Abstract
A 16-year-old female Japanese cat was presented with a single mammary-gland nodule approximately 3 cm in diameter. Histologically, the nodule consisted of necrotizing granulomatous panniculitis, vasculitis, and mastitis, and contained free and clustered protozoal organisms. The organism was present in the cytoplasm of macrophages, fibroblasts, endothelial cells, and mammary-gland epithelia. The organism was positive for anti-Toxoplasma gondii and anti-Neospora caninum antibodies. Electron microscopy showed single and grouped tachyzoites, with morphologic features similar to those of T. gondii. Polymerase chain reaction and deoxyribonucleic acid sequence analysis was consistent with T. gondii infection. This is the first report of cutaneous toxoplasmosis in a Japanese cat.
Toxoplasma gondii is a coccidian parasite of the phylum Apicomplexa, which infects all warm-blooded animals, including humans. 3 Domestic cats and other Felidae are the definitive hosts, whereas, both felids and nonfelids serve as intermediate hosts. Transmission of Toxoplasma can occur via ingestion of oocysts from feline feces, ingestion of cysts present in the tissue of infected intermediate animals, and transplacentally via tachyzoites. 3, 8 Systemic toxoplasmosis has been reported in most species of domestic animals, but cutaneous toxoplasmosis is rare. 1, 5, 13 In this report, we describe a feline case of a single nodule in the mammary gland caused by T. gondii and document the differential diagnosis made between T. gondii and Neospora caninum by using several diagnostic methods.
A 16-year-old female domestic Japanese cat was presented to the veterinary hospital (Inukai Cats Hospital, Kitami city, Japan) with a history of dyspnea, anorexia, lethargy, and a single mammary-gland nodule approximately 3 cm in diameter at the right fifth mammary gland (Fig. 1). The cat had been vaccinated against panleukopenia, viral rhinotracheitis, and caliciviral disease, and was kept outdoors. Laboratory results revealed anemia and leukopenia, and enzyme-linked immunosorbent assay (ELISA) serologic assays for feline leukemia virus (FeLV), feline immunodeficiency virus (FIV), and T. gondii detecting immunoglobulin (Ig) G 11, 15 were negative. The mammary-gland nodule was excised, because of a suspected mammary-gland tumor, fixed in 10% neutral-buffered formalin solution, and embedded in paraffin, according to a conventional method. Sections (3-μm thick) were stained with hematoxylin and eosin (HE). Histopathologic analysis revealed that the lesions were located in the deep dermis to subcutis, exhibited severe necrotizing granulomatous panniculitis, vasculitis (Fig. 2), and mastitis, and contained free and clustered protozoal organisms. The organisms were round and ovoid in shape, and were often present in extracellular fluid and in the cytoplasms of mammary-gland epithelial cells (Fig. 3), macrophages, fibroblasts (Fig. 4), and endothelial cells in free and clustered forms. The protozoal organisms were light basophilic and contained a dark-staining round nucleus. Clusters of organism with mild infiltration of lymphocytes were also found in the peripheral nerves (Fig. 5). In the stroma, collagenolysis, edema, severe hemorrhages, and secondary inflammation were observed. The infiltrate was composed of a variable admixture of hemosiderin-laden macrophages, histiocytes, lymphocytes, plasma cells, neutrophils, and cell debris. The epidermis showed partly acanthosis and hyperkeratosis, but the organism was not observed. For immunohistochemical examination, sections were deparaffinized and checked for endogenous peroxidase activity with 0.3% hydrogen peroxidase. The sections were incubated with commercially available antibodies against T. gondii (polyclonal goat antibody, VMRD Inc., USA) and N. caninum (polyclonal goat antibody; VMRD) at a dilution of 1 : 1000 for 3 hours at 37°C. Bound antibodies were detected by the peroxidase-streptavidin-biotin method (Histofine SAB-PO goat kit, Nichirei, Tokyo, Japan). The organisms were more strongly stained for T. gondii antibody (Fig. 6) than for N. caninum antibody. For electron microscopy, the 10% formalin-fixed mammary-gland nodule was cut into 1-mm blocks, fixed in 1% buffered osmium tetroxide, and embedded in epoxy resin. Sections (about 70-nm thick) were stained with uranyl acetate and lead citrate, and were examined by using transmission electron microscope (H–700, Hitachi, Tokyo, Japan). Protozoal organisms were mainly found in the cytoplasm of the fibroblasts and macrophages, and measured 2–4 μm in width and 3–6 μm in length, and were crescent or arc shaped. Dividing forms that contain 2 nuclei was also observed. The nucleus of the organism was mainly located near one pole of the cell. Its cytoplasm contained round granules, lipid bodies, a conoid, micronemes, and rhoptries. The rhoptries were osmiophilic, vase-shaped, gland-like structures, and their narrow parts were situated at the pointed end of the organism, and their broader bases were situated near the nucleus (Fig. 7). Up to 4 to 8 rhoptries were seen in 1 plane of the longitudinally cut section. Electron microscopic characteristics were similar to those of tachyzoites of T. gondii.
