Abstract
Self-renewal of the intestinal epithelium originates from stem cells located at the crypt base. Upregulation of various stem cell markers in intestinal epithelial neoplasms indicates a potential role of stem cells in tumorigenesis. In this study, the immunoreactivity of potential intestinal stem cell markers (Sry box transcription factor 9 [Sox9], homeodomain-only protein [Hopx], survivin) and tuft cell marker doublecortin-like kinase 1 (DCLK1) in normal canine intestine and intestinal epithelial neoplasms was investigated. Formalin-fixed paraffin-embedded (FFPE) small and large intestine as well as intestinal neoplasms (55 colorectal adenomas [CRAs], 17 small intestinal adenocarcinomas [SICs], and 12 colorectal adenocarcinomas [CRCs]) were analyzed immunohistologically. Potential stem cell markers Sox9, Hopx, and survivin were detected in the crypts of normal canine small and large intestine. DCLK1+ tuft cells were present in decreasing numbers along the crypt-villus axis of the jejunum and rarely detectable in large intestine. In canine intestinal epithelial tumors, nuclear Sox9 immunoreactivity was detectable in 84.9% (CRA), 80% (CRC), and 77% of epithelial neoplastic cells (SIC). Hopx and survivin were expressed within cytoplasm and nuclei of neoplastic cells in benign and malignant tumors. DCLK1 showed a cytoplasmic reaction within neoplastic cells. The combined score of Hopx, DCLK1, and survivin varied among the examined cases. Overall, malignant tumors showed lower DCLK1 scores but higher Hopx scores in comparison with benign tumors. For survivin, no differences were detectable. In conclusion, stem cell markers Sox9, Hopx, and survivin were detectable at the crypt base and the immunoreactivity of Sox9, DCLK1, survivin, and Hopx was increased in canine intestinal adenomas and adenocarcinomas compared with normal mucosa.
Keywords
Gastrointestinal cancer is among the most prevalent human cancers with very high mortality. 9 Although gastrointestinal tumors are reported rare in many domestic animals, dogs and cats show the highest tumor incidence among the domestic species. 65 In dogs, most neoplastic intestinal lesions occur in the large intestine as it has been reported in humans.9,35,48,59,65,69 As in humans, canine intestinal carcinomas have a poor prognosis due to local recurrence, metastasis to regional lymph nodes, and peritoneal metastases.18,36,65
The intestinal epithelium has the ability to rapidly regenerate during basal homeostasis and in response to injury due to the presence of intestinal epithelial stem cells (IESCs), which are located at the base of intestinal crypts beneath the +4 position, counted relative to the bottom of the crypts.31,62,89 From those so-called crypt base stem cells, the rapidly dividing transit-amplifying cells develop, which then migrate apically toward the villus where enterocytes are shed.2,16,47 Proliferating cells are detectable from the crypt base up to the crypt-villus transition zone. Epithelial cells along the villus are postmitotic and fully differentiated. 26
IESCs are known to express Sry box transcription factor 9 (Sox9), leucine-rich repeat-containing G-coupled receptor 5, and markers of proliferation, whereas potential reserve stem cells, located just above the IESCs at the +4 position, demonstrate homeodomain-only protein (Hopx) immunoreaction.3,4,11,72,75 Furthermore, survivin is reported as a factor involved in maintenance of IESCs and gut homeostasis.54,55 Besides studies on human and murine intestine, there are only single reports in which IESCs expressing Sox9 and/or Hopx were demonstrated in normal equine and porcine small and large intestinal tissue by immunofluorescence microscopy.29,30
Doublecortin-like kinase 1 (DCLK1) is known to be expressed by tuft cells.25,33,45,60 Furthermore, these cells are known to express Sox9, β-tubulin, and cytokeratin 18.28,38 Tuft cells represent a chemosensory cell type that is involved especially in tissue repair and type 2 immunity.27,45,57,63,71,87 Recently, the presence of Sox9- and DCLK1-expressing cells in canine intestinal organoids and intestinal biopsies from healthy dogs was confirmed by in situ hybridization. 11
Results from studies in human colorectal carcinomas (CRCs) strongly suggest that IESCs located at the bottom of intestinal crypts are the cells of origin of the majority of the tumors. 78 In human CRCs, altered mRNA and protein expression of various stem cell markers has been reported.10,13,43,50,52,53,76,79 Among these markers, Sox9, Hopx, and DCLK1 were reported to be expressed by a special subpopulation of cancer cells, called the cancer stem cells (CSCs).22,34,43,51,58,83,86 In studies of human CRCs, the overexpression of Sox9 and DCLK1 was correlated with tumor progression, advanced tumor stage, or poor differentiation.10,13,43,50,52,53,76,79 In addition, survivin marks CSCs and plays an important role in initiation and progression in several human tumors including colorectal cancer.1,15,37,41 So far, there are only single reports about the protein expression of Sox9 and survivin in canine tumors such as prostate carcinomas and various canine skin neoplasms.5 –7,20 The protein expression of stem cell markers in the normal canine intestine or in canine intestinal tumors, however, has not been described.
One aim of this study was to characterize the immunohistochemical expression of the potential stem cell markers Sox9, Hopx, DCLK1, and survivin in the normal canine small and large intestine. A further aim was to analyze the immunohistochemical expression of those stem cell markers in canine intestinal adenomas and adenocarcinomas.
Materials and Methods
Animals
For the examination of marker expression in normal canine gut, tissue samples from dogs without signs of gastrointestinal disorders were used. The dogs were presented at the Small Animal Clinic, University of Veterinary Medicine Hannover, with diseases unrelated to the gastrointestinal tract. Euthanasia was elected in agreement with the owners due to poor prognosis in course of disease. The animals were sent to the Department of Pathology, University of Veterinary Medicine Hannover within 2 hours after euthanasia for necropsy (Supplemental Table S1). Full-thickness specimens of jejunum and/or colon were obtained during necropsy. Tissue samples from all animals were fixed in 10% neutral-buffered formalin and embedded in paraffin according to routine protocols.
Formalin-fixed paraffin-embedded (FFPE) intestinal samples from dogs with intestinal epithelial tumors from 2017 to 2019 were selected from the archive of the Department of Pathology, University of Veterinary Medicine Hannover. Selected cases comprised 84 epithelial tumors (55 adenomas, 29 adenocarcinomas) and in 4 of these cases the respective regional lymph nodes were included. Samples were surgical and endoscopic biopsies taken under general anesthesia by practitioners or by clinicians of the Clinic for Small Animals, University of Veterinary Medicine Hannover, and had been sent to the Department of Pathology for diagnostic histopathology (Supplemental Table S2).
