallor, poor staining of cellular detail, nuclear changes such as pyknosis or karyolysis, and/or loss of cells, with variable gitter cell infiltrates. In some areas of necrosis, neurons retained cellular staining detail and morphology. Within areas of hemorrhage, some sections had evidence of plump and prominent endothelial cells lining small capillaries (endothelial hypertrophy) and/or erythrophagocytosis. While a considerable portion of these cases had bilaterally symmetrical, minimal to moderate areas of vacuolation in the brainstem (19/93 eagles; 20.4%), all other histologic lesions within the brain were asymmetric and randomly distributed.
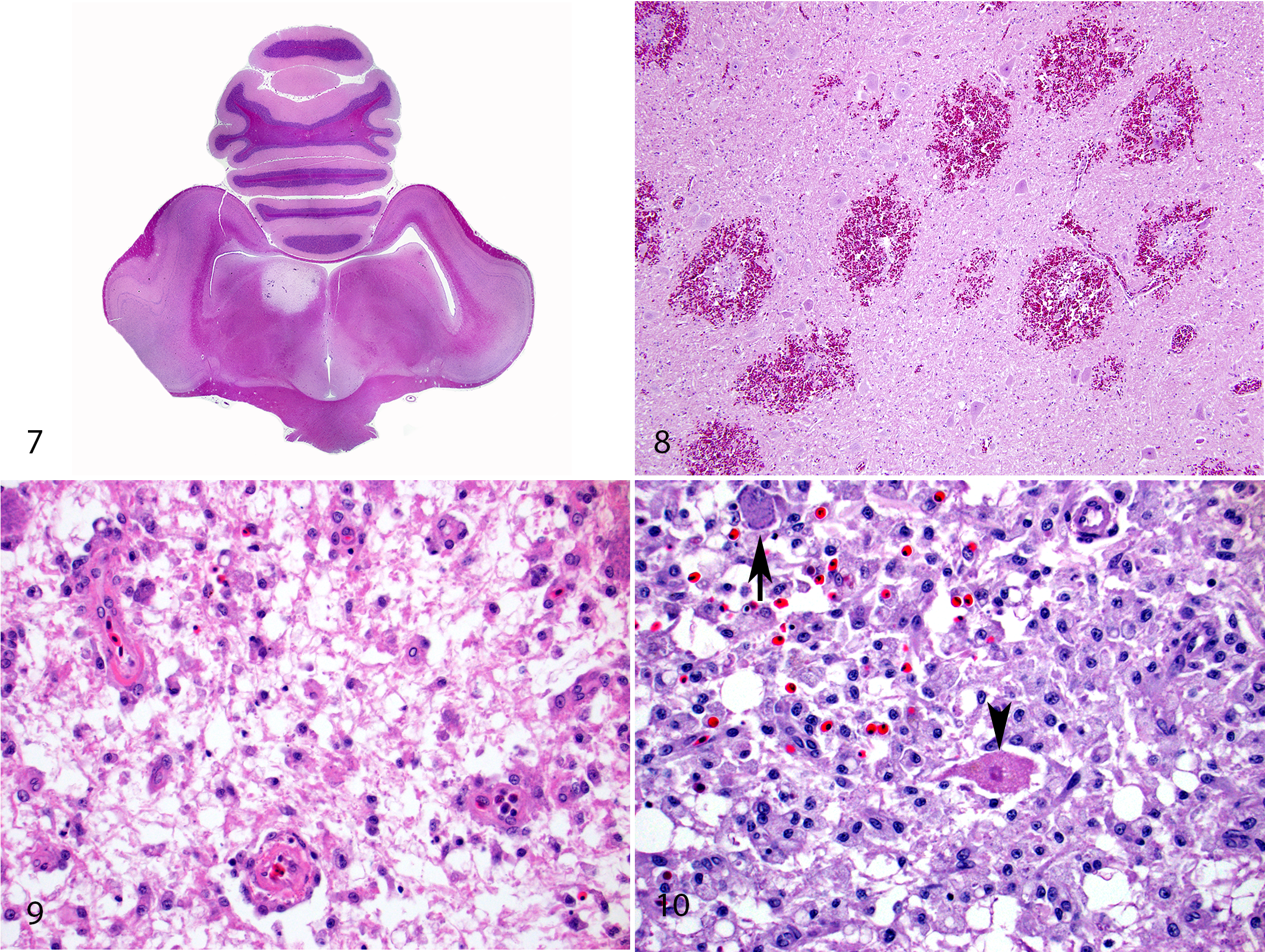
Gross and histologic lesions were slightly more common in the brain of birds receiving chelation (18.6% and 69.8%) vs birds without treatment (10.0% and 58.0%).
Fibrinoid arteriolar necrosis was the primary histologic lesion within the eyes and was limited to the choriocapillaris (Fig. 11). Lesions were unilateral or bilateral and ranged from a single vessel affected per section to extensive segments of choriocapillaris with vessels expanded with eosinophilic, hyaline material. Contrasting to vascular lesions within the brain and heart, fibrinoid arteriolar necrosis within the choriocapillaris was frequently accompanied by heterophilic infiltrates. Ocular lesions were only detected in birds that also had heart and/or brain lesions.
