Abstract
Cutaneous mast cell tumors (cMCTs) account for approximately 20% of skin neoplasms in cats. As there is no grading system for these tumors, prognosis is difficult to estimate. Although the typical presentation is a benign tumor that can be cured by surgical excision, a small but important proportion of feline cMCTs is biologically aggressive and can spread to local lymph nodes, precede the onset of disseminated cutaneous disease, or be associated with visceral involvement. A number of macroscopic and histologic features were retrospectively evaluated in cases of feline cMCTs treated with surgical excision with or without medical therapy. Cats were divided into 2 groups based on the clinical outcome. Group 1 included cats alive with no mast cell tumor–related disease at 1000 days from surgery; group 2 included cats developing histologically confirmed metastatic or cutaneous disseminated disease. The criteria allowing the best differentiation between the groups were used to develop a grading scheme. Groups 1 and 2 were composed by 48 (76%) and 15 (24%) cases, respectively. Tumors were classified as high grade if there were >5 mitotic figures in 10 fields (400×) and at least 2 of the following criteria: tumor diameter >1.5 cm, irregular nuclear shape, and nucleolar prominence/chromatin clusters. According to this scheme, the 15 (24%) high-grade cMCTs had significantly reduced survival time (median, 349 days; 95% CI, 0–739 days) as compared with the 48 low-grade tumors (median not reached; P < .001). Further studies are warranted to validate this grading system and test reproducibility on a larger case series.
Cutaneous mast cell tumors (cMCTs) represent the second-most frequent skin neoplasm in cats, accounting for approximately 20% of feline cutaneous tumors. 1
The majority of feline cMCTs are clinically benign, but up to 22% of cases may show an aggressive behavior and ultimately lead to lymph node metastases, visceral organ involvement, and/or cutaneous dissemination. 1
There is currently no grading system for feline cMCT, and the identification of tumors with malignant behavior remains a challenge. The mitotic activity is regarded in the majority of studies as the only reliable histologic parameter to predict prognosis; 4,8,10,13,15,16 nevertheless, a significant degree of overlap in mitotic index was observed between benign and malignant cMCTs, 13 suggesting that a combination of multiple parameters might allow a better differentiation.
The attempts to identify further prognostic indicators were unconvincing. These included classification into subtypes, 5,8,20 growth pattern, 7 application of the Patnaik grading system, 3,14 evaluation of cytoplasmic granules, and characterization of the accompanying inflammatory infiltrate. 3,15 Similarly, the evaluation of molecular parameters, such as Kit receptor dysregulation and the detection of cell cycle markers, yielded poorly reproducible results. 4,16
The aim of the current study was to select a number of parameters that, when combined into a basic grading system, could allow a satisfactory identification of feline cMCTs with an aggressive biological behavior.
Materials and Methods
Case Selection
Cases were retrieved from the medical database of the Department of Veterinary Medical Sciences, University of Bologna, Italy. Cats with a history of surgically removed cMCT with available clinical and follow-up information were considered for inclusion in the study. Required information included demographic data, tumor characteristics (location, number, largest diameter), status of surgical margins, development of local recurrence or other histologically confirmed primary or metastatic mast cell tumors (MCTs) following surgery, progression-free interval (PFI), overall survival (OS), and cause of death. Cats with multiple cMCTs were eligible for inclusion only if all the nodules had been surgically removed and were available for histologic examination.
Cases were divided in 2 groups based on the clinical outcome. Cats alive and free from MCT-related diseases at 1000 days from surgery were classified in group 1, whereas cases were classified in group 2 upon the presence of at least 1 of the following: (1) microscopically confirmed lymph node metastasis (ie, histologically or cytologically), (2) microscopically confirmed visceral involvement following cMCT removal, and (3) onset of microscopically confirmed cutaneous disseminated mast cell disease (>10 nodules). Cats that had an uncertain cause of death or were deceased for causes unrelated to MCT within 1000 days from surgery were excluded from the analysis.
Histologic Examination
All histologic samples had been fixed in 10% neutral-buffered formalin and paraffin embedded at the time of submission. Four-micrometer sections were stained with hematoxylin and eosin and toluidine blue or Giemsa to confirm the previous diagnosis. For atypical cMCTs, immunohistochemical negativity to CD18 (1:20, clone Fe3.9F2; P. F. Moore, Leukocyte Antigen Biology Laboratory, University of California, Davis) and positivity to CD117 (1:100, rabbit polyclonal, A4502; Dako, Glostrup, Denmark) were also required for inclusion.
Hematoxylin and eosin–stained slides were blindly reviewed (S.S.). Based on the available literature, different histologic features were evaluated in each tumor, including growth pattern, subcutaneous invasion, histotype (Suppl. Figs. S1 and S2), nuclear:cytoplasmic ratio, irregular nuclear shape (nuclear pleomorphism), nucleolar prominence, chromatin clusters, anisokaryosis, multinucleated cells, pyknosis/karyorrhexis, mitotic count, neoplastic emboli, eosinophils, and lymphoid aggregates. Definitions of these parameters are provided in Table 1. In case of multiple cMCTs, the tumor with the highest grade was included in the survival analysis.
