Abstract
Mast cell tumor (MCT) is a frequent cutaneous neoplasm in dogs that is heterogeneous in clinical presentation and biological behavior, with a variable potential for recurrence and metastasis. Accurate prediction of clinical outcomes has been challenging. The study objective was to develop a system for classification of canine MCT according to the mortality risk based on individual assessment of clinical, histologic, immunohistochemical, and molecular features. The study included 149 dogs with a histologic diagnosis of cutaneous or subcutaneous MCT. By univariate analysis, MCT metastasis and related death was significantly associated with clinical stage (P < .0001, r P = –0.610), history of tumor recurrence (P < .0001, r P = –0.550), Patnaik (P < .0001, r P = –0.380) and Kiupel grades (P < .0001, r P = –0.500), predominant organization of neoplastic cells (P < .0001, r P = –0.452), mitotic count (P < .0001, r P = –0.325), Ki-67 labeling index (P < .0001, r P = –0.414), KITr pattern (P = .02, r P = 0.207), and c-KIT mutational status (P < .0001, r P = –0.356). By multivariate analysis with Cox proportional hazard model, only 2 features were independent predictors of overall survival: an amendment of the World Health Organization clinical staging system (hazard ratio [95% CI]: 1.824 [1.210-4.481]; P = .01) and a history of tumor recurrence (hazard ratio [95% CI]: 9.250 [2.158-23.268]; P < .001]. From these results, we propose an amendment of the WHO staging system, a method of risk analysis, and a suggested approach to clinical and laboratory evaluation of dogs with cutaneous MCT.
Mast cell tumor (MCT) is the second most frequently diagnosed malignancy in dogs, 9,18 representing 16% to 21% of all canine cutaneous neoplasms. 26 Nevertheless, canine MCT is highly heterogeneous in clinical presentation and biological behavior with a variable potential for recurrence and metastasis. Thus, there is a need to identify prognostic factors that predict outcome. 26,31,34
Current prognostic factors include: (1) clinical features, such as clinical stage, 4,26,46,47 breed, 26,28 anatomic site, 19,26 and history of tumor recurrence (ie, the dog presented with a recurrent tumor rather than a primary tumor) 6,29,33,44 ; (2) histological criteria considered for the 3-tier Patnaik grading system (grades 1, 2, and 3) 33 and the 2-tier Kiupel grading system (low and high grade), 20 although some histological features may represent isolated prognostic factors, as shown for the mitotic count 27,35 ; (3) immunohistochemical evaluation of Ki-67 labeling index 40 and KITr expression 14,20,48 and evaluation of metalloproteinases (MMP-2 and MMP-9) 23 and fibroblast activating protein 15 ; (4) molecular features, emphasizing gain-of-functions mutations in c-KIT oncogene, such as internal tandem duplications in exon 11 2,49,50,53 as well as in exons 8 and 9 24,43 (which may also predict a favorable response to tyrosine kinase inhibitors currently licensed for use in dogs in United States and Europe, ie, masitinib and toceranib). 17,25 Despite all these prognostic factors, the decision-making process remains challenging for the small animal practioner. 47 It would be desirable to classify dogs with MCT as low, intermediate, or high risk for tumor recurrence or metastasis since this would be of value for clinical decisions as well as patient selection in clinical trials. The aim of this study was to develop a classification system for dogs with MCT based on the mortality risk, considering the assessment of clinical, histological, immunohistochemical, and molecular features.
Materials and Methods
Patients
This prospective-retrospective study was conducted from 2006 to 2016 and approved by the Ethics Committee on Animal Use of Universidade Federal de Minas Gerais, Brazil (protocol number 384/2013). Inclusion criteria was histological diagnosis of cutaneous or subcutaneous MCT attended in a primary case veterinary hospital, resulting in 316 cases. Exclusion criteria was lack of histological sample or staging information (ie, fine needle aspiration of regional lymph nodes and abdominal ultrasound) or patients who were lost to follow-up in less than 6 months after surgery, resulting in 149 dogs (84 prospectively enrolled). Patients who died due to MCT or unrelated causes were included, even if followed for less than 6 months. Follow-up consisted of phone calls and rechecks during treatment or every 4 to 6 weeks after treatment in the first 6 months and every 2 to 3 months after that.
Surgical and Medical Management
All dogs were evaluated by electrocardiogram, complete blood count, coagulation profile (time of activated partial thromboplastin and time of prothrombin), and serum biochemistry (total protein and albumin, blood urea nitrogen, creatinine, alkaline phosphatase, alanine transferase, aspartate transferase, and gamma-glutamyl transferase) prior to anesthesia. Patients were submitted to full staging, including physical exam, fine needle aspiration of lymph nodes, and abdominal ultrasound. Patients with suspected metastastes on lymph node evaluation had them removed for further staging.
Prednisone or prednisolone was used at the clinician’s discretion at a dose of 40 mg/m2 daily for 7 to 10 days prior to surgery to promote cytoreduction or reduce peritumoral inflammation, as previously suggested. 9,39,47 According to staging, prognostic and clinical presentation after the cytoreductive treatment, the patients were treated by surgical resection (excisional biopsy) or incisional biopsy. Excional biopsy involved an elipitical incision with 2- to 3-cm lateral margins, followed by deep dissection aiming to include at least 1 muscular fascia. 12 The excised tissues were fixed in 10% neutral buffered formalin for subsequent histopathologic, immunohistochemical, and molecular evaluation. After surgery, treatments were chosen according to the oncologist's experience and prognostic factors evaluated by the laboratory findings.
