Abstract
Acquired outpouches of the intestinal tract are referred to as pseudodiverticula or false pulsion diverticula. In contrast to true diverticula, in which the wall contains all layers of the intestinal tract, the wall of pseudodiverticula lacks the tunica muscularis. Smooth muscle hypertrophy of the small intestine is commonly considered a cause of pseudodiverticulosis in animals due to increased intraluminal pressure. This study reports pseudodiverticula associated with idiopathic smooth muscle hypertrophy of the small intestine in lagomorphs. Four domestic rabbits had single or multiple (up to 200) pseudodiverticula of various size in the small intestine. In all cases the tunica muscularis was diffusely thickened, significantly exceeding reference thickness of 14 rabbits (mean, 112.3 µm; range, 26.3–389.0 µm). Clinical signs were considered to be caused by severe necrosis and inflammation of the wall of large pseudodiverticula, leading to perforation with subsequent peritonitis and mesenteric and omental abscess formation in 2 cases.
Keywords
Acquired false pulsion diverticula or pseudodiverticula of the intestine are outpouches of the tunica mucosa and submucosa communicating with the lumen of the intestinal tract. In congenital true diverticula, such as persistent Meckel diverticulum, the wall consists of all layers of the intestinal tract, whereas the wall of pseudodiverticula lacks the tunica muscularis. 11 Pseudodiverticula are mainly found at the mesenteric surface of the intestine 5,6,8,14 but can rarely be observed at the antimesenteric side. 7,13 Pseudodiverticulosis—the disease associated with pseudodiverticula formation—has been described in the small intestine of animals, most frequently in horses 5,6 and pigs 6 and rarely in sheep, 6 cats, 2 an elk, 14 and a wallaby. 8 Small intestinal pseudodiverticula have not been reported in lagomorphs to date. However, colonic pseudodiverticula have been induced experimentally in rabbits by a low-residue diet in combination with pharmacologic stimulation of intestinal motility. 9 In humans, pseudodiverticula of the small intestine are rare. 10,12 In contrast, pseudodiverticula of the large intestine (also referred to as diverticular disease) are quite common in humans but not in other species. 15
Hypertrophy of the tunica muscularis of the small intestine (MHSI) occurs concurrently with most pseudodiverticula of animals, including horses, 5,6 cats, 2 pigs, 6 and sheep. 6 MHSI has not been reported in rabbits. The term muscular hypertrophy (MH) is commonly used, although it has been proven that thickening of the tunica muscularis results from concurrent hypertrophy and, to a lesser degree, hyperplasia. 2,4 Two forms of MH can be recognized: (1) idiopathic or primary, if a predisposing cause cannot be identified; (2) secondary, if MH is associated with chronic partial stenosis of an aboral intestinal segment and presumably results from an increased force of intestinal contractions. 2,5
Here, we describe clinical and pathologic features of 4 domestic dwarf rabbits with small intestinal pseudodiverticula associated with MHSI. In addition, reference values for muscular thickness were established in 14 unaffected rabbits.
Rabbit No. 1 was a 4.5-year-old castrated male rabbit with a body weight of 1.7 kg. The animal had been inappetent and apathetic for 2 days. Palpation and abdominal radiographs (Suppl. Fig. S1) and sonography (Suppl. Fig. S2) revealed masses in the midabdomen that were connected to the intestinal tract. The rabbit’s condition deteriorated rapidly, and it died spontaneously. Postmortem examination revealed moderate amounts of intestinal content in the abdominal cavity and severe diffuse fibrous adhesions of the peritoneum (chronic peritonitis). Three abscesses were present in the omentum majus and mesentery. A microbiological examination from a mesenteric abscess (see supplemental material for microbiological methods) detected Escherichia coli, Enterococcus faecium, and bacteria of the Bacteroides fragilis group. E. faecium and the B. fragilis group can be considered part of the gut microbiota in rabbits. Although E. coli can be a commensal resident of the gastrointestinal tract, distinct pathovars have the capacity of causing significant diarrhea in rabbits. Gross examination also revealed >200 pouches throughout the small intestine originating from the mesenteric side of the intestinal wall (Figs. 1, 2). Pouches greatly varied in size, ranging from 0.1 to 3.8 cm, with the smaller pouches being covered by mesenteric fat. The lumina of all pouches were connected to the intestinal lumen and impacted with intestinal content (Fig. 3). The largest pseudodiverticulum in the ileum was surrounded by a 0.5-cm-thick fibrotic capsule and connected to the abdominal cavity via a perforation of the pouch wall (Suppl. Figs. S5, S6).