Histopathology
Tissue samples from all animals were fixed in 10% formalin and embedded in paraffin according to routine protocols. Hematoxylin-eosin stain was applied to 2-µm sections of FFPE tissues after deparaffinization and rehydration. Two pathologists (W.R. and M.H.T.) independently analyzed the slides based on the World Health Organization classification for gastrointestinal neoplasms. 43 The mitotic count was measured in 10 high-power fields (HPF, objective magnification 40×; area per HPF: 0.16 mm²) and the result was divided by 10, as described by Woldemeskel and colleagues. 85 The depth of invasion and the presence of lymphatic invasion or metastases were recorded in all cases of adenocarcinomas.
Immunohistochemistry
Immunohistochemistry was performed according to established protocols using the avidin-biotin complex method and 3,3′-diaminobenzidine (DAB) with minor modifications. 80 Briefly, 2-µm-thick sections of FFPE material on Superfrost PlusTM slides (Gerhard Menzel GmbH, Braunschweig, Germany) were deparaffinized in Roti® Histol (Carl Roth, Karlsruhe, Germany) and rehydrated in descending alcohol series. After blocking endogenous peroxidase by 0.5% hydrogen peroxidase for 30 minutes, heat-induced antigen retrieval was performed (citrate buffer at pH 6, 95°C, 20 minutes). After cooling, unspecific binding was blocked with normal goat serum for 30 minutes at room temperature, and the sections were incubated with primary antibodies (Supplemental Table S3) at 4°C overnight (β-tubulin, DCLK1, Hopx, Ki-67, Sox9) or 75 minutes at room temperature (active caspase-3, cytokeratins, survivin, vimentin).
An Avidin/Biotin Blocking Kit (Vector Laboratories, Burlingame, CA) was used according to manufacturer’s instructions to decrease background staining. After applying the appropriate biotin-labeled secondary anti-bodies (goat antirabbit or goat anti-mouse, respectively), the slides were incubated with the avidin-biotin complex, each step lasting for 30 minutes at room temperature. For some antibodies, biotinylated tyramine followed by another incubation with avidin-biotin complex was used for signal enhancement (each 15 minutes; Supplemental Table S3). Thereafter, slides were labeled with DAB and counterstained with hematoxylin before coverslipping. Normal murine intestinal FFPE samples served as positive controls for expression pattern of DCLK1, Sox9, and Hopx in normal canine intestine. No animals were killed for the purpose of this study. Canine squamous cell carcinoma served as positive control for survivin, 6 and canine lymph nodes were used for Ki-67 and caspase-3.
The immunoreactivity of antibodies (Sox9, DCLK1, Hopx, survivin) was shown by western blot analysis of frozen normal canine intestinal tissues (Supplemental Figures S1–S4). Furthermore, the expression pattern of Sox9 and DCLK1 was confirmed by in situ hybridization of normal canine small and large intestine as well as representative neoplasms. RNAScopeTM probes for canine DCLK1 and Sox9 were used according to the manufacturer’s protocol (Supplemental Figures S5–S12). 11
For negative controls, primary antibodies were replaced by either rabbit serum or appropriate murine immunoglobulins, and slides were labeled according to the respective protocols.
To confirm the presence of tuft cells, the coexpression of Sox9 and DCLK1 was analyzed by immunofluorescence. After incubation with the DCLK1 primary antibody, a fluorochrome-labeled goat anti-rabbit secondary antibody ((Alexa FluorTM 488; 1:200; 30 minutes; room temperature) was added. For Sox9 detection, an Alexa FluorTM 555-labeled antibody was applied to the same slides. Slides were counterstained by 4′,6-diamidino-2-phenylindole (1:200,000; 3 minutes).
Quantitative Evaluation of Sox9, Ki-67 Index, and Active Caspase-3 Expression in Normal Canine Intestine and Intestinal Neoplasms
Expression of the markers was evaluated on slides from normal jejunal and colonic tissue of 5 dogs each. From each section (normal intestine and intestinal neoplasms), 5 photomicrographs were randomly taken with an Olympus BX-51 digital camera microscope (objective magnification 40×) and the cellSens imaging software (Olympus Soft Imaging Solutions GmbH, Münster, Germany). Positive and negative epithelial cells were counted with the ImageJ-based imaging processing package called Fiji (http://fiji.sc/Fiji). 74 Percentages of positive cells were calculated.
Quantitative Evaluation of DCLK1 Expression in Normal Intestinal Mucosa
From each dog without gastrointestinal disorders, 10 to 15 photomicrographs (depending on sample size; objective magnification: 20×) were randomly taken from every slide representing each aspect of the gut mucosa (villus, crypt-villus transition zone, and crypt in small intestine; surface epithelium and crypt in colon). Examined slides originated from the jejunum of 8 dogs and from the colon of 5 dogs, and DCLK1 positive tuft cells were counted per millimeter of epithelial lining as described above. Immunofluorescence photomicrographs were taken with an Olympus IX70 digital camera microscope (objective magnification 40×) and the cellSens imaging software.
Semiquantitative Analysis of Hopx, DCLK1, and Survivin Expression in Intestinal Epithelial Tumors
Intestinal neoplasms were analyzed for the percentage of labeled epithelial cells, labeling intensity, and intracellular distribution of signal. A modified scoring system was based on the labeling intensity (0 = no labeling, 1 = weak intensity, 2 = moderate intensity, 3 = marked intensity) and percentage of labeled epithelial cells (0 = <5%; 1 = 5%–25%, 2 = 26%–50%, 3 = 51%–75%; 4 = >75%). 5 Both scores were multiplied to create a combined score ranging from 0 to 12. For DCLK1, positively labeled intramural neurons were set as reference for marked labeling (score 3). Intestinal samples of 5 dogs were evaluated for normal survivin and Hopx expression pattern and localization of positive cells.
Statistical Analysis
All results are presented as means of data sets. Statistical analysis was performed by SAS Enterprise guide 7.1 (SAS Institute Inc., Cary, NC). Shapiro-Wilk test showed that data sets lack normal distribution (P < .05). Therefore, Wilcoxon 2-sample test (Mann-Whitney U test) was applied to analyze differences between expression levels of Sox9, Ki-67, and caspase-3 as well as mitotic count. The 5% level (P ≤ .05) was assumed to be statistically significant. To test whether the categorical data sets (eg, invasiveness, DCLK1) influenced the quantitative target value (eg, Sox9), variance analyses (Kruskal-Wallis test) were performed. To calculate the association of categorical data sets (eg, Hopx, survivin, DCLK1, invasiveness), Fisher exact test was applied. Correlation of quantitative data sets was analyzed by Spearman rank correlation coefficient ρ (Sox9, Ki-67, capase-3, mitotic count).
Results
Description of Cases
Fifty-five intestinal adenomas were identified, restricted to the large intestine. Tubular adenomas represented the major subtype of colorectal adenoma (CRA) with 54/55 of all cases. Only 1 villous adenoma was present. The mean age of dogs with CRA was 7.3 years (range: 2–13 years; Supplemental Table S2).