Histological Analysis
Tumor tissue was fixed in formalin for 48 hours and 4-μm-thick sections were prepared routinely and stained with hematoxylin-eosin and toluidine blue. Histopahological examination was performed under light microscopy by a single veterinary pathologist (LM). Tumors were graded using the Patnaik 33 and Kiupel 20 systems. In addition, a combined Patnaik/Kiupel grade was assigned by combining these 2 grades. Neoplasms were also evaluated to characterize the tumor extent (subcutaneous, dermal, ulcerated, or within the muscle), architectural pattern (more than 50% in cords or in sheets), intratumoral necrosis, edema, hemorrhage, collagen hyalinization, cystic change of apocrine glands, desmoplasia, and eosinophilic infiltrate in a semi-quantitative analysis (absent, mild, moderate, or intense). Other assessments were the surgical margins (clean, narrow, or compromised), number of binucleated cells, and mitotic count. Multinucleated cells (more than 2 nuclei) were counted for grading according to the Kiupel system. 20 Mitotic figures, binucleation, and multinucleation were counted in fields (FN = 22 mm) with higher numbers of these events. Patients presenting with multiple lesions were classified according to the most undifferentiated pattern (ie, the highest grade, highest mitotic count and Ki-67 labeling index, abnormalities in KITr expression, and mutations in c-KIT oncogene, when present).
Immunohistochemistry
For this technique, 4-μm-thick sections were obtained from representative paraffin blocks of each case and placed into gelatin-coated slides. Slides were deparaffinized and rehydrated in an alcohol series. Heat-induced antigen retrieval was performed (Target Retrieval Solution Citrate pH 6, Dako Cytomation) under pressurized heat (20-25 mmHg, 125°C, 2 minutes). Endogenous peroxidase was blocked by immersion in 3% hydrogen peroxide, and protein blockage was performed with a Protein Block solution (Thermo Scientific Ultravision). Primary antibodies CD117 (polyclonal, 1:800, Dako Cytomation) and Ki-67d (MIB-1, 1:25, Dako Cytomation) were incubated at 4°C for 16 hours. Secondary antibody (Advance HRP Link, Dako Cytomation) was incubated in a humid chamber for 30 minutes, and the reaction was amplified by the polymer (Advance HRP Enzyme, DakoCytomation). The immunoreaction was revealed with the chromogen 3,3–diaminobenzidine tetrahydrochloride (Liquid DAB + Substrat Chromogen System, Dako Cytomation) and counterstained with Harris hematoxylin. Sections from canine MCT known to express Ki-67 and CD117 were used as positive controls. Negative controls were assessed using normal serum instead of the primary antibodies.
The immunolabeling pattern for KITr was evaluated by counting membrane, focal, or diffuse cytoplasmic immunoexpression of CD117 (KIT patterns I, II, or III, respectively) in 10% of the tumor fragment at 400× magnification. Each MCT was assigned the highest staining pattern present in at least 10% of the neoplastic cell population or present in large clusters of neoplastic cells within the tumor. 21 Ki-67 labeling index was determined as the percentage of positive nuclei in at least 500 neoplastic cells in 3 to 5 hot-spots (ie, 3-5 400× fields with higher number of labeled nuclei). Each nucleus with evidence of immunolabeling was considered positive for Ki-67. 1,37
Screening of Mutations in the c-KIT Oncogene
The PCR reaction for amplification of the fragment of interest in the c-KIT oncogene (exon 11 – nucleotides 1661 to 1787 of chromosome 13) was performed by Progen (Vetpat Laboratory) using DNA extracted from paraffin-embedded tumor by the proteinase K method. The primers (Invitrogen) were: forward 5’-ATCTGTCTCTCTTTTCTCCCCC-3’ and reverse 5’-TGGGGTTCCCTAAAGTCATTGT-3’. The product was 225 bp in the absence of mutations (native c-KIT). Reactions were carried out in a GenPro thermocycler (BIOER Technology), with a maintenance at 95°C for 5 minutes, then 30 cycles of 94°C for 45 seconds for denaturation, 63°C for 45 seconds for annealing, 72°C for 1 minute for extension, then maintained at 72°C for 10 minutes for molecular stabilization. The amplified material was separated by electrophoresis at 100 V with free amperage. The positive and negative controls were canine healthy skin samples and water, respectively.
Assessment of Response
Clinical outcome was defined as a categorical variable in 3 levels: (1) no local recurrence, metastasis, or tumor-related death; (2) MCT local recurrence or metastasis but no tumor-related death; (3) MCT local recurrence or metastasis and tumor-related death. The disease-free interval (DFI) was considered from the beginning of therapy for patients that achieved complete remission or those for whom surgery was amenable. The overall survival (OS) was determined for all patients and defined as the time from the beginning of therapy until tumor-related death. Patients lost to follow-up or with unrelated deaths were censored. Cytology was used to confirm the diagnosis in case of progressive disease and appearance of new lesions.
Statistical Analysis
Statistical analysis was performed using the Stata Statistical Software (v. 12.0, Stata Corp). Differences were considered significant when P < .05.
A matrix correlation considering prognostic factors and clinical outcome was conducted with Pearson test. Significant correlations were considered strong when they occurred in over 49% of the studied population (r > 0.7), moderate when occurring in 9% to 49% (0.3 < r <0.7), and weak when occurring in less than 9% of the population (r < 0.3).
A ROC curve was used to set a cut-point for quantitative variables that were significantly correlated to clinical outcome. The cut-point was considered with maximum specificity without compromising sensitivity for predicting the clinical outcome.
