Abstract
A large number of studies have investigated feline mammary tumors in an attempt to identify prognostic markers and generate comparative analyses with human breast cancer. Nevertheless, a retrospective base of assessments and the lack of standardization in methodology and study design have caused weakness in study results, making comparison difficult. We examined feline mammary tumor publications and evaluated postulated prognostic parameters according to the recently published “Recommended Guidelines for the Conduct and Evaluation of Prognostic Studies in Veterinary Oncology.” Using these criteria, we determined with statistically significant reliability that prognostic parameters for feline mammary tumors are tumor grading and lymph node/lymphovascular invasion. Furthermore, tumor subtype, size, and staging are worthy of further standardized investigation. We present statistical significance for each studied parameter as well as its relevance to disease progression and survival. Our evaluation suggests that marker expression (ie, Ki67, HER2, ER) may provide relevant information applicable for therapeutic predictions; however, consensus efforts and protocol standardization are needed. We identify and discuss major points of concern—such as sample preservation and selection, standardization of immunohistochemical protocols, and evaluation of results—to provide support for subsequent reliable analyses.
Approximately 200 manuscripts have been published on feline mammary tumors (FMTs) since the first Symposium on Mammary Neoplasia in the Dog and Cat was held at the ninth annual British Small Animal Veterinary Association congress in London in 1966. 91 Mammary tumors are common in cats and constitute approximately 17% of all feline neoplasms. 26,61 Most are hormone-independent carcinomas (80%–90%) of the simple type (involvement of a single neoplastic cell component: epithelial luminal) that have an aggressive biological behavior. 24,59,68 Reported rates of metastases range from 50% to 90% and most frequently affect the regional lymph nodes (83%), lungs (83%), liver (25%), and pleura (22%). 22,23 Survival after primary tumor detection averages 12 months in untreated cats but varies with clinical staging following tumor excision. 19,22,25
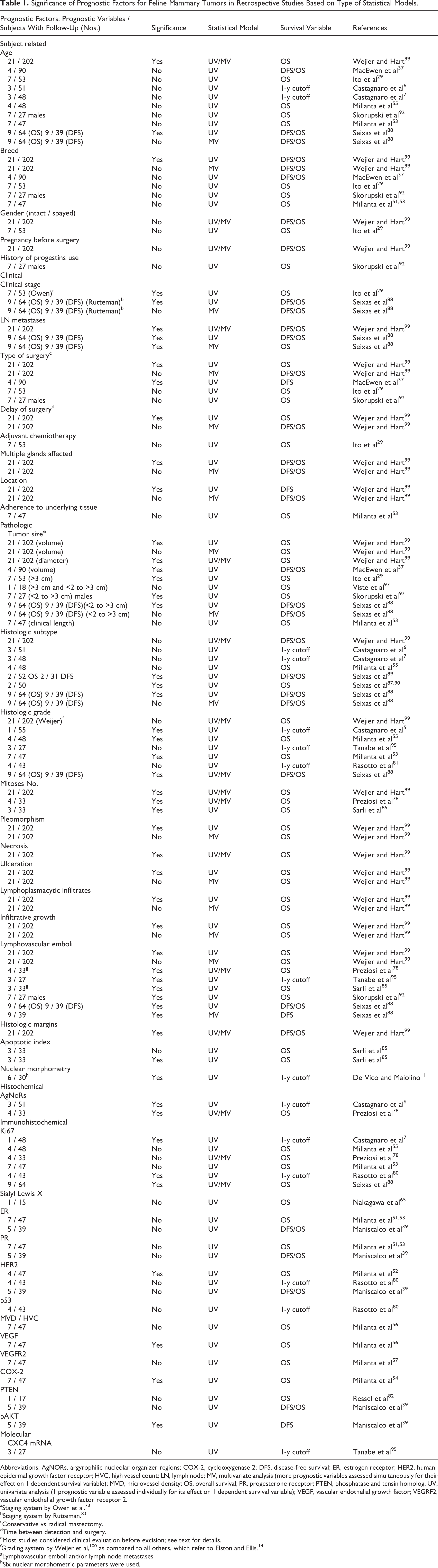
Clinical, pathologic (macro- and microscopic), and molecular features that have been studied to identify prognostic indicators, as well as therapeutic predictive markers of FMTs, include (1) stage based on tumor size, lymph node status, and metastases; (2) histologic grade; and (3) molecular markers, such as cell-cycle associated molecules, estrogen and progesterone receptors (ER and PR), and epidermal growth factor receptor 2 (HER-2).5,10,28,39 –45,51,53,70,71,80,88,100 Unfortunately, clinical follow-up data were included in only 15% of all studies, almost all of which were retrospective; thus, the most powerful predictor of clinical behavior was frequently lost—namely, disease-free survival (DFS; time from surgery to the development of recurrences and/or metastases). In addition, methods for collecting follow-up information are often unclear, 29,87,99 and the number of cases included is often small (15–65 animals in papers after 1990). 88 Furthermore, numerous retrospective studies indirectly associate a prognostic role with the investigated parameter on the basis of its correlation with previously published prognostic factors (ie, grade). This practice does not allow for a correct assessment of investigated parameters in determining if they have a direct effect on tumor progression and final outcome. 98 Approximately 3% of all studies have investigated therapeutic predictive markers of FMTs. 18,31,32,38,46,104 In addition, most published prognostic factors of FMTs have been evaluated through univariate analyses (ie, Kaplan Meyer curve). Multivariate analyses may be lacking or result in loss of prognostic significance of the investigated parameter (Table 1). 28
Significance of Prognostic Factors for Feline Mammary Tumors in Retrospective Studies Based on Type of Statistical Models.
