Abstract
Controversies remain regarding the cell type from which human prostate cancer originates, and many attempts have been made to identify the cellular origin of canine prostate cancer but without definitive proof. This study aims to evaluate the expression of luminal (androgen receptor [AR], cytokeratin [CK]8/18) and basal (CK14, CK5) cell markers in different histologic subtypes of canine prostatic carcinoma (PC) and to suggest the most likely tumor-initiating cells. Normal prostates (n = 8) were characterized by AR+CK8/18+ luminal cells and few CK5+ basal cells, while CK14 was absent. Similar pattern was observed in all 35 prostates with benign prostatic hyperplasia, except few scattered CK14+ basal cells in 13 samples (37.14%). AR was localized in the nucleus of both normal and hyperplastic cells. In 34 samples of PC, the following growth patterns were identified: cribriform (44.12%), solid (32.35%), small acinar/ductal (20.59%), and micropapillary (2.94%). Most PCs expressed AR and CK8/18, while CK5 and CK14 expression was observed in 25% and 20% of cases, respectively. AR revealed a variable intracellular distribution, both nuclear and cytoplasmic. Solid PC was characterized by an undifferentiated or aberrant phenotype with a reduced expression of AR and CK8/18, increased number of CK14+ cells, and 7 antigen expression patterns. This study demonstrated a predominance of differentiated luminal cell types in canine prostatic tumors, although the role of basal cells in prostate carcinogenesis should also be considered. Moreover, few scattered CK5+ cells in AR+CK8/18+ tumors identified the existence of intermediate cells, from which neoplastic transformation may alternatively commence.
Keywords
The dog is the only species other than humans that regularly develops prostate cancer (PC) spontaneously, although with a lower incidence (0.2%–0.6%). 2 PC is the most commonly diagnosed tumor in men and is an increasing cause of morbidity and mortality in the Western world. Human PC is initially androgen sensitive and relapses after androgen deprivation therapy as a hormone-refractory, highly undifferentiated, and heterogeneous tumor. 32 In dogs, PC is not responsive to androgen deprivation therapy and has an undifferentiated morphology and aggressive behavior, resembling the refractory phase of human PC. 21 Therefore, the dog is generally regarded as a suitable animal model for the study of the initiation and promotion of the molecular mechanisms of human PC.
For human PC, previous studies have reached differing conclusions regarding the cell types of origin, with the basal, intermediate, or other cells within the luminal compartment possessing stem cell characteristics and producing tumors with different biological properties. 5 In addition, the ability of inflammation to enhance basal-to-luminal differentiation in vivo 20 suggests that alteration of the tissue microenvironment could influence the cell of origin. 13 Many attempts have been made to identify the cellular origin of canine PC but remain inconclusive thus far.
Within the prostatic epithelium, basal and luminal cells can be distinguished on the basis of their localization, morphology, and phenotypic features. Luminal secretory cells express high levels of androgen receptor (AR) and cytokeratins (CKs) 8 and 18. Basal cells specifically display high expression of CK5 and CK14, but little or no AR, CK8, and CK18. 6,36,40 A third group of cells is characterized by an intermediate phenotype between basal and luminal cells. Intermediate basal cells within the basal cell compartment have high expression of CK5 without CK14 (CK5++/CK8+), while intermediate cells in the luminal layer are characterized by colocalization of CK18 and the basal cell marker (CK5+/CK18++). 33,35,37,44 Another protein strongly expressed in prostatic basal cells is p63, belonging to the p53 family of genes. 16
To understand the pathogenesis of canine PC, it would be necessary to identify the cell of origin in this species. Therefore, in this study, we aimed to characterize different histologic subtypes of canine prostatic carcinomas through the expression patterns of different markers (CK8/8, CK5, CK14, AR) and to hypothesize the cellular origin of this type of tumor. Prostatic basal cells were identified using CK5, proven to show similar sensitivity and specificity to p63. 1 Moreover, CK5 performed optimally in formalin-fixed paraffin-embedded tissues and slide storage, while p63 demonstrated some shortcomings and staining variations 17 and loss of immunostaining intensity in stored slides. 4,17
Materials and Methods
Samples and Histopathology
Seventy-seven formalin-fixed, paraffin-embedded canine prostatic samples were retrieved from the archives of the School of Veterinary Science–Diagnostic Pathology Service of the University of Queensland and the Department of Veterinary Science and Public Health, the University of Milan. This project was approved by the University of Queensland Animal Ethics Committee (approval no. ANFRA/SVS/406/13).