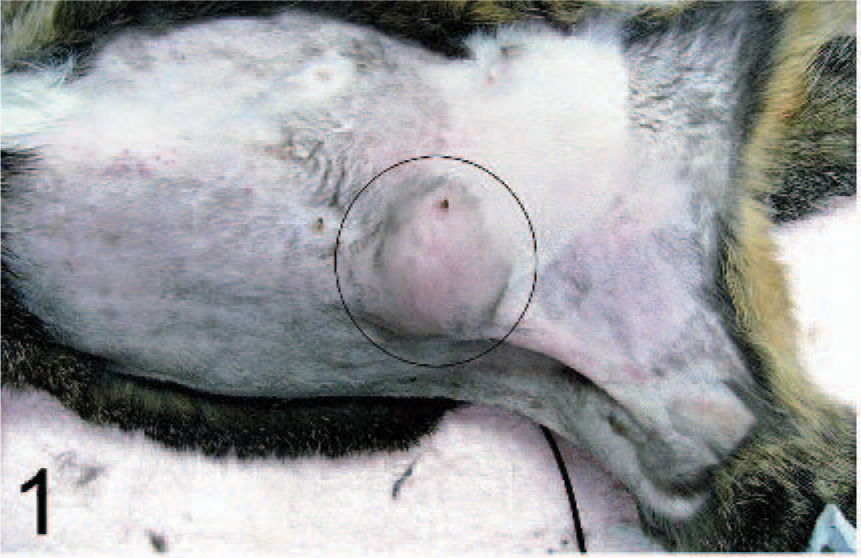
Mammary gland; Japanese cat. A single nodule is present at the right fifth mammary gland (circled).
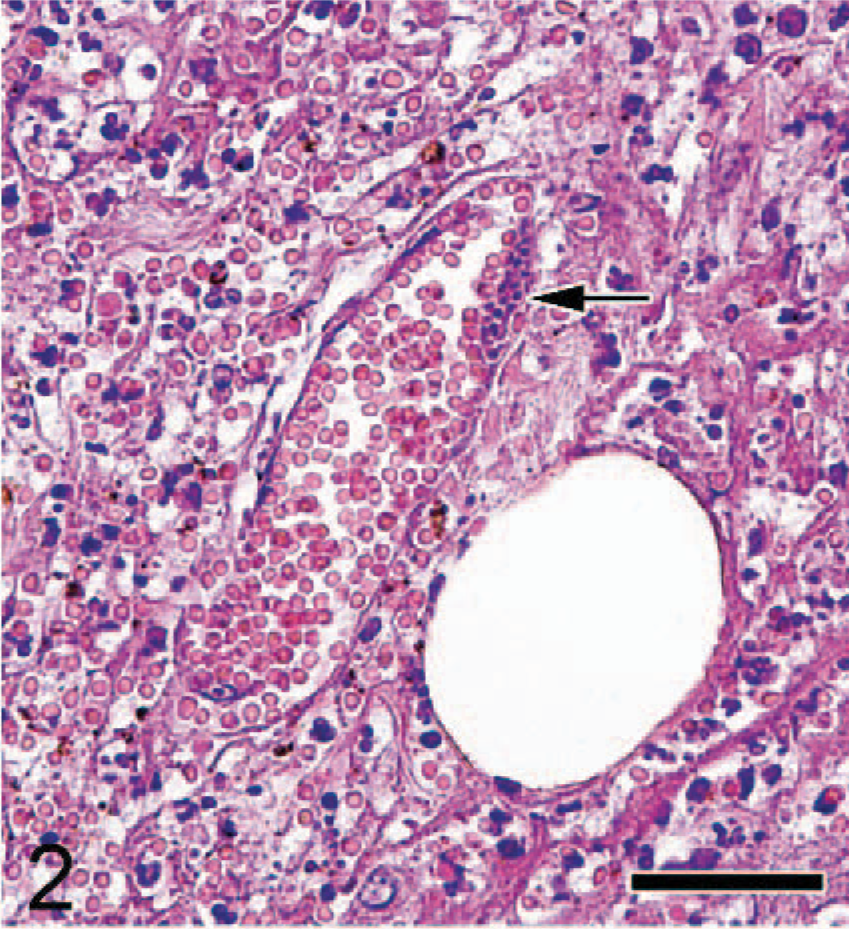
Subcutaneous tissue; Japanese cat. Vasculitis with severe hemorrhages and