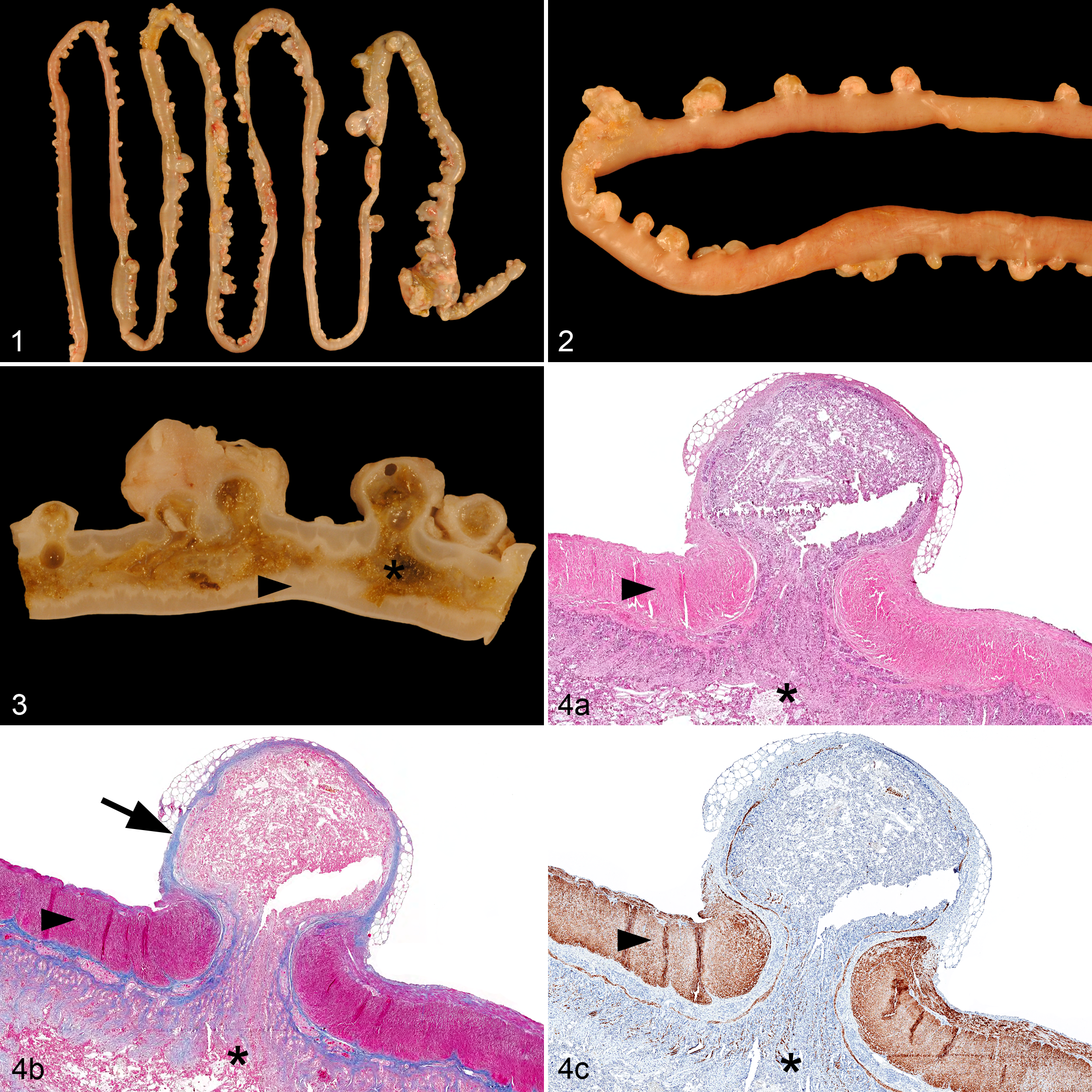
Rabbit No. 2 was a 2-year-old intact female rabbit with a body weight of 2.0 kg. The owners reported inappetence for 4 days, and the clinical examination identified multiple abdominal masses by palpation and sonography. A laparotomy revealed that the abdominal masses were located in the mesentery and often had a fibrotic connection to the small intestine. The peritoneum had a diffusely rough surface with multiple adhesions to abdominal organs, and scant plant material was detected in the abdominal cavity. The rabbit was euthanized intraoperatively and the entire intestinal tract examined. Grossly, the mesenteric masses in proximity to the small intestine had a thick fibrotic capsule and were impacted with intestinal content. In contrast, masses with a thick fibrotic capsule located in the mesentery rather distant to the small intestine were filled with pus and necrotic debris, consistent with mesenteric abscesses.
Rabbit No. 3 was a 5-year-old intact female rabbit with a body weight of 1.6 kg that showed inappetence, production of few smaller fecal pellets, and teeth grinding. Abdominal radiographs (Suppl. Fig. S3) and sonography revealed a mass located in the mid- to caudal abdomen. During exploratory laparotomy, a connection of the mass with the mesenteric surface of the ileum was identified, and fibrinous exudate suggested an inflammatory process. Due to poor prognosis, the rabbit was humanely euthanized intraoperatively by intravenous pentobarbital injection. Postmortem examination revealed a 5.0 × 4.7 × 3.4–cm large globular mass impacted by intestinal content and connected to the lumen of the ileum. Additionally, there were 17 other small intestinal pseudodiverticula with a maximum diameter of 0.3 cm, completely embedded in mesenteric fat.
Rabbit No. 4 was a 3-year-old male rabbit that had a body weight of 1.84 kg. The rabbit had been inappetent, and an abdominal mass was discovered clinically. A laparotomy revealed a single large mass in the mesentery with a fibrotic connection to the jejunal wall. Due to poor prognosis, the rabbit was humanely euthanized intraoperatively. Gross examination revealed that the mass was impacted by intestinal content.