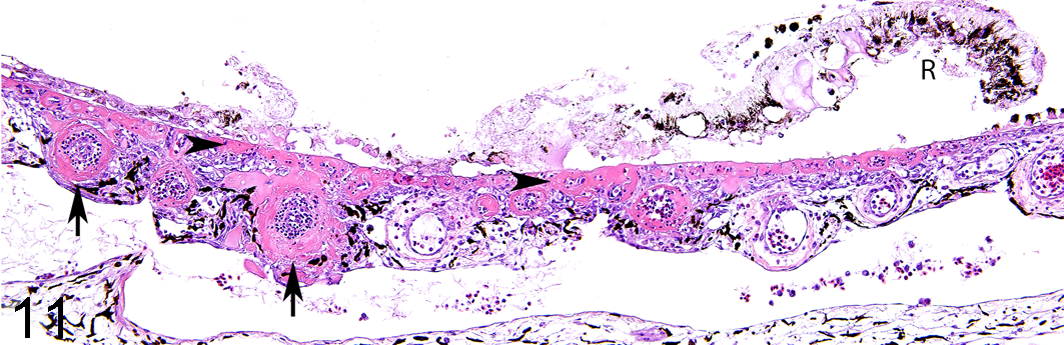
Lead toxicity, eye, bald eagle. The walls of multiple choroidal arterioles are circumferentially or partly expanded by homogeneous eosinophilic material (interpreted as proteinaceous plasma-like fluid; arrows). Similar material is present around the capillaries beneath Bruch’s membrane (arrowheads). The retina (R) is largely detached and degenerate. Blood lead concentration = 5.44 ppm. Hematoxylin and eosin.
The ventriculus was examined histologically in 53 cases, and ventriculitis was present in 9 of them. Ventriculitis was erosive or ulcerative with occasional bacterial colonies and/or hemorrhage. Metal fragments were present at necropsy in 2 of 9 birds with ventriculitis and detected in an additional bird on clinical radiographs.
Fibrinoid necrosis of small- and medium-caliber arteries was sporadically observed within other tissues and organs, including the adrenal gland (1 bird), mesentery (1 bird), pancreas (2 birds), small intestine (3 birds), and colon (1 bird). Within the affected colon, the fibrinoid arterial necrosis was associated with mucosal infarction.