clusters of organisms (arrow) in the endothelial cells are present. HE. Bar = 30 μm.
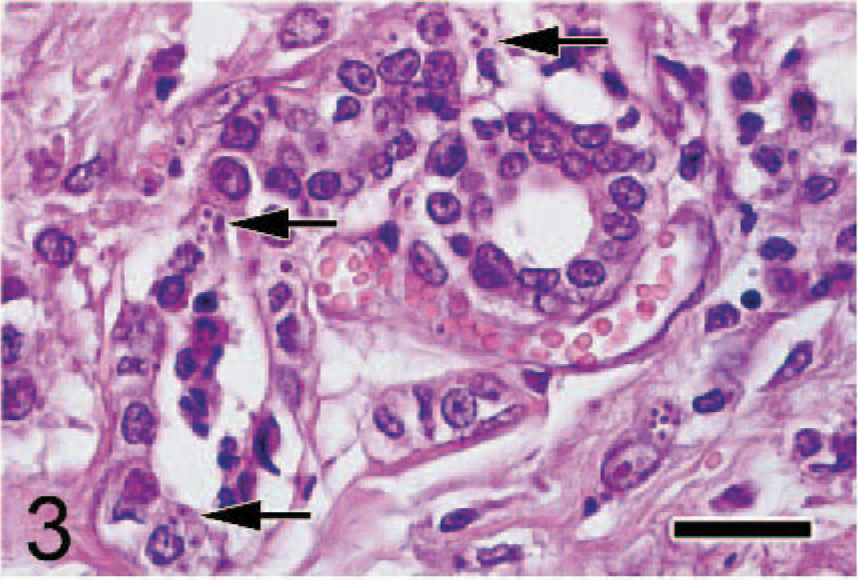
Mammary gland; Japanese cat. Free organisms (arrows) in the cytoplasms of mammary gland epithelial cells are present. HE. Bar = 26 μm.
