Abstract
Canine prostatic carcinoma is a relevant model for human prostatic carcinoma. Survivin is proposed as a biomarker of malignancy in human prostatic cancer. Sox9 is a stem cell marker required for prostate development and expressed in several adult tissues. The aims of the present study were to evaluate the patterns and expression levels of 2 putative stem cell markers, survivin and Sox9, in canine benign prostatic hyperplasia (BPH) and prostatic carcinoma to investigate their potential as stem cell markers. Immunohistochemistry with specific antibodies was performed on 3 samples of normal prostate gland, 18 samples of canine BPH, and 16 samples of prostatic carcinoma. The basal cell layer of normal and hyperplastic prostatic lobules had nuclear Sox9 immunolabeling and nuclear and rarely cytoplasmic survivin immunostaining, identifying them as potential stem cell markers. Significantly more frequent survivin and Sox9 expression (≥10% of nuclei) was observed in prostatic carcinoma as compared with BPH. The potential coexpression of survivin with Sox9, androgen receptor, and p63 was also investigated in selected BPH and prostatic carcinoma cases with immunofluorescence, and a partial colocalization was observed. Results indicate that Sox9 and survivin could be considered markers of stemness in canine prostate cells. Given its role in proliferation, cells in the basal cell layer with nuclear survivin expression are likely to be transit-amplifying cells that maintain some stem cell proprieties.
Canine prostatic carcinoma is considered a relevant model for studying advanced hormone-refractory prostatic carcinoma in men. 1,17 Besides man, the dog is the only species to spontaneously develop prostate cancer, and these cancers have certain features in common: age of onset, frequency of invasion, sites of metastases, and histologic features. 17
It has been proposed that cancer contains a minor population of cells that can self-renew while giving rise to tumor cells: cancer stem cells (CSCs). 33 Human 14 and canine 24 prostatic tumors appear to contain a subpopulation of cells with stem cell features. CSCs are highly tumorigenenic, have a high metastatic potential, and show a relatively high resistance to traditional cancer therapies. 23 These findings have prompted the emergence of a new field of study in cancer treatment involving the targeting of CSCs; accordingly, this requires the identification of new CSC markers.
Survivin represents a well-known cancer therapy–resistance factor that is overexpressed in several tumor types. 2 A role for survivin was proposed in tumor initiation and progression as well as in the maintenance of CSCs. 4,29 It was proposed that survivin be used as a biomarker for malignancy in early screening for human prostatic cancer. 30 The overexpression of survivin was implicated in the development of prostatic carcinoma, leading investigators to evaluate the efficacy of survivin inhibitors as a new therapeutic option. 35 High expression of survivin was observed in putative CSCs isolated from prostatic adenocarcinoma in a murine model. 19,20 However, no data have been published regarding the expression and significance of survivin in canine prostatic tumors.
Sox9 is a stem cell marker expressed in several adult tissues and is required for human prostate development. 14,32 It contributes to the development of human prostatic carcinoma 7 and is therefore considered a potential prognostic marker in human prostatic carcinoma. 37 However, although Sox9 is expressed in a large proportion of prostate cancers, its relevance to prognosis varies depending on the molecular environment: It was recently demonstrated that loss of Sox9 expression was associated with prostatic carcinoma recurrence in ERG-positive and PTEN-deleted prostate cancers. 6
Based on the hypotheses that survivin and Sox9 could represent markers of CSCs and malignancy in canine prostatic tumors, the present study aimed to characterize the immunohistochemical patterns and levels of expression of survivin and Sox9 in canine benign prostatic hyperplasia (BPH) and prostatic carcinoma.