Kidneys were examined histologically in 90 birds. Seven eagles had renal tubular degeneration or necrosis. The changes were minimal to mild in all but 1 bird. Intranuclear inclusion bodies were not observed within tubular epithelial cells in any of the cases. Acid-fast stain was applied to histologic sections of the kidneys of 6 birds with high renal lead concentration and degeneration or necrosis of renal tubular epithelial cells; acid-fast inclusions were not apparent in any of these sections.
Lead concentrations in the initial blood sample (Fig. 12) collected at clinical presentation, as well as lead concentrations within tissues collected at necropsy, are reported in Fig. 13. Liver, kidney, and bone lead concentrations for birds receiving chelation treatment and those not given chelation treatment are shown in Tables 4, 5, and 6.
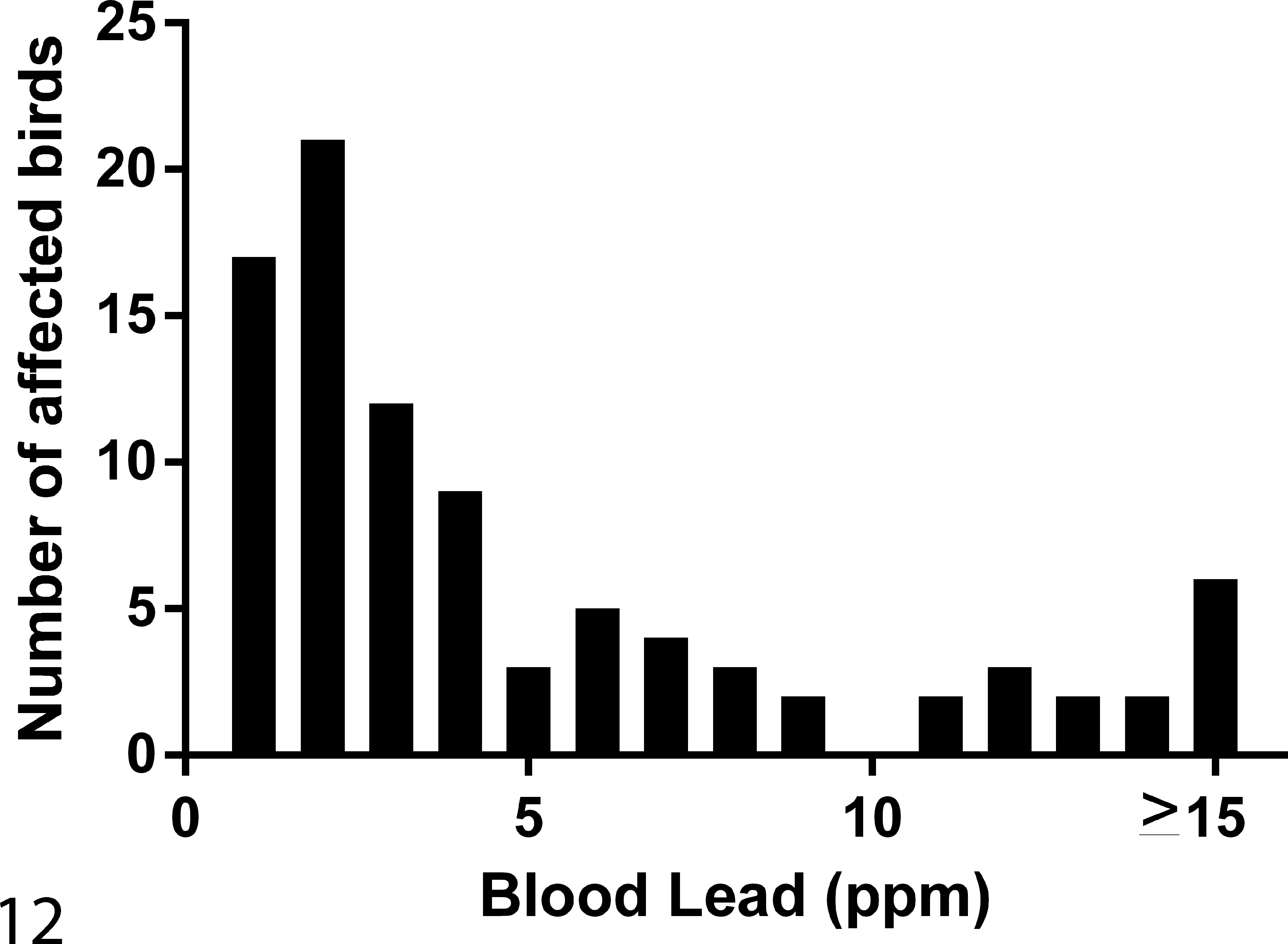