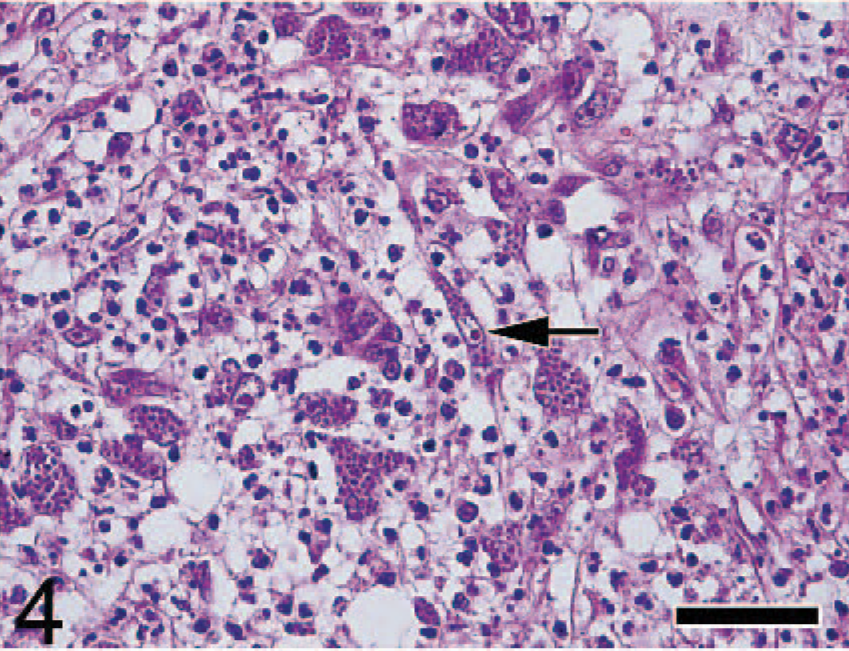
Subcutaneous tissue; Japanese cat. Free and clustered organisms are present within macrophages and fibroblasts (arrow). HE. Bar = 24 μm.
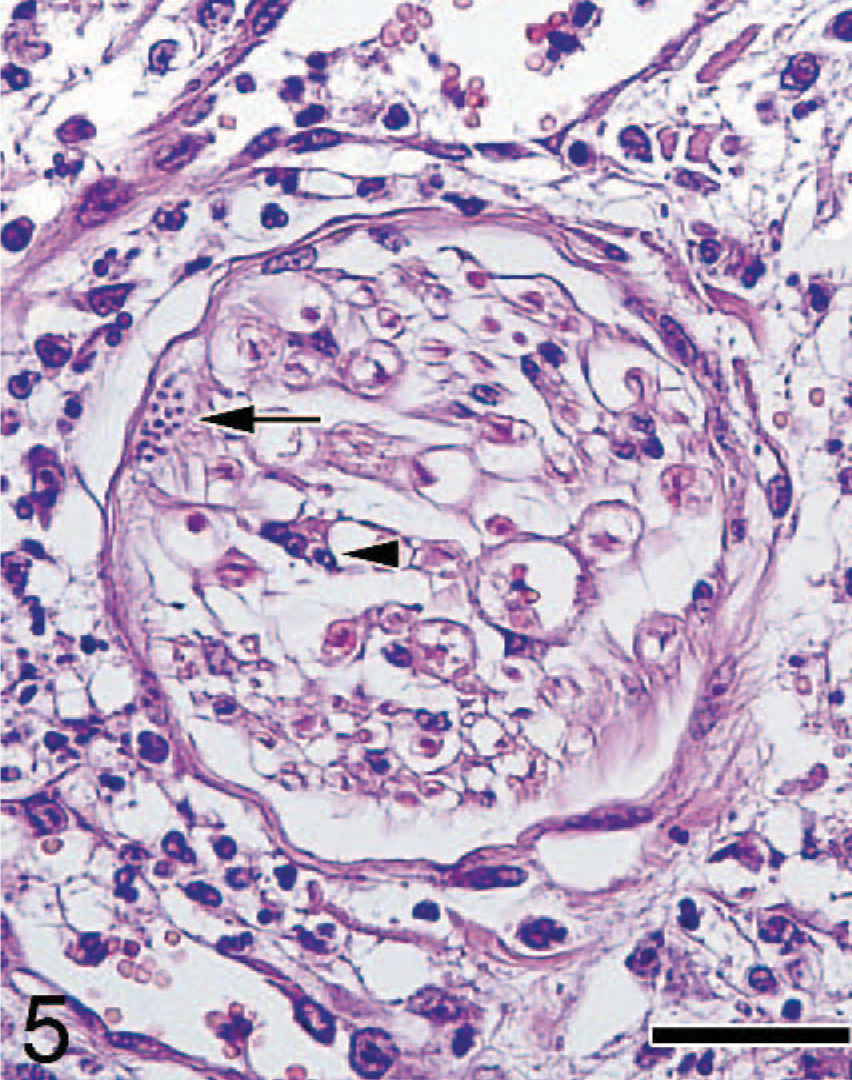
Subcutaneous tissue; Japanese cat. Clusters of organism (arrow), with mild infiltration of lymphocytes (arrowhead) were observed in the peripheral nerves. HE. Bar = 30 μm.