The overall survival of patients in this study was estimated by the Kaplan-Meier curve, and the logrank test of Cox-Mantel was used to compare the curves within each prognostic factor. Cox proportional hazard model was performed along with factor analysis.
Results
This study included 149 dogs, aging from 3 to 16 years old (9.6 ± 3.1; mean ± SD). Aproximately 19.5% (29/149) were mixed-breed dogs, and the remaining were of 34 breeds, including Labrador Retriever (n = 30), Poodle (n = 10), Golden Retriever (n = 7), Pinscher (n = 7), Pit-bull (n = 6), Boxer (n = 5), Dachshund (n = 5), and Cocker Spaniel (n = 5).
MCT were considered solitary in 95.3% of cases (142/149). The anatomic sites included the limbs (70/142, 49.3%) and trunk (39/142, 27.5%) as well as scrotum (9.9%), vulva (2.8%), perineum (2.8%), face (2.8%), prepuce (2.1%), oral mucosa (2.1%), and pinna (0.7%).
Clinical Outcomes and Associated Parameters
Of the 149 cases, 97 (65.1%) had no local recurrence, metastasis, or tumor-related death. Local tumor recurrence was not observed in this study, even in patients with narrow (n = 14) or incomplete surgical margins (n = 84). Progressive disease with concomitant metastasis to the skin and/or lymph node occurred in 52 cases. Of these, 6 of 52 (11.5%) remained alive at the end of the study, and 46 were considered tumor-related deaths (ie, 46/149, 30.8%, tumor-related deaths in the full population). Tumor-related death resulted from euthanasia in 42 cases due to disease progression and clinical deterioration. Four cases died because the owner refused euthanasia; of these, 1 had metastasis to iliac lymph nodes and paraneoplastic syndromes including anaemia and gastritis in addition to paralytic ileus, and 3 developed marked anaemia related to bone marrow invasion (with no metastases to the liver or spleen, detected through ultrasound or autopsy). Threrefore, clinical outcome was defined as (1) no local recurrence, metastasis, or tumor-related death; (2) MCT metastasis but no related death; (3) MCT metastasis and related death. Significant correlations between parameters are shown in Table 1 and as Supplemental Material. Regarding clinical features, the clinical outcome was not related to breed, sex, age, or location of the tumor (multicentric, trunk, limb, face/muzzle, mucosa, scrotum, prepuce/vulva/perineum), but it was moderately correlated to WHO clinical stage (P < .0001, r P = –0.610) and history of tumor recurrence (whether it was a primary tumor or a recurrence) (P < .0001, r P = –0.550).
Statistically Significant Correlations Among Clinical and Pathologic Parameters in 149 Dogs With Mast Cell Tumors.
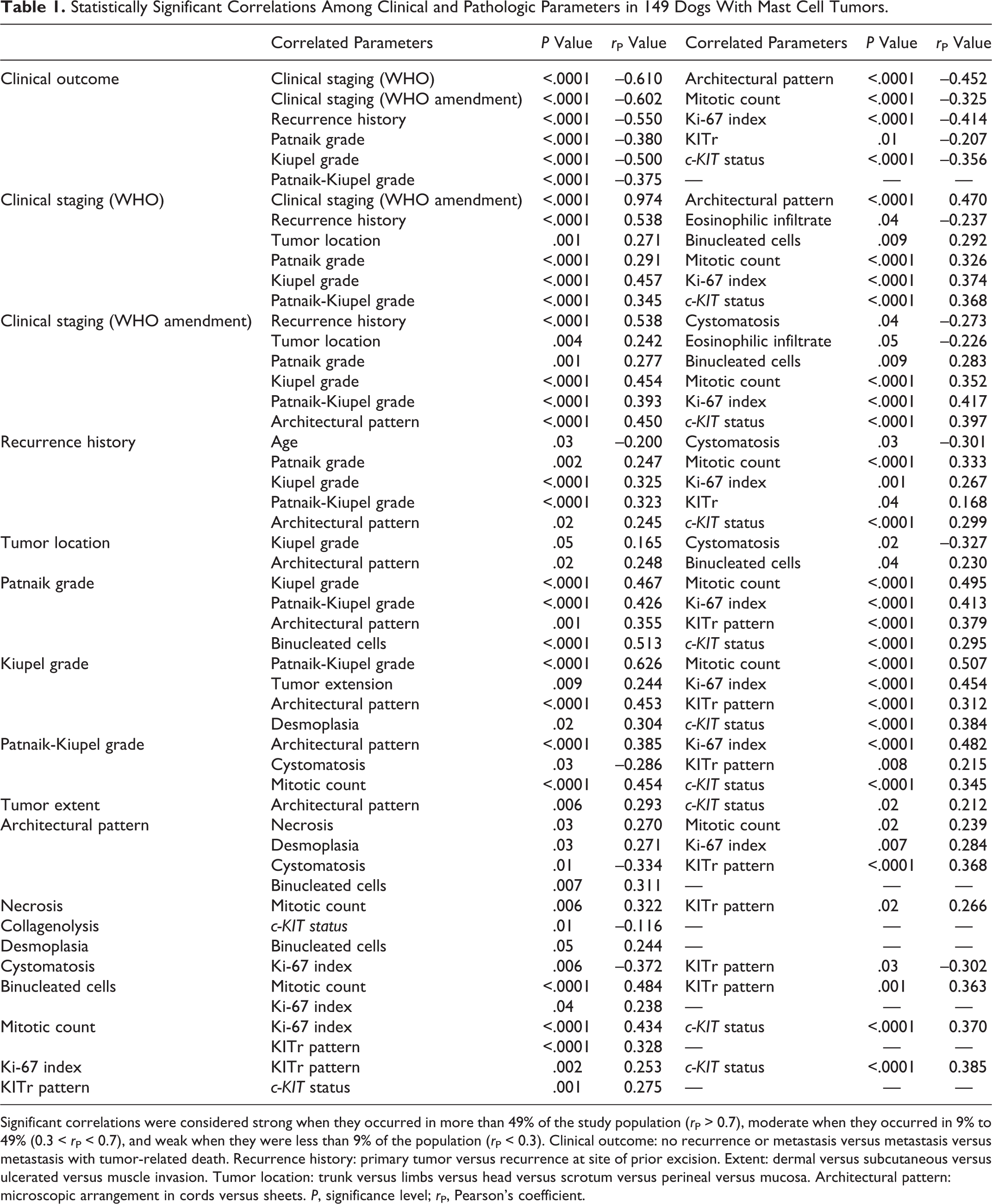