Abbreviations: AgNORs, argyrophilic nucleolar organizer regions; COX-2, cyclooxygenase 2; DFS, disease-free survival; ER, estrogen receptor; HER2, human epidermal growth factor receptor; HVC, high vessel count; LN, lymph node; MV, multivariate analysis (more prognostic variables assessed simultaneously for their effect on 1 dependent survival variable); MVD, microvessel density; OS, overall survival; PR, progesterone receptor; PTEN, phosphatase and tensin homolog; UV, univariate analysis (1 prognostic variable assessed individually for its effect on 1 dependent survival variable); VEGF, vascular endothelial growth factor; VEGRF2, vascular endothelial growth factor receptor 2.
aStaging system by Owen et al. 73
bStaging system by Rutteman. 83
cConservative vs radical mastectomy.
dTime between detection and surgery.
eMost studies considered clinical evaluation before excision; see text for details.
gLymphovascular emboli and/or lymph node metastases.
hSix nuclear morphometric parameters were used.
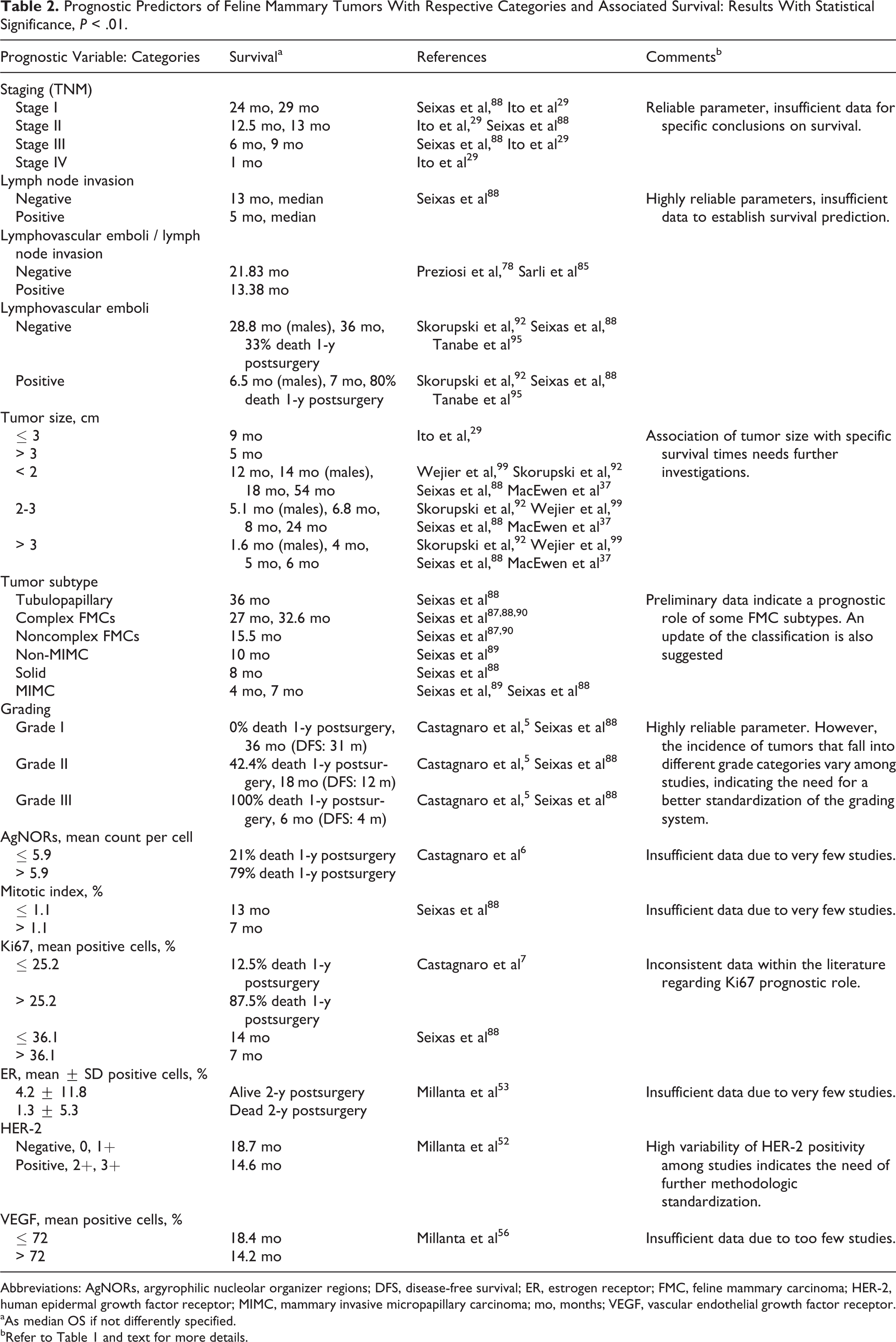
We conducted a detailed literature review of FMT and evaluated the different postulated prognostic parameters (Table 1) according to the recently published “Recommended Guidelines for the Conduct and Evaluation of Prognostic Studies in Veterinary Oncology.” 98 These guidelines reflect the current consensus of veterinary pathologists and oncologists on how to assess the best prognostic parameters for animal neoplasms. Using these criteria, we determined which prognostic parameters were statistically significant, and we provide recommendations for the prediction of behavior of FMTs (Table 2).