The study included 29 intestinal adenocarcinomas (16 acinar, 7 papillary, 3 mucinous, 1 signet-ring carcinoma, 2 undifferentiated), of which 17 were present in the small intestine and 12 in the large intestine. In the small intestine, 11/16 acinar, 1/7 papillary, 2/3 mucinous, 1 signet-ring carcinoma, and 2 undifferentiated adenocarcinomas were detected. In the large intestine, 5/16 acinar, 6/7 papillary, and 1/3 mucinous adenocarcinomas were present. The mean age of dogs with small intestinal adenocarcinoma (SIC) was 10.3 years (range: 7–13 years), and with CRC was 8.1 years (range: 3–12 years; Supplemental Table S2).
Of the 17 SICs, 1 showed invasion only into the submucosal tissue, 4 showed invasion into the muscle layers, and 12 showed transmural invasion. In 5 cases, invasion into lymphatics was present with lymph node metastases in 2 of these cases. Lymph nodes were available only in these 2 cases.
Of 12 large intestinal adenocarcinomas, 3 had only submucosal invasion, 1 showed invasion into the muscle layer, and 4 showed transmural growth. Three tumors showing transmural invasion revealed invasion into lymphatics and in 2 of these cases lymph node metastases were present. In the other one, no lymph node was submitted for evaluation. Four samples had only submucosal tissue; therefore, depth of invasion was not evaluated.
Transmural invasion was detected in 11/16 acinar, 1/16 papillary, 2/16 mucinous, 1/16 signet-ring, and 1/8 undifferentiated carcinomas. Lymph node metastases were present in 3/4 acinar and 1/4 undifferentiated carcinomas.
Expression of Sox9 in Normal Canine Intestine and Intestinal Neoplasms
As in murine intestine, canine intestinal epithelial cells expressed Sox9 within the nuclei in the basal crypt zones of small and large intestine (Supplemental Figure S13). In addition, there were scattered Sox9+ cells within the transition zone and only few cells in the villus area. In the jejunum, these scattered cells were also labeled by DCLK1, as shown by immunofluorescence. At the crypt base, Sox9+ cells did not coexpress DCLK1. Overall, Sox9 expression was detected in 32.8% (range: 21.9%–43.0%) of small intestinal and 37.2% (range: 37.8%–45.3%) of large intestinal epithelial cells.
In intestinal neoplasia, Sox9 showed a diffuse nuclear labeling. Sox9+ tumor cells were distributed equally throughout the benign and malignant epithelial neoplastic cells (Figs. 1–3). In CRAs, the mean percentage of positive cells was 84.9% (range: 75.5%–91.1%). In CRCs and SICs, labeling intensity of infiltrating tumor cells did not show detectable decrease at the invasive front. In CRC, Sox9+ cells represented an average of 80.0% (range: 70.3%–89.4%) and in SIC 77.0% (range: 62.7%–88.5%).
Expression of Hopx in Normal Canine Intestine and Intestinal Neoplasms
Hopx was expressed in nuclei and cytoplasm of single epithelial cells at the base of intestinal crypts. Positive cells were rarely detected at the base of intestinal crypts (Supplemental Figure S14). The immunoreactivity resembled the expression pattern observed in murine intestinal samples.
Hopx was expressed by neoplastic cells of intestinal adenomas and adenocarcinomas. Intestinal adenomas showed an intense diffuse cytoplasmic labeling with low to moderate numbers of labeled nuclei within the neoplastic cells. Hopx expression varied among individual cases (Figs. 4–6; Table 1). In all adenocarcinomas except for the papillary subtypes, expression was increased within deep invasive nests compared with the luminal aspects of the adenocarcinomas. However, in papillary adenocarcinomas, the intraluminal proliferations showed a similar labeling pattern compared with the CRAs, whereas the invasive tumor cells lacked or showed only weak expression of Hopx.
Hopx’s, DCLK1’s, and survivin’s combined immunolabeling scores in 55 canine colorectal adenomas, 17 small intestinal adenocarcinomas, and 12 colorectal adenocarcinomas.
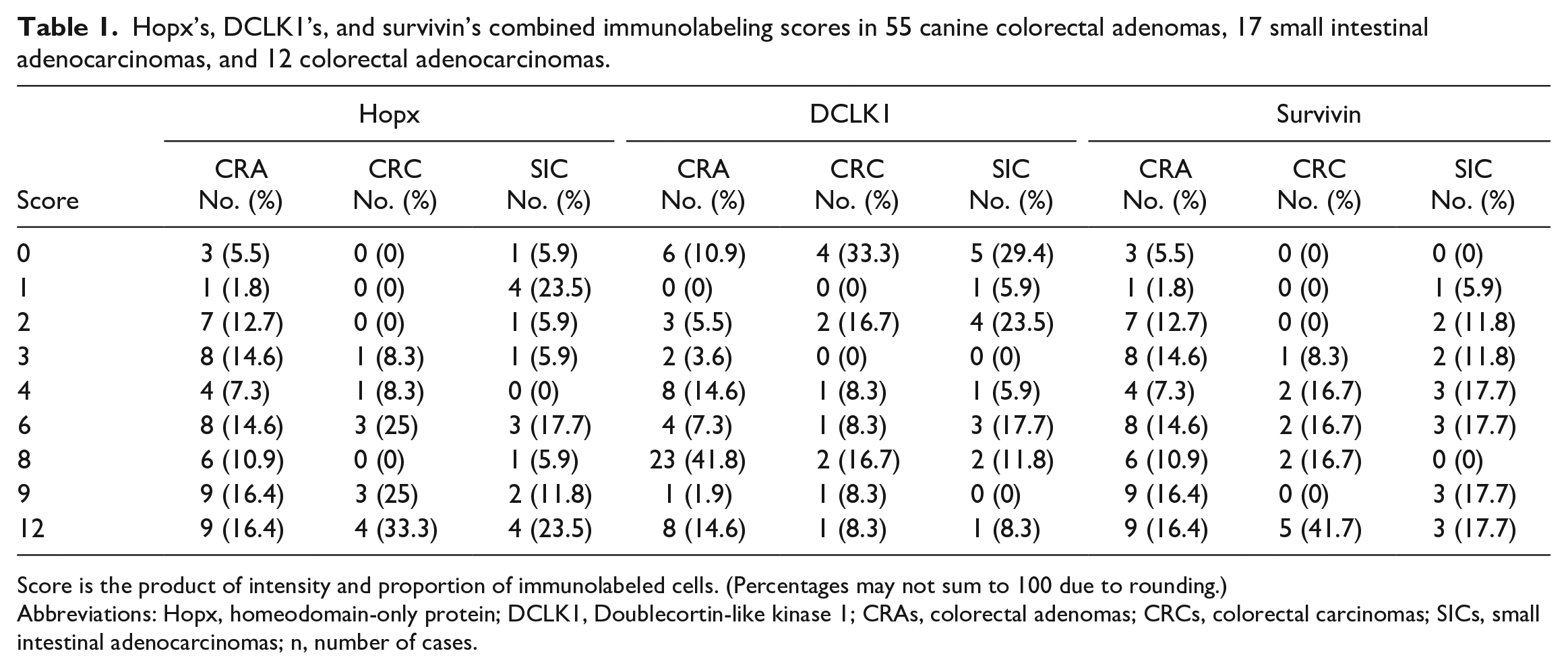
Score is the product of intensity and proportion of immunolabeled cells. (Percentages may not sum to 100 due to rounding.)