Histologic Parameters Evaluated in 63 Feline Cutaneous Mast Cell Tumors.

Development of the Grading System
The preferred characteristics of the grading system were (1) a 2-tier mode, with no intermediate grade; (2) a limited number of parameters and (3) an algorithmic approach, to simplify the application; (4) the use of numeric parameters, to improve reproducibility; and (5) the possibility of application regardless of the availability of peritumoral tissues.
For the development of the grading system, demographic information, tumor characteristics, and the histologic parameters were compared between groups 1 and 2. Analysis of variance and chi-square/Fisher’s exact tests were used to exclude the parameters that were not significantly different between the groups. For quantitative variables (tumor diameter, mitotic count), the optimal cutoff values between groups 1 and 2 were determined by receiver operating characteristic curves.
The parameters with statistically significant P values were combined in various algorithms, and these combinations were tested for their ability to correctly identify cats from group 2. Binary logistic regression was performed to confirm the ability of the selected parameters to distinguish group 1 from group 2 tumors.
The prognostic significance of the grading scheme was investigated by means of the Kaplan-Meier survival plots; the log-rank test was used to compare PFI and OS between low- and high-grade tumors. The prognostic utility of the grading system was compared with that of mitotic count alone by multivariable Cox regression analysis.
For survival analysis, the date of surgery, as the day of submission of the sample to the laboratory, was used as the entry point. Cats that developed further MCT-related disease (PFI) or were reported dead because of MCT (OS) were recorded as events. The remaining cats were censored to the date of death (if dead for unrelated causes) or to the last follow-up (if death had not occurred). Survival estimates are presented as medians with corresponding 95% CIs.
Statistical analysis was performed with SPSS 24 (IBM, Somers, NY). Significance was set at P < .05.
Results
Demographic, clinical, and histopathologic details of the 63 cases of feline MCT included in this study are provided as Supplemental Table S1.
Group 1
Forty-eight cases were included in group 1. Represented breeds were domestic shorthair (n = 40), domestic longhair (n = 3) ragdoll (n = 1), Siamese (n = 1), Persian (n = 1), Maine coon (n = 1), and Chartreux (n = 1). There were 26 males (54%) and 22 females (46%). The mean ± SD age was 8.5 ± 3.8 years. Nodules were single in 42 cases (88%), while the remaining cats had 2 (n = 5, 10%) and 5 (n = 1, 2%) nodules. Thirty tumors (53%) were located on the head (ear pinna, n = 9; eyelid, n = 6); 20 (35%) were on the trunk and 7 (12%) on the limbs. Three tumors (5%) were located in sites with a negative prognosis in dogs (digits, n = 2; lip, n = 1). Median diameter of the largest nodule was 1 cm (interquartile range [IQR], 0.5–1.3 cm). Surgical margins were clean (≥1 mm) in 39 cases (81%) and infiltrated in 9 (19%). Surgery was the only treatment in 44 cases (92%), whereas 4 cats received additional treatment (corticosteroids, n = 3; toceranib, n = 1).
At data analysis closure, 34 cats (71%) were alive with a median follow-up time of 1317 days (IQR, 1152–1531 days), whereas 14 cats (29%) had died of MCT-unrelated causes (chronic renal failure, n = 6; traumatic death, n = 2; squamous cell carcinoma, n = 2; fibrosarcoma, n = 1; lymphoma, n = 1; cholangiocarcinoma, n = 1; FIV, n = 1) after a median OS of 1412 days (95% CI, 1185–1939 days).
During the follow-up time, 5 cats (10%) developed a local recurrence of the excised tumor (median time to recurrence, 109 days; 95% CI, 9–324 days). Three had been diagnosed with infiltrated margins and 1 with clean margins. Nine cats (19%) developed other single (n = 6) or multiple (n = 3) cMCTs at distant sites. In the latter group, 2 cats developed 2 cMCTs, and 1 developed 6 other nodules. The median PFI was 252 days (95% CI, 30–520 days).
Group 2
Fifteen cats were included in group 2. There were 11 domestic shorthair, 2 Siamese, 1 Turkish Angora, and 1 Chartreux: 4 males (27%) and 11 females (73%). The mean age was 10.6 ± 3.5 years. At presentation, 12 cats (80%) had a single nodule, whereas 3 (20%) had 2 nodules. Two tumors (11%) were located on the head (periorbital region), 11 (61%) on the trunk, and 5 (28%) on the limbs. Three tumors (17%) were located in sites with a negative prognosis in dogs (inguinal region, n = 3). Median tumor diameter was 2.5 cm (IQR, 1.5–3 cm). Surgical margins were adequate in 11 cases (73%) and infiltrated in 4 (27%).