In rabbit Nos. 1 and 3, a connection of the intestinal lumen with the lumen of the mesenteric masses (ie, pseudodiverticula) was grossly evident. However, in rabbit Nos. 2 and 4, outpouching of the intestinal lumen was discovered by histopathology in only 5 distinct locations or a single location, respectively (Suppl. Figs. S11–S14). Histopathology revealed that the small intestinal outpouches were consistently located in the mesentery where blood vessels and lymphatics penetrate the intestinal wall (Suppl. Figs. S15–S18). The mucosal and submucosal layers, including the lamina muscularis mucosae, were herniated through the tunica muscularis (Fig. 4a). Some small pseudodiverticula were partially covered by an attenuated outer layer of longitudinal muscle. However, larger pseudodiverticula were completely devoid of the tunica muscularis and had prominent fibrotic walls (Suppl. Figs. S7–S9). The content of the pseudodiverticula consisted of digested plant structures, abundant amounts of bacteria, and moderate amounts of yeast consistent with Cyniclomyces guttulatus. The epithelium of noninflamed pseudodiverticula resembled normal mucosa of the small intestine, but larger pseudodiverticula were generally lined by an almost completely necrotic epithelium (Suppl. Fig. S10). There was moderate to severe infiltration with heterophils and/or lymphocytes in large pseudodiverticula (pseudodiverticulitis) of each rabbit. Additionally, the tunica muscularis was severely thickened throughout the entire small intestine, consistent with severe diffuse MHSI (Figs. 4c, 5). Also, all 4 rabbits had mild to moderate submucosal fibrosis (Fig. 4b) and moderate diffuse lymphoplasmacytic and/or heterophilic enteritis, and 2 rabbits had moderate to severe dilatation of the mucosal lymph vessels. All rabbits had severe chronic peritonitis. Rabbit Nos. 1 and 2 had diffuse peritonitis with mesenteric abscess formation, while in rabbit Nos. 3 and 4, peritonitis was focally distributed and centered on the pseudodiverticulum.