Abbreviations: Hopx, homeodomain-only protein; DCLK1, Doublecortin-like kinase 1; CRAs, colorectal adenomas; CRCs, colorectal carcinomas; SICs, small intestinal adenocarcinomas; n, number of cases.
Expression of Survivin Within Intestinal Epithelial Neoplasia
In control dogs, cells at the crypt base expressed survivin within nuclei. Expression pattern was diffuse in the majority of epithelial nuclei, but some cells showed granular to clumped labeling of chromatin, distinct labeling of nuclei, and prominent labeling of the mitotic spindle (Supplemental Figure S15). Survivin+ cells were detected at the crypt base, but also in the upper aspects of crypts and less frequent in the crypt-villus transition zone.
Survivin was expressed in the cytoplasm and nuclei of varying numbers of neoplastic cells of benign and malignant lesions. In addition, an intense labeling of the mitotic apparatus was present (Figs. 7–9; Table 1).
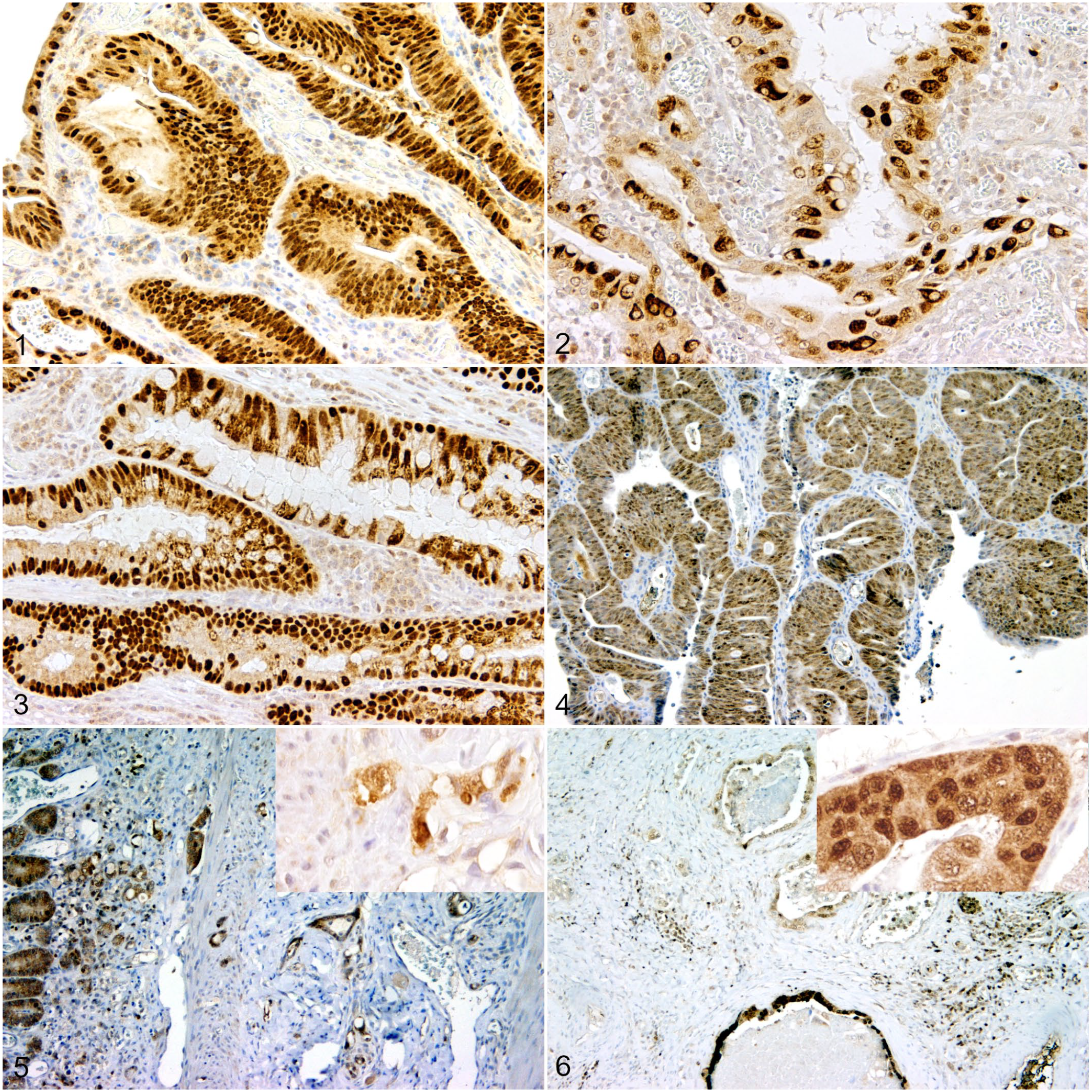
Intestinal neoplasms, dogs. Sox9 immunohistochemistry, 3,3′-diaminobenzidine (DAB) chromogen.
Expression of DCLK1 Within the Normal Canine Intestine and Intestinal Epithelial Neoplasms
DCLK1 was expressed in the cytoplasm of single cells, as it was in murine intestine. In dog samples, DCLK1+ cells were detectable mainly within the small intestine. The density in the jejunum decreased along the crypt-villus axis (Supplemental Figure S16a–c). The mean density of DCLK1+ tuft cells density was 2.60 cells/mm of epithelial lining in the crypt area 1.72 tuft cells/mm in the crypt-villus transition zone, and 0.91 cells/mm at the villus. Presence of tuft cells was confirmed by the coexpression of DCLK1 and Sox9 by immunofluorescence (Supplemental Figure S17a–c). Cells that showed similar localization and morphology as DCLK1+ cells additionally expressed β-tubulin and cytokeratin 18. In the large intestine, single DCLK1+ cells were present mainly beneath the cells at the base of the crypts.