In 3 cats (20%), histologically confirmed regional lymph node metastases were present at diagnosis. In other 2 (13%), diagnosis of nodal metastasis was performed 95 and 384 days following cMCT removal. Two of these cats and 6 others (n = 8, 53%) developed the cutaneous disseminated form after a median of 141 days from cMCT removal. Nine cats (60%)—including 1 with lymph node metastasis, 2 with the cutaneous disseminated form, and 1 with both—were diagnosed with a splenic MCT after a median of 340 days from cMCT removal. Additional treatment was administered in 8 cases (53%) and included masitinib (n = 1), toceranib (n = 1), lomustine (n = 1), vincristine (n = 1), and corticosteroids (n = 6). Splenectomy was performed in 2 cats. Lymphadenectomy and surgical removal of additional cutaneous nodules were performed in 5 and 4 cases, respectively. Median PFI was 95 days (95% CI, 1–262 days). Fourteen cats died or were euthanized because of MCT. Only 1 cat with a cutaneous disseminated disease was alive and in clinical remission at 2580 days from surgery. Median OS was 349 days (95% CI, 0–739).
Statistical Analysis and Grading System
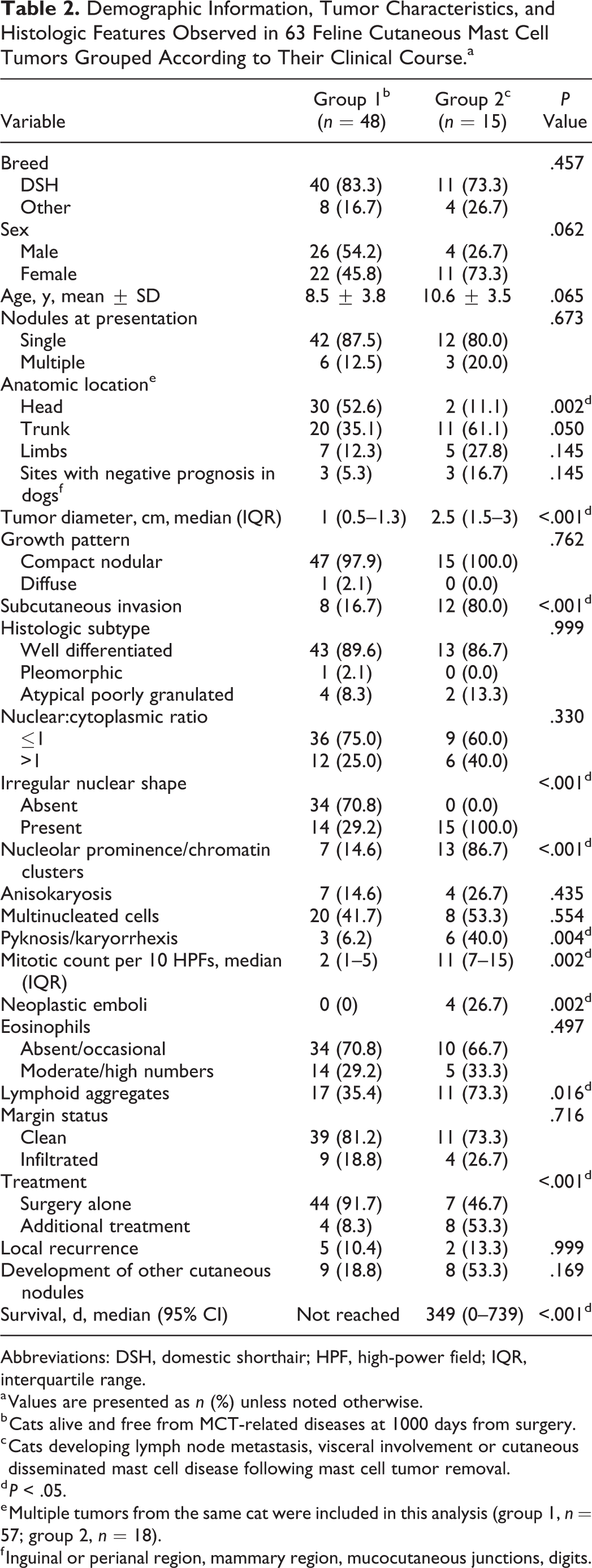
From the analysis of background information, the tumors with aggressive biological behavior were more likely to be larger (P < .001) and were rarely located on the head (P = .002; Table 2). Among histologic parameters, those significantly associated with group 2 tumors were subcutaneous invasion, irregular nuclear shape, prominent nucleoli or clusters of chromatin, presence of pyknosis or karyorrhexis, higher mitotic counts, and presence of vascular emboli and lymphoid aggregates. According to receiver operating characteristic curves, the best cutoff value for tumor diameter was 1.5 cm (sensitivity, 80%; specificity, 80%; area under the curve, 0.85; 95% CI, 0.73–0.96; P < .001), and the best cutoff value for mitotic count was 5 of 10 high-power fields (sensitivity, 100%; specificity, 79.17%; area under the curve, 0.92; 95% CI, 0.86–0.99; P < .001).