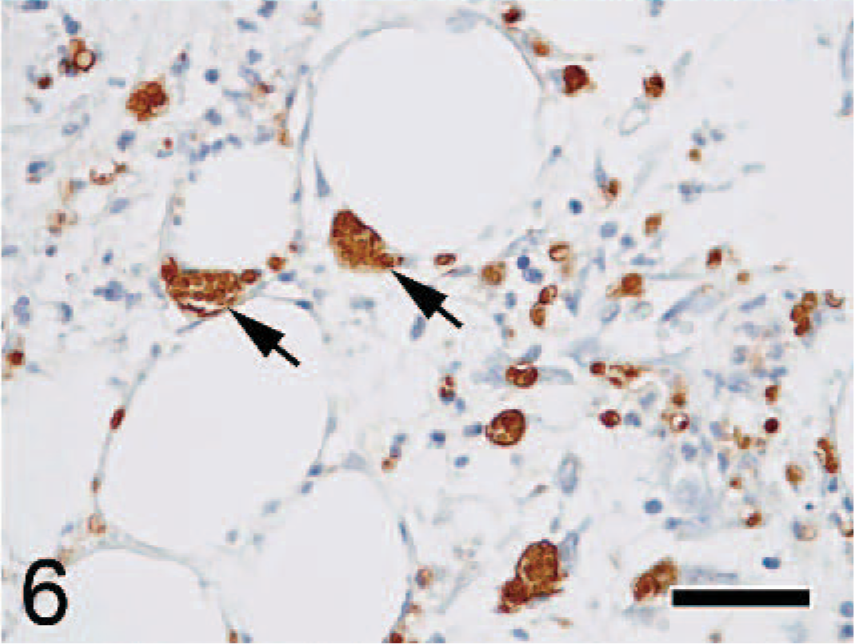
Mammary gland; Japanese cat. Organisms exhibited strong positive staining for T. gondii antibody (arrows). Immunohistochemistry. Bar = 30 μm.
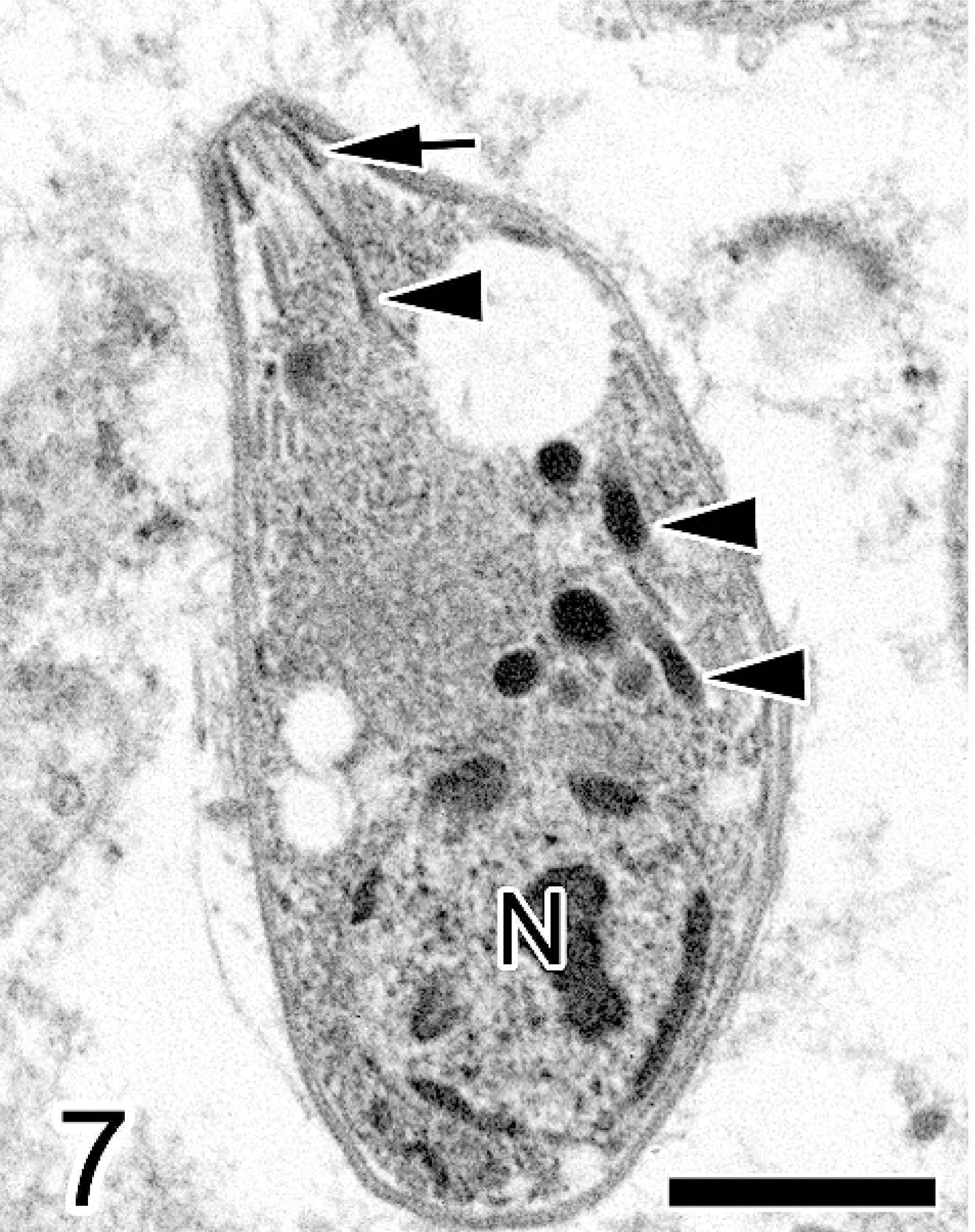
Mammary gland; Japanese cat. The organism is crescent shaped, and the nucleus (N) is located near one pole of the cell. Rhoptries (arrowheads) and a conoid (arrow) are present. Electron microscope. Bar = 1 μm.
The cat received glucocorticoid steroid treatment for 1 month (intraoral injection with prednisolone at 2.5 mg/cat twice a day, one time and intravenous injection at 15 mg/cat once a day), over 4 times, after surgical excision of the mammary-gland nodule; however, the cat died before further diagnostic procedures could be carried out. At necropsy, pulmonary edema, yellow pleural effusion, and nodular hyperplasia of the spleen were observed. Pathologic findings consisted of mild interstitial pneumonia, congestive pulmonary edema, multifocal necrotizing hepatitis, lymphadenitis, and myocarditis. A few organisms in the lungs, spleen, and liver were observed upon histologic and immunohistochemical examination.