Prognostic Predictors of Feline Mammary Tumors With Respective Categories and Associated Survival: Results With Statistical Significance, P < .01.
Abbreviations: AgNORs, argyrophilic nucleolar organizer regions; DFS, disease-free survival; ER, estrogen receptor; FMC, feline mammary carcinoma; HER-2, human epidermal growth factor receptor; MIMC, mammary invasive micropapillary carcinoma; mo, months; VEGF, vascular endothelial growth factor receptor.
aAs median OS if not differently specified.
bRefer to Table 1 and text for more details.
Signalment: Subject-Related Parameters
Age
The mean age for the development of FMTs was between 10 and 12 years, with increasing risk up to 14 years of age. Carcinomas were more likely than benign mammary tumors to occur in older cats. 26,28,60,99 While there is some indication that prognosis is worse in older cats, the data are inconsistent. A cutoff value of 10 years was not associated with recurrence, disease-free interval, or survival time. 29,37 Age also had no significant association with 1- and 2-year postsurgery survival (51 and 48 cats, respectively). 6,55 Age was not a significant prognostic predictor for FMTs in male cats. 92 Conversely, in one study, age was related to survival, with decreased survival rates in older subjects; however, the study consisted of a large set of variables (n = 35) in 220 tumors, which lowered the overall power of analysis. 99 In this particular study, the method for collection of survival data was not precisely described, and mortality rate and results for all 35 variables were not reported. 99 Old age associated with shorter survival (DFS and overall survival [OS]) was found in one study based on univariate analysis; however, prognostic significance was lost on multivariate analysis. 88
Gender and Reproductive Status: Intact/Spayed
While queens are most commonly affected, mammary tumors have also been described in male cats 2,28 –30,34 –36,76,92,94 where their aggressive clinical behavior appears to be similar to that reported in females. 92 There is no difference in the biological behavior of FMTs in intact versus spayed cats. 29,99 One study indicated a 91% and 86% reduction in the risk of developing FMTs in cats spayed before 6 months and 1 year of age, respectively. 72 An increased risk of mammary tumor development is observed in female 24,58 and male 92 cats under regular and prolonged progestin treatment.
Breed
Siamese cats are at increased risk for developing mammary carcinomas (relative resk = 2) 27 and have a higher risk of developing mammary tumors at a younger age (9 years) than other breeds (14 years). 26 In another study, Siamese cats also commonly developed mammary invasive micropapillary carcinomas (MIMC) and were the second-most represented breed (34%) after domestic shorthair cats (60%) for developing invasive feline mammary carcinomas (FMCs) of other subtypes. However, no data were included for reliable incidence studies regarding the frequencies of the different breeds within the feline population examined. 89 Longhair breeds (Siamese and Persian, 36 of 202 subjects) had a poorer prognosis when compared to all other breeds in univariate analysis, but this significance was lost on multivariate analysis. 99 Breed was not prognostically significant in male cats with mammary tumors. 29,92
Clinical Parameters
Clinical Staging
Staging of FMCs is based on the TNM classification of malignant tumors system 73 and is widely accepted in clinical settings despite slight variations in how the system has been applied. 19,34,83,88 There is variability in the methodology of tumor measurement and application of size categories, as well as controversies on how and which lymph nodes should be evaluated, all of which are indications that there needs to be standardization of the TNM system for FMTs. 4,5,19 Staging has infrequently been included in reports describing prognosis of FMCs, since information about the size of the tumor and the presence of distant metastases are often unavailable. For this reason, tumor size and lymph node status are frequently treated as separate variables. In spite of these limitations, the prognostic significance of clinical staging of FMCs has been investigated (Table 2). In a univariate analysis of a cohort of 53 cats, the median OS of animals with stage I, II, III, and IV FMTs was 29, 12.5, 9, and 1 months, respectively. 