Abstract
Megaesophagus appears to be more common in Friesian horses than in other breeds. A prevalence of approximately 2% was observed among Friesian horses presented to the Wolvega Equine Clinic and the Utrecht University Equine Clinic. In this study, morphologic changes in the esophagi of Friesian horses with megaesophagus were compared with those of 6 control horses. Of 18 horses with clinically observed megaesophagus, only 12 animals had esophageal dilation at necropsy, usually involving the thoracic portion. Muscular hypertrophy of the distal esophagus was present in only one-third of the affected horses, indicating that this change is not the most relevant cause of megaesophagus in Friesians. Increased deposition of clumped and disorganized collagen was present in these clinically affected horses mainly in the non-dilated portion of the esophagus. At necropsy, a decrease in neural elements and elastin was present principally in horses with megaesophagus. Mild degeneration and necrosis of the tunica muscularis along the entire length of the esophagus were present in clinically affected horses and encountered only rarely in control animals. There were no significant differences among affected and control horses with respect to inflammation, mineralization, or the number of cells of Cajal. The increased occurrence of megaesophagus in the Friesian breed compared with other horse breeds, together with the presence of abnormal collagen in very young foals, supports the hypothesis that megaesophagus is hereditary in Friesians.
Megaesophagus is a chronic dilation of the esophagus. Several different underlying pathophysiologies for megaesophagus have been proposed, including the loss of interstitial cells of Cajal and idiopathic muscular hypertrophy of the distal esophagus (IMHO). 6
Megaesophagus has been reported with higher frequency in Friesians than in other horse breeds. 3,6 The percentage of Friesian horses with megaesophagus presented to Utrecht University Equine Clinic is high (2.2%) in comparison to esophageal disease in other breeds (0.5%). 6 This suggests that it could be a hereditary trait as comparable to dwarfism and hydrocephalus in Friesians. 7,10 In some cases of clinical megaesophagus, dilation was not present at necropsy. 6 To better understand these features, it is important to characterize the histopathologic elements of this disease in detail. Therefore, the morphologic characteristics of megaesophagus in Friesian horses were analyzed, with emphasis on esophageal elements important for normal esophageal function.
Materials and Methods
Case Selection
Eighteen Friesian horses presenting to the Faculty of Veterinary Medicine at Utrecht University and to Wolvega Equine Clinic, Oldeholtpade, the Netherlands, between 2009 and 2012 that had been diagnosed clinically with megaesophagus (supported by endoscopy and contrast radiography) were euthanized and necropsied. Three nonaffected Friesian horses euthanized for other reasons (Suppl. Table S1) served as controls because they were not closely related to affected horses based on a preliminary screening of pedigrees. In addition, 3 warmblood horses (Suppl. Table S1) that presented for necropsy for orthopedic disease were non-Friesian controls.
Histopathology
For every horse, at least 3 transverse sections of esophagus were collected at necropsy. In horses with esophageal dilation, the first sample was taken at the widest point of esophageal dilation (Fig. 1, location B). The second and third samples were taken at the oral and aboral sides of the dilation at least 10 cm from the dilation (Fig. 1, locations A and C, respectively). In horses without dilation, samples were taken at 3 anatomically similar locations.