DCLK1 showed a cytoplasmic reaction in CRA, CRC, and SIC. Labeling intensity varied among individual slides and tumor subtypes. In general, a lower number of immunolabeled cells was detected in adenocarcinomas than in adenomas (Figs. 10–12; Table 1).
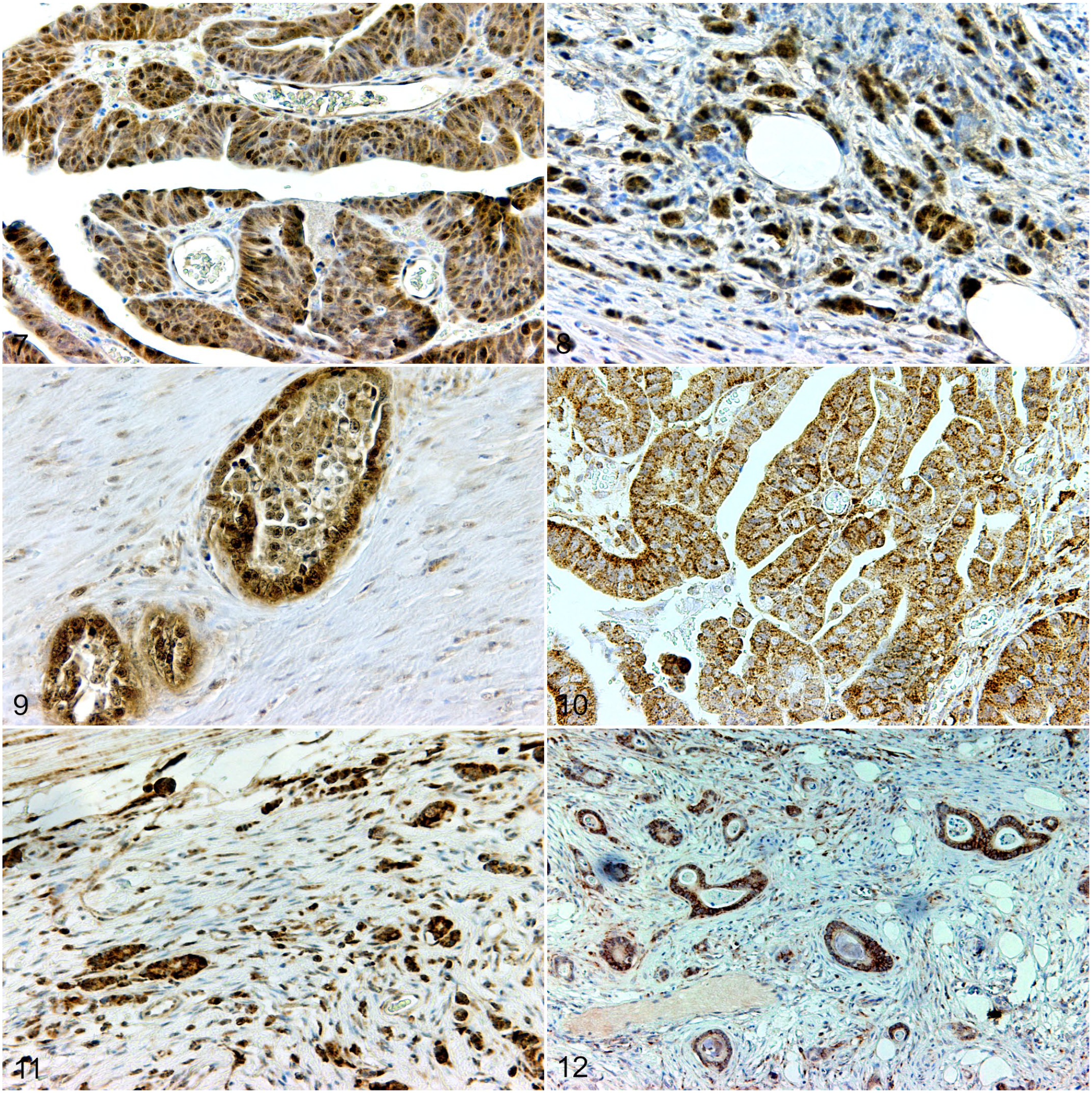
Intestinal neoplasms, dogs. Survivin immunohistochemistry. 3,3′-diaminobenzidine (DAB).
Evaluation of Ki-67 Index and Active Caspase-3 Expression in Normal Canine Intestine and Intestinal Neoplasms
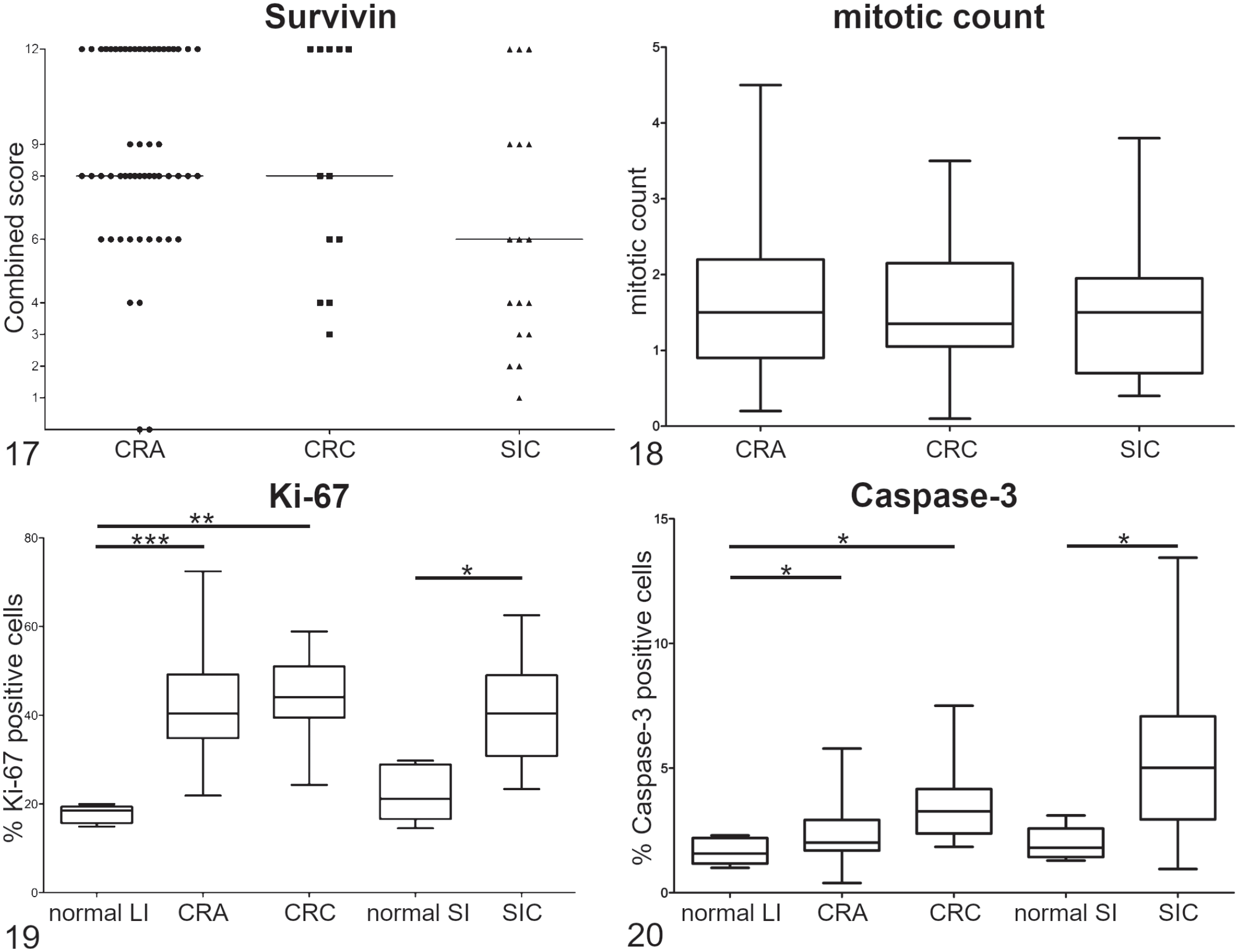
Ki-67 immunoreactivity was restricted to the crypts of normal canine small and large intestine. Normal small intestine showed immunoreactivity in 22.4% of cells (range: 14.5%–29.8%), whereas in normal large intestine 17.7% (range: 14.9%–20.0%) were positive for Ki-67. Ki-67 index in adenomas was 42.3% (range: 21.9%–72.4%), in CRC was 43.7% (range: 24.3%–58.9%), and in SIC was 39.5% (range: 23.4%–62.5%). The mean mitotic count of CRA was 1.7 mitoses (range: 0.2–4.5) per HPF (objective magnification 40×, area per HPF: 0.16 mm²), CRC was 1.7 mitoses per HPF (range: 0.1–3.5), and SIC was 1.5 mitoses per HPF (range: 0.4–3.8).