Five-micron-thick sections were stained with hematoxylin and eosin for the histopathologic examination. The samples were classified as normal prostates, benign prostatic hyperplasia (BPH), and prostatic carcinoma.
Prostatic carcinoma diagnosis was limited to those cases showing no evidence of urinary bladder involvement. The classification of prostatic carcinoma was adapted from Lai et al 21 and from the World Health Organization’s human classification of tumors of the urinary system and male genital organs. 43
Immunohistochemistry
Immunohistochemistry was performed using an avidin-biotin-peroxidase staining procedure (Vectastain Standard Elite; Vector Labs, Burlingame, CA) and the following antibodies: rabbit polyclonal anti-human AR (Santa Cruz Biotechnology; 1:1000), mouse monoclonal anti-human CK8/18 (Novocastra, 1:600), mouse monoclonal anti-human CK5 (Novocastra, 1:300), mouse monoclonal anti-human CK14 (Thermo Scientific, Lab Vision, 1:1500). All incubations were performed at room temperature unless otherwise stated.
Following deparaffinization and rehydration of the sections, endogenous peroxidase was blocked by hydrogen peroxide 3% in distilled water for 45 minutes. For AR, CK5, and CK14, antigen retrieval was achieved by submerging the sections in 0.1M sodium citrate buffer pH 6.0 (Vector Labs) and heating in a microwave oven for 15 minutes. For CK8/18, the sections were incubated with proteinase K (Novocastra) for 5 minutes. All sections were then preincubated with 5% bovine serum albumin (Vector Labs) for 30 minutes and avidin/biotin blocking solution (Vector Labs). Sections were finally incubated with the primary antibodies at 4°C overnight, using the antibody concentration as indicated above in phosphate-buffered saline. After the slides were washed 3 times for 5 minutes in TBS, sections were incubated with the appropriate biotinylated secondary antibody diluted 1:200 for 30 minutes: goat anti-mouse IgG for CK8/8, CK5, and CK14 and goat anti-rabbit IgG for AR (Vector Labs). Following incubation with peroxidase-coupled AB complex, peroxidase activity was visualized by 3,3′diaminobenzidine (Abcam) for 5 minutes. Slides were then counterstained with Mayer’s hematoxylin, dehydrated, and mounted. Negative controls comprised slides incubated with an isotype-matched nonspecific antibody. The following positive controls were used: normal canine skin for CK5, CK14, and CK8/18 and normal prostate for AR. Since canine prostate carcinomas may arise from glandular or ductular epithelium of the prostate or from urothelium of the prostatic urethra, tumor morphology and nuclear AR expression have been used to differentiate prostatic and urothelial carcinomas. Downes et al 8 demonstrated that in prostate carcinomas, AR is strongly expressed in the nuclei of neoplastic cells, while urothelial tumors are negative and have cytoplasmic staining or occasional weak nuclear staining.
Assessment of the Immunohistochemical Labeling
A semiquantitative assessment was performed analyzing 10 high-power fields at 400× magnification. For each sample, the labeling intensity and the percentage of positive neoplastic cells were analyzed. The labeling intensity was recorded as negative, weak, moderate, or strong. Samples were divided according to the percentage of neoplastic cells in 5 groups: group 0, no positive cells; group 1, >0 to <10% positive cells; group 2, ≥10% to <25%; group 3, ≥25% to <50%; group 4, ≥50% to <75%; group 5, ≥75%. After the 10 selected fields were averaged, a final mean ± SD of the percentage of positive neoplastic cells was obtained for each sample.
Statistical Analysis
Differences among normal prostates, BPH, PC, and each subtype of PC were assessed by the chi-square test and considered to be significant at P < .05. Only those carcinomas confirmed to be prostatic through the nuclear expression of AR were included in the statistical analysis (case Nos. 44, 46–49, 51–55, 56, 59, 60, 62–77).
Results
Histopathology
The histopathologic examination revealed 8 normal prostates (case Nos. 1–8), 35 BPHs (case Nos. 9–43), and 34 PCs (case Nos. 44–77).