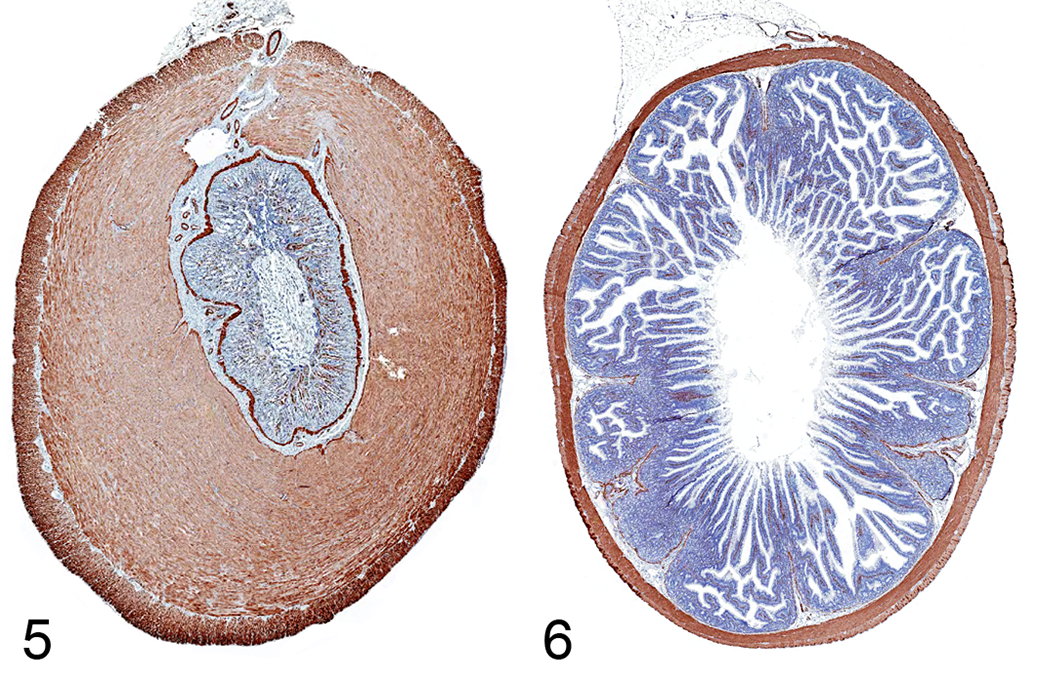
Thickness of the small intestinal tunica muscularis was measured on transverse sections stained by immunohistochemistry with primary antibodies for desmin (see supplemental materials for methods). For rabbit Nos. 1–4, the tunica muscularis had a maximum thickness of 2340 µm (median: 1015 µm), 2281 µm (median: 1078 µm), 2922 µm (median: 1443 µm), and 1613 µm (median: 945 µm), respectively. For reference values, the small intestinal tunica muscularis thickness of 4 dwarf rabbits and 10 rabbits reared for meat devoid of gastrointestinal disorders was measured systematically, rendering a mean thickness of 112.3 µm (95% CI, 107.8–117.03 µm; range, 26.3–389.0 µm) (Fig. 6). Differences of the muscular thickness was statistically significant (P < .001; unpaired t test; Suppl. Fig. S4) between the 4 presented cases and the 14 reference cases.