Activated caspase-3 was detected in 2.4% of tumor cells in CRA (range: 0.4–5.8), 3.5% (range: 1.8–7.5) in CRC, and 5.5% (range: 1.0–13.4) in SIC. In normal canine intestine, active caspase-3 was expressed by 2.0% (range: 1.3%–3.1%) of small intestinal and 1.7% (range: 1.0%–2.3%) of large intestinal epithelial cells.
Statistical Analysis
The frequency of immunolabeling for Sox9 was not only significantly different when comparing normal mucosa with CRA (P = .0002), CRC (P = .0019), or SIC (P = .001), but also when comparing expression of CRA and CRC (P = .011). No significant differences were detected between CRC and SIC (P = .30; Fig. 13).
Combined scores of Hopx expression in CRAs, CRCs, and SICs did not reveal any significant differences (Fig. 14).
In normal canine small intestine, DCLK1 expression was significantly different when comparing crypt and transition zone (P = .03), transition zone and villus (P = .0019), as well as crypt and villus (P < .0001). No differences were detectable between the different tumor types for combined score of DCLK1 (Figs. 15, 16).
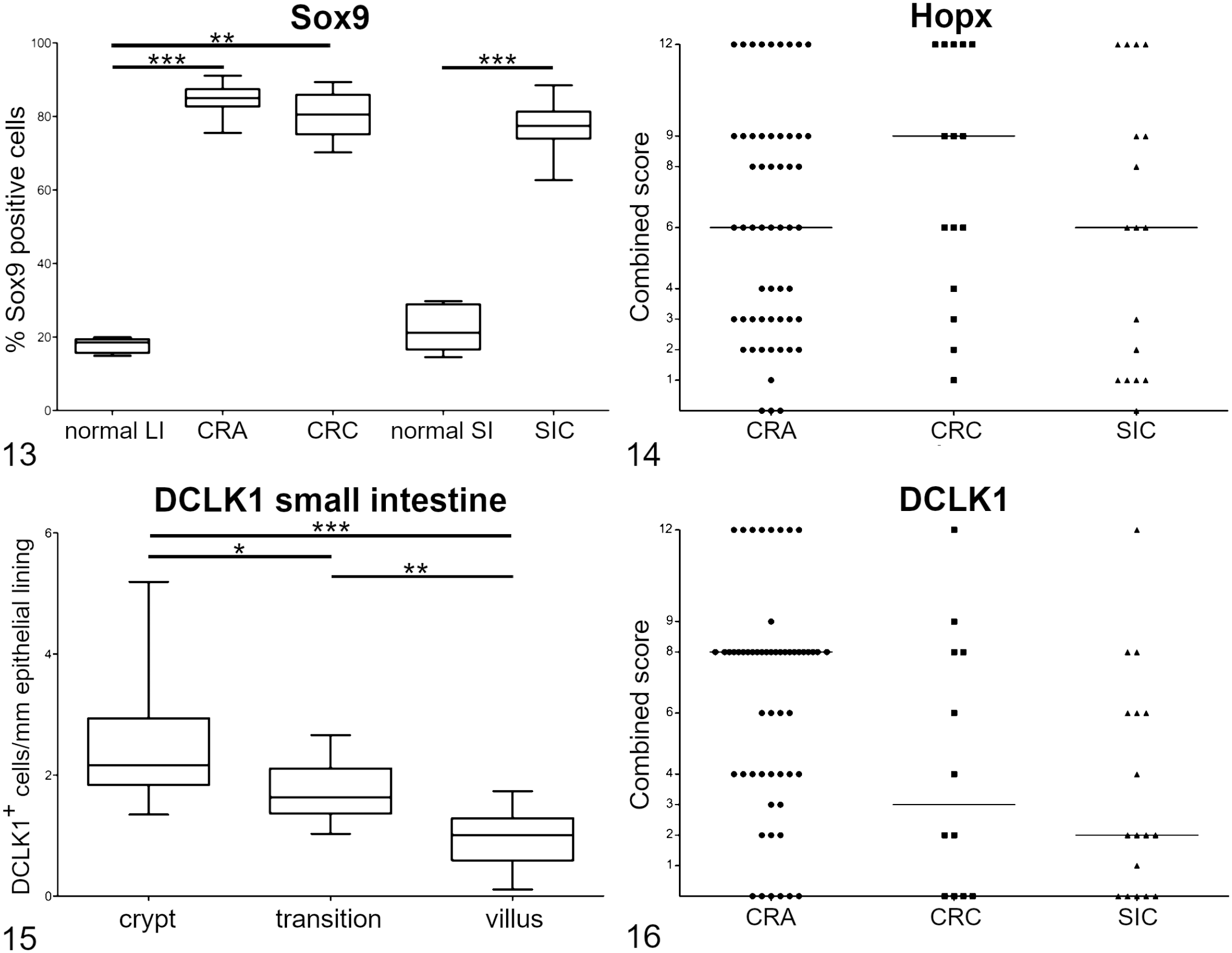
Survivin’s combined scores and mitotic count did not show significant differences between the examined tumors (Figs. 17, 18).