Four growth patterns of PC were differentiated:
Cribriform: ducts completely extended by neoplastic cells with the formation of regular fenestrae, often associated with central necrosis (comedonecrosis; n = 15 of 34; case Nos. 44–58).
Solid: pleomorphic neoplastic cells arranged as solid nests or occasional individual cells (n = 11 of 34; case Nos. 59–69)
Small acinar/ductal: with variably sized microacini arranged within a dense fibrous stroma (n = 7 of 34; case Nos. 70–76)
Micropapillary: formation of papillary projections of neoplastic cells within extended ducts (n = 1 of 34; case No. 77)
Immunohistochemistry
Normal Prostates
A strong cytoplasmic immunostaining for CK8/18 was extensively observed in the luminal cells of all samples (case Nos. 1–8), while the expression of CK5 was limited to few scattered basal cells. Expression of CK14 was absent. In all samples, strong nuclear expression of AR was consistently detectable in the nucleus of most luminal cells.
Benign Prostatic Hyperplasia
A strong cytoplasmic immunostaining for CK8/18 was extensively observed in the luminal cells of all the 35 prostates with BPH (case Nos. 9–43). Few scattered basal cells positive for CK5 were also recognizable. The expression of CK14 was observed in 13 samples (case Nos. 14, 16, 18, 19, 21–23, 25, 30, 35, 37, 41, 43) with a percentage of positive cells ranging from 0.2% to 0.6%. Strong nuclear AR immunostaining was detected in most luminal cells in all samples.
Prostatic Carcinomas
Twenty-five (case Nos. 44–52, 56–58, 63, 64, 66, 67, 69–77), 33 (case Nos. 44–59, 61–77), and 20 (case Nos. 44–54, 60, 61, 63–65, 69, 71, 72, 74) cases of PC expressed CK5, CK8/18, and CK14, respectively.
All 34 cases of PC expressed AR. In 10 neoplasms, positive signal was nuclear (case Nos. 49, 55, 56, 60, 70) or cytoplasmic (case Nos. 45, 50, 57, 58, 61), while in 24 cases (Nos. 44, 46–48, 51–54, 59, 62–69, 71–77), immunolabeling was detectable in both compartments. In these cases, a prevalent nuclear or cytoplasmic distribution was observed in 9 (case Nos. 44, 47, 48, 52, 63, 68, 71–73) and 3 (case Nos. 53, 54, 74) cases, respectively.
To better characterize PC, the expression of the different markers per growth pattern was also analyzed. The frequency of expression of the different antigens in each growth pattern is summarized in Supplemental Table 1.
All 15 cribriform patterns expressed AR and CK8/18. The percentage of positive neoplastic cells ranged from 11.43 (case No. 56) to 85 (case No. 53) for AR and from 61.67 (case No. 55) to 96.25 (case No. 47) for CK8/18. The distribution of AR was nuclear in 3 of 15 cases (Nos. 49, 55, 56), cytoplasmic in 4 (case Nos. 45, 50, 57, 58; Fig. 1), and nucleocytoplasmic in 8 (case Nos. 44, 46–48, 51–54). AR- and CK8/18-positive neoplastic emboli were observed in 1 (case No. 45) and 2 samples (case Nos. 44, 49), respectively. CK5 expression was detected in 12 of 15 cases (Nos. 44–52, 56–58): localized in the basal cell layer in 7 cases (Nos. 44, 46, 50–52, 57, 58), in randomly scattered neoplastic cells in 4 cases (Nos. 45, 47, 48, 56), or in both in 1 case (No. 49). The CK5-positive basal cell layer was present in 2 cases (Nos. 44, 52) characterized by small cribriform lesions without comedonecrosis and absent in large lesions with central necrosis (Fig. 2). CK5-positive neoplastic emboli were observed in 1 case out of 12 (case Nos. 44; Fig. 3). The percentage of neoplastic CK5-positive cells ranged from 1.33 (case No. 48) to 35.71 (case No. 47). Eleven of 15 cases (Nos. 44–54) expressed CK14 with a variable cytoplasmic staining in basal cells in 3 of 11 cases (Nos. 45, 46, 51; Fig. 4), in randomly scattered cells in 6 cases (Nos. 44, 47, 48, 50, 53, 54), and in both in 2 cases (Nos. 49, 52). The percentage of neoplastic positive cells ranged from 0.2 (case Nos. 48, 53) to 7.3 (case No. 44).