Distribution of blood lead concentrations in bald eagles included in this study.
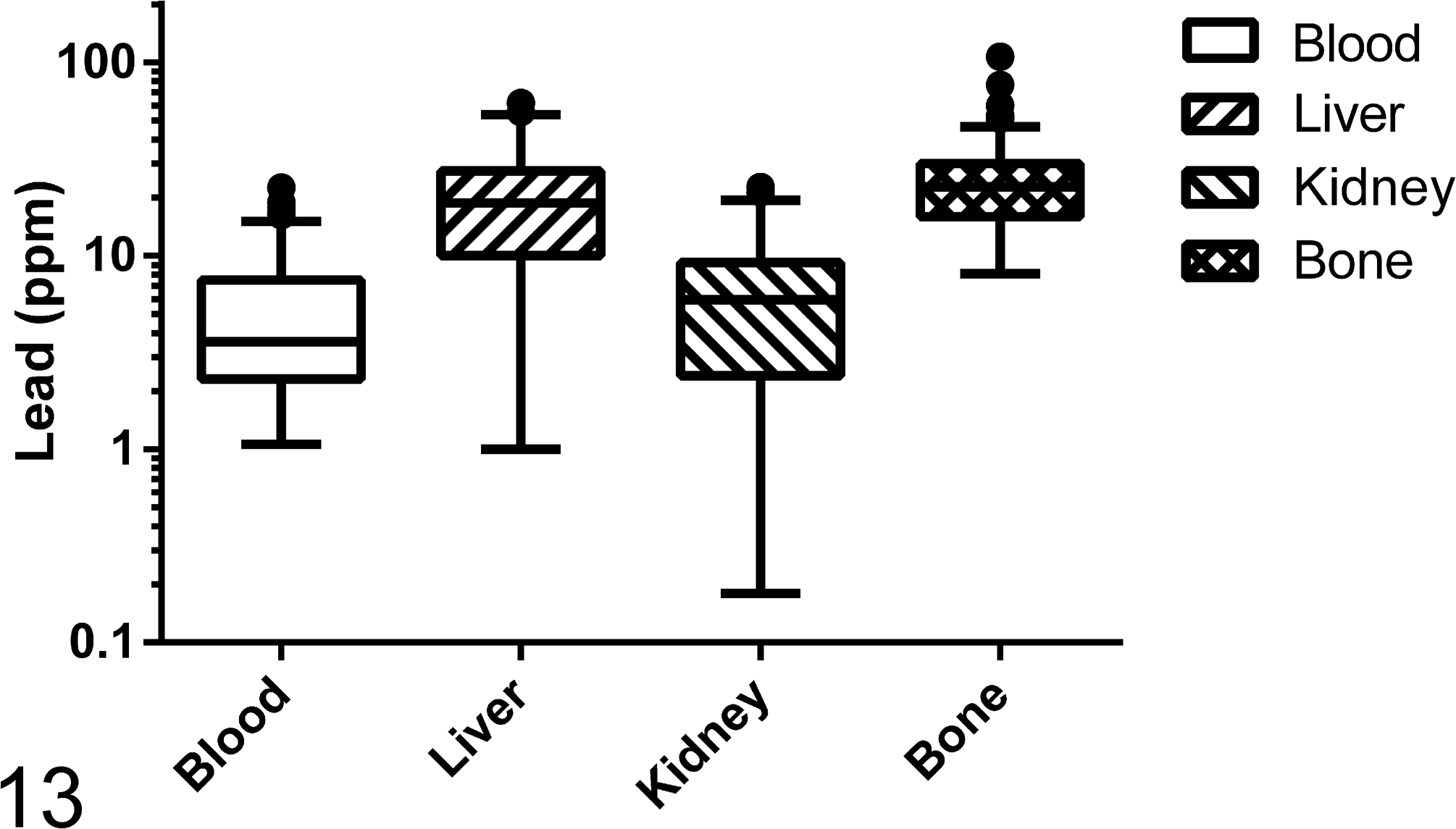
Lead concentrations in blood, liver, kidney, and bone. The concentrations in liver and kidney are reported on a wet weight basis. The line represents the median, the box encloses the first and third quartiles, and the whiskers represent the range. Individual data outside of that range were deemed “outliers” by Tukey’s rule as determined by the graphing software.
Lead Levels in Liver for Bald Eagles Receiving/Not Receiving Chelation Treatment.
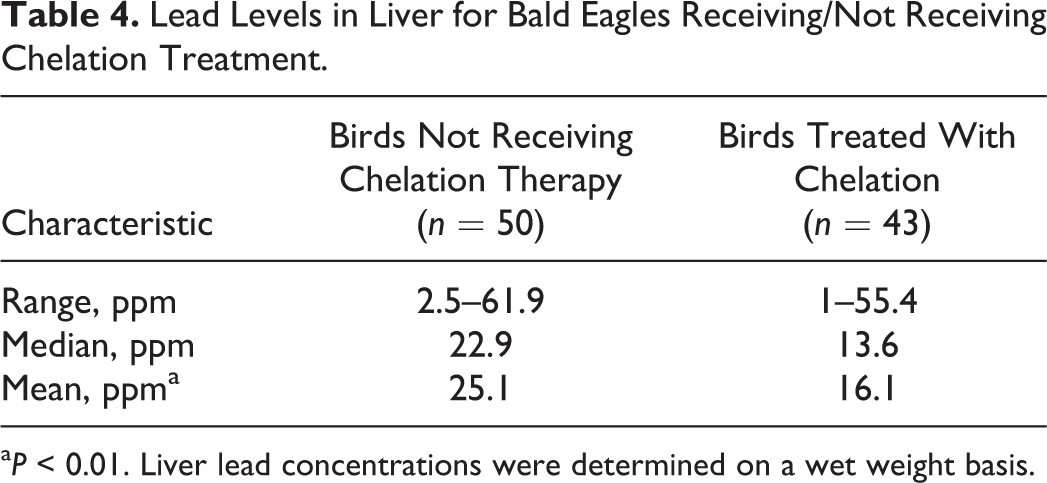
aP < 0.01. Liver lead concentrations were determined on a wet weight basis.
Kidney Lead Concentrations in Bald Eagles Receiving/Not Receiving Chelation Treatment.

aP < 0.01. Kidney lead concentrations were determined on a wet weight basis.
Tibiotarsal Bone Lead Concentrations for Bald Eagles Receiving/Not Receiving Chelation Treatment.
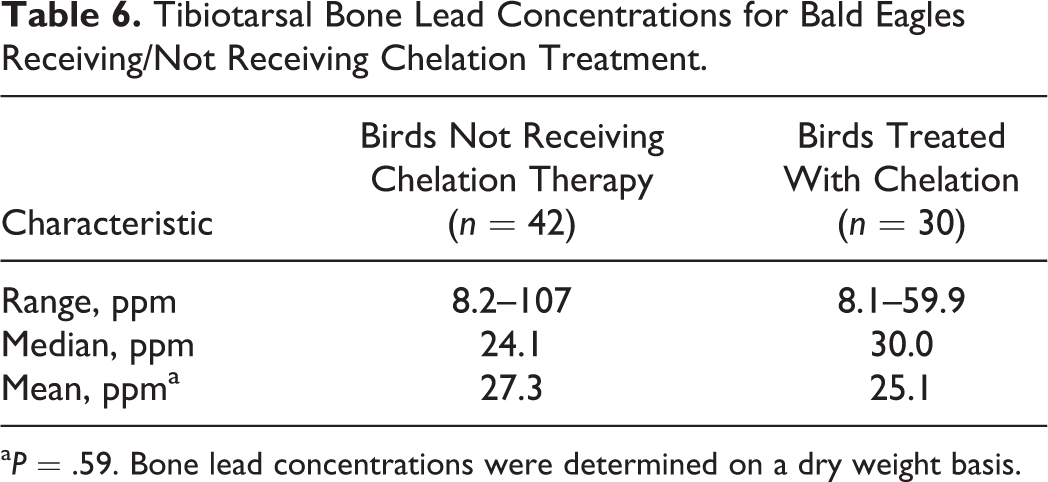
aP = .59. Bone lead concentrations were determined on a dry weight basis.