Significant correlations were considered strong when they occurred in more than 49% of the study population (r P > 0.7), moderate when they occurred in 9% to 49% (0.3 < r P < 0.7), and weak when they were less than 9% of the population (r P < 0.3). Clinical outcome: no recurrence or metastasis versus metastasis versus metastasis with tumor-related death. Recurrence history: primary tumor versus recurrence at site of prior excision. Extent: dermal versus subcutaneous versus ulcerated versus muscle invasion. Tumor location: trunk versus limbs versus head versus scrotum versus perineal versus mucosa. Architectural pattern: microscopic arrangement in cords versus sheets. P, significance level; r P, Pearson’s coefficient.
Among histological criteria, clinical outcome (ie, MCT metastasis or related death) was correlated to Patnaik grade (P < .0001, r P = –0.380) and Kiupel grade (P < .0001, r P = 0.500), predominant architectural pattern (ie, cords vs sheets) of neoplastic cells (P < .0001, r P = –0.452), and mitotic count (P < .0001, r P = –0.325). However, in this study, there was no correlation of clinical outcome with presence of neoplastic cells at the surgical margin, tumor extent (ie, subcutaneous, dermal, ulcerated, or within the muscle), or the presence or intensity of edema, necrosis, hemorrhage, collagenolysis, cystic change of apocrine glands, desmoplasia, eosinophilic infiltrate, and number of binucleated cells. Histological features are shown in Figs. 1 and 2.
On immunohistochemical evaluation, a correlation with clinical outcome was obtained for Ki-67 labeling index (P < .0001, r P = –0.414) and KITr pattern (P = .01, r P = –0.207) (Figs. 3–7).
Internal tandem duplications in exon 11 of the c-KIT oncogene was found by PCR in 10.7% (16/149) of dogs in this study, and it was associated with a higher risk of MCTs metastasis or related-death (P < .0001, r P = –0.356). Only 2 patients (1.3%) presented with deletions in the PCR amplicon, and both of these had confirmed lymph node metastasis and died within 30 and 134 days of treatment.
The ROC curve established a cut-point for the mitotic count of 2 mitotic figures in 10 HPF (AUC = 0.8163; 95% CI, 0.741-0.891; OR = 1.944; sensitivity = 84%; specificity = 56%) and for the Ki-67 labeling index of 5.6% Ki-67-positive cells in 500 neoplastic cells (AUC = 0.786; 95% CI, 0.706-0.867; OR = 1.777; sensitivity = 92%; specificity = 42%).
Survival Time and Tumor-Related Death
The mean follow-up time for the 149 patients was 358 days, and the median overall survival was 1020 days.
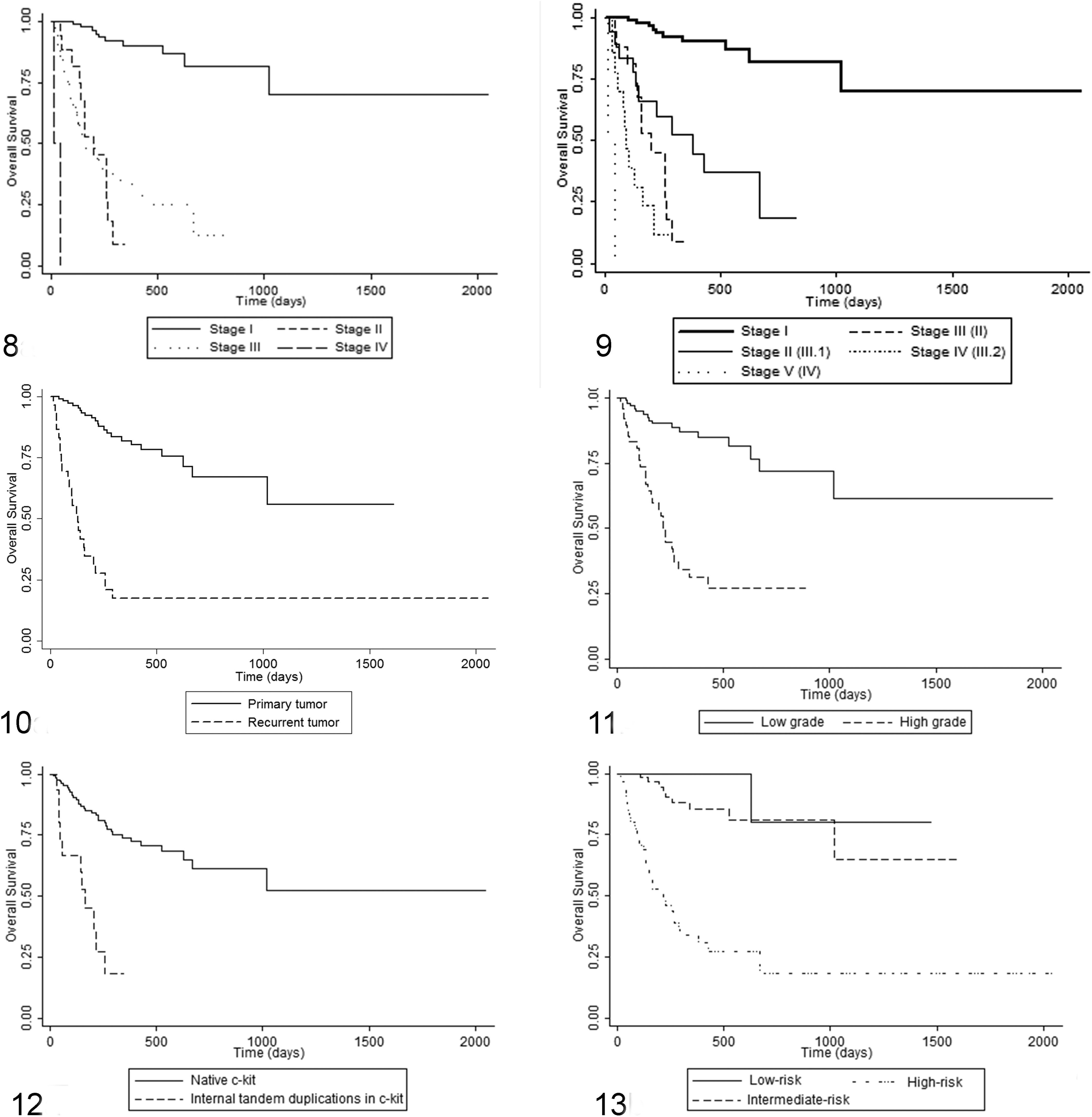
Clinical staging, as proposed by the World Health Organization, 32 resulted in 64.4% (96/149), 12.8% (19/149), 21.5% (32/149), and 1.3% (2/149) of patients in stages I, II, III, and IV, respectively. Median survival was not reached for stage I; median survival was 203, 164, and 15 days for stages II, III, and IV, respectively, and these were significantly different (P < .0001), although there was no difference between stages II and III (Fig. 8). An amendment to the WHO staging system (Table 2) was done to divide stage III into III.1 (new stage II; multiple tumors with no regional lymph node involvement; n = 18; OS = 381 days) and III.2 (new stage IV; large/infiltrative or multiple tumors with regional lymph node involvement; n = 14; OS = 90 days). The OS was significantly different between all stages (P < .0001) and was significantly higher in stage III.1 compared to stages II (new stage III) or III.2 (Fig. 9).