28 Similarly, based on a slightly modified staging system, (stage I, II, and III), 83 clinical stage was significant for predicting DFS (12, 14, and 3 months; 39 cases) and OS (24, 13, and 6 months; 64 cases). 88 Via univariate analysis, DFS was significantly affected by lymph node invasion in stage III FMTs, whereas tumor size (< 2 cm and 2–3 cm) used to differentiate stage I from stage II FMTs did not affect DFS, and multivariate analysis found that staging was not a significant independent prognostic parameter. 88
Lymph Node Status and Distant Metastases
Numerous studies on FMT cats with metastatic and nonmetastatic disease at the time of diagnosis were included in the population evaluated in this study, which may significantly affect evaluation of prognostic factors. Most reports do not indicate how, or if, animals were evaluated for the presence of metastases and if the presence of metastasis was a selection criterion. 5,7,56,99 Lymph node metastasis, assessed by histologic evaluation of the draining lymph node, was an independent prognostic factor. 88 Lymph node invasion was independently associated with OS (< 9 months) in 64 cats with mammary carcinomas but not with DFS, although the latter information was available for fewer cases (39 of 64). 88 In other studies, lymph node invasion was associated with a worse OS; however, in these studies, lymph node invasion was evaluated in conjunction with lymphovascular invasion, making it difficult to determine whether these two parameters were independent predictors of OS. 77,78,84,85
Type of Surgery
Cats with nonmetastatic mammary carcinomas treated with radical mastectomy had a longer DFS than did those undergoing more conservative surgery that consisted of excision of the involved and immediately adjacent mammary glands. 37 Similarly, the surgical procedure was statistically significant for OS in a univariate analysis of 202 cats with mammary carcinomas, but significance was lost on multivariate analysis. 99 No significant differences in OS were found between cats undergoing partial versus radical mastectomy in a study of 53 queens or in 27 male cats with FMCs. 29,92
Pathologic Parameters: Gross and Histologic
Tumor Size
Tumor size, based on tumor diameter, is included in the TNM staging system and is of prognostic significance in FMTs. 34,73,83,88,97 In older studies, tumor size has been commonly treated as a continuous variable. 37,73,99 However, information on how tumor measurement was taken is often lacking (ie, by clinician, after fixation, after processing on the histologic slide). 29,37 Some studies included tumor volume (1–8 cm3, 9–27 cm3, and > 28 cm3) rather than tumor diameter. Tumor volume correlated with DFS of > 1600 days, 1200 days, and 800 days and OS of > 2500 days, 1300 days, and 1000 days. 37 In one study of 202 FMCs, both tumor diameter and tumor volume, measured clinically and included as continuous variables, were significantly correlated to OS based on univariate analysis; however, only tumor diameter remained an independent prognostic variable upon multivariate analysis. 99 Since tumor volume is more difficult to measure and requires evaluation of completely excised, nonsectioned fresh tissue, later studies chose tumor diameter for prognostic evaluation. A cutoff of 3-cm tumor diameter was found to be prognostically significant for OS; cats with larger tumors (> 3 cm) had a mean OS of 5 months, compared to a mean OS of 9 months for cats with smaller tumors. 29 Recently, tumor diameter classified as < 2 cm, 2–3 cm, and > 3 cm, measured at the time of clinical evaluation, was prognostically significant in the univariate analysis of 64 cats with FMCs, but this significance was lost on multivariate analysis (see Table 1), 88 or the OS times were different (see Table 2). 92,99 Large tumor size was also a negative prognostic predictor in male cats with mammary carcinomas. 92 Standardized methodology should be applied, and even though tumor size has been established as a prognostically significant parameter, size categories or cutoff values were not statistically established by receiver operator curves but rather arbitrarily selected.