Blood concentrations greater than 4 ppm were associated with histologic lesions in the heart (χ2 = 4.31, P = .06). This association was not observed in other tissues. Using logistic regression, the strongest predictor of heart lesions was the presence of elevated liver lead concentrations (P = .01). This association was strengthened when only chelated animals were considered for this analysis. There were no identified predictors for eye or brain lesions. The strongest correlations between tissue (blood, liver, kidney, and bone) lead concentrations were documented between lead liver and lead kidney values (r = .69, Pearson correlation coefficient). Blood concentrations correlated modestly with liver (r = 0.39) and kidney concentrations (r = 0.49). These correlations were observed irrespective of birds receiving chelation therapy or not. Linear regression analysis graphically supports these findings, providing the following R2 values for the correlation of blood and liver concentrations (0.1730), blood and kidney concentrations (0.2405), liver and kidney concentrations (0.4130), and blood and bone concentrations (0.00001; Fig. 14).
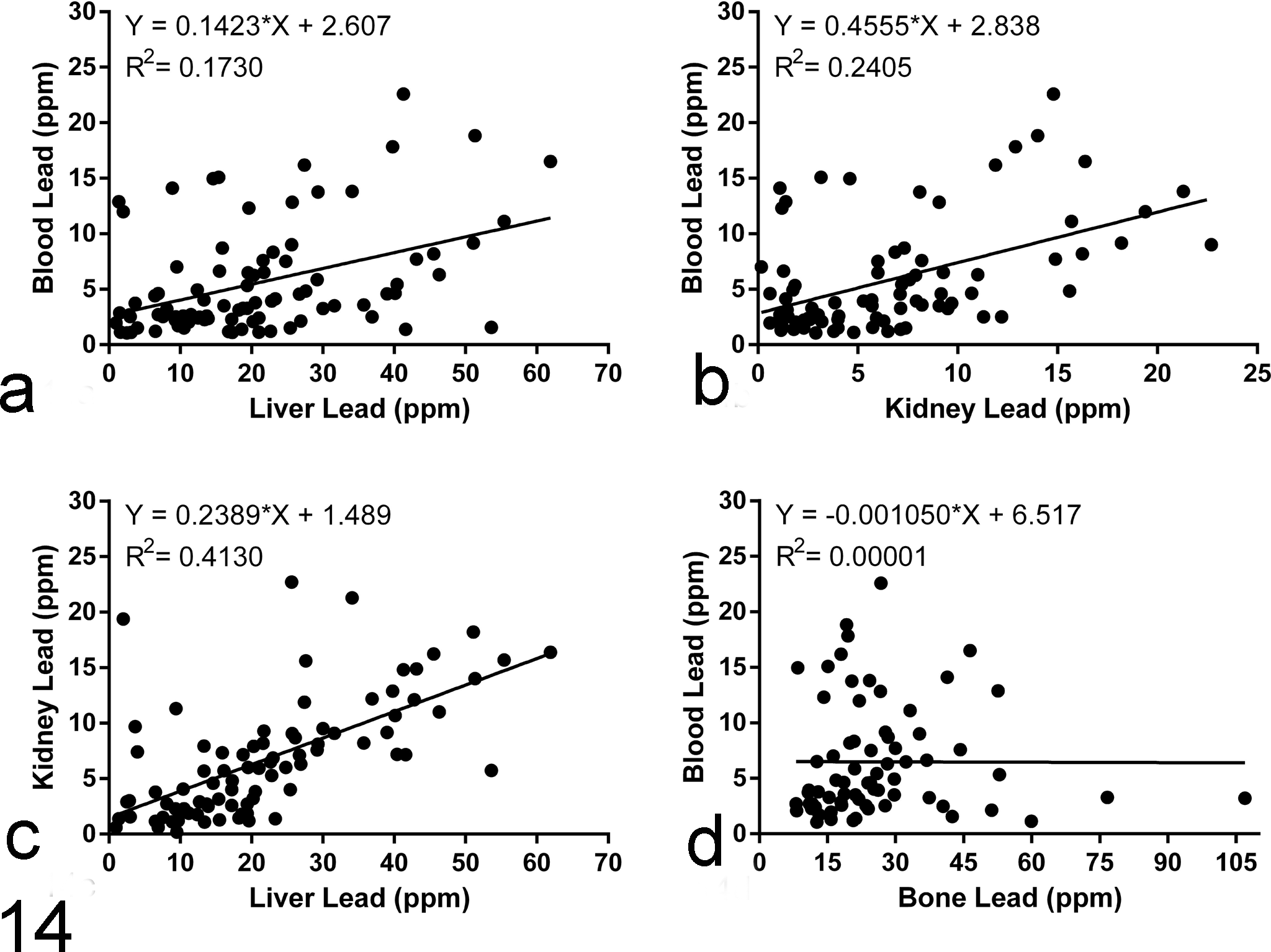
Correlation of lead concentrations among different tissues. Linear regression was performed on plots of lead concentrations from (a) blood vs liver, (b) blood vs kidney, (c) kidney vs liver, and (d) blood vs bone. Lead concentrations in liver and kidney are strongly correlated to one another. Lead concentrations in liver and kidney correlate modestly with the blood lead concentration.