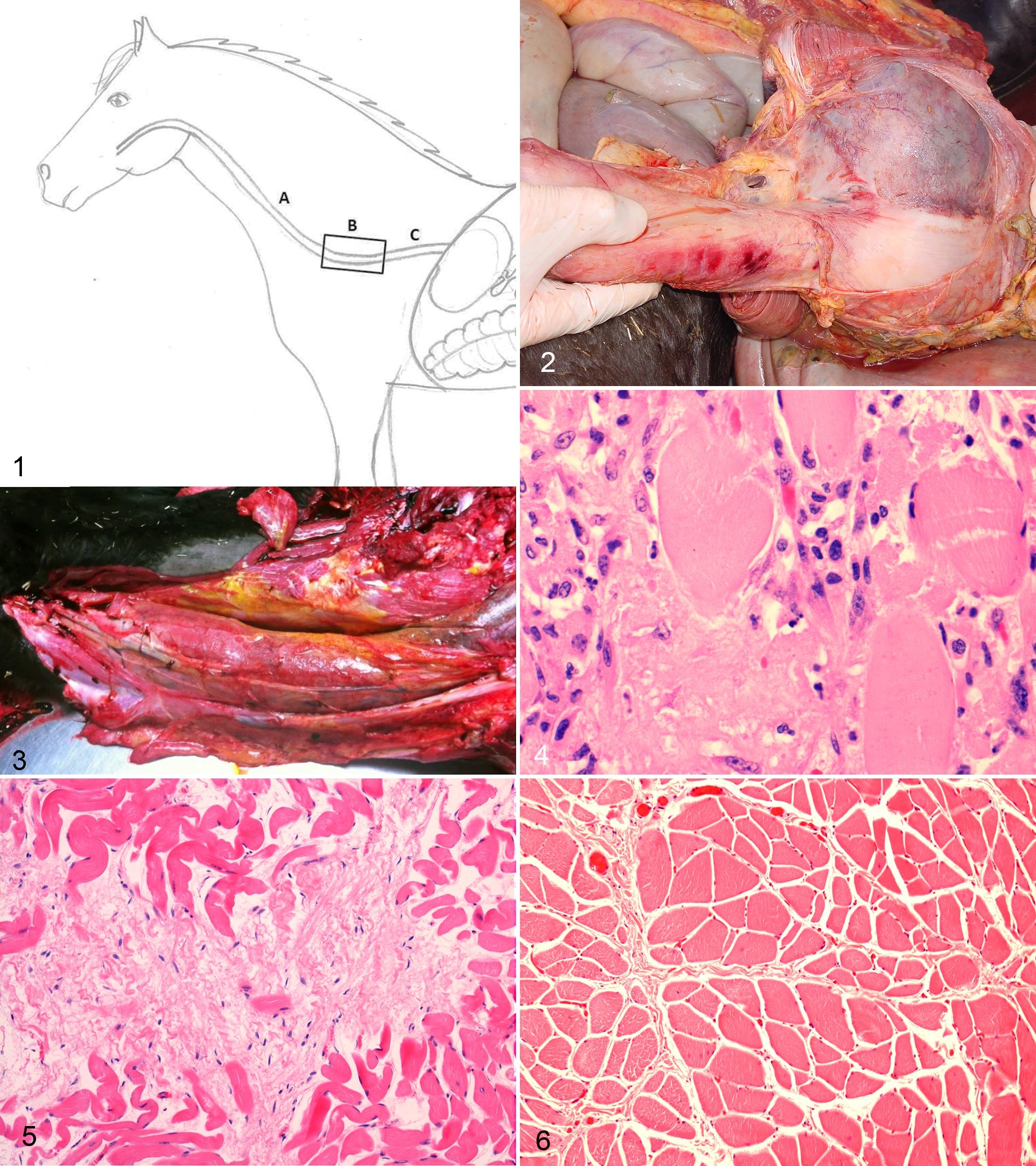
Esophagus with the 3 locations sampled: A. Oral side of dilation. B. Site of esophagus where dilations usually occur in Friesian horses. C. Aboral side of dilation.
Masseter and semitendinosus muscles were collected from 13 of the 18 horses with megaesophagus and all control horses to assess possible evidence of primary systemic muscular disease. The vagal nerve and brainstem (nucleus dorsalis and nucleus ambiguus) of the same Friesians were sampled and assessed for neural lesions.
Tissues were fixed in 10% buffered formalin, paraffin embedded, sectioned, and stained with hematoxylin and eosin (HE). HE staining was used to assess the degree of degeneration and necrosis of the tunica muscularis, inflammation, and mineralization within all layers of the esophagus; the number of neurons; relative amount of degenerate and necrotic neurons; number of ganglia; and number of neurons per ganglion in the tunica muscularis (myenteric plexus). Van Gieson’s stain (Klinipath 64089, Duiven, the Netherlands) was used to visualize the relative quantity of collagen in the mucosa and tunica muscularis.
Histologic findings were semiquantified. Presence of degeneration and/or necrosis, inflammation, mineralization, and collagen deposition were scored as absent (0), mild (grade 1), moderate (grade 2), and severe (grade 3). The number of neurons, ganglia, and neurons per ganglion was assessed as previously described. 4,8 Results from affected Friesians were compared with age-matched controls.
The non-dilated sections of the esophagus were evaluated for distal muscular hypertrophy. The striated muscle cells located on the oral (proximal) side of the dilation (Fig 1, location A) and the smooth muscle cells located on the aboral (distal) side of the dilation (Fig. 1, location C) in the tunica muscularis were analyzed for the presence of hypertrophy and hyperplasia. Two randomly chosen fields were photographed at a magnification of 400× and scored for the number of cells and the width of 20 randomly chosen cells per field. Results from affected Friesian were compared with controls.