Ki-67 expression was significantly different when comparing normal small intestinal mucosa with SIC (P = .0061), and normal large intestinal mucosa with CRA (P = .0003) or CRC (P = .0019). No significant differences were found between CRA and CRC (P = .41), or between CRC and SIC (P = .32; Fig. 19).
Labeling for caspase-3 was significantly different when comparing normal mucosa with either CRC (P = .0072) or SIC (P = .015), and between CRA and CRC (P = .0062). Differences between normal mucosa and CRA (P = .103) and between CRC and SIC (P = .088) did not reach significant levels (Fig. 20).
Correlation of Stem Cell and Epithelial Cell Markers as well as Proliferation Indices
There was a weak negative correlation of Sox9 and Ki-67 labeling in CRA, ρ(CRA) = −0.145, a weak positive correlation in CRC, ρ(CRC) = 0.119, and no correlation in SIC, ρ(SIC) = 0.027.
In CRC and SIC, a weak-to-moderate positive correlation of Ki-67 and caspase-3 expression was detected, ρ(CRC) = 0.168; ρ(SIC) = 0.453. In CRA, hardly any correlation was seen, ρ(CRA) = 0.088. For Sox9 and caspase-3, a weak negative correlation was found in CRC and SIC, ρ(CRC) = −0.154; ρ(SIC) = −0.299, but a weak positive one in CRA, ρ(CRA) = 0.100. No association of Sox9 with invasiveness or the combined scores of Hopx, DCLK1, nor survivin were present.
Discussion
The expression of intestinal stem cell markers Sox9, Hopx, and survivin as well as tuft cell marker DCLK1 is of high interest in human colorectal cancer regarding prognosis and as possible therapeutic targets.1,10,14,15,32,34,43,49 –51,53,56,58,76,79,82 Shared molecular characteristics in human and canine sporadic CRCs, such as frequent mutations of adenomatous polyposis coli gene, suggest sporadic canine CRCs as an appropriate model for human disease. 88
In this study, the presence and distribution of intestinal stem cells and tuft cells within the canine small and large intestine was demonstrated immunohistologically using antibodies for potential stem cell markers Sox9, Hopx, and survivin and tuft cell marker DCLK1. In addition, we evaluated their expression in canine benign and malignant intestinal neoplasms. The detection of the expected canine proteins was confirmed by western blot analysis with no evidence of detection of non-target and nonspecific antigens. Furthermore, reaction pattern in normal canine intestine and intestinal tumors resembled those reported in normal human, murine, porcine, and equine intestine as well as in human and murine intestinal tumors.
As known for humans and mice and for pigs and horses, Sox9 expression in normal canine intestine was mainly present at the crypt base of small and large intestine as well as in single cells along the small intestinal crypt-villus axis.3,4,26,29,30,51,70 Sox9 reaction of canine crypt cells resembles that with Sox2, another stem cell marker, in a recently published study. 61 The positivity of canine crypt cells for Sox261 and Sox9 suggests that these cells are intestinal stem cells. Those single Sox9+ cells in the upper crypts of the canine intestine were identified as tuft cells by their coexpression of DCLK1. 28
Hopx was expressed by single cells in normal canine intestinal mucosa just above the crypt base as it is reported in mice and pigs, probably marking the quiescent stem cell population.30,75 Hopx+ quiescent or reserve stem cells regenerate the intestinal crypts in case of intestinal injury as they reactivate after the insult to regenerate intestinal epithelium.30,62,75
Survivin represents a protein of the inhibitor of apoptosis gene family. In normal human and murine intestinal mucosa, survivin mRNA and protein are detectable within the crypts and sporadically in the intestinal villi.44,73 In this study, survivin expression was found in the lower half of crypts in canine small and large intestine. The distribution of survivin+ cells in normal canine intestine, as shown in our study, indicates its role as potential stem cell marker as is reported in human and murine intestine as well as in canine skin and prostate.5,6,44,73
DCLK1, formerly interpreted as a marker for intestinal stem cells, has been identified to mark intestinal tuft cells. 25 This study showed that canine tuft cells share morphological characteristics with human and murine tuft cells. Furthermore, our investigation revealed that canine tuft cells express DCLK1 as well as Sox9, β-tubulin, and cytokeratin 18 as is reported for human and murine tuft cells.26,38,39 In canine large intestine, tuft cells are rarely detectable, which is in consensus with studies in mice and men that reported a proximo-distal gradient of tuft cells in the murine and human intestine.17,40,45 In mice and humans, they are present without predilection between crypts and villi.40,45 Immunohistological examination of DCLK1 expression in canine small intestine revealed a decrease of tuft cell frequency along the crypt-villus axis. In contrast to our findings, a uniform expression of DCLK1 mRNA has been reported in canine intestinal crypts and villi, and in vitro in jejunal enteroids. 11 Whether these differences relied on the different detection methods or environmental influences remains unclear. It has been shown that the composition of microbiota can influence the number of tuft cells in murine intestine. 40
In human CRCs, an overexpression of stem cell markers Sox9 and survivin as well as tuft cell marker DCLK1 was revealed, whereas Hopx has been shown to be downregulated in poorly differentiated CRCs.24,53,56,66,68,86
Sox9 expression was significantly increased in canine CRAs, CRCs, and SICs compared with normal intestinal mucosa. These findings are in accordance with human and murine studies showing a higher mRNA and protein level of Sox9 in intestinal cancer.10,51,53,56,70 Sox9 overexpression has been shown to be involved in tumorigenesis of human colorectal cancer in vitro and in vivo. 56 Increased immunoreactivity of Sox9 seen in the adenoma and carcinoma cases of this study suggests a similar role in canine intestinal neoplasia as well. These findings are in accordance with a recent publication showing a strong immunoreactivity of canine colorectal adenocarcinomas with Sox2, which is discussed to have a tumor initiation potential. 61 The increased Sox9 expression in CRAs indicates that dysregulation of Sox9 might be an early event in tumorigenesis, for example, in tumor initiation, as has been discussed for human adenomas. 51 In human duodenal adenomas and adenocarcinomas, Sox9 expression is associated with proliferating cells. 70 In our study, increased immunoreactivity of Ki-67 was also detected, although Ki-67 index was much lower than Sox9 levels indicating that Sox9 dysregulation occurs not only in proliferating cells, but in the majority of tumor cells.