For differential diagnosis of T. gondii and N. caninum, nested polymerase chain reaction (PCR) and deoxyribonucleic acid (DNA) sequence analyses were performed on the paraffin-blocked mammary-gland nodule and spleen samples after fixation in 10% formalin solution for 3 days. Feline tissue was used as a negative control for PCR, and cultured T. gondii (RH strain) was used as a positive control. DNA extraction of samples from tissue was performed by using a DNA Isolator PS kit (Wako, Tokyo, Japan). Four oligonucleotide primers for toxoplasmatiid and T. gondii were used for PCR and nested PCR. The first PCR was performed as described previously. 17 Primers CT-nF (5′-AGCGTGATAGTATCGAAAGG-3′) and CT-nR (5′-TCAGTATCCCAACAGAGACA-3′) were used for nested PCR. The 25-μl reaction mixture contained 1 μl of the first PCR product; 2.5 μl of 10× dilution of PCR buffer that contained 15 mM MgCl2 (Applied Biosystems, Foster, CA); and 2.5 μl of 2 mM dNTP mix (Applied Biosystems), 1.25 U Taq DNA polymerase (Applied Biosystems), and 25 pmol of each primer for PCR. Amplification conditions for nested PCR were the following: initial denaturation at 94°C for 3 minutes, 40 cycles at 94°C for 1 minute, 55°C for 1 minute, and 72°C for 2 minutes, followed by a final extension at 72°C for 7 minutes. The final PCR products were subjected to electrophoresis in a 2.0% agarose gel by using TBE buffer (89 mM Tris-base, 89 mM boric acid, 2 mM ethylenediamine tetraacetic acid). DNA sequence analysis was performed as described previously. 9 The visualized sizes of the PCR bands were identical to those of the T. gondii positive control (expected size of 146 bp) (Fig. 8). Base composition of the DNA products resulted in 100% homology (data not shown) with that of T. gondii recorded in the GenBank. Based on these results, the organism was identified as T. gondii.
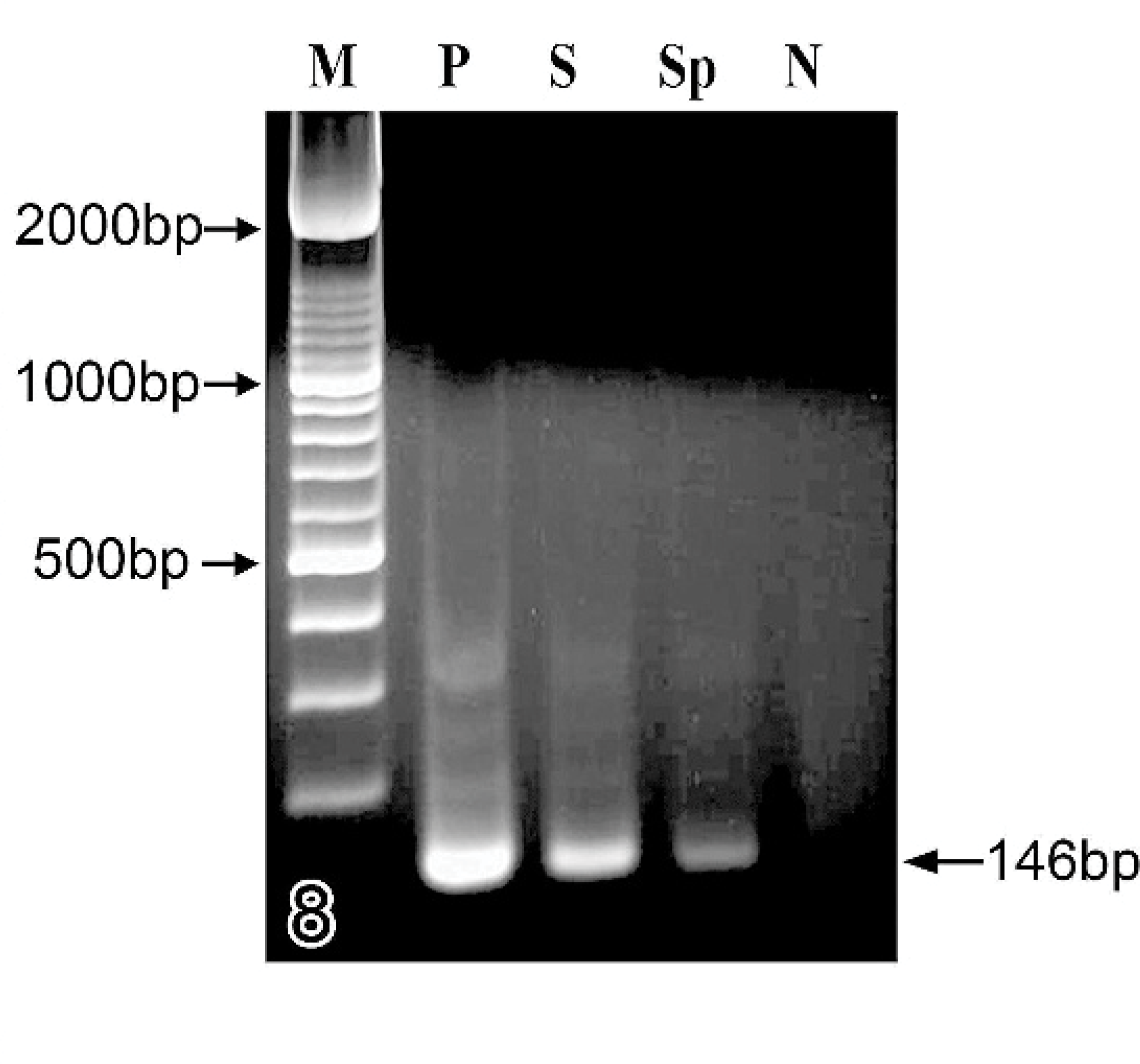
Results of PCR analysis of tissues embedded in paraffin blocks; Japanese cat. DNA from skin (lane 3, S) and spleen (lane 4, Sp), DNA size maker (lane 1, M), positive control (lane 2, P), and negative control (lane 5, N).
It has not been fully investigated why some infected cats develop clinical toxoplasmosis and others remain asymptomatic. The age of the cat, route and stage of T. gondii administered, concurrent infections, and immunodeficiencies are known to affect the outcome of clinical toxoplasmosis. 3 Although young kittens are more susceptible to clinical toxoplasmosis than adult cats, 6 there is no evidence to indicate that primary toxoplasmosis in aged cats is more severe than that in young mature cats. In cats, clinical toxoplasmosis has been documented concomitantly with infections with FeLV, FIV, and feline infectious peritonitis (FIP). 