Immunohistochemistry
The immunohistochemical stains were applied according to the manufacturer’s instructions. Elastin was visualized using a monoclonal antielastin antibody BA-4 (Leica Biosystems, Diegem, Belgium). Smooth muscle abundance was examined using mouse anti–smooth muscle actin (SMA) (1:1200, lot MU1281008; Biogenex, Duiven, the Netherlands) as primary antibody and horse anti–mouse/biotin (1:125, lot W1123; Vector Laboratories, Amsterdam, the Netherlands) as secondary antibody. Cells of Cajal were evaluated using polyclonal rabbit anti–human CD117 (1:100, lot W2206; DAKO, Eindhoven, the Netherlands) as primary antibody and biotinylated anti–rabbit IgG (1:250; Vector Laboratories) as secondary antibody.
Nervous tissue was examined using monoclonal mouse anti–human neurofilament protein (1:160, lot 00006119; DAKO) as primary antibody and biotinylated anti–mouse IgG (1:125; lot W1123; Vector Laboratories) as secondary antibody. Nervous tissue and the cells of Cajal were evaluated by using a grid (hundred squared; E-PI 10×/20; Carl Zeiss, Feldbach, Switzerland) that was placed at 6 different, randomly chosen locations of the sections of the esophagus, excluding the epithelial layer. The number of grid squares containing immunohistochemically positive-labeled tissue using a 10× objective (100× magnification) was counted.
Negative controls were prepared from serial sections in which the primary antibody was omitted and dilution buffer was applied in its place. Small intestine was used to ensure the specificity of the primary antibodies.
Phenotyping
Patient records of Friesian horses with megaesophagus that were presented to the Faculty of Veterinary Medicine at Utrecht University and to Wolvega Equine Clinic, Oldeholtpade, the Netherlands, between 1999 and 2012 were reviewed. In total, 89 Friesian horses had clinical signs, endoscopic findings, and/or contrast radiography indicating megaesophagus. Specific data, including sex, age at onset of symptoms, and clinical signs, were recorded for all 89 cases.
Statistical Analysis
Clinically affected horses (Nos. 1–18) were compared with control horses (Nos. 19–24). In addition, Friesians with macroscopically visible dilation (Nos. 1–12) were compared with Friesians without a grossly visible dilation (Nos. 13–18) and control horses. Comparisons were made for either all sampled locations of the esophagus (A+B+C) or at (A+C) or at (B) alone. Hypertrophy and hyperplasia were assessed for smooth muscle at location C and striated musculature at location A only.
Results were analyzed using the Kruskal-Wallis test (nonparametric analysis of variance) (SigmaPlot 12.5; Systat Software, San Jose, CA). A difference of P < .05 was considered significant.
Results
Findings are summarized in Supplemental Tables S1 and S2.
Necropsy Findings
Only 12 of 18 horses (Nos. 1–12) had macroscopic megaesophagus, whereas the remaining 6 horses (Nos. 13–18) had a macroscopically normal esophagus. The first group ranged in age from 2 months to 26 years, and the second group ranged from 14 days to 1 year.
Esophageal dilation presented as a gradual luminal widening toward a centrally located broad point. Eight horses had dilation that was mainly located in the thoracic part of the esophagus (Fig. 1, location B) over a distance of 10 to 150 cm (Fig. 2). In 6 of 8 cases (Nos. 1, 2, 5, 6, 8, and 12), the dilation extended distally to the cardia (aboral side, Fig. 1, location C). In 4 of 8 cases (Nos. 2, 4–6), the dilation extended proximally to the cervical portion (oral side, Fig. 1, location A) of the esophagus. Two horses (Nos. 10, 11) had esophageal dilation over its full length (Fig. 3). In 4 (22%) of the affected horses (Nos. 6, 8, 10, and 12) with gross dilation, concentric hypertrophy of the tunica muscularis was present in the most distal portion of the esophagus over a distance of 10 to 30 cm. None of the clinically affected horses and controls had esophageal muscular hypertrophy.
Histologic Examination
There was more degeneration and necrosis in the esophagus (Fig. 4) of clinically affected horses (Nos. 1–18) at locations (A+B+C) than in control horses (P < .05). In most of these clinically affected horses, changes were mild and mainly seen in the striated muscle of the tunica muscularis. In control horses, degeneration and necrosis were present in 2 cases.