Tumor Subtype
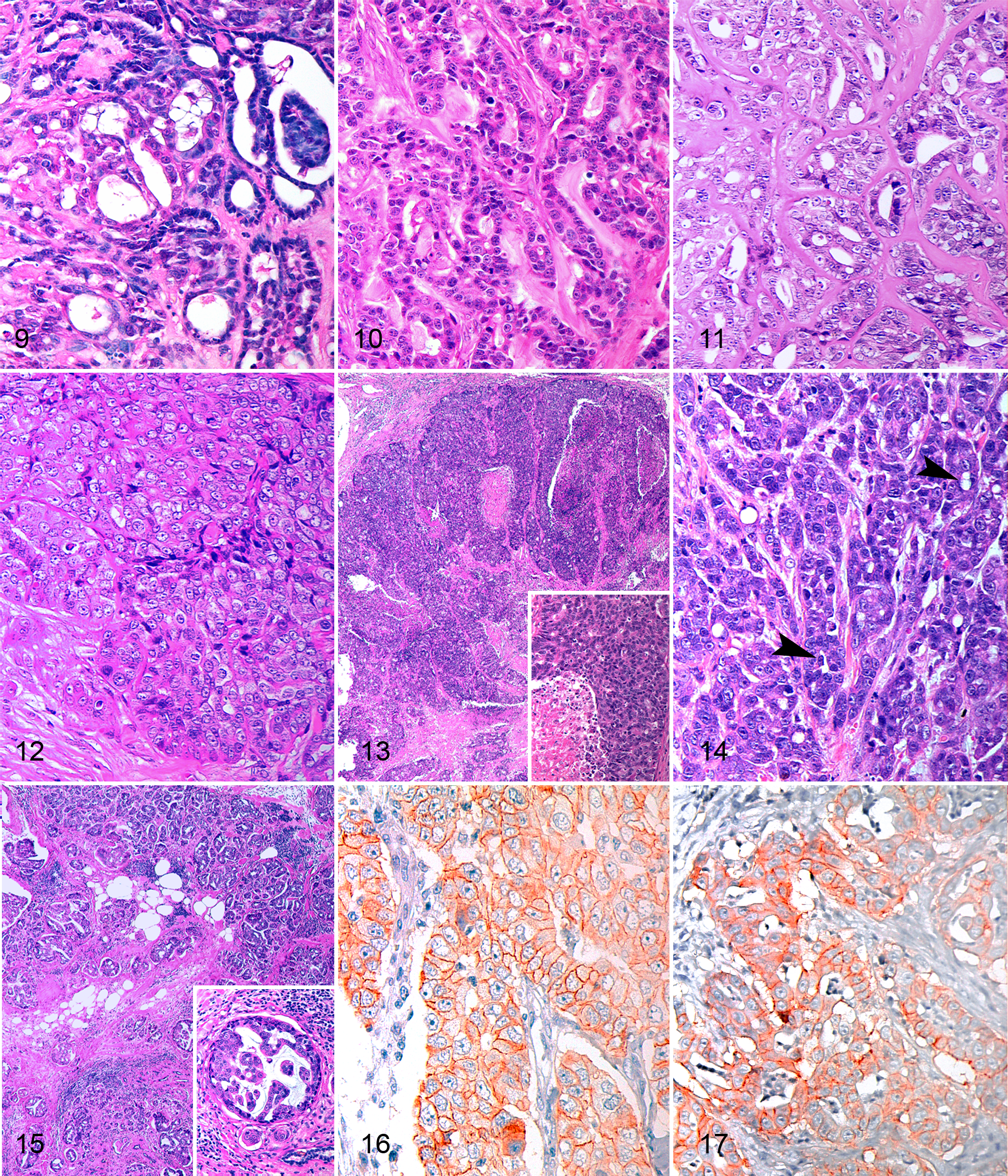
In human breast cancer, histopathologic examination remains the gold standard for diagnosing the tumor and predicting patient outcome. 16 The morphologic diagnosis represents the initial fundamental step and has to follow a standardized classification system to ensure accurate evaluation of markers that may predict tumor behavior. 98 Currently, FMTs are classified according to World Health Organization guidelines, 59 but some recent modifications have been suggested (see Figs. 1–17; specifically, Fig. 1, Figs. 4–6, and Figs. 9–15 for examples of common and more recently identified tumor subtypes). “Complex” mammary carcinomas in female cats have been recently reported 87,89,90 ; however, these complex FMCs do not have the myoepithelial proliferation that typically characterizes their canine counterparts but rather consist of tubules and nests of luminal epithelial cells subtended by cuboidal or fusiform, clear or eosinophilic, myoepithelial cells that stain with calponin and p63. 59,87,90 These neoplasms have recently been reclassified as feline mammary ductal adenomas/carcinomas rather than “complex” carcinoma (Figs. 1, 4–6). 105 A second entity—feline inflammatory mammary carcinoma—has been recognized and has many similarities to its human and canine counterparts. 75 Furthermore, feline MIMC (Fig. 15) has been described as a specific entity in 16 queens, 89 and a case of lipid-rich carcinoma has been reported with no evidence of metastasis. 33
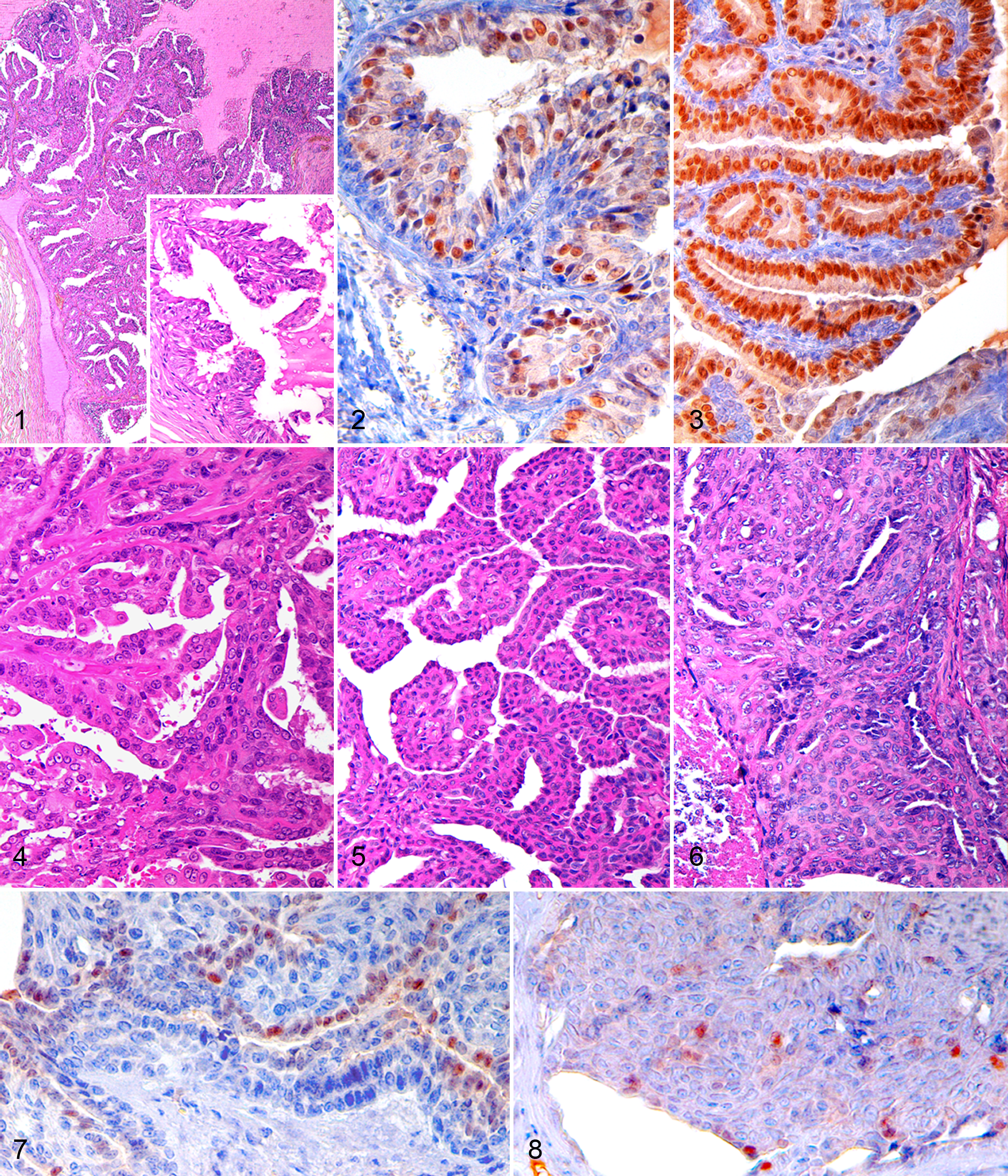
Some authors have determined that morphologic subtypes of FMCs were of no prognostic value. 6,55,59 Yet, the prognostic significance of several recently described FMCs subtypes has been evaluated, 87,89,90 and cats with MIMCs had significantly shorter median OS (4 months) and median DFS (5 months) when compared to cats with other FMTs (10 months both for median OS and median DFS). 89 An MIMC in a male cat also demonstrated aggressive behavior (OS of 10 months and DFS of 7.5 months). 21 Feline ductal carcinomas (complex carcinomas) had a mean OS of 32.5 months and were significantly less aggressive than simple tumors. 87,90 In a recent study of 64 cats that had clinical follow-up data for 2 years postdiagnosis, cats with tubulopapillary and complex mammary carcinomas had a significantly longer OS compared to cats with solid carcinomas and MIMCs. 88 Similarly, DFS was much longer in cats with complex carcinomas; however, the significance of the tumor subtype was not maintained upon multivariate analysis. 88
Grading
The most widely accepted grading system for FMCs was adapted from the grading system for human breast cancer 14 and is based on 3 parameters: pleomorphism, tubule formation, and mitotic count. Lack of standardization about how these parameters are precisely assessed and interobserver variability have resulted in inconsistent assignment of grades among FMC cohorts. Pleomorphism may include evaluation of the neoplastic cell or may be restricted to evaluation of nuclear morphology, yet no details have been provided regarding how this parameter should be assessed. Mitoses have been variably counted at the periphery of the tumor, in the most mitotically active areas of the tumor, in random or consecutive fields. 5,15,55,88 Examples of the variable morphology for the 3 grades are represented in Figures 9–11. As an example, the differential diagnosis between an adenoma and a representative grade I carcinoma (Fig. 9) might be highly influenced by a nonstandardized subjective evaluation.