5, 10, 12 In the present case, the results of ELISA serologic essays for T. gondii (IgG), FeLV, and FIV were negative upon presentation, and there was no histologic evidence of FIP virus infection. This cat had been repeatedly treated with glucocorticoid steroid (prednisolone) for 1 month after surgical excision of the mammary-gland nodule; therefore, it was concluded that the glucocorticoid steroid treatment caused immunosuppression 7 and resulted in acute systemic spread of T. gondii.
Reports of toxoplasmic dermatitis are rare in the cat and dog. 2, 13, 18 In humans, macular, hemorrhagic, and even exfoliative lesions were reported in congenital toxoplasmosis, 1 whereas, in the acquired form, the lesions were described as maculopapular, hemorrhagic, lichenoid, 14 hyperemic nodules, and erythema multiforme-like. 1, 4 Cutaneous nodules with ulcers caused by vasculitis were described in 2 subjects that belonged to a group of 100 cats affected by histologically confirmed clinical toxoplasmosis. 5 Tachyzoites were identified by direct microscopy and immunohistochemistry staining from visceral organs but not from the skin. Recently, disseminated cutaneous nodules with clinical toxoplasmosis were described in 2 reports, and the lesions were characterized by a necrotic dermatitis/panniculitis, with vasculitis 2 and lymphoplasmacytic dermatitis, 13 respectively. These histologic changes are similar to those of the present case, with the exception of mastitis. Our case was initially presented with a single mammary-gland nodule. Malignant mammary tumors are frequently found in the aging cat as multiple or single nodules. Therefore, before surgical excision, differential diagnosis should be considered, not only mammary-gland tumor but also both infectious and noninfectious inflammatory causes.
Polyclonal antibodies for T. gondii and N. caninum resulted in positive immunohistochemical staining. Low specificity for antibodies against these 2 organisms was already described. 2 Immunohistochemistry positivity obtained with the antibody against N. caninum was considered a cross-reaction. Electron microscopy is useful for the differential diagnosis of T. gondii and N. caninum, but they are sometimes indistinguishable, because their tachyzoites share many ultrastructural characteristics. 16 Therefore, the results of the PCR and DNA sequence analyses were considered the most diagnostically accurate of the analyses conducted.