A further objective was to study the colocalization of survivin with Sox9, p63, and androgen receptor (AR) in a subset of canine prostatic hyperplastic and neoplastic lesions. p63 was identified as a prostate basal cell marker required for normal prostatic development 27,31 and seems to be required for maintainance of progenitor/stem cells. 27 It was also shown that p63 is discontinuously expressed in the basal cells of normal canine prostatic acini and hyperplastic lobules but dowregulated in canine prostatic carcinoma. 28
The androgen/AR axis controls the growth and development of prostate tissue as well as prostatic carcinoma progression, and AR differentially influences the characteristics of normal stem cells and prostate CSCs. 9,13 Recently it was shown that AR is expressed in canine BPH and, to a lesser extent, in prostatic carcinoma. 9,28 In the prostate, survivin expression seems to be regulated by androgen. Survivin expression/overexpression in human prostatic carcinoma was proposed as one of the molecular mechanisms of progression to androgen independence. The overexpression of survivin appeared to be sufficient to induce androgen-independent growth of androgen-dependent cells. 36
Material and Methods
Histologic Examination
The present study was carried out with 37 formalin-fixed, paraffin wax–embedded samples of canine prostate tissues from different dogs: 16 BPH, 16 primary prostatic carcinoma, and 1 necropsy sample of normal canine prostate gland retrospectively collected from the university archives, with unknown fixation time. Fresh material from 4 additional dogs—2 normal prostate glands and 2 BPH cases—was formalin fixed for a maximum of 24 hours. These additional samples were used specifically to investigate the pattern of Sox9 expression with immunohistochemistry and the possible effects of formalin fixation time on the immunostaining.
Histologic diagnosis was performed with hematoxylin and eosin–stained slides, according to World Health Organization guidelines. 16 Prostatic carcinoma samples were further classified according to the histologic subtypes 26 and the Gleason-like grading system. 25
Immunohistochemistry
Immunohistochemistry was performed with specific primary antibodies (Suppl. Table S1) per a previously described technique. 3,5 Briefly, deparaffinized and rehydrated sections were incubated with 3% H2O2 in absolute methanol for 45 minutes to inhibit endogenous peroxidase activity and then rinsed in 0.05M Tris-buffered saline (TBS; pH 7.6) for 5 minutes. Antigen retrieval was performed by heat treatment in Tris-EDTA buffer, pH 9.0, in a microwave oven for 5 minutes (4 cycles). After the last treatment, sections were left for 20 minutes in the buffer for cooling. To reduce nonspecific binding, slides were then incubated with normal goat serum (code MR*HRP-650; Biospa, Milan, Italy) for 10 minutes at room temperature before overnight incubation with the primary antibody in a humidified chamber at 4°C. After rinsing with TBS, immune complexes were treated at room temperature for 30 minutes with secondary biotinylated goat anti-mouse or anti-rabbit antibodies (1:200 dilution; Vector Laboratories, Burlingame, CA) and subsequently visualized with an avidin-biotin complex method (Vectastain ABC Kit; Vector Laboratories) for 30 minutes. Peroxidase activity was detected by a 5-minute application of 0.1% H2O2 in 3-3′-diaminobenzidine solution (code D5905; Sigma-Aldrich, St Louis, MO), followed by counterstaining with Mayer’s hematoxylin (Merck, Darmstadt, Germany) for 1 minute before rinsing, dehydrating, and mounting. A negative control was performed in all instances by omitting the primary antibody and incubating tissue sections with TBS and/or replacing it by an antibody of irrelevant specificity (rabbit anti-human von Willebrand factor polyclonal antibody from DAKO, Glostrup, Denmark). Sections of canine tissues known to display expression of the investigated molecules, as indicated in Supplemental Table S1, were used as positive controls.