The degree of tumor differentiation in conjunction with tumor size and lymph node status has been considered prognostically useful in human breast cancer and canine mammary tumors. 20,79,81 Similarly, in FMCs, grade was associated with 1- and 2-year postsurgical outcomes. 5,55,74,88 In one of these studies, 55 feline infiltrative mammary carcinomas were analyzed and classified into grades I, II, and III according to the World Health Organization classification (12.7%, 60%, and 27.3%, respectively). 5 Mitoses were counted at the periphery of the tumors, where cell proliferation was likely to be higher. Only 1-year postsurgical survival was assessed through univariate analysis, and there was a significant difference in the 1-year postsurgery survival in grade I (0% death), grade II (42.4% deaths), and grade III tumors (100% death). 5 Similarly, grade was predictive of survival for 48 feline invasive mammary carcinomas in 2 studies, with 33.3% of the study animals having grade I tumors, 47.9% with grade II, and 18.7% being grade III FMCs. 53,55 COX regression analysis identified grade as the strongest independent predictor of survival (DFS and OS) in a 2-year postsurgery follow-up study. 88 Of 92 carcinomas analyzed, 5.4% were grade I, 46.7% grade II, and 47.8% grade III. Cats with a grade III mammary carcinoma had a median OS of 6 months and a median DFS of 4 months, while cats with grade I tumors had an OS of 36 months and a DFS of 31 months. 88
It is important to recognize that detection of lymphovascular neoplastic emboli is not included in the grading of FMCs. It is not clear based on previous studies if and how many grade III tumors had lymphovascular invasion and if neoplastic emboli were found in any grade I or II FMCs. Lymphovascular invasion should be used as a morphologic feature to warrant inclusion into grade III FMCs.
Other Morphologic Parameters
Other relevant histologic parameters that have been considered for their prognostic role include nuclear and cellular atypia (pleomorphism), necrosis/apoptotic index, cutaneous ulceration, lymphoplasmacytic infiltrates, infiltrative growth, lymphovascular emboli, adhesion to the underlying tissue, and tumor margins.
In one study, pleomorphism, necrosis, chronic inflammation around the tumor, lymphatic emboli, infiltrative growth, incomplete surgical margins, and ulceration, together with the mitotic count, were predictive of survival (DFS or OS). 99 The extent of necrosis and incomplete surgical margins retained their prognostic value after multivariate analysis. Lymphatic emboli were strongly associated with DFS (5 months when present, compared to 14 months when absent) and OS (7 months when present, compared to 36 months when absent) based on univariate and multivariate analyses. 88,95 In 27 male cats with mammary carcinomas, the median OS was 29 months when neoplastic emboli were absent, compared to 6 months in cases with lymphovascular invasion. 92 In some multivariate analyses, lymphatic invasion of neoplastic cells lost prognostic significance when cats with metastatic tumors were included. 99
The apoptotic index (TUNEL) was not significantly associated with OS. 85 “Nuclear grade” was specifically evaluated, as assessed by image analysis for nuclear enlargement, anisokaryosis, and nucleolar abnormalities, but only nuclear shape was significantly associated with 1-year postsurgical survival. 11
Histochemical and Immunohistochemical Parameters
Proliferative Markers
The growth fraction is generally assessed by the percentage of Ki67-positive neoplastic cells and the proliferation time by the mean AgNOR (argyrophilic nucleolar organizer region) count per neoplastic cell. PCNA count can be also applied as a measure of the growth fraction (S-phase index). The mitotic index represents a phase index.