Demographic Information, Tumor Characteristics, and Histologic Features Observed in 63 Feline Cutaneous Mast Cell Tumors Grouped According to Their Clinical Course.a
Abbreviations: DSH, domestic shorthair; HPF, high-power field; IQR, interquartile range.
a Values are presented as n (%) unless noted otherwise.
b Cats alive and free from MCT-related diseases at 1000 days from surgery.
c Cats developing lymph node metastasis, visceral involvement or cutaneous disseminated mast cell disease following mast cell tumor removal.
d P < .05.
e Multiple tumors from the same cat were included in this analysis (group 1, n = 57; group 2, n = 18).
f Inguinal or perianal region, mammary region, mucocutaneous junctions, digits.
Based on these results, tumors were classified as high grade in the presence of >5 mitotic figures per 10 high-power fields and at least 2 of the following 3 criteria: tumor diameter >1.5 cm, irregular nuclear shape, and nucleolar prominence/chromatin clusters (Figs. 1–7; Suppl. Figs. S3–S6). The selected parameters all had statistically significant odds ratios favoring a diagnosis of high-grade tumor (Table 3). In the 9 cases (14%) with multiple cMCTs, the tumors from the same cat had a similar morphologic appearance and belonged to the same grade.
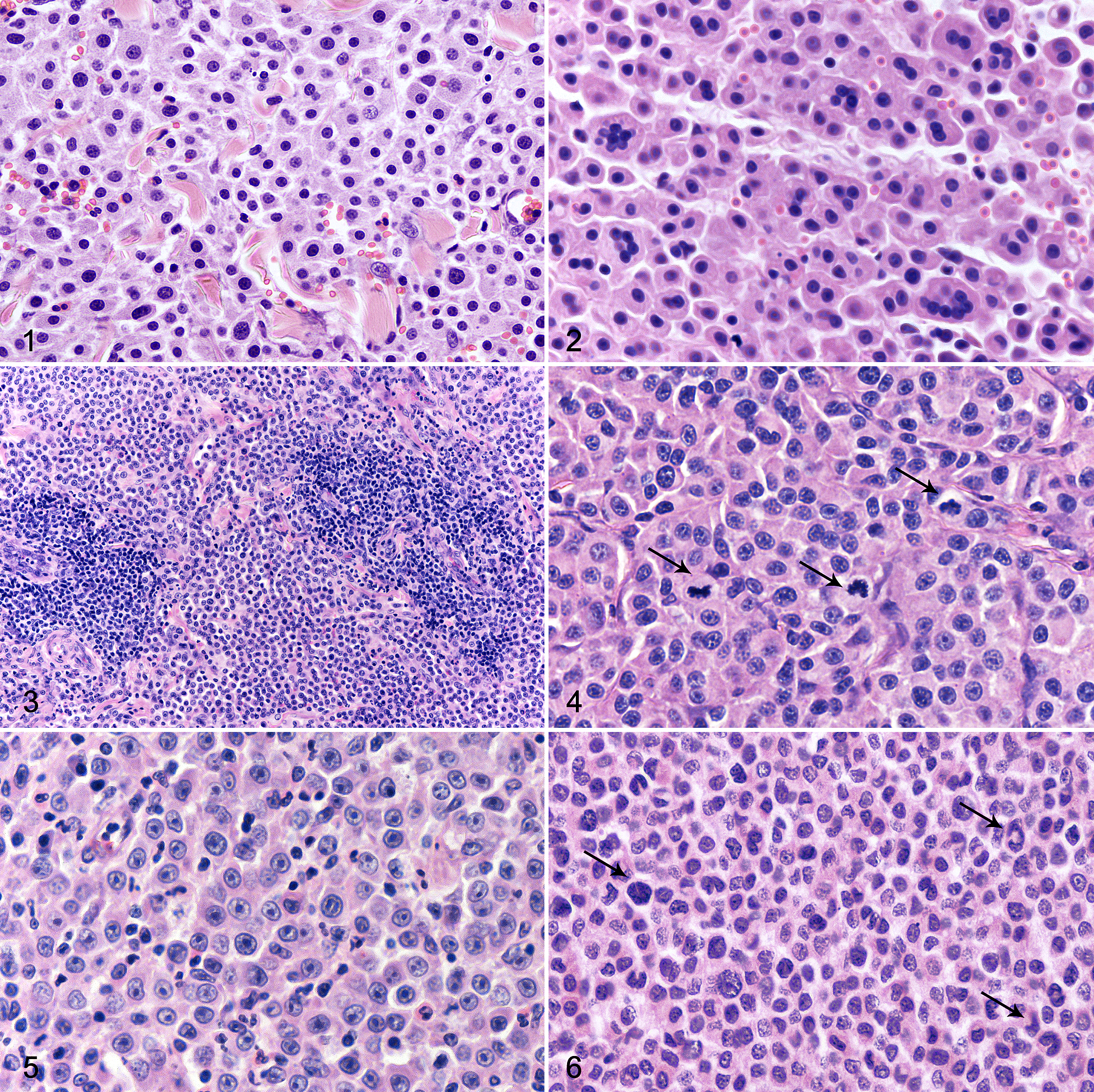
Mast cell tumor, skin, cat.