Cribriform carcinoma, prostate, dog.
All 11 tumors with solid patterns expressed AR with a more variable intensity of staining and percentage of positive cells compared to the cribriform pattern. The percentage of positive neoplastic cells ranged from 0.5 (case No. 60) to 78.5 (case No. 62). In 1 case (No. 60), AR positive staining was nuclear, while in another single case (No. 61), it was cytoplasmic. In the remaining 9 cases (Nos. 59, 62–69) both nuclear and cytoplasmic expression was detectable. AR-positive emboli were observed in 1 case (case No. 69). CK8/18 was observed in 10 of 11 tumors (case Nos. 59, 61–69) with variable percentages of neoplastic cells, ranging from 0.6 (case No. 59; Fig. 5) to 96.25 (case No. 62). CK5 was expressed in randomly scattered cells in 5 of 11 cases (Nos. 63, 64, 66, 67, 69; Fig. 6). One case of 5 (case No. 64) contained intravascular neoplastic cells that were positive for CK5. CK14 was expressed in 6 of 11 cases (Nos. 60, 61, 63–65, 69; Fig. 7), with positive intravascular neoplastic cells observed in 2 of 6 cases (Nos. 64, 69; Fig. 8). The percentage of neoplastic positive cells ranged from 6.67 (case No. 66) to 12 (case No. 64) for CK5 and from 0.3 (case No. 63) to 55 (case No. 61) for CK14.
All 7 tumors with small acinar/ductal patterns expressed AR in a high percentage of neoplastic cells and with a prevalent nuclear (1 of 7 cases; case No. 70) or nucleocytoplasmic (6 of 7 cases; case Nos. 71–76) distribution. The percentage of positive neoplastic cells ranged from 58 (case No. 70) to 82.4 (case No. 71).
CK8/18 expression was observed in all 7 samples, with a percentage of positive neoplastic cells ranging from 28 (case No. 74) to 96.25 (case No. 71). A low number of randomly scattered neoplastic cells expressed CK5 in all cases. CK14 expression was revealed in 3 of 7 cases (Nos. 71, 72, 74) with mild cytoplasmic staining. The percentage of neoplastic positive cells ranged from 0.17 (case No. 71) to 17 (case No. 70) for CK5 and from 0.3 (case No. 72) to 12 (case No. 74) for CK14.
The tumor with papillary growth pattern was AR, CK8/18, and CK5 positive with a nucleocytoplasmic staining for AR and a randomly scattered distribution of CK5. CK14 expression was not detected. This growth pattern was present in only 1 tissue section, and no significant differences in the expression of the different markers could be detected.
The final mean ± SD of the percentage of positive cells for each antigen in normal, hyperplastic, and carcinomatous prostates is summarized in Table 1, with the number of cases for each group of percentage of positive cells in Supplemental Table 2.
Percentage of Positive Cells for Each Antigen in Normal, Hyperplastic, and Carcinomatous Prostates, Mean ± SD.
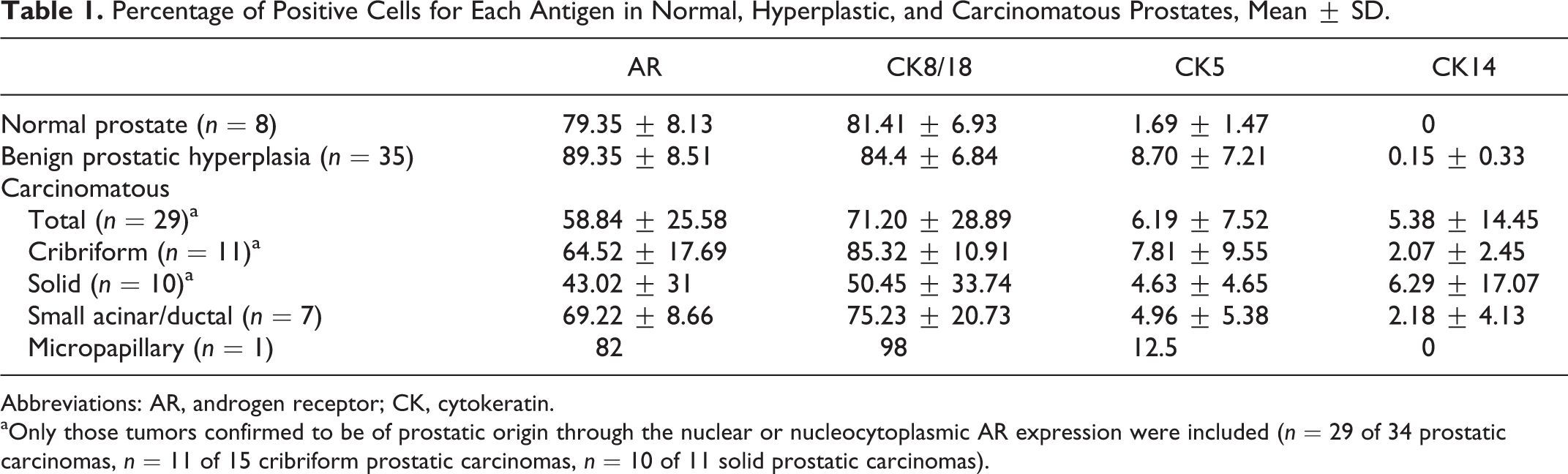
Abbreviations: AR, androgen receptor; CK, cytokeratin.