Double Immunofluorescence
Double immunofluorescence was used to qualitatively investigate survivin-Sox9, survivin-p63, and survivin-AR nuclear coexpression in 16 selected cases (8 BPH and 8 prostatic carcinoma) based on our previous published study 26 with specific antibodies (Supplemental Table S1). Tissue samples were treated as described for the immunohistochemical procedure. A sequential protocol was used for double staining. Primary antibodies were applied overnight at 4°C. The first secondary antibody, biotinylated goat anti-rabbit (for survivin, 1:200 dilution; Vector Laboratories) was applied and incubated for 30 minutes at room temperature, and slides were then incubated with fluorescein-conjugated avidin (1:100 dilution in 0.1M NaHCO3, 0.15M NaCl buffer, pH 8.2–8.5; Vector Laboratories) for 10 minutes at room temperature. An avidin/biotin blocking step was performed by incubating slides for 15 minutes with avidin and then biotin (Avidin/Biotin Blocking Kit, code SP-2001; Vector Laboratories) at room temperature. A further protein blocking step was performed by incubating slides with normal goat serum (Vector Laboratories) for 15 minutes at room temperature, before the overnight application of the second primary antibody. The second secondary antibody—biotinylated goat anti-rabbit (for Sox9, AR) or anti-mouse (for p63, 1:200 dilution; Vector Laboratories)—was applied and incubated for 30 minutes at room temperature. Slides were then incubated with Texas red–conjugated avidin (Vector Laboratories) diluted 1:100 in a buffer composed of 0.1M NaHCO3 and 0.15M NaCl (pH 8.2–8.5) for 10 minutes at room temperature. Nuclei were counterstained with 4′,6-diamidino-2-phenylindole (DAPI; Vector Laboratories).
Quantification of Immunolabeling and Statistical Analysis
Nuclear and cytoplasmic survivin and nuclear Sox9 immunolabeling was assessed semiquantitatively by 2 pathologists (L.B. and M.R.) in 10 randomly selected high-powered fields (40×) and graded as a percentage of all neoplastic cells: 0, absent; 1 (low), >0% and <10% of cells; 2 (moderate), 10% to <25%; 3 (high), 25% to <50%; 4 (very high), ≥50%. Labeling intensity of cytoplasmic survivin was graded as no (0), weak (–/+), moderate (+), or strong (++) labeling.
Fisher exact test was used to compare semiquantitatively assessed immunoreactivity in BPH and prostatic carcinoma. For this purpose, the survivin cytoplasmic score and the survivin and Sox9 nuclear score were evaluated. The cases were grouped according to the nuclear score as follows: <10% positive nuclei (absent and very low number of positive nuclei) versus ≥10% positive nuclei (low/moderate and high number of positive nuclei). This choice depended on the distribution of nuclear score values for each protein and on several statistical analyses performed. Analyses were performed with SPSS 24 (IBM, Chicago, IL), with P < .05 considered to be significant.
Results
Histologic Examination Histopathology and Immunohistochemistry
Histologic features are reported in Supplemental Table S2. In half of the prostatic carcinoma cases (8 of 16), a single histologic pattern was seen (4 papillary, 3 solid/undifferentiated, and 1 small acinar/ductal); a mixed pattern was observed in the other 8 cases. Necrosis was observed in 12 of 16 (75%) cases, and 2 of 16 (12.5%) were characterized by abundant fibrous tissue (formerly termed “scirrhous prostatic carcinoma”).
The immunohistochemistry results are summarized in Table 1 and Supplemental Table S2.
Expression of Survivin and Sox9 in Normal Prostate, Benign Prostatic Hyperplasia, and Prostatic Carcinoma of Dogs.a

a Data show immunohistochemistry grades. 0, 0% of cells; 1, >0% and <10%; 2, 10% to <25%; 3, 25% to <50%; 4, ≥50%. Labeling intensity of cytoplasmic survivin was graded as no (0), weak (–/+), moderate (+), or strong (++) labeling. Dashes (—) indicate no data.
b Maximum 24 hours of formalin fixation time.
