Abstract
Esophageal intramural pseudodiverticulosis is a benign disease characterized by numerous, small outpouchings from the esophageal epithelium. Esophageal intramural pseudodiverticulosis has scarcely been reported with only 200–300 cases worldwide. The etiology of esophageal intramural pseudodiverticulosis is also unclear; however, there is an associated increased risk with diabetes mellitus, gastroesophageal reflux disease, esophageal candidiasis, and chronic alcohol and tobacco abuse. Esophageal intramural pseudodiverticulosis has a characteristic appearance on esophagogastroduodenoscopy. Treatment of esophageal intramural pseudodiverticulosis has historically been limited to symptom management with acid suppression, anti-fungal therapy, and endoscopic dilation in areas of stricture. This report is a case of a 52-year-old female status post two esophageal stricture repairs with dilation over prior 2 years, who presented with non-remitting solid food dysphagia and food impaction found to have esophageal intramural pseudodiverticulosis with concomitant jackhammer esophagus and esophageal candidiasis.
Keywords
Introduction
Esophageal intramural pseudodiverticulosis (EIP) is a benign disease characterized by numerous, small outpouchings from the esophageal epithelium. These outpouchings represent dilated excretory ducts of mucinous glands protruding into the esophageal lumen.1–4 Although EIP was first described by Mendl 5 in 1960, only 200–300 cases have been reported worldwide.1,6 A review of 14,350 radiologic esophageal examinations revealed the presence of EIP in only 0.15% of cases. 2 The pathophysiology of EIP remains to be elucidated, but current evidence suggests that the pseudodiverticulae may be secondary to submucosal duct compression from chronic esophageal inflammation or hypermobility.7–9 The etiology of EIP is also unclear; however, there is an associated increased risk with diabetes mellitus, gastroesophageal reflux disease (GERD), esophageal candidiasis, and chronic alcoholism.10–13 EIP has also been associated with functional esophageal disorders such as jackhammer esophagus and achalasia.14,15 EIP is most common in men in their sixth and seventh decades of life.16–19 The most common clinical presentation is intermittent or progressive dysphagia often associated with esophageal stricture.3,4 EIP has a characteristic appearance on esophagogastroduodenoscopy (EGD) and histopathology typically reveals chronic inflammation with dilated submucosal glands and excretory ducts without evidence of infection or malignancy.7,8 Treatment of EIP has historically been limited to symptom management with acid suppression, anti-fungal therapy, and endoscopic dilation in areas of stricture.7,19,20 Here, we report a patient with a past medical history significant for acute esophagitis and esophageal stricture status post balloon dilation who presented with recurrent esophageal dysphagia due to EIP and jackhammer esophagus.
Case presentation
A 52-year-old female presented with esophageal dysphagia. The patients’ past medical history was significant for GERD, constipation, hypertension, and arthritis. The patient had a 23 pack-year smoking history, history of alcohol abuse, and daily non-steroidal anti-inflammatory drugs (NSAIDs) use for her joint pain. A brief timeline of events follows (Table 1). On initial presentation, she had two episodes of food impaction. EGD showed severe circumferential ulceration in the distal esophagus with low-grade narrowing (Figure 1). Esophageal biopsy revealed reflux esophagitis without evidence of eosinophilic esophagitis and gastric biopsy revealed Helicobacter pylori-negative chronic gastritis. The lower esophagus was dilated from 15 to 18 mm with a controlled radial expansion (CRE) balloon. Proton-pump inhibitor (PPI) therapy was initiated, and the patient reported improvement of symptoms on routine follow-up.
Timeline of events.

EGD: esophagogastroduodenoscopy; PPI: proton-pump inhibitor; CRE: controlled radial expansion.