aOnly those tumors confirmed to be of prostatic origin through the nuclear or nucleocytoplasmic AR expression were included (n = 29 of 34 prostatic carcinomas, n = 11 of 15 cribriform prostatic carcinomas, n = 10 of 11 solid prostatic carcinomas).
The frequency of antigen expression pattern in each tumor and each growth pattern is summarized in Table 2 and Supplemental Table 3.
Frequency of Antigen Expression Patterna in Prostatic Carcinomas as a Whole and for Each Growth Pattern.

Abbreviations: AR, androgen receptor; CK, cytokeratin.
aIn descending order of percentage of positive cells.
bPositive cells for both markers (group 1), <10%.
Statistical Analysis
The final percentage of AR- and CK14-positive cells was significantly different between prostatic carcinomas and both normal prostates and hyperplastic prostates (P < .05). The difference in CK5 expression was statistically significant between prostatic carcinomas and normal prostates (P < .05).
Regarding the different PC subtypes, the following statistically significant differences were observed: for AR and CK14 expression, between solid PC and both cribriform and acinar PC; for CK8/18 expression, between cribriform and solid PC.
Discussion
All canine prostatic tumors, including a variety of growth patterns, express AR and most of them express CK8/18. This reflects a predominance of differentiated cell types in the canine prostate tumors, corresponding with observations from human prostatic carcinoma. 26 However, CK5 and CK14, the indicators for basal cells, are also expressed in our canine tumors (25% and 20% of tumors, respectively). Furthermore, the expression of CK5 in scattered neoplastic cells of AR+CK8/18+ tumors identified the existence of cells with an apparent intermediate cell phenotype in canine PC.
In normal prostates, no CK14 expression was observed in the basal cells. This corresponds to the results of LeRoy et al 24 and Grieco et al, 15 but it is in contrast to what is described in the human prostate, where CK14-expressing cells are more prominent. 40 Compared to CK14, CK5 is more abundantly expressed in the canine prostate, mainly in scattered cells at the periphery of the acini. This finding confirms that CK5-positive acinar basal cells, although low in number, constitute the major proliferative component in the canine prostate. 23 In general, the normal canine prostate has a more differentiated phenotype (lack of CK14-positive cells, few CK5-positive basal cells) compared to the human prostate, with less immature epithelial cells.
Canine prostates undergoing hyperplastic changes reveal an increased number of CK5-positive cells and the occurrence of CK14 immunoreactivity, while maintaining a differentiated phenotype. This finding may indicate an active regeneration of the epithelium, with CK5+/CK14+ cells replicating and expanding the epithelial compartment of the gland.