Survivin Expression in Normal Gland, BPH, and Prostatic Carcinoma
Survivin+ nuclei were present among the basal/reserve cell layer of normal (Fig. 1) and hyperplastic prostatic lobules: 0% to 10% of cells in the 11 of 16 (68.75%) cases with positive labeling (Fig. 2). Although only one normal gland was analyzed, normal prostatic tissue surrounding the BPH lesions was also evaluated and showed the same pattern of expression of the molecule. Most BPH cases (12 of 16, 75%) showed patchy cytoplasmic immunostaining, with a low to moderate expression (0%–25% of neoplastic cells) in most cases (10 of 12, 83,3%). In contrast, prostatic carcinomas had more diffuse cytoplasmic expression as well as nuclear expression of survivin. Cytoplasmic staining was observed in all prostatic carcinoma samples, with half of the cases (8 of 16) showing low to moderate expression (0%–25% of neoplastic cells) and the other half, high expression (>25% of neoplastic cells). Positive nuclei were present in all except 1 case of prostatic carcinoma, with most (9 of 16, 56%) showing moderate to high expression (10% to >50% of positive nuclei; Fig. 3), present mainly among the neoplastic cells with basal cell morphology in the areas with tubular-papillary pattern.
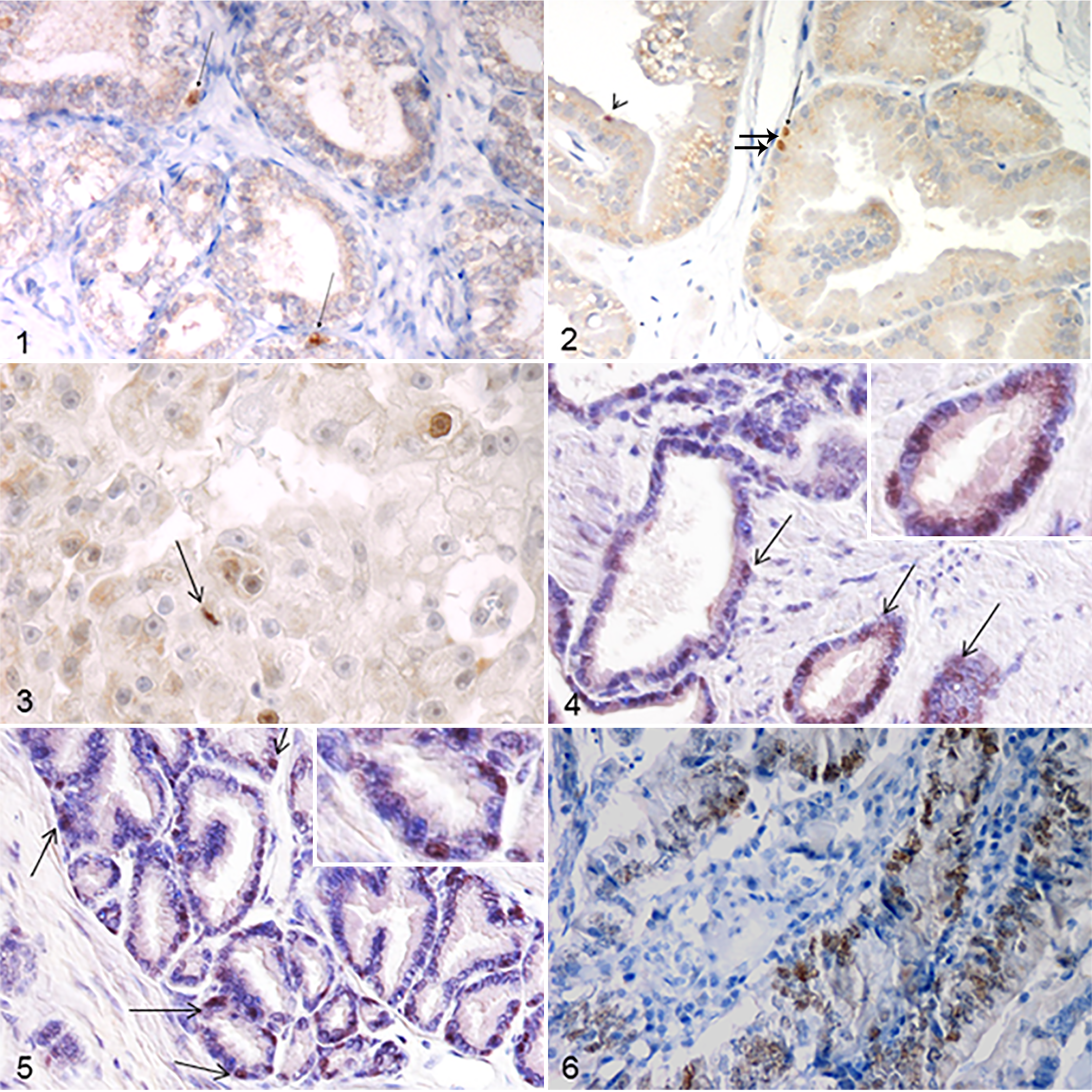
Sox9 Expression in Normal Gland, BPH, and Prostatic Carcinoma