Discussion
This study describes lesions associated with severe lead toxicity in a large series of bald eagles and provides evidence for a high frequency of macroscopically and microscopically detectable lesions. With the exception of 1 previous study (n = 484 bald eagles), 11 reports of lead intoxication–associated lesions in bald eagles have otherwise been limited to small case numbers (n = 1–6). 6,8,13,23,29
We infer that severe, segmental fibrinoid necrosis of small- to medium-caliber arteries is likely the primary underlying insult in lead-intoxicated eagles. Most other gross and histologic lesions described herein are consistent with ischemia, likely secondary to vascular injury. Absence of fibrinoid arterial necrosis in sections of heart or brain with concurrent ischemic lesions may be an effect of sampling since only individual or small numbers of arteries are usually affected. A multisystemic fibrinoid necrosis of arterioles has previously been described in a bald eagle with lead toxicity. 8 It is unknown whether the preferred involvement of cardiac, neural, and ocular arteries in the current study may indicate an increased sensitivity of these vessels to lead. This selective vascular injury does not appear to be directly associated with the amount of blood flow, as renal arterioles would be expected to be frequently affected if that were the case.
Endothelial injury may occur secondary to various toxic, infectious, immune-mediated, or inflammatory processes. No infectious agents were present in histologic sections. With the exception of the choriocapillaris, fibrinoid arterial necrosis was usually not associated with inflammation. Thus, inflammation does not appear to be the primary cause of vascular injury in lead toxicity in bald eagles. The reason for the disparity with respect to the presence and absence of a heterophilic inflammatory component of the vascular lesion in the eye vs other tissues is unknown.
Arterial injury may be due to a direct toxic effect of lead on vessels. Experimental lead toxicity in rats results in vascular injury in the brain, leading to edema and often hemorrhage, particularly in the cerebellum of young animals. 3,15,31 Endothelial cell necrosis and alterations in interendothelial tight junctions within the developing central nervous system of rats occur with lead toxicity. 31 Proposed mechanisms of lead toxicity include (1) oxidative cellular damage secondary to generation of free radicals and depletion of antioxidant reserves and (2) mimicking or inhibition of bivalent cations such as calcium, affecting cell function. 9,12 Lead may lead to altered calcium homeostasis within rat capillary endothelial cells, 16 and chronic exposure can result in oxidative stress in the brain of rats. 1
Cardiac abnormalities suggestive of acute or chronic ischemia, including cardiomyocyte degeneration, necrosis, and/or fibrosis, occurred frequently (72 of 93 birds, 77.4%), and have previously been reported. 8,11,23,30 The only other study with high enough numbers to assess lesion prevalence in lead-intoxicated bald eagles reported the frequency of myocardial degeneration and necrosis at 36.4%. 11 The reason for the discrepancy in the prevalence is uncertain, but the usually good to excellent postmortem preservation of our cases vs cases submitted to the NWHC may account at least for some of the difference. Although multifocal myocardial degeneration and necrosis can be due to causes other than lead toxicity, this lesion is highly predictive of lead toxicity in our bald eagle necropsy material (data not shown). Other hitherto unreported lesions in the heart of bald eagles with lead toxicity were left ventricular dilation and hydropericardium observed in multiple birds. These lesions are interpreted to be the consequence of reduced myocardial contractility associated with the myocardial degeneration and necrosis.
With only 2 previous reports of lead-associated neural lesions, lesions compatible with lead toxicity were surprisingly common within the brain of the examined bald eagles. 6,23 Central nervous system (CNS) lesions were not reported in the only other study of a larger series of bald eagles. 11 While many birds of our study had lesions in both the heart and brain, 10 bald eagles had lesions in the brain without concurrent histologic cardiac abnormalities. It is therefore prudent to examine brain histologically in cases of possible lead toxicity in bald eagles.
As in the heart, most neural parenchymal lesions were randomly distributed and consistent with ischemia secondary to a selective or segmental vascular injury. Ischemia secondary to vascular injury in the brain may result from thrombosis, vasogenic edema impairing diffusion of oxygen and nutrients, and/or with cytotoxic edema of astrocytes. Thrombosis was occasionally present but not a consistent feature, although it may have been missed in the sections examined. Edema was a prominent feature within the brain and likely contributed to neural ischemic lesions. Therefore, multiple mechanisms of ischemic injury may exist, or the mechanisms may vary between organs in lead toxicity in bald eagles.