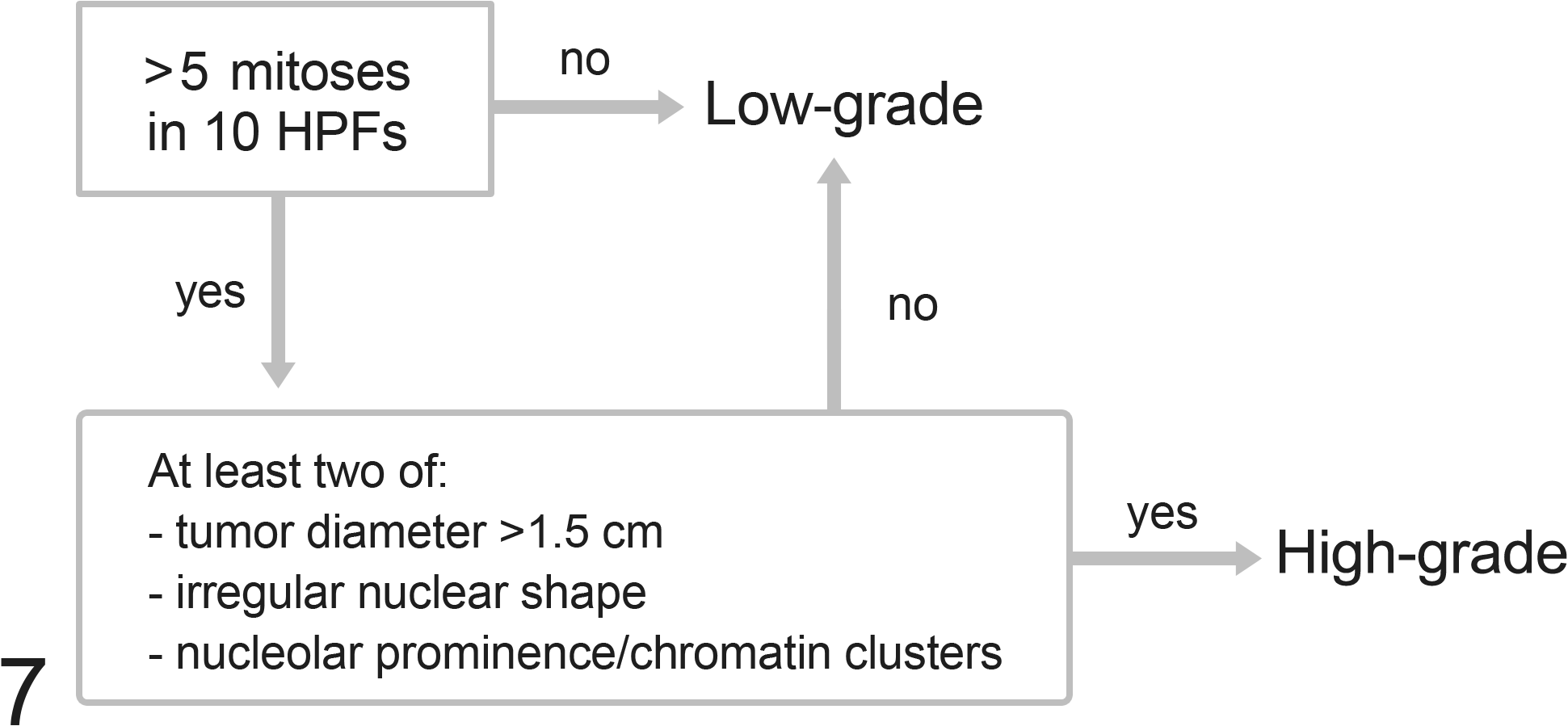
Algorithm of the grading system proposed for feline cutaneous mast cell tumors. Although not included in the grading system due to low frequency, a feline cutaneous mast cell tumor with vascular invasion should be considered high grade even in the absence of other histologic malignant features. HPF, high-power field.
Variables Favoring a Diagnosis of High-Grade Feline Cutaneous Mast Cell Tumor.
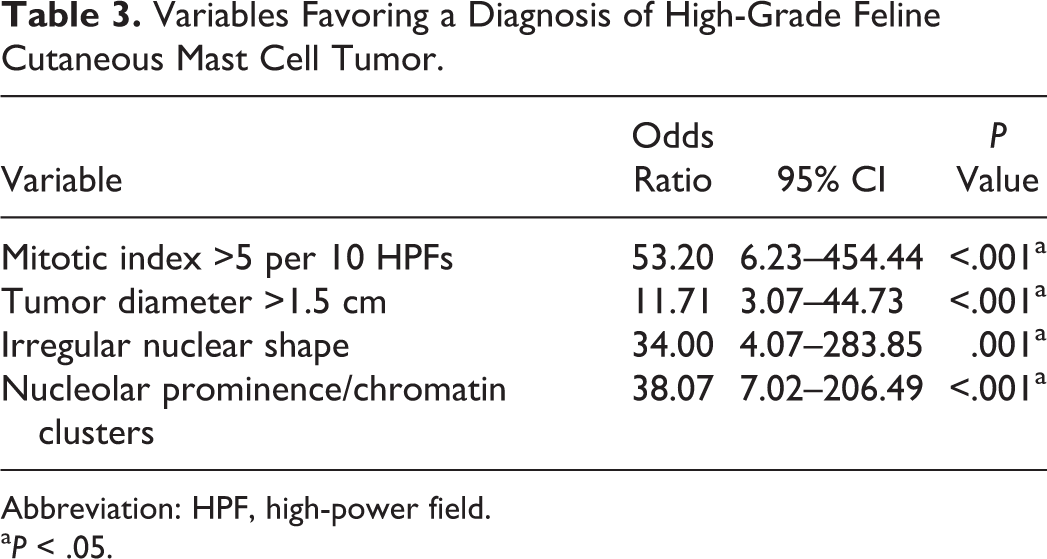
Abbreviation: HPF, high-power field.
a P < .05.
According to this grading system, there were 48 (76%) low-grade and 15 (24%) high-grade tumors. One cMCT from group 1 and all but 1 cMCT in group 2 were high grade. Notably, the only low-grade cMCT in group 2 belonged to the cat that was still alive at the end of the study.
Median OS was 349 days (95% CI, 0–739) for high-grade cMCTs and not reached for low-grade cMCTs (P < .001; Fig. 8). In a multivariable analysis model with the mitotic count, the grading system was the only parameter retaining prognostic significance (hazard ratio = 47.05, 95% CI = 4.96–445.85, P = .001).
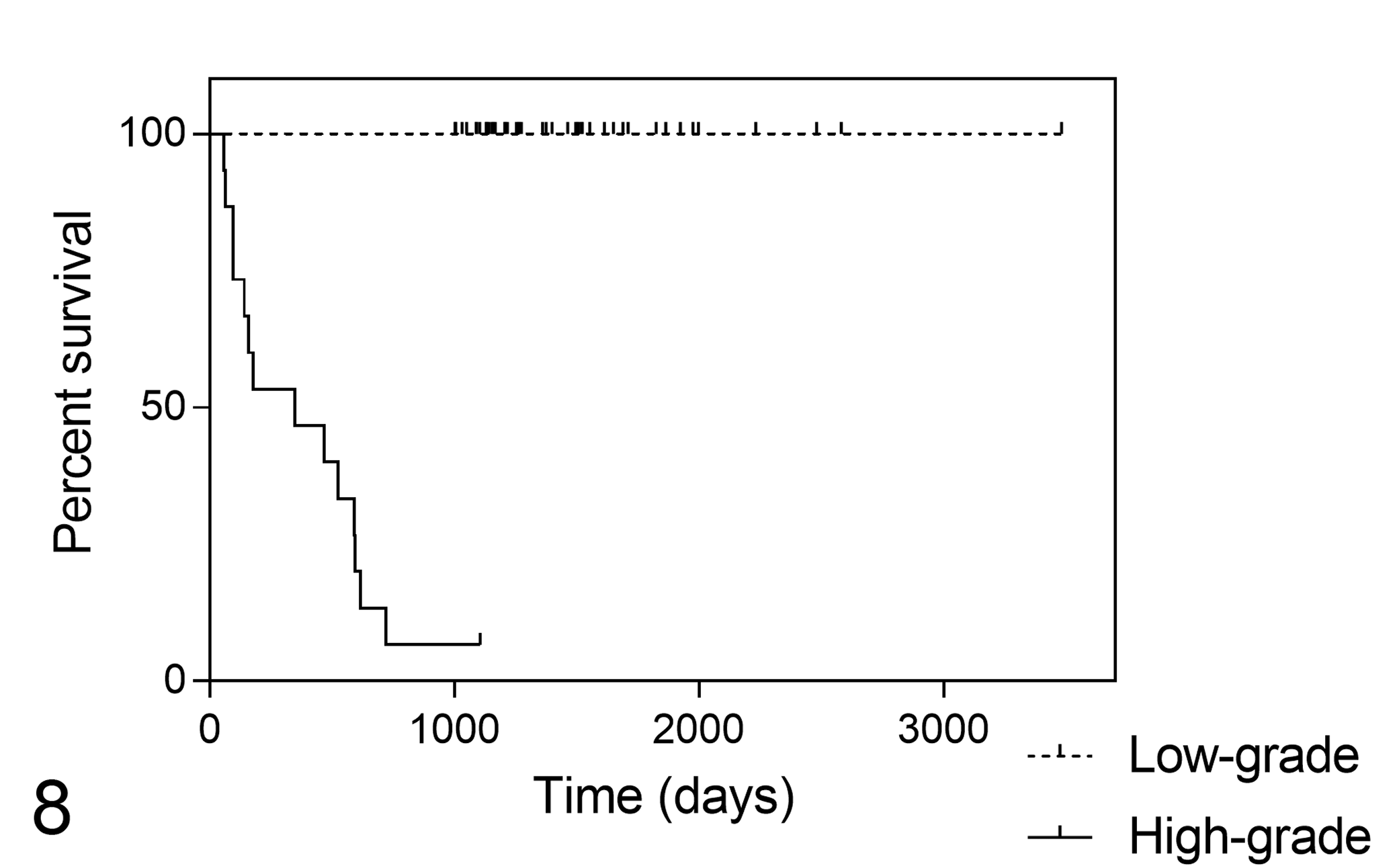
Kaplan-Meier survival curves for mortality due to cutaneous mast cell tumor in 63 cats, stratified according to the proposed grading system (P < .001).
Discussion
Several questions about feline MCT remain unanswered, at both a clinical and pathologic level, including how to identify the cases deserving further medical attention. One of the biggest limitations for assessing cMCT biological behavior is the lack of standardized staging procedures, impairing the possibility to confirm visceral involvement and/or nodal metastasis. Another limit is the poor consistency of long-term follow-up information, with consequent difficulties in assessing the actual cause of death of these cats. This may derive from the assumption that almost all feline cMCTs are clinically benign. Actually, the reported incidence rate of systemic spread following surgical excision is up to 22%, which is not substantially different from the metastatic rate observed in the canine counterpart. 12,17,19
We tried to overcome the difficulties related to staging and reliability of follow-up information by performing a strict selection, at the cost of reducing the number of cases and losing cases of potential interest. Indeed, cats alive or deceased for MCT-unrelated cases were included only upon availability of an exceptionally long follow-up time (1003–3481 days), to exclude the possibility of malignant biological behavior even in the absence of proper clinical monitoring, whereas all the cats with an uncertain cause of death were excluded from the analysis. Further studies will be necessary to test the present grading system on a larger number of cases.
Due to the aforementioned lack of uniform staging procedures, in the 9 cats developing splenic MCT, we cannot completely exclude the presence of visceral involvement at the time of cMCT removal. The veterinary literature is divided in opinion on the subject of concomitant cutaneous and visceral MCTs and on which should be considered the primary form in these cases. Nevertheless, this study aimed to define prognostic criteria to identify the cMCTs that might be associated with a wider disease extension, regardless of the primary or metastatic nature of the cutaneous lesion.