The mitotic index—the number of mitoses per 10 fields (400×)—has been considered predictive of survival (DFS or OS) in univariate and multivariate analyses. 85,88,99 In one study in which a mitotic rate was prognostic via univariate analysis, 88 the authors reported that if the mitotic rate was below 1.1%, the OS was 14 months, whereas if the mitotic rate was above 1.1%, OS was 7 months. The DFS was 12 months when the mitotic rate was below 1.09%, and it was 5 months when above 1.09%. Similarly, mitotic index was significantly associated with OS in 33 nonmetastatic FMCs. 85
A Ki67 value < 25.2% of positive neoplastic cells (median value of all cases, calculated as positive nuclei in 1000 neoplastic cells) was significantly associated with increased survival in a 1-year postsurgery follow-up study (Table 2). 7 Similar results were found by other authors. 81 Several other studies investigated the prognostic role of the Ki67 index in FMCs with discordant results. 55,78,88 In a 2-year postsurgery follow-up study, Ki67 was an independent prognostic parameter for OS but was not significantly associated with DFS. 88 OS was 7 months with Ki67 > 36.1% and 14 months with Ki67 < 36.1%. 88 In 2 studies of 48 and 33 cats with mammary carcinomas, the Ki67 indices were not prognostically significant. 55,78 The variation may reflect exclusion of low-grade tumors in some studies. 55
The prognostic significance of the AgNOR count—expressed as mean number per neoplastic cell in 100 cells at 100× magnification—was evaluated in 51 FMCs in a 1-year postsurgical follow-up study. 6 No indication of distant metastases was reported. Fewer than 5.9 AgNORs per cell was significantly associated with longer survival.
Hormone Receptors and HER-2
The reported data on ERα and PR expression in FMTs are scarce and controversial, possibly because of the use of different techniques, protocols, scoring systems, statistical analyses, as well as variability in reporting results (see Figs. 2, 3, 7, and 8 for examples of ER and PR expression in FMTs).
Specific and robust data on ER and PR as prognostic indicators in FMTs are lacking (Tables 1 and 2). Only 2 retrospective studies on 7 cats with invasive FMCs found no correlation between ER and PR expression and OS in a 2-year follow-up period. 51,53 In these studies, ER-negative FMCs had a higher death rate when compared to ER-positive FMCs. Cats that were alive 2 years after surgery (31.9%) had a significantly higher percentage of ER-positive neoplastic cells (4.2% ± 11.8%) compared to cats that died (1.3% ± 5.3%). 51,53 Conversely, in a study on 39 female cats, ER and PR were not significantly correlated with survival. 39
There is significant variability in HER-2 immunohistochemical expression in FMCs. 4,10,39,52,71,80,102 These discrepancies are likely related to technical factors and problems related to the interpretation of immunohistochemical results. 39,71,80 Figures 16 and 17 are examples of HER-2 expression in FMTs.
The role of HER-2 in feline mammary carcinogenesis is also contradictory. Rasotto et al did not demonstrate any significant correlation between HER-2 expression and the 1-year clinical outcome, 80 whereas Millanta et al found HER-2 expression correlated with a shorter OS in a 2-year follow-up study: the average OS was 18.7 months for HER-2-negative FMCs and 14.6 months for those overexpressing HER-2 (Tables 1 and 2). 52 A larger multilaboratory prognostic study might help to better elucidate the role of this marker in FMCs and determine its significance as a prognostic marker.
In human medicine, classic morphologic classification systems are yielding to new schemes based on molecular and immunophenotypic markers. 63 These newer classification schemes have divided human breast cancers into prognostically significant categories, including ER-positive luminal A and B subtypes, HER-2 overexpressing subtype, normal breast, and basal-like subtype. 93 A variety of immunohistochemical markers have been proposed to define the basal-like subtype. 1 Expression of one or more high molecular weight basal cytokeratins (CK5/6, CK14, and CK-17) 93 and/or epidermal growth factor receptor are most commonly accepted to identify basal-like differentiation. 69 A large percentage of basal-like human breast cancers lack expression of ER and PR and do not overexpress HER-2 (“triple negative” immunophenotype). 8 Such triple-negative breast cancers exhibit aggressive behavior and are further complicated by the lack of targeted therapy. Furthermore, in humans, breast cancer susceptibility 1 (BRCA1) and 2 (BRCA2) genes are mutated in a large number of early-onset breast and ovarian cancers. 50,103 Breast cancers with BRCA1 mutations and, less commonly, BRCA2 mutations have a triple-negative immunophenotype and a distinct basaloid morphology. 17 More than 75% of tumors in women with BRCA1 mutation have a triple-negative immunophenotype, a basal-like subtype, or both. 96
In contrast to humans, while aberrant expression of ER and PR and overexpression of HER-2 have been reported, the majority of FMTs are ER negative 4,101 and lack HER-2 overexpression when a protocol is applied strictly matching the Food and Drug Administration–approved test in humans, 80,101 potentially suggesting a more aggressive behavior in a large percentage of feline tumors. However, only a few studies have evaluated triple-negative FMCs. 28,101 Interestingly, in a recent study, immunohistochemistry (IHC) for ER, PR, and HER-2 identified 14 of 24 (58%) FMCs as triple negative, and 11 of the 14 (79%) had a basal-like subtype appearance. 101 While sequencing of a subset of these triple-negative basal-like FMCs and testing for loss of heterozygosity of BRCA1 and BRCA2 did not identify mutations similar to those described in humans, further in-depth evaluation is needed to elucidate a potential role of BRCA in tumorigenesis and its prognostic significance. 101 Despite the fact that adequate prognostic data are lacking, the ability to classify FMCs using similar molecular and immunophenotypic markers as described for human breast cancer suggests the potential comparative, prognostic, and therapeutic value of applying such systems.