Severe circumferential ulceration with low-grade narrowing in distal esophagus.
Sixteen months later, the patient returned with 2 months of solid food avoidance due to trouble swallowing. The patient also reported increased frequency of heartburn from once or twice weekly to every day despite adherence to daily PPI therapy. Patient reported increased severity of heartburn when she drank soda. Otherwise, patient reported normal appetite and no significant weight changes. She denied nausea, vomiting, chest pain, abdominal pain, or change in bowel habits. Physical exam revealed a healthy-appearing, obese woman in no acute distress. Family history was significant for gastritis and recurrent vomiting. The patient reported she desired relief of the food stuck in her mid-chest.
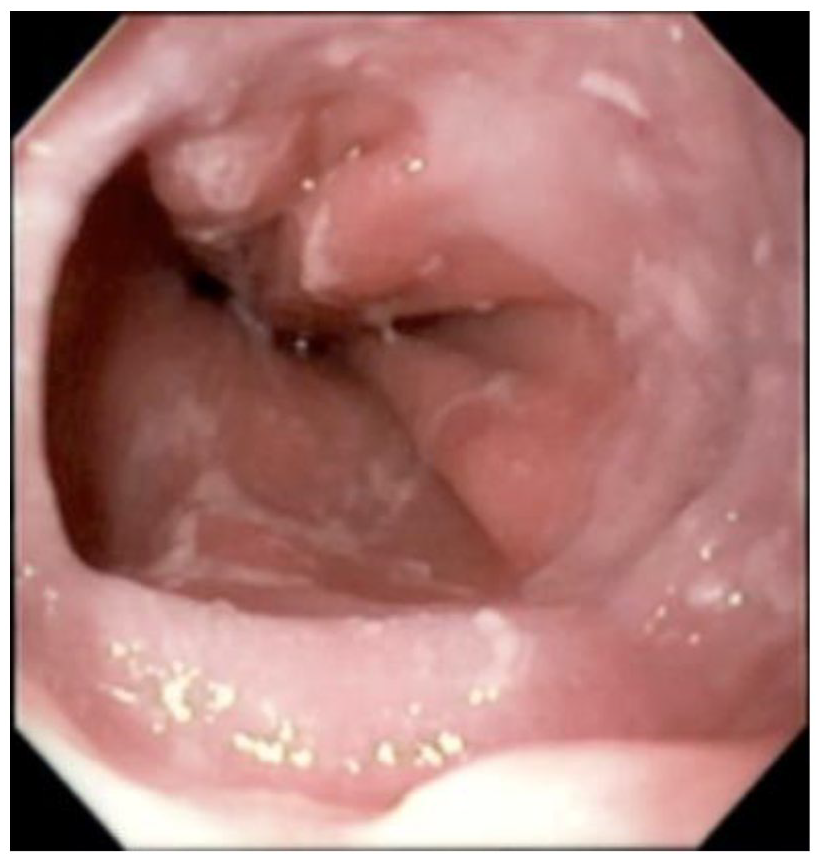
Subsequent EGD revealed fine rings and diffuse yellow-white exudate (Figure 2). No esophageal stricture, ring, or hiatal hernia were seen to suspect a mechanical cause of the dysphagia (Figure 3). However, multiple openings in the esophageal mucosa were noted, suspicious for EIP (Figure 4). Random biopsies of the middle and upper esophagus also showed active esophagitis and were positive for focal Candida spp. In contrast to the H. pylori-negative gastritis at initial presentation, random gastric biopsies (Figure 5) revealed reactive gastropathy with focal H. pylori colonization. In addition, the patient underwent high-resolution esophageal manometry (HRM), which revealed strong contractions of the esophageal body with normal lower esophageal sphincter relaxation (Figure 6) with a distal contractile integral of 11,576 mm Hg s cm (normal range = <8000 mm Hg s cm). HRM findings were consistent with hypercontractile peristalsis (“Jackhammer esophagus”) based on the Chicago v3.0 classification. 19 Patient was initiated on a 14-day course of 200 mg Diflucan once daily for esophageal candidiasis. Patient was also treated for H. pylori colonization with standard quadruple therapy for 14 days, consisting of four times daily administration of bismuth subsalicylate 262 mg, metronidazole 250 mg, and tetracycline 500 mg, along with pantoprazole 40 mg twice daily. Despite adherence to therapy, patient reported symptoms of dysphagia and heartburn were not fully resolved at 1-month follow-up. Due to lack of clinical improvement, decision was made to repeat EGD to monitor for resolution of esophageal mucosal damage and undergo empiric esophageal dilation. Repeat EGD revealed normal-appearing esophageal mucosa with regular Z-line. Esophageal dilation with 16 mm Savary was successfully performed. Patient reported full resolution of her symptoms at 2-week follow-up. Patient remained asymptomatic at her most recent appointment 7 months after empiric dilation.
Diffuse yellow-white exudate.

No esophageal stricture, ring, or hiatal hernia at GE junction.

Multiple openings in the esophageal mucosa.

Visualization of stomach, random gastric biopsies taken.