There was significantly more collagen deposition between and separating the large muscle fibers (Fig. 5) of the muscularis (P < .05) in both the entire group of clinically affected horses (Nos. 1–18) and the subgroup of horses with megaesophagus at necropsy (Nos. 1–12) for locations (A+C) compared with the controls. In the lamina propria of clinically affected horses (Nos. 1–18), there was more collagen deposition than in the control horses (P < .01) at locations (A+B+C). Some increased collagen deposition was also present in 2 control horses but was quite subtle and scored as grade 1, whereas in the Friesians with megaesophagus at necropsy (Nos. 1–12), changes were scored as grade 2 to 3. In addition, the collagen fibers in affected horses were morphologically different from those in the control horses; they were disorganized, were less condensed, and presented as small and clumped (Fig. 6).
A significant decrease in elastin in the lamina propria (P < .05) was present in the clinically affected horses (Nos. 1–18) compared with controls for locations (A+B+C). Horses without megaesophagus at necropsy had significantly less elastin (P < .05) for locations (A+B+C) and (A+C) compared with Friesians with dilation.
A decrease in the number of neurons, ganglia, and neurons per ganglion was present only in the myenteric plexus of the horses with macroscopic esophageal dilation (Nos. 1–12) compared with nonaffected Friesians (Nos. 13–18) (P < .05) at locations B, C, and (A+B+C). Degenerate neurons were rare in all of the groups.
Neurofilament staining showed a significant decrease of nervous tissue at locations (A+B+C) of clinically affected horses (Nos. 1–18) compared with controls. This decrease in nervous tissue at locations (A+B+C) was also significant when the subgroup (Nos. 1–12) was compared with grossly nonaffected Friesians (P < .05). There were no significant differences in the number of Cajal cells among the different groups.
Inflammation was present in several cases of the clinically affected (Nos. 1–18) and control horses. Inflammation consisted in most cases of low numbers (grade 1) of lymphocytes, plasma cells, macrophages, and a few neutrophils. A small amount (grade 1) of mineralization was present within the tunica muscularis of the esophagus in 5 clinically affected horses (Nos. 5, 9, 10, 12, and 15). Differences among the groups with respect to inflammation and mineralization in the mucosa, submucosa, and muscularis were not significant.
There were no significant differences in hypertrophy and hyperplasia scores of the tunica muscularis between the clinically affected Friesians (Nos. 1–18) and controls. There were no significant differences in hypertrophy and hyperplasia of the smooth musculature (location C) among the groups. There was significantly less hypertrophy and hyperplasia of the striated muscle (location A) in the Friesians without macroscopically visible dilation (Nos. 13–18) compared to Nos. 1–12. No significant histologic abnormalities were present in the vagal nerve, brainstem (nucleus dorsalis and nucleus ambiguous), or the semitendinosus and masseter muscles.
Phenotyping
In total, 4108 Friesians were presented to the Utrecht University Equine Clinic between 1999 and 2012. Ninety-two were clinically diagnosed with megaesophagus (prevalence, 2.2%). In the same period, a total of 18 204 non-Friesian horses were presented to the Utrecht University Equine Clinics; 122 of these had esophageal dysfunction (prevalence, 0.67%). None of these horses were diagnosed with megaesophagus.
Age was documented in 73 of the 89 clinically affected Friesian horses (Suppl. Table S1). Of these, most were younger than 5 years, and the oldest horse with megaesophagus was 26 years (range, 1 day to 26 years; mean, 3.8 years). The reported onset of clinical signs varied from a few days to several years. There was no sex predilection (n = 40 females, n = 35 males, and n = 5 geldings; for 9 horses, sex was not recorded) (Suppl. Table S1).
Clinical signs included recurrent episodes of esophageal obstruction (n = 17), coughing (n = 12), bilateral (alimentary) nasal discharge (n = 11), ptyalism (n = 5), dysphagia (n = 4), visible or palpable swelling of the cervical esophagus (n = 3), fever (n = 3), regurgitation (n = 3), anorexia (n = 3), bruxism (n = 2), and lethargy (n = 2). In a few cases, the horses developed the habit of resting the head on a door post or another structure after having eaten (n = 3), probably to generate a straight and sloping position of the esophagus, thus facilitating passage of food.
The prevalence of megaesophagus among Friesian horses presented to the Wolvega Equine Clinic was 2.1%; esophageal problems in non-Friesian breeds was 0.6%.