Sox9+ nuclei were observed in scattered cells among the basal/reserve cell layer of 2 of 3 normal prostatic glands (Fig. 4) and 5 of 15 hyperplastic lobules (Fig. 5). Of these positive cases, 2 normal glands and 2 BPH cases were freshly sampled, while the other 3 BPH cases were from the archive and the fixation time was not known, suggesting that prolonged fixation may interfere with Sox9 immunostaining. 34 Among prostatic carcinomas, 9 of 16 cases were positive, and most (7 of 9) showed a moderate to high expression (>25% of neoplastic cells with nuclear immunolabeling; Fig. 6).
Double Immunofluorescence
From a qualitative evaluation of the immunofluorescence-stained slides, only a partial colocalization of survivin and p63 (survivin+/Sox9+) was observed. In BPH, survivin–/p63+ nuclei were more numerous (5%–10%); survivin+/p63– nuclei were scattered and rare; and survivin+/p63+ nuclei were few (<5% of cells were double positive) and observed among basal cells (Fig. 7). In contrast, in prostatic carcinoma, survivin+/p63– nuclei were more numerous (10%–25%), and survivin+/p63+ nuclei were scattered and few (<5% of cells were double positive). p63 cytoplasmic immunostaining was also evident (Fig. 8).
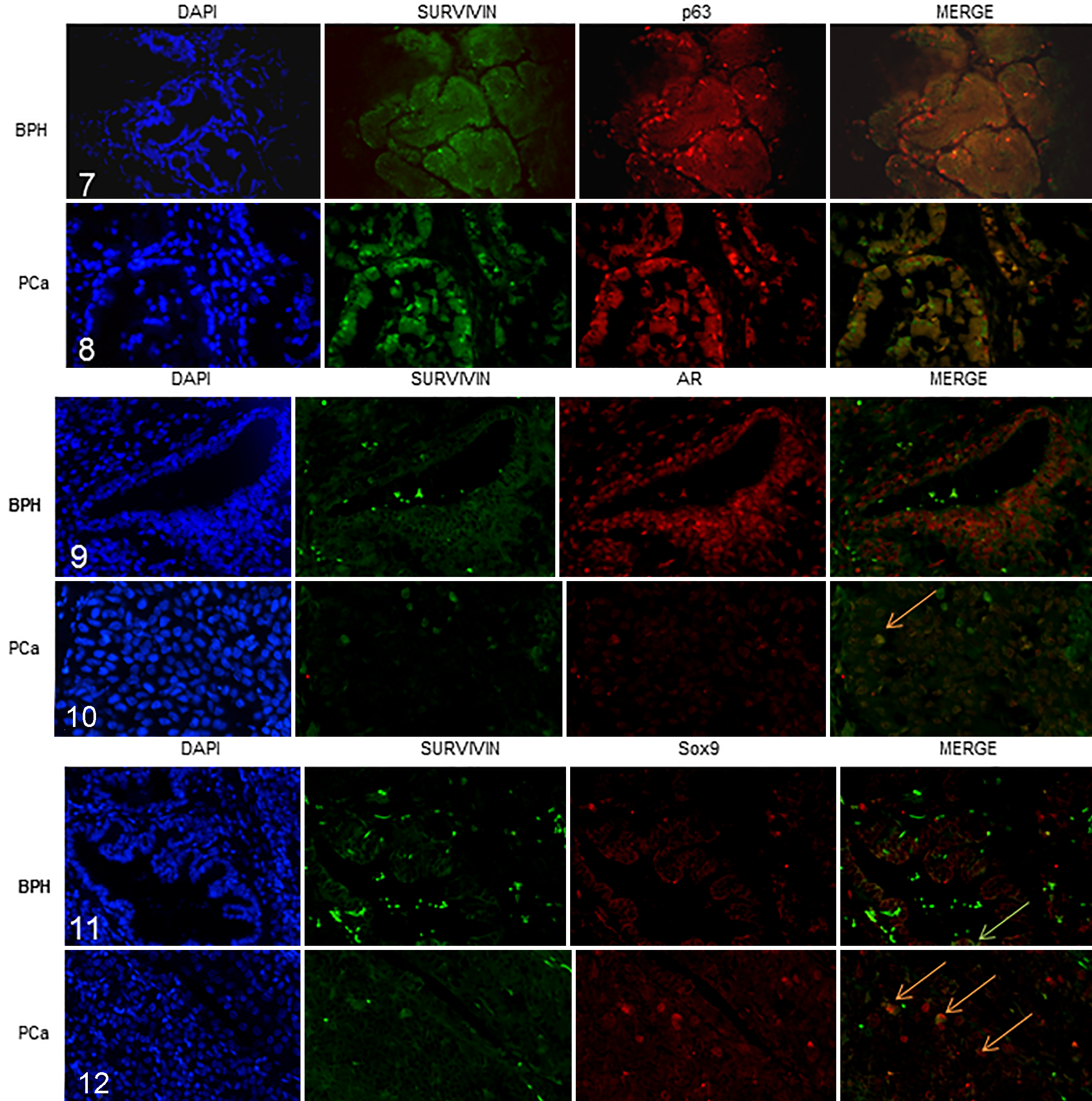