Proposed Amendment to the WHO System of Clinical Staging of Cutaneous and Subcutaneous Mast Cell Tumors in Dogs.

Previous WHO stage II became stage III. Previous WHO stage III was divided in III.1 (current stage II) and III.2 (current stage IV). Previous WHO stage IV became stage V.
A history of tumor recurrence (ie, the dog presented with a recurrent tumor rather than a primary tumor) was present in 30 of 149 (20.1%) cases. MCT-related death occurred in 24 of 30 (80.0%) dogs with recurrent MCT (median survival of 131 days), compared to 22 of 119 (18.5%) of dogs with initial (nonrecurrent) MCT (median survival was not reached for this group) (P < .0001; Fig. 10).
For the Patnaik grading system, grade 1, 2, and 3 tumors were found in 4.0% (6/149), 79.9% (119/149), and 16.1% (24/149) of cases. Tumor-related mortality occurred in 0% (0/6) of grade 1, 25% (30/119) of grade 2, and 67% (16/24) of grade 3 cases. Median survival of 203 days was reached only for grade 3 cases, differing from low and intermediate grade tumors (P < .0001).
For the Kiupel grading system, high-grade MCT were found in 32.9% (49/149) and low-grade found in 67.1% (100/149) of cases. Tumor-related mortality occurred in 61% (30/49) of high grade and 16% (16/100) of low grade MCT. A median survival of 213 days was reached only for high-grade cases (P < .0001; Fig. 11).
Using the Patnaik/Kiupel grading systems together resulted in 6 grade 1/low grade, 94 grade 2/low grade, 25 grade 2/high grade, and 24 grade 3/high grade, and tumor-related mortality occurred in 0%, 17% (16/94), 56% (14/25), and 67% (16/24), respectively. Median OS was not reached for grade 1/low grade and grade 2/low grade, and these were not significantly different. There was also no difference in OS between grade 2/high grade (OS = 226 days) and grade 3/high grade (OS = 203 days). Nevertheless, low grade differed from high grade (P < .0001).
There were no MCT-related deaths in cases with a diffuse cord-like achitectural pattern, and the median survival was therefore not reached. In contrast, cases with sheet-like architectural pattern reached the median survival at 337 days (P < .0001).
Using the cut-points defined by the ROC curves, the median survival was reached only for patients whose tumors had values that exceeded the cut-point: 260 days for cases with more than 2 mitotic figures in 10 hpf (P < .0001) and 524 days for Ki-67 labeling index of more than 5.6% (P < .0001). Two samples were excluded from the survival analysis because they lacked immunolabeling for Ki-67 even in the internal positive control (epithelial cells).