Prognostic relevance of Sox9 expression is discussed controversially in human oncology. On one hand, overexpression of Sox9 by intestinal tumors is correlated with an unfavorable disease outcome,51,56 whereas other investigators neglect the significance of Sox9 as a prognostic marker in human colorectal and duodenal neoplasia.10,70 Because this was a retrospective study, data about clinical staging and follow-up results for the canine cases were not available.
In contrast to descriptions about human colon cancer, 51 no decrease of Sox9-immunolabelled tumor cells at the invasive front of CRCs and SICs was detected in our canine cases. Low expression of Sox9 at the invasive front in human colon cancer is significantly associated with high risk of relapse. 53 Furthermore, decrease of Sox9 expression at the invasive front was discussed to be associated with development of epithelial-mesenchymal transition. 53 The reason for the contradictory results in our cases remains undetermined. One reason might be the lack of epithelial-mesenchymal transition in our cases due to less advanced tumor stages, but this should be elucidated in further studies.
In human CRCs, downregulation of Hopx mRNA and protein is reported to be present in poorly differentiated CRCs compared with well-differentiated ones indicating Hopx to be a prognostically negative marker.34,43 Hopx downregulation is associated with hypermethylation of the CpG promoter region of its gene. However, demethylation agents restore the function of Hopx thereby leading to decreased proliferation, angiogenesis, and invasion.34,43 In all examined intestinal neoplasms of this study, Hopx’s combined scores varied within the groups. Overall, CRCs showed a higher median of combined scores than CRAs and SICs. In adenocarcinomas, no decrease in Hopx labeling intensity was detected at the invasive front; instead, deep invasive nests often showed an intense immunoreactivity. As described for humans,34,43 epigenetic silencing might also be a possible explanation for the decrease of Hopx expression in malignant tumors compared with the benign lesions. However, in contrast to humans there is no evidence that Hopx has any prognostic relevance in canine colorectal tumors. Whether the increased Hopx protein expression in canine CRCs has an influence on proliferation and invasion of tumor cells remains undetermined. Low combined scores were detectable within all tumor subtypes, including well-differentiated and poorly differentiated ones.
Survivin is known to be upregulated in human colorectal neoplasia.15,37,41,46 Dysregulation is detected in adenomas and adenocarcinomas; therefore, it has been considered as an early event in colorectal carcinogenesis. 37 In some studies, increased protein and mRNA expression of survivin was correlated with poor differentiation, advanced stage, and/or poor prognosis in human CRC,37,77,81 whereas others neglect those correlations and/or highlight the importance of subcellular distribution of survivin as prognostic marker.15,46,66,67
Within our canine cases, increased expression of survivin was also detected in all neoplastic lesions compared with normal intestine. Interestingly, survivin score was nearly similar in CRAs and CRCs and higher in colorectal lesions than in SICs, but a high variability of the combined scores was present within these tumors.
Over the past years, DCLK1+ cells have reached special interest in intestinal oncology research. Initial assumptions interpreted these cells as quiescent stem cells that might play a role as CSCs.12 –14,21 –23,58,76,83 Although more recent studies identified the DCLK1+ cell population within the intestinal epithelium as tuft cells, 25 DCLK1 is still of interest in gastrointestinal oncology. Nakanishi et al 64 have shown DCLK1 to discriminate between normal IESCs and CSCs. Very recent studies demonstrated that DCLK1 plays an important role in epithelial-mesenchymal transition and is associated with drug resistance as well as poor prognostication of colorectal cancer.24,32,49,52,68,84 In this investigation, DCLK1 was diffusely expressed in canine intestinal adenomas and infrequently detectable in intestinal adenocarcinomas. In general, papillary CRCs showed a higher expression of DCLK1 in the luminal aspects of the tumor than in the invasive areas. The underlying mechanism and the possible role of these findings regarding the prognosis and biological behavior in canine intestinal tumors remain undetermined and should be evaluated in future studies. The lack of increased DCLK1 at the invasive front might indicate a lower number of potential CSCs that might result in a less aggressive behavior of canine intestinal adenocarcinoma.
The results of this study showed that in benign and malignant canine intestinal neoplasms, increased immunoreactivity for stem cells markers Sox9, Hopx, and survivin as well as DCLK1 compared with normal intestinal mucosa is detectable. These findings indicate that IESCs might be involved in the initiation, promotion, and progression of canine intestinal neoplasms. However, as there are only small differences between benign and malignant tumors, no conclusion can be drawn regarding the importance of stem cells for tumor recurrence, metastasis, and therapy resistance, as is discussed in humans.14,24,34,43,51,56,66,67
In this study, caspase-3 showed an increased immunoreactivity in all tumor types, but no significant association was detected with the investigated intestinal stem cell markers. An increase of caspase-3 expression is frequently detected in canine mammary tumors and osteosarcomas but no association with clinico-pathological parameters has been shown.8,19 In human CRCs, however, high caspase-3 activity is associated with a higher risk of disease relapse.42,90
In conclusion, this immunohistochemical study showed that the potential stem cell markers Sox9, Hopx, and survivin are expressed in the normal canine intestine, as it is reported in other species. Furthermore, the presence of tuft cells was confirmed in normal canine small intestine by demonstrating their coexpression of DCLK1 and Sox9. We found an increased immunoreactivity of the intestinal stem cell markers in canine benign and malignant intestinal neoplasms, as it is reported in humans, without significant differences observed between tumor types or locations. The prognostic relevance of the higher protein expression in canine intestinal neoplasms should be investigated in further studies including clinical and follow-up data of patients, such as tumor staging, relapse-free period, and/or overall survival time.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858221079666 – Supplemental material for Sox9, Hopx, and survivin and tuft cell marker DCLK1 expression in normal canine intestine and in intestinal adenoma and adenocarcinoma
Supplemental material, sj-pdf-1-vet-10.1177_03009858221079666 for Sox9, Hopx, and survivin and tuft cell marker DCLK1 expression in normal canine intestine and in intestinal adenoma and adenocarcinoma by Wencke Reineking, Ida E. Schauerte, Johannes Junginger and Marion Hewicker-Trautwein in Veterinary Pathology
Footnotes
Acknowledgements
The authors are grateful to Georg Beythien, Kerstin Rohn, Jana-Svea Harre, Julia Baskas, Caroline Schütz, Petra Grünig, and Christiane Namneck for their excellent technical assistance. The statistical support of Dr. Fritjof Freise, Institute for Biometry, Epidemiology and Information Processing, University of Veterinary Medicine Hannover, Germany, is gratefully acknowledged.
Author Contributions
W.R. and M.H.T. planned the study, analyzed, and interpreted the data. W.R. and I.S. performed the experiments and prepared the article. M.H.T. und J.J. supervised the experiments and revised the article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