Within a subset of bald eagles in the current study (20.4%), bilaterally symmetrical status spongiosus or vacuolation was present within the brainstem in the absence of arterial necrosis and hemorrhage. The symmetrical distribution of these changes differed from the aforementioned randomly distributed lesions within the brain. A concurrent selective toxic effect on neurons and/or glial cells within the brainstem is suspected. Lead-induced neuronal injury may be due to a direct toxic effect on neurons or occur secondary to astrocytic dysfunction, altered neurotransmission, and/or excitotoxicity within the brain. 27,34 Experimental studies of lead toxicity in rats have suggested that apoptosis may play a role in the death of cells within the hippocampus 35 and neurons of the cerebellum. 28 Astrocytes have an affinity for lead and have been shown to take up and sequester lead. 7,19 Neurons appear to be more sensitive to the effects of lead than astrocytes in vivo, although lead inhibits aerobic metabolism in both cell types. 19 As neurons have a high metabolic rate, hypoxia due to vascular lesions may also contribute to neuronal toxicity.
Fibrinoid necrosis of choroidal vessels was apparent in 27% of the birds. To the authors’ knowledge, only 1 case of similar vasculopathy in the eye of a bald eagle with lead toxicity has been reported. 8 The fact that fibrinoid arteriolar necrosis in the choriocapillaris only occurred in animals that also had brain and/or heart lesions suggests that the eye lesions may represent a later stage of lead toxicity. Interestingly, increasing lead concentrations in blood, liver, kidney, or bone were not significantly correlated with a higher likelihood of eye lesions in our series.
Erosive or ulcerative ventriculitis was occasionally present in bald eagles (17.0%). Fibrinoid arterial necrosis is possibly an underlying mechanism of the ventriculitis. Fibrinoid arterial necrosis of the media of the ventriculus and proventriculus has been described in experimental lead poisoning of bald eagles. 30 Chemical or mechanical mucosal irritation associated with the presence of lead particles within the gastrointestinal tract also may cause irritation of the ventricular mucosa. As in our study, the absence of lead particles within the gastrointestinal tract of most of the bald eagles is common due to natural casting mechanisms of raptors and/or dissolution or mechanical breakdown of the particles within the gizzard followed by systemic absorption. 10,25
Renal tubular degeneration and necrosis was rare in bald eagles in the current study and, when present, was minimal to mild. In agreement with previous studies, intranuclear lead inclusion bodies were not detected in the bald eagles of our study. 11,20,30
The presence of a full gallbladder or bile staining of the proventriculus or ventriculus, reported as being common in eagles with lead intoxication, 11 was not included in the present analysis as these changes are considered nonspecific for lead toxicity, occurring in other disease states with gastric stasis or anorexia. Emaciation and/or muscle atrophy has previously been described in association with chronic lead toxicity in bald eagles. 25,30 Only around half (48%) of eagles were classified as underweight or emaciated in the present study. The presence of emaciation suggests chronic lead toxicity, but it is possible that acute lead toxicity occurred in some birds that were already emaciated when they ingested lead.
The demographics of the reported population of bald eagles are similar to previous studies. Most birds in this study were adults. Adult bald eagles have been previously reported to have greater odds of elevated lead concentrations or lead poisoning. 5,11 The predominance of adults in this study may reflect a cumulative effect of repeated lead exposure.
Anemia occurs secondary to lead toxicity in numerous species due to inhibition of enzymes involved in heme synthesis and, in some cases, hemolysis. 10 Although anemia occurred in 66% of birds in this study, anemia is unlikely to be associated with the pathogenesis of the underlying vascular and ischemic lesions, with arterial necrosis frequently occurring in the absence of anemia. Birds with acute toxicoses are more likely to have a mild anemia compared to a more severe anemia in chronic toxicoses. 32 Reduced hematocrit has been reported in experimental lead toxicity of bald eagles and Andean condors, with concurrent reduction in δ-aminolevulinic acid dehydratase (ALAD) activity, an enzyme required for heme synthesis and hemoglobin production. 18,29 While hemosiderosis within the liver and spleen may occur with anemia associated with destruction of defective erythrocytes, this nonspecific finding was not included in statistical analysis.
Blood lead concentrations greater than 4 ppm were associated with a greater likelihood of lesions within the heart but not within the brain or eyes. Using logistic regression analysis, elevated liver lead concentration was the strongest predictor of heart lesions. Future investigation of lesions in eagles with less severely elevated blood lead concentrations is necessary to determine additional correlations between lead concentrations and gross and histologic lesions.