Regarding KITr expression, the median survival was not reached in the group with a membranous KIT pattern (KIT I), but it was 1020 and 224 days for cytoplasmic focal (KIT II) or diffuse (KIT III) expression, respectively. However, statistical significance was found only between KIT I and KIT III (P = .008).
In a survival analysis, excluding those with deletions in the c-KIT amplicon (n = 2), the median survival was not reached for patients without mutations (n = 131), but it was 164 days for those with internal tandem duplications in exon 11 of the c-KIT oncogene (n = 16; P < .0001; Fig. 12). MCT related-deaths occurred in 69% (11/16) of patients with such mutations.
In the factor analysis, 11 variables failed to show an association with clinical outcome (P values above .6), including tumor extent (.67), necrosis (086), colagenolysis (.72), desmoplasia (.74), cystomatosis (.75), inflammatory infiltrate (.77), surgical margins (.89), and patient age (.83), Patnaik grade (.62), Ki-67 labeling index (.75), and KITr pattern (.66) although the last three were correlated to clinical outcome in an univariate analysis. The multivariate analysis with Cox proportional hazard model demonstrated that only 2 of the variables were independent predictors of OS: both WHO stage and the proposed amendment of the WHO clinical staging system (hazard ratio [95% CI]: 1.824 [1.210-4.481]; P = .01) and the history of tumor recurrence (hazard ratio [95% CI]: 9.250 [2.158-23.268]; P < .001).
From the analysis of correlations, OS, and multivariate analysis, a model of the risk of recurrence or metastasis was developed for canine MCT (Table 3). In this model, dogs diagnosed with MCT were assigned to the high-risk group if they presented with advanced stage disease (stage II, III.2, and IV or III, IV and V, according to the proposed amended of the WHO staging system) and/or a history of tumor recurrence. Cases were assigned to the intermediate risk group if they had no history of tumor recurrence, WHO stage I or II (according to the proposed amended of the WHO staging system shown in Table 2), plus any one of: combined Patnaik/Kiupel grade 2/high or grade 3/high, sheet-like architectural pattern, mitotic count higher than 2 in 10 HPF, Ki-67 labeling index above 5.6%, KIT pattern III, or internal tandem duplications in exon 11 of the c-KIT oncogene. In this study, none of these parameters were independent even if patients with recurrent tumors and advanced stage disease were withdrawn. Cases were assigned to the low-risk group if they had none of the prognostic factors listed in Table 3. Of the 149 cases, 58 were in the high-risk group; these had a median survival time of 213 days, and 36 of 58 (62%) had MCT-related deaths. The intermediate risk group included 64 cases, and 9 of 64 (14%) had MCT-related deaths. The low-risk group included 27 cases, and 1 of 27 (4%) had MCT-related death. Median survival was not reached in low- or intermediate-risk groups but differed from that of the high-risk group (P < .0001, Fig. 13).
Negative Prognostic Features Used to Establish the Risk of Metastasis and Tumor-Related Death in Dogs With Cutaneous or Subcutaneous Mast Cell Tumor.

High-risk patient: stage III disease or higher (using the amendment to the WHO system shown in Table 2) and/or recurrent mast cell tumors. Intermediate-risk patient: stage I or II disease (using the amendment to the WHO system shown in Table 2) and primary tumor (no history of recurrence) but with at least 1 other negative prognostic features listed. Low-risk patient: none of the negative prognostic features listed.
Discussion
An extensive list of prognostic factors was used in this study, allowing the definition of the risk of recurrence or metastasis in canine MCT. Numerous correlations were present, but only a few of them were related to clinical outcome. Other correlations were also identified. Mitotic count was correlated to Ki-67 labeling index, as expected, 3,36 but also to the number of binucleated cells and presence/intensity of intratumoral necrosis. Mutations in the c-KIT oncogene were positively correlated to clinical stage, history of tumor recurrence, Patnaik and Kiupel grading systems, mitotic count, and Ki-67 labeling index. After a multivariate analysis, only the WHO staging and proposed amended system and history of tumor recurrence were independent predictors of poor outcome in canine MCT.
Clinical staging, as proposed by the World Health Organization, 32 allows evaluation of the extent of the disease, and it has been recommended for canine MCT once diagnosis is established. 4,26 Clinical staging is directly related to prognosis of canine MCT 10,26,52 as observed in this study. However, the true influence of stages II and III is long debated in the literature, and it is hard to define. This is related to: (1) difficulty in the diagnosis of lymph node metastases and (2) uncertainty of the importance of multiple lesions and multicentric presentations, especially when there is no concomitant involvement of regional lymph nodes. 10,26,52 Here we propose that stage III is divided into III.1 in the absence of lymph node involvement and III.2 when there is a confirmed metastasis in the lymph node. In this study, a better outcome was observed in dogs in stage III.1 compared to stage II. Therefore, we propose an amendment of the canine MCT staging system, based on 5 tiers, which may improve the definition of clinical stages and their progression. Although the WHO staging was also a significant independent variable associated with clinical outcome, the amended staging system better predicted the outcome of dogs with MCT.
The relevance of canine MCT staging is often questioned, but its importance was clearly demonstrated in this study. Nonetheless, the probability of metastasis may be extremely low in some patients,
46
and a complete stating is rarely necessary.
4
Considering this, a protocol for staging is proposed in 3 levels: Simple/basic staging: detailed physical examination searching for new cutaneous lesions but also for regional lymph node involvement, following the lymphosome territory proposed for dogs.