Pathologic findings of the 4 rabbits are consistent with acquired small intestinal pseudodiverticula (syn. false pulsion diverticula). Rabbit Nos. 1–3 had multiple outpouches, while a single pseudodiverticulum was discovered in rabbit No. 4. In 2 cases, besides the masses being impacted with intestinal content, a connection to the intestinal lumen was not grossly evident. However, histopathologic examination clearly confirmed a small epithelial-lined junction connecting the small intestine with the mesenteric masses. Based on the delicate size of the connection in some cases and the preexisting intestinal epithelial lining of the pseudodiverticula being completely necrotic, those lesions may be easily misdiagnosed as an intestine-associated abscess.
Pseudodiverticula presumably emerge due to increased intraluminal pressure and/or increased weakness of the intestinal wall. 2,5,6,13 Also, a significantly increased intracolonic pressure was measured in rabbits with experimentally induced colonic pseudodiverticula. 9 A predisposed area for outpouch development is the location in which blood vessels penetrate the intestinal wall at the mesenteric surface. 5,6 It is assumed that the tunica mucosa and submucosa are herniated through the tunica muscularis at the vessel entry sites following increased intraluminal pressure. In most equine and feline cases, increased intraluminal pressure resulting in pseudodiverticula was considered to emerge from MHSI. 2,5,6 Pseudodiverticula with MHSI were found by Bettini et al 2 in 2 of 9 cats and by Chaffin et al 5 in 6 of 11 horses. Similarly, in the present 4 cases, mucosal herniation most likely resulted from severe MHSI. Pseudodiverticula were also reported to be directly associated with intestinal lymphoma in 3 horses, 13 potentially resulting from a weakening of the intestinal wall in those areas. In contrast to that in animals, human pseudodiverticulosis of the small intestine is generally not associated with MHSI but rather caused by degeneration of smooth muscle cells (visceral myopathy) or neuronal and axonal degeneration (visceral neuropathy). 10 The hypertrophic tunica muscularis in the present cases leads to the assumption of MHSI causing pseudodiverticula formation in rabbits. A predisposing cause for MHSI—specifically, partial stenosis due to intestinal neoplasia, displacement of the intestinal tract, mucosal cicatrization, impaction, or endoparasitism 2,5 —was not found in any of the 4 rabbits. Therefore, MHSI was considered primary. Whether the chronic lymphoplasmacytic enteritis predisposed to or was secondary to MHSI remains unknown. At least in rats and cats, idiopathic MHSI has been suspected to be caused by chronic intestinal inflammation. 2,3
Reported secondary complications of pseudodiverticula in animals include perforation with subsequent peritonitis, 6,8,13 as well as intramural bleeding. 1 In the 4 presented cases, severe necrosis and inflammation of the wall of large pseudodiverticula were considered the most likely explanation for the clinical signs. The rapid deterioration in rabbit No. 1 was likely due to pseudodiverticulum perforation with subsequent peritonitis and septic shock. Also in rabbit No. 2, chronic leakage of intestinal content led to severe peritonitis with formation of mesenteric abscesses.
In conclusion, we report pseudodiverticula of the small intestine associated with severe MHSI in lagomorphs. An underlying cause of the MH was not evident and was therefore considered idiopathic. The clinical relevance of the lesions pertains to secondary complications, including severe necrosis and inflammation of the pseudodiverticula, leading to pseudodiverticular perforation with subsequent peritonitis and formation of mesenteric abscesses in 2 cases.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985818790789 - Pseudodiverticula of the Small Intestine Associated With Idiopathic Smooth Muscle Hypertrophy in Domestic Rabbits (Oryctolagus cuniculus)
Supplemental Material, DS1_VET_10.1177_0300985818790789 for Pseudodiverticula of the Small Intestine Associated With Idiopathic Smooth Muscle Hypertrophy in Domestic Rabbits (Oryctolagus cuniculus) by Christof A. Bertram, Kerstin Müller, Lesley Halter, Christiane Nastarowitz-Bien, Anne-Katrin Schink, Antina Lübke-Becker, Ellen von Czapiewski, and Robert Klopfleisch in Veterinary Pathology
Footnotes
Acknowledegments
We thank Nancy Ann Erickson for correction of this manuscript.
Supplemental material for this article is available online.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