The proposed grading system was primarily based on the number of mitoses, which is an acknowledged prognostic factor in feline cMCT. 4,8,10,13,15,16 This parameter was used as a filter to isolate the potentially high-grade cases on which to evaluate the other 3 parameters. The obtained grading system, as included in a multivariable analysis model with the mitotic count, was the only factor retaining prognostic significance, indicating an improved value as compared with the evaluation of mitoses alone.
The option to include a nonhistologic parameter, such as tumor diameter, in a grading system can be questionable. Nevertheless, this is objective information, which the pathologist can generally acquire from the specimen submission form. If it is missing, a comment can be added in the histologic report, indicating that the tumor should be considered high grade if the diameter exceeds 1.5 cm.
Lymphocytic infiltrate, subcutaneous invasion, pyknosis/karyorrhexis, and neoplastic emboli, although significantly different between the groups, were not included in the grading system. Although easy to identify, the lymphocytic infiltrate was not considered specific enough, as it was detected in more than one-third of tumors with low malignancy. Lymphocytes are also apparently correlated with tumor histotype, being reported more frequently in pleomorphic and atypical cMCTs, and it was hypothesized that they may reflect an immunologic response against the tumor. 3 The evaluation of subcutaneous invasion is possible only in surgical samples submitted entirely to the laboratory. Additionally, in our series of feline cMCTs, the presence of subcutaneous infiltration was likely attributable to an enlarged nodule size. In fact, almost all the tumors in this study, including those belonging to group 2, showed a compact growth, with a minimal tendency to infiltration. Finally, tumor cell necrosis (pyknosis/karyorrhexis) and neoplastic emboli were not included in the grading system due to their low frequency among tumors with aggressive behavior (40% and 27%, respectively), resulting in a poor sensitivity of these parameters; additionally, emboli may be missed when samples lack enough peritumoral tissue. Nevertheless, a feline cMCT with vascular invasion should be considered high grade even in the absence of other histologic malignant features.