41
This should include cytological evaluation and/or histological assessment through an incisional or excisional biopsy. This may be performed during an incisional biopsy of the primary tumor itself or during the definitive excision. Cytological evaluation of lymph nodes for dogs presenting with MCT is limited as up to 24% of healthy dogs may have an increased population of normal mast cells in the lymph node,
5
but there are cytological criteria available that can be used.
22
A histological classification of the level of involvement of the regional lymph node in dogs with MCT may further improve clinical staging and refine clinical and therapeutic decisions.
51
If a lymph node is confirmed to have metastasis (high-risk patient) or if the primary tumor is located in a site with potential drainage to internal lymph nodes, complete staging is recommended. Complete staging: besides the basic staging, complete staging should include imaging, especially abdominal ultrasound (for perineal MCT) and thoracic radiographs (for neck and trunk MCT),
4,46
but also computed tomography if the previous images suggest possible metastatic lesions. In these cases, an extended staging should be performed.
4
Extended staging: cytological evaluation of suspected metastatic lesions, performed through a fine needle aspiration (FNA), guided by ultrasound or computed tomography. Additionally, a bone marrow assessment may be recommended, although there is also a lack of criteria for diagnosing bone marrow metastasis, and such involvement is rare in canine MCT and usually associated with severe and terminal disease.
10,26,47,52
Recurrent canine MCT are associated with a poor prognosis, with related death rates dramatically reaching 86% to 100% of cases, as reported in previous studies. 6,33 Although recent studies in dogs with recurrent MCT have demonstrated effective disease control with surgery and adjuvant therapies, 44 mortality rates remain high. Tumor-related mortality occurred in 80% of patients with recurrent MCT in this study, even though adjuvant treatments were recommended and performed in most cases. 6,33
In this study, in contrast to the literature, differences in the clinical outcome were not associated with breed and tumor site. Probably due to epidemiological differences in the distribution of canine breeds, we did not observe a greater frequency of MCT in breeds presenting the Bulldog as a common ancestral. 26,28 The wide variation of breeds in this study compromised evaluation of overall survival according to many breeds, and additional studies are necessary for defining such association.
Although tumor localization was not associated with clinical outcome in this study, it may affect the prognosis of some patients. 7,38,42 A poor prognosis is reported for canine MCT located in the mucosa, muco-cutaneous junctions, prepuce, scrotum, vulva, and perineum; however, clinical outcomes similar to other locations may be obtained for cases with tumors at these sites if prognosis and treatment are properly evaluated and applied. 7,38 It is reported that canine MCT located on the muzzle may also have high probability of metastasis, but they may also have a good outcome under proper treatment. 19 The only muzzle MCT included in this study presented with an inoperable lesion and confirmed lymph node metastasis. It was a high-grade tumor with an internal tandem duplicaiton in exon 11 of the c-KIT oncogene, evaluated from an incisional biopsy. Treatment with TKI in this patient resulted in complete remission and prolonged disease-free interval (240 days), and the patient was still alive at the end of this study.
In this study, MCT metastasis and/or related deaths occurred in 40.7% of patients with compromised or exiguous surgical margins and 16% of those with tumor-free margins, regardless of treatment; however, no significant correlation was found. It is suggested that evidence of tumor-free histologic margins was not correlated with DFI or OS for canine MCT, and such information should not indicate a worse outcome. 29,30 However, additional studies are needed to verify the true impact of histological margins on the prognosis of canine MCT.
The Patnaik grading system 33 has been criticized for giving too much importance to extent of the tumor and including subjective criteria that result in the inclusion of more than 40% of MCT in an intermediate grade (grade 2), gathering a wide and diverse group of lesions with distinct biological behavior. 16 In fact, 80.5% of MCT in this study were classified as a grade 2. Furthermore, tumoral extent (ie, restricted to dermis or subcutaneous tissue, skin ulceration within deeper layers including muscle) was not correlated with the clinical outcome for patients in this study. The Kiupel 2-tier grading system 20 reduces the subjectivity of the Patnaik grading system and has shown to be highly reproducible by different pathologists. 33,36 A study of 162 cases demonstrated superiority of the Kiupel grading system. Furthermore, in the present study, the group with combined Patnaik/Kiupel grade 2/low grade MCT did not reach the median survival, while the group with grade 2/high grade MCT reached a median OS of 698 days (P = .001). However, there was also a difference between grade 2/high grade and grade 3 MCT/high grade (P = .027). 36 Prognostic superiority of this system was confirmed in this study, and Patnaik grade failed as a relevant prognostic tool in the factor analysis. A Kiupel high grade is sufficient for inclusion of dogs with MCT in an intermediate-risk group, whatever the Patnaik grade. Nevertheless, 19 of 49 (39%) of patients with high-grade tumors were still alive at the end of this study, and there were 16 of 100 (16%) patients with low-grade tumors that sucumbed to the disease, suggesting that other prognostic factors must be used along with the Kiupel grading system.
Among histological features, the architectural pattern (microscopic arrangement as cords vs sheets) and the mitotic count were also associated with the clinical outcome in this study. These data are included (with variable importance) in the Patnaik 33 and Kiupel grading systems, 20 but they should be evaluated separately. The mitotic count evaluated in 10 HPF is probably one of the best prognostic factors for grade I and II MCT in early stages of disease at first diagnosis (nonrecurrent MCT). 11,35,45 Longer survival rates are reported for patients with MCT that have less than 5 mitotic figures in 10 HPF. 35 Nevertheless, a cut-point of 5 was associated with a high specificity but low sensitivity, with many patients succumbing to MCT despite of a mitotic count lower than 5. 11,45 A system with 2 cut-points of mitotic counts was proposed: 0, associated with low risk; 1–7, associated with intermediate risk; and >7, associated with high risk. 11 In fact, in this study, a low sensitivity for the cut-point of 5 was observed (aproximately 50%; OR = 6045), and therefore a new cut-point of 2 is proposed to have a higher sensitivity, as reported by other authors. 11,45 We propose that MCT with a mitotic count ≥2 should be considered at greater risk of adverse outcome.