p53
p53 mutations have been identified in FMCs, but a significant correlation with aggressive behavior has not been established. 47 –49 Different immunohistochemical protocols, scoring systems, and the absence of a standardized cutoff value for p53 positivity may be responsible for this variability. 62,64,66,67,80
In one study with follow-on data, there was no association between p53 mutations and 1-year postsurgical survival. 80 Furthermore, no significant correlation has been found between p53 expression and histologic grade, ER status, Mdm2, cyclin A, cyclin D, TopBP1 (topoisomerase IIb binding protein 1), and HER-2 expression (Table 1). 62,66,80 Standardization of the IHC protocol and scoring system and more extensive follow-up studies are required to evaluate the role of p53 in FMCs.
Other Markers
The role of cyclooxygenase-2 (COX-2) as an indicator of tumor aggressiveness and prognosis has been documented in FMCs. 54 When COX-2 is used as a therapeutic target, controversial results have been obtained on the basis of COX-2 expression and its predictive significance as a response to anti-inflammatory therapies. 3,9,12,13,86 As such, standardization of IHC protocols for COX-2 is necessary. COX-2 and VEGF were evaluated in association with other parameters (microvessel density, VEGFR2, Ki67; HER-2, CD31, von Willebrand Factor, ER, and PR). 51,53,54,56,57 High COX-2 expression was significantly associated with shorter OS. 54 VEGF was directly associated with survival: OS averaged 18.4 months for FMCs with <72.1% VEGF-positive cells, whereas OS was 14.2 months for those carcinomas with >72.1% VEGF-positive cells. VEGF expression was also significantly higher (>72.1% VEGF-positive neoplastic cells) in those animals that died by the end of the 2-year follow-up period in comparison with those that were still alive (<72.1% VEGF-positive neoplastic cells). 56 In these studies, microvessel density and VEGFR2 (flk-1/KDR) expressions were also assessed, but they did not correlate with survival. 56,57
Two studies investigated the deregulation of the PI3K/AKT/PTEN pathway in FMTs. PTEN expression was investigated by IHC in 17 FMCs but was not a significant prognostic factor. 82 In a second study, 39 FMTs (27 FMCs and 12 benign FMTs) were followed for 2 years, and p-AKT positivity was significantly associated with a shorter DFS. 39
A single study on 15 FMCs evaluated the expression Sialyl Lewis X as a ligand of E-selectin. 65 Sialyl Lewis X was expressed in 60% of FMCs, but it was associated neither with survival nor with other tumor parameters tested. 65
Finally, one study investigated the prognostic role of the chemokine receptor CXCR4 in 65 FMCs by in situ hybridization. 95 This receptor is overexpressed in human breast cancer and described as possibly directing the organ-specific metastasis of neoplastic cells. CXCR4 expression was not associated with 1-year postsurgery survival in FMTs. 95
Conclusions
Given our review of the literature on FMTs, we found 2 areas of concern that need to be addressed. The first concerns methodology. In veterinary oncology, particularly with cats, it is often difficult to follow the diseases as they progress, and as such, most studies are retrospective and lack significant data associated with survival. There is variability in terms of study design, and this weakens the significance of the results. Efforts need to be made to apply the American College of Veterinary Pathologists’ recommended guidelines and to set up prospective investigations that fulfill these guidelines. 98
The second area concerns those parameters prognostic for FMTs. Lymph node involvement, lymphovascular invasion, tumor size, and tumor grade (including mitotic count) are the most reliable parameters, but histologic tumor subtype and clinical staging require further study as prognostic indicators. Before being accepted as valid, there first needs to be consensus on the classification of FMTs, along with standardization of those parameters that may be of prognostic significance. An updated FMT classification with detailed descriptions is required, as was recently done for canine mammary tumors. 20 In addition, specific survival times should be reported for each studied variable, in contrast to the current state of affairs in which studies fail to report survival correlations (Table 2).
Tumor grading is one of the most reliable prognostic parameters and is significant for DFS and OS in multivariable analysis. Studies that compared tumor size found that smaller tumors are correlated with better prognosis than larger tumors; however, the methods for measuring and categorizing tumor size as part of the TNM grading system, as well as related survival rates, need to be standardized. In addition, lymphovascular invasion and lymph node involvement are also extremely important prognostic indicators that must be further evaluated when biopsy samples are examined, as they are integral to a useful grading system. Data on regional lymph node status are not often included in retrospective FMC studies. Lymph nodes should be evaluated for metastases via standard methodology (ie, cytology, histology). The lack of these data may significantly affect prognosis and median survival. 98 By standardizing future retrospective and prospective studies, preferably in conjunction with veterinary oncologists, we will be able to establish those factors that are of prognostic importance and expand the molecular evaluation of FMTs. This would allow targeted specific therapies to be used and would enhance the value of FMTs as a model of breast cancer in women.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