A limit of this study was the low number of atypical and pleomorphic cMCTs, thereby preventing the possibility to assess the consistency of the proposed grading system on these specific histologic subtypes. The atypical and pleomorphic subtypes represent a minority of feline cMCT, and their intrinsic prognostic significance is still debated. Atypical cMCTs are composed of a poorly demarcated, deep dermal, or subcutaneous proliferation of histiocyte-like mast cells admixed with lymphoid aggregates and eosinophils. Atypical mast cells only show faint, irregular, and mostly orthochromatic cytoplasmic granules. These tumors are reported to occur more frequently as multiple nodules in juvenile cats, especially of Siamese breed, and they have been associated with a benign clinical course or spontaneous regression, similar to urticaria pigmentosa. 20 However, atypical cMCTs are likely to be under- or misdiagnosed because of their histologic similarity to nonneoplastic mixed inflammatory processes (eg, eosinophilic granuloma), and immunohistochemistry is required for differential diagnosis with feline progressive histiocytosis and other neoplasms, including melanoma and cutaneous lymphoma. Consequently, little is known about their biological behavior.
In the present study, there were 6 atypical cMCTs: 4 in group 1 and 2 in group 2. All of them were correctly graded. While this suggests that an atypical morphology is not in itself of prognostic importance, the low number of cases does not allow conclusions on the applicability of the grading system for this subtype. Additionally, atypical cMCTs tend to be larger than mastocytic cMCTs, and nuclear shape may be irregular; as such, they are theoretically more likely to be graded high.
The prominent features of feline pleomorphic cMCTs are mononuclear or multinucleated tumor giant cells 3 to 20 times larger than neighboring neoplastic cells. 8 In the present study, only 1 cat in group 1 was diagnosed with a pleomorphic cMCT, based on the presence of moderate anisokaryosis (up to 3-times variation in nuclear diameter) in a minority of cells. Feline pleomorphic cMCTs were variably reported to have benign or malignant behavior, partly due to recent confusion between the terms “pleomorphic” and “diffuse.” 1,2,6,8,18 One case is not enough to speculate; however, according to this study, anisokaryosis should not be considered a malignant feature in itself. A recent study by Melville et al identified a new rare subtype of feline cMCT with well-differentiated mast cells together with prominent multinucleated cells, showing a more aggressive behavior. 13 In the present study, multinucleation (ie, ≥3 nuclei) not associated with anisokaryosis was a rather common feature, observed in as many as 30% of tumors but with no significantly different distribution between groups 1 and 2. As a whole, this seems to prevent the application of the Kiupel grading system for canine MCTs to the feline counterpart. 9
The cats with multiple tumors were included in this study only if all the nodules had been surgically removed and were available for histologic examination, at the price of eliminating most cases of multicentric cMCTs, which are more likely to be malignant. 11 Nevertheless, in the 9 included cases with multiple nodules, the tumors from the same cat had a similar morphologic appearance and were graded equally, suggesting that in the case of multiple tumors, the histologic assessment of 1 nodule (possibly the largest) might be sufficient.
Finally, the proposed grading system predicted survival but not PFI. It must be noted that some cats are predisposed to the onset of multiple cMCTs during life and that local recurrence may develop if tumors are incompletely excised, without this necessarily being an indication of malignancy, as confirmed by our data. Clearly, it is essential to grade each new nodule occurring over time because if the first cMCT was low grade, those arising later may not be.
In conclusion, the present study proposes a grading system that identified most of the aggressive cMCTs in a population of 63 cats with available long-term follow-up information. This system relies on the number of mitoses, tumor size, and nuclear parameters (nuclear shape, nucleoli, and chromatin). Further research will be necessary to confirm its prognostic utility on a different population of cats, assess reproducibility, and test it on a larger number of atypical and pleomorphic subtypes that were not sufficiently represented in this study.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985818800028 - Grading Cutaneous Mast Cell Tumors in Cats
Supplemental Material, DS1_VET_10.1177_0300985818800028 for Grading Cutaneous Mast Cell Tumors in Cats by Silvia Sabattini, and Giuliano Bettini in Veterinary Pathology
Supplemental Material
Supplemental Material, DS2_VET_10.1177_0300985818800028 - Grading Cutaneous Mast Cell Tumors in Cats
Supplemental Material, DS2_VET_10.1177_0300985818800028 for Grading Cutaneous Mast Cell Tumors in Cats by Silvia Sabattini, and Giuliano Bettini in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