Ki-67 labeling index was moderately related to the mitotic count for canine MCT, and both have a similar association with tumor behavior. 3 Ki-67 protein (recognized by the monoclonal antibody MIB-1) is expressed in every phase of the cell cycle except G0. 37 Different cut-points were proposed in the literature, but the 1.8% value is the most accepted one for Patnaik grade 2 MCT. 27 In the current study, a cut-point of 5.6% was associated with the risk of recurrence or metastasis, with a sensitivity of 92%. This cut-point approaches that established by another study, which reported a mortality rate of 14.3% and 55.6% in dogs with a KI-67 labeling index lower or higher than 7%, respectively. 40 It is the authors’ experience that patients with MCT and Ki-67 labeling indices above 1.8% often have long-term control of the disease, even in the absence of adjuvant therapies. In this study, a 1.8% cut-off had high sensitivity (98%) but poor specificity (10%), with an OR of 1.088. It must be noted that the cut-points used in this study have high specificity but low sensitivity and must not be used as a separate prognostic factor.
A decreased OS in patients with KIT pattern III was also observed, confirming its relationship to the prognosis of canine MCT. 14,21,48,49 However, among relevant prognostic factors, it might be considered a less informative one since in this study, differences were found only between patients with a membranous pattern (KIT I) and those with a cytoplasmic diffuse pattern (KIT III). 8,13 Recent studies 8,13 also demonstrated a weak association between expression of KITr and prognosis. In this study, Ki-67 and KITr failed as relevant prognostic features in the factor analysis. Immunohistochemistry is a complementary technique to histology and a solid partnership between the clinical oncologist and pathologist is necessary for better results.
Furthermore, gain of function mutations in the c-KIT oncogene, especially the internal tandem duplications in exon 11, had a significant impact on the outcome of dogs with MCT and may even drive treatment toward the use of TKIs. These are in fact the most frequent mutations in this oncogene in canine MCT and lead to constitutive activation of KITr. 49,50 However, the overall incidence of such mutations is low in canine MCT (approximately 10% in this study), especially if low-risk MCT are also considered. 2,43,49,50,53 Although mutations may occur in patients with aberrant KITr expression, those are separate events, and 80% of patients with abnormalities in KITr expression did not have mutations in this locus. 49 In a study performed with dogs with internal tandem duplications (n = 8), a DFI of 130 days was reached, while those without mutations (n = 39) had a DFI of 345 days. 43 Nonetheless, screening for mutations may not be done in all cases of canine MCT, particularly if resources are limited and there are no prognostic features to indicate an intermediate or high risk of recurrence or metastasis. Although it was not included in this study, screening for exons 8 and 9 mutations has been recommended for high- to intermediate-risk MCT as those mutations may confer gain of function by increasing the affinity of the extracellular domain to the stem cell factor. Nevertheless, the prevalence of such mutations in exons 8 and 9 are around 4.7% and 6.4% for exon 8 and 14.9% for exon 9, and their impact on prognosis and treatment is still not well established. 24,43
Despite the wide range of treatments, comparison of different treatments was not a goal of this study. Considering the clinical experience and information about treatment and prognosis of canine MCT, 26 the best available treatment was ethically offered for each case.
This study allowed a classification of dogs with MCT according to the risk of recurrence or metastasis. An amendment of the WHO clinical staging system can be used, considering that patients with multiple lesions without regional lymph node involvement (III.1, new stage II) usually have a better prognosis than those within the previous stage II, meaning single lesion with lymph node involvement (now stage III). Patients with large and infiltrative MCT or multiple MCT with involvement of the lymph node (III.2) remain with a worse prognosis and therefore are included in stage IV. Therefore, previous stage IV becomes now stage V. Clinical staging and history of tumor recurrence proved to be independent factors in this study, and a high risk may be assumed for patients presenting with advanced stage disease (II, III.2, and IV) or a history of tumor recurence, while an intermediate risk may be assigned for patients whose tumors present a primary tumor (no recurrence) in stage I or III.1 and at least 1 of a Patnaik/Kiupel grade 2/high grade or grade 3/high grade, mitotic count above 2, Ki-67 labeling index above 5.6%, KIT pattern III, or internal tandem duplications in c-KIT exon 11. Although the presence of exiguous or compromised surgical margins was not related to clinical outcome, controlled studies with precise evaluation of margins are needed to verify its true impact on the prognosis of MCT in dogs. Other studies have suggested that a worse prognosis may be associated with certain breeds (ie, Shar-pei) and tumor sites (mucosa, scrotum, prepuce, perineum, and vulva), but these require further study. These findings may be of value in preventing tumor recurrence by directing patients with intermediate to high risk to receive adjuvant therapies according to the oncologist’s experience and clinical condition of each patient.
Footnotes
Supplementary material for this article is available online.
Authors’ Note
Institution where the work was conducted: Universidade Federal de Minas Gerais (RH, GL, LM, GC, RA)
Acknowledegments
We thank FEPMVZ (Fundação de Estudo e Pesquisa em Medicina Veterinária e Zootecnia; Belo Horizonte, MG – Brazil), Vetpat Laboratory and Progen Biotecnologia (São Paulo, SP – Brazil).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