HRM showing strong esophageal body contractions.
Discussion
EIP is characterized by numerous flask-shaped out-pouching in the esophageal wall. The pathology of EIP is thought to be due to excretory duct obstruction caused by inflammation of epithelial cells and surrounding mucosal or submucosal fibrosis and dysmotility.3,7–9,21–22 Although scarcely reported, early pathological changes of EIP may be more common than currently appreciated. An autopsy study of 100 grossly normal esophageal specimens revealed that 67% had chronic submucosal inflammation associated with mucinous glands and ducts, 14% had excretory duct dilation, and 7% had developed esophageal cysts. 23 Nevertheless, no relationship between chronic submucosal inflammation and duct dilation has been elucidated.
Eighty percent of EIP cases present with progressive dysphagia due to esophageal stricture, but others may present as worsening GERD, vomiting, or chest pain resembling an acute cardio-pulmonary process. 24 The risk of developing EIP is increased with underlying conditions such as diabetes mellitus, GERD, HIV infection, as well as Crohn’s disease, tuberculosis, and Mallory–Weiss syndrome.5,10–13 Alcohol and tobacco use are leading risk factors for the development of EIP. 12 One recent study reported that from a sample of EIP patients, 76% suffered from alcohol abuse and 57% had past or ongoing nicotine abuse. 25 Nevertheless, esophageal dysmotility because of jackhammer esophagus, hypoperistalsis, and achalasia have also been associated with increased risk of EIP.8,9 Our patient had a history significant for GERD and chronic alcohol abuse, as well as having jackhammer esophagus identified on manometry. Furthermore, although EIP is a benign pathology, there is some evidence of an association with esophageal cancer. One study showed EIP in 4.5% of patients with esophageal carcinoma, which is significantly higher than the 0.09% of control patients found to have EIP. 26
EGD classically shows multiple pits in the esophageal wall that are 1–4 mm in length and 1–2 mm in width.3,19 There may be 5 to more than 20 pseudodiverticula that are diffuse or segmental in distribution. 27 EIP is typically also associated with strictures in proximal esophageal locations. Nevertheless, food impaction without definite stricture may be a common finding on EGD. 25 Our case was uncommon in that, although empiric esophageal dilation led to resolution of symptoms, there was no evidence of esophageal stricture or other cause for the dysphagia reported by the patient.
Our esophageal biopsy results were also consistent with Candida spp. Numerous infectious cases of EIP with fistulation, abscesses, and tract formation have been reported; however, the prevalence of infections in EIP is unknown.28–30 Approximately 50% of EIP patients have evidence of Candida infection, although its role in etiopathogenesis remains unclear. 24 It is unclear if esophageal candidiasis is a cause or consequence of EIP, and our patient did not have a known history of fungal infection. Nevertheless, oral azole therapy is typically provided to EIP patients with evidence of Candida, with most reports demonstrating symptomatic improvement. 20 Our patient was provided with a 14-day course of Diflucan, which led to healed esophageal mucosa on repeat EGD.
Currently, symptomatic treatment is the goal of therapy for patients with EIP. Endoscopic dilation is the principal treatment for most cases of EIP with strictures. Most patients who undergo endoscopic dilation experience symptomatic relief. However, most patients have recurrence of symptoms and require an average of nine dilations during follow-up. 31 Thus, endoscopic dilation is not an ultimate treatment for EIP and has no effect on pseudodiverticula present. Given the association with GERD, some patients with EIP may achieve symptom relief with PPI therapy. 27 Due to the association between EIP and the numerous co-morbidities mentioned above, most patients stand to benefit from multimodal therapies to control underlying disease processes.
Conclusion
In summary, we present a case of a patient with EIP without evidence of esophageal stricture and with co-present esophageal candidiasis, H. pylori chronic gastritis, and jackhammer esophagus, whose recurrent dysphagia and food impaction successfully resolved after empiric esophageal dilation. Our case emphasizes the importance of considering co-morbid conditions in the treatment EIP and pursuing empiric esophageal dilation even in the absence of evident esophageal stricture.
Footnotes
Acknowledgements
The authors are very appreciative to the patient for the opportunity to learn as well as thankful to the gastroenterology department at our hospital for providing support for completing this report.
Author contributions
S.V. contributed to the investigation, coordination, writing (original and final draft), reviewing, and editing. L.D.G. contributed to the writing (original and final draft), reviewing, and editing. C.V.L contributed to the writing (original and final draft), reviewing, and editing. T.J. contributed to the reviewing and editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