In BPH cases, a higher number of survivin–/AR+ cells (>50%) was observed as compared with survivin+/AR+ cells (5%–10%), while no survivin+/AR– cells were observed (Fig. 9). Nuclear immunolabeling in prostatic carcinoma was characterized by numerous survivin+/AR+ cells (25%–50%), and survivin+/AR– cells were also evident (10%–25%; Fig. 10). Several neoplastic cells showed intense aberrant AR cytoplasmic expression as well as cytoplasmic immunolabeling for survivin.
In BPH cases, survivin+/Sox9– cells were more numerous (10%–25%) than survivin+/Sox9+ cells (<5% of double-positive cells), while no survivin–/Sox9+ cells were observed (Fig. 11). In the prostatic carcinoma cases, a higher number of survivin+/Sox9+ double-immunostained cells was observed (Fig. 12), with intense nuclear and faint cytoplasmic immunolabeling and a few (<5%) survivin+/Sox9– and survivin–/Sox9+ cells.
Statistical Analysis
Nuclear survivin and Sox9 expression both appeared to be higher (≥10% of neoplastic cells with positively labeled nuclei) in prostatic carcinoma as compared with BPH cases (P < .01 for both). No significant differences were observed in cytoplasmic survivin expression between BPH and prostatic carcinoma cases.
Discussion
This study is the first to characterize the immunohistochemical expression of survivin and Sox9 in normal, hyperplastic, and neoplastic canine prostate. The main aims were to verify if Sox9 and survivin are expressed by canine prostatic epithelial cells and if they could represent prostatic stem cell markers.
The nuclear immunostaining observed in the basal cell layer of normal prostate gland is consistent with Sox9 as a stem cell marker in canine prostate, similar to what has been reported in the human literature. 7 In contrast to our predictions, the first round of testing with Sox9 immunohistochemistry, with cases selected from the university’s archives, showed no positive cells in the normal glands and only a few positive samples among the BPH cases. A second round of testing, with freshly sampled prostatic tissue fixed in formalin for 24 hours, revealed positive staining in all 4 cases, and it was more intense and evident than that observed in samples with longer fixation time. This suggests that prolonged formalin fixation time decreased Sox9 antigen detection, indicating that this antigen is more vulnerable to degradation than the other investigated molecules, such as survivin, p63, and AR. 34
The role of survivin in stem cells 15 might indicate that the positive cells found in the basal cell layer of normal glands are stem cells. However, since survivin also has a role in proliferation, it is possible that these cells represent partially differentiated, proliferating, transit-amplifying cells that maintain some stem cell properties.
The prostate basal cell marker p63 was already shown to be involved in epithelial development by regulating stem cell/transit-amplifying cells, their differentiation, and cell death. 8,21 Results obtained from the immunofluorescence study showed that in BPH, survivin and p63 are expressed in different cells, with only partial, minimal colocalization. Our hypothesis is that they mark transit-amplifying cells in these lesions—an undifferentiated population of cells in transition between stem cells and differentiated cells, with intermediate features. This would support the previously reported theory that at least 2 biologically distinct populations of basal cells exist in the canine prostate gland 18 and that these cells can be involved in the development/maintenance of BPH.
Other recent studies characterizing the expression of stem cell markers showed that coexpression of CD44 and CD133 detects stem cells in canine prostatic cancer cell lines. 22,24 Further studies should be done to investigate whether survivin and Sox9 are coexpressed with these markers.
In human prostatic tissue, survivin expression is regulated by androgen stimulation. 36 In the canine prostatic tissue studied here, the presence of survivin+/AR– cells in prostatic carcinoma, with the increased expression of survivin in prostatic carcinoma versus BPH, suggests a role for survivin in the progression to androgen independence in the dog as well. The survivin+/AR– cells could represent a subpopulation of androgen-independent tumor cells. These may be important in the early stages of development of prostate cancer and, if present in human prostatic carcinoma, may be the cause of the failure of androgen ablation therapy that occurs in most human advanced cases. 10 Our findings suggest the presence, in canine prostatic carcinoma, of a subpopulation of neoplastic cells with low levels of AR and high levels of survivin and some features of stem cells (prostatic stem cells or transit-amplifying cells). This supports the possibility that this tumor derives from basal/stem cell–like epithelial cells expressing low levels of AR. 11,17
The origin of CSCs within solid tumors has not yet been clarified and would appear to vary according to tumor type: tumor cells could originate from normal tissue stem cells or from partially differentiated transit-amplifying cells that undergo oncogenic transformation to produce CSCs. 12 Further studies should be done to better characterize this subpopulation and its features of stemness and to investigate the possible regulation of the AR-activated pathways on survivin expression in canine prostatic tissue and prostatic neoplasms.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985818794161 - Survivin and Sox9: Potential Stem Cell Markers in Canine Normal, Hyperplastic, and Neoplastic Canine Prostate
Supplemental Material, DS1_VET_10.1177_0300985818794161 for Survivin and Sox9: Potential Stem Cell Markers in Canine Normal, Hyperplastic, and Neoplastic Canine Prostate by Laura Bongiovanni, Francesca Caposano, Mariarita Romanucci, Valeria Grieco, Daniela Malatesta, Chiara Brachelente, Marcella Massimini, Cinzia Benazzi, Rachel E. Thomas, and Leonardo Della Salda in Veterinary Pathology
Footnotes
Acknowledgements
We thank the Veterinary Pathology Diagnostic Centre of the Utrecht University for providing tissue blocks of canine prostatic lesions, as well as Marina Baffoni and Saskia Essen-van Dorresteijn for their technical support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