Discussion
Our study confirmed the predilection of Friesan horses for megaesophagus compared with non-Friesians, as reported before by several studies. 2,3,6 Histopathologic abnormalities were also seen in Friesian foals, making a genetic basis for this disease very likely. In non-Friesian horses, megaesophagus is rare and in most cases develops as a sequel to a previous esophageal obstruction or laceration. 3,5,6
A subset of horses with esophageal dysfunction did not have esophageal dilation at necropsy. It is possible that esophageal dilation is slowly progressive over several years since this subset includes only young horses (up to 1 year old) or foals. Slow progression is supported by the presence of histopathologic abnormalities in this subgroup of young Friesians. Esophageal dilation was almost always in the thoracic cavity. However, as reported in a previous case, 6 there were 2 young Friesians with esophageal dilation along its entire length. This suggests that the primary cause of megaesophagus in Friesians affects the organ as a whole. Interestingly, concurrent gastric overload or gastric rupture, 2 conditions that seem to have an increased frequency in Friesians, was not present in any of the Friesians in the current study.
An important difference among affected horses and controls was more abundant and abnormal collagen between the muscular layers of the esophagus in affected horses. This abnormal collagen was also present in Friesians that had only functional esophageal problems without grossly visible esophageal dilation. This suggests a causative role for collagen in this disease and supports findings of a previous study that suggested a role for collagen type IV. 6
Esophageal dilation is most often encountered in the thoracic portion, which may be a predilection site due to a combination of factors, including gravity, the absence of external pressure of adjacent muscle, and the presence of negative pressure during inspiration.
Horses with grossly visible dilation had a significant decrease in the number of neurons and ganglia of the myenteric plexus chiefly in the dilated part of the esophagus compared with grossly nonaffected Friesians. There was no significant decrease compared with controls, making the decrease in these neural elements unlikely as a primary defect. Neither loss of myenteric plexi or inflammation was reported. 6 Although a primary functional defect causing megaesophagus cannot be excluded, a primary neuronal defect could presumably lead to a diffusely dilated esophagus, which was rarely observed in the present study.
The interstitial cells of Cajal play a major role in generating and coordinating gastrointestinal motility patterns. 9 Loss of neurogenic input at the level of the interstitial cells of Cajal in the Friesian breed has been suggested to underlie the development of megaesophagus. 11 However, no differences in the number of interstitial cells of Cajal were present among affected and control horses in our study. This makes a role for interstitial cells of Cajal in this disease less likely, although a functional defect cannot be excluded.
In a recent study of 852 horses of different breeds, 6 only 6 horses, all of which were Friesians, had megaesophagus. In that study, 5 of the 6 cases (83%) had muscular hypertrophy of the distal esophagus, and a possible link with this change was proposed. The present study identified only 4 of 18 horses (22%) with muscular hypertrophy of the distal esophagus. Idiopathic muscular hypertrophy in non-Friesian horses is usually seen in older animals. 1 Results of this study indicated that megaesophagus is frequently encountered in Friesians aged 2 years or younger. Esophageal dilation was present in both smooth muscle and striated esophageal muscle, suggesting that esophageal dilation in Friesians has little or no association with distal muscular hypertrophy.
In conclusion, megaesophagus occurs more frequently in Friesian horses than in other breeds and occurs most frequently during the first 5 years of life. Most dilations are in the mid to caudal thoracic esophagus. Friesian horses can have severe functional problems of the esophagus without a grossly visible esophageal dilation. There is increased, aberrant collagen in chiefly the nondilated parts of the esophagus of affected horses. This collagen can be identified in very young foals, suggesting megaesophagus has a hereditary component in Friesians.
Footnotes
Acknowledgements
We thank Johan van Amerongen, Louis van den Boom, and the technicians of the histology laboratory of the Veterinary Pathology Diagnostic Center for their technical assistance. Many thanks to Chana Callens and Sarah Loomans for their accurate technical work. We also thank the Royal Friesian Studbook for the financial support and all referring veterinarians. In addition, we thank Rachel Thomas for editing English usage.
Author Contribution
Conception or design: MP, AG, CMdB, WB, PRvW, WS, TP, BJD, IW, CD. Data acquisition, analysis, or interpretation: MP, AG, VS, CMdB, WB, PRvW, WS, TP, BJD, IW, CD. Drafting the manuscript: MP, AG. All authors participated in critically revising the manuscript, gave final approval, and agree to be accountable for all aspects of work to ensure integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Transportation of the Friesian horses to Utrecht University was financed by the Royal Friesian Studbook (KFPS).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