According to the literature, clinical signs of lead toxicity in bald eagles presenting in good body condition with elevation of blood lead concentrations to 1 to 4 ppm are considered to represent acute toxicoses compared to chronic intoxication associated with poor body condition and a lesser increase in blood lead concentrations (0.6–1 ppm). 32 Histologically, lesions within the heart may represent a spectrum of chronicity of changes from myocardial degeneration and necrosis (acute) to fibroplasia and fibrosis (subacute to chronic). Comparison of lesions in bald eagles with a blood lead concentration of 0.6 to 1 ppm and greater than 1 ppm in the context of clinical signs and necropsy findings may assist in further defining chronicity of lesions.
Blood lead concentrations were correlated with liver and kidney lead concentrations, particularly for the bald eagles that were not treated with chelation. In addition, kidney and liver lead concentrations were moderately correlated, with liver lead concentrations typically being approximately 3-fold higher than kidney lead concentrations. Lead concentrations within the blood, kidney, and liver are thought to be more representative of recent lead exposure while high lead concentrations in bone are considered to be reflective of chronic and cumulative exposure. 10 The lack of correlation between blood and bone lead concentrations in this study is therefore not unexpected. However, lead is also known to be released from bone such as during formation of the eggshell in female birds, when calcium and inadvertently lead are mobilized.
Chelation therapy was attempted in 43 of 93 birds and was associated with lower tissue lead concentrations. The median liver lead concentration was considerably higher in birds not treated with chelation (22.9 ppm) than those treated with chelation (13.6 ppm). This difference was also apparent in median kidney lead concentrations for birds not chelated (7.4 ppm) and chelated (1.8 ppm). Some birds treated with chelation therapy received only 1 treatment dose; therefore, the effect on lead concentrations within the liver and kidney in some treated birds may have been minimal. The difference between treated and nontreated groups was less apparent for tibiotarsal bone lead, consistent with bone representing a more chronic storage of lead. For birds receiving chelation treatment, bone lead concentration may be more sensitive in detecting previous exposure to lead, but further studies are needed to confirm this.
Chelation therapy had only minor effects on frequency of lesions. Somewhat unexpectedly, gross and histologic lesions in the heart and brain occurred slightly more frequently in birds receiving chelation treatment compared to birds that were not treated. This tendency of lesions to be slightly higher in treated birds may be due to these birds surviving longer, allowing more time for lesions to develop.
Liver lead concentrations greater than 6 ppm (wet weight) are thought to be associated with clinical toxicity and greater than 10 ppm with severe poisoning. 10,22 However, liver and kidney lead concentrations below 6 ppm may sporadically occur in eagles with severe or fatal lead toxicity based on our results (3 cases). Interpreting tissue lead concentrations in the context of histologic lesions may increase the sensitivity of diagnosing lead toxicity.
The described gross and histologic lesions in these birds are attributed to lead toxicity. Infectious diseases such as West Nile fever were excluded due to the lack of inflammation associated with the lesions in heart and brain. 40 Also, the choroid lesions in the lead-intoxicated eagles differ from the lesions present in the choroid of eagles with West Nile disease in that West Nile virus–induced lesions are characterized by lymphoplasmacytic infiltration of the choroid without evidence of vascular necrosis. 40 In addition, all birds were submitted well outside of the West Nile virus season in the upper Midwest. 2 Euthanasia may induce tissue changes that could resemble some of the lesions described such as edema. 17 However, gross and histologic lesions occurred at a similar frequency in birds euthanized (63.3% and 91.1%) and not euthanized (64.3% and 100%). Furthermore, the authors (A.G.A. and A.W.) have not observed the described combination of lesions in bald eagles euthanized for reasons other than lead toxicity (data not shown). Bald eagles with severe and fatal lead intoxication frequently have gross and histologic lesions within the heart, as well as histologic lesions within the brain and eyes that are recognizable by pathologists in well-preserved carcasses. Diagnostic histologic criteria that indicate probable lead toxicity in bald eagles include fibrinoid necrosis of small- and medium-caliber arteries and secondary parenchymal ischemia in the absence of infectious agents or significant inflammation. Such lesions indicate testing of tissue lead concentration, preferably in the liver and/or kidney, for a definitive diagnosis of lead toxicity. Correlation of clinical signs with the presence and distribution of lesions may allow further insight into the pathogenesis of lead toxicity in bald eagles.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985818813099 - Lead Intoxication in Free-Ranging Bald Eagles (Haliaeetus leucocephalus)
Supplemental Material, DS1_VET_10.1177_0300985818813099 for Lead Intoxication in Free-Ranging Bald Eagles (Haliaeetus leucocephalus) by Leah K. Manning, Arno Wünschmann, Anibal G. Armién, Michelle Willette, Kathleen MacAulay, Jeff B. Bender, John P. Buchweitz, and Patrick Redig in Veterinary Pathology
Footnotes
Acknowledgements
We thank the histology team at University of Minnesota, Tom Arendt for his toxicology work at the University of Minnesota Veterinary Diagnostic Laboratory, and Andreas Lehner at Michigan State University.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
Note
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