One of the most interesting findings in canine prostatic carcinomas is the expression of CK5 in a randomly scattered or basal and patchy pattern. Although the loss or discontinuity of the basal cell layer has long been the hallmark of adenocarcinoma in humans, few cases of unequivocal Gleason pattern 3 adenocarcinoma have been shown to retain basal cells, both as patchy or continuous basal cell staining. 27 Moreover, from our experience and as shown in this study, the canine basal cell layer is not continuous; therefore, its examination does not provide an indication of the prostatic lesion, as it does in humans. Furthermore, a strong and diffuse expression of another basal cell marker, p63, in human adenocarcinoma is a recently recognized phenomenon. 28 Fonseca-Alves et al 9 observed a moderate number of p63-positive cells in canine prostatic carcinoma, and Grieco et al 15 reported 1 case of CK5-positive canine undifferentiated carcinoma. Occasional PC cells can express basal cell markers in a non–basal cell distribution, including some high-grade PCs and distant metastases. 10,14,45 Since prostate basal cells are less well differentiated and proliferate more frequently, 3 they are more likely than luminal cells to accumulate genetic alterations. In a few cribriform lesions observed in this study with positive basal CK5 staining in small lesions without comedonecrosis, the CK5 basal staining pattern can suggest basal cell retention in early carcinoma. However, this does not explain the increased frequency of CK5 expression in other growth patterns. Since the distribution of CK5-positive cells seems to be a variable yet consistent finding across different histologic subtypes of canine carcinomas, their roles as tumor-initiating cells should be considered.
In comparison to normal and hyperplastic prostates, neoplastic cells revealed a variable distribution of the AR (nuclear, cytoplasmic, or nucleocytoplasmic). With reference to the cytoplasmic distribution observed, mutations in the AR might render the AR unable to enter the nucleus. The cytoplasmic AR expression has also been found in human PCs and is considered to be an independent predictor of recurrence after androgen depletion. 7 In the latter study and in others, 30,31 cytoplasmic AR expression was seen more often in androgen-independent than hormone-sensitive PCs. In addition, a significantly reduced expression of AR has been observed in canine tumors compared to normal prostates and BPH, suggesting possible mutations with loss of expression or de-differentiation of neoplastic cells.
In this study, we demonstrated specific immunohistochemical features of the solid subtype of canine prostatic carcinoma: heterogeneous and general reduction of AR and CK8/18 expression but increased number of CK14+ cells. All these features are suggestive of a less differentiated growth pattern containing more neoplastic cells with an undifferentiated or intermediate phenotype. Moreover, 7 antigen expression patterns have been described in canine solid PC, and this confirms their aberrant differentiation.
Taken together, our results allow us to hypothesize different models of the cell of origin of canine PC. Many conflicting theories have been formulated in the human counterpart so far. Luminal cells are generally accepted as the cells of origin for human PC 26,29 and human pathologists diagnose the disease based on the absence of basal cell markers. 42 Evidence from the mouse implicates both luminal cells 18,39,41 and basal cells 22,25,38 in PC initiation. The basal cell hypothesis suggests that cells in the basal compartment possess self-renewal capability and can generate basal cells, intermediate or transient amplifying cells with an intermediate phenotype, and luminal cells. 12,34 The second type of tumor-initiating cells could be the rare multipotent luminal cells that could generate both basal and luminal cells in vivo. Although they share features with luminal cells, they are distinguished from most other luminal cells by their retention of Nkk3.1 protein after castration (so-called castration-resistant Nkk3.1-positive cells). 39 Alternatively, neoplastic transformation may commence in differentiated progenitor cells (intermediate cells) that co-opt self-renewal programs form tissue stem cells. 5 Finally, even mature differentiated postmitotic prostatic luminal cells could be reprogrammed by oncogenic pressure into tumor-initiating cells. 19 Considering the frequent expression of CK5 and CK14 in our canine samples, we believe that the oncogenic activation can occur in both compartments (basal and luminal) and the intermediate cells can play a role in tumor initiation when a CK5+CK8/18+ phenotype is prevalent. Further investigations with colocalization of CK5, CK14, and CK8/18 are required to produce a deeper understanding of the role of intermediate cells in canine prostatic carcinogenesis. Androgen deprivation and androgen-independent tumor progression in men are associated with an increase of intermediate cells, indicating that these cell types are androgen independent for their survival. 11 Identification of their cellular biological characteristics in canine PC appears crucial for further understanding of their role in prostate carcinogenesis. The identification of cell types of origin for canine PC is significant, since distinct cell populations within a tissue may give rise to different cancer subtypes distinguished by their histopathologic phenotypes and patient outcomes. Therefore, additional molecular studies may add provide more important evidence to this ongoing debate.
Footnotes
Authors’ Note
Syeda Hasina Akter and Fabian Zhi Xiang Lean contributed equally to the paper. S.H. Akter contributed to design, data analysis and interpretation; F.X.Z. Lean contributed to conception, data acquisition and proof writing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
