Abstract
Chronic liver disease is an important cause of illness in horses, and treatment is mainly supportive. Research into new treatment modalities for humans has shown promising data regarding metallothionein (MT), which has been shown to possess regenerative, antifibrotic, and anti-inflammatory properties. This study aimed to examine the relationship between hepatic MT expression and the histopathologic markers of hepatic inflammation, fibrosis and bile duct proliferation, as well as cellular regeneration in 77 selected cases of chronic liver disease in horses. We hypothesized that higher MT expression would be associated with increased heptocellular proliferation and decreased fibrosis, inflammation, and bile duct proliferation. Hepatocellular MT expression was evaluated with immunohistochemistry. Additionally, cellular regeneration was evaluated with immunohistochemistry for Ki-67, a protein expressed during all active stages of the cell cycle. The severity of inflammation and fibrosis was scored, and bile duct proliferation was assessed by counting bile duct profiles. MT expression was observed in 73 of 77 (94.8%) cases of chronically diseased livers. Ki-67 expression was seen in resident Kupffer cells (n = 42, 54.6%), lymphocytes (n = 39, 50.7%), bile duct epithelium (n = 10, 13.0%), and hepatocytes (n = 8, 10.4%). MT expression was significantly associated with Ki-67 staining in bile duct epithelium and Kupffer cells. Additionally, median MT expression was higher in cases containing lymphocytic infiltrates as compared with cases with no lymphocytic infiltrate (P < .05). These findings are the first known report of MT expression within chronic equine hepatic disease.
Keywords
Liver disease is an important cause of illness in horses. It affects all ages, breeds, and sexes and may result in fatality in up to 25% of cases. 17 The exact etiology of chronic hepatic disease in adults is rarely identified; however, some causes include aflatoxicosis, 13 pyrrolizidine alkaloid toxicity, 48 serum hepatitis, 2 alsike clover, 35 and cholangiohepatitis. 40 Because of the lack of specificity in clinical signs, the diagnosis of liver disease is largely based on biopsy and serum biochemical analyses. 7 In general, most signs of hepatic failure appear suddenly, regardless of the cause and duration of the underlying disease, after the loss >75% of the functional capacity of the liver. 7 Irrespective of the cause, the prognosis of hepatic disease depends on the severity of histologic lesions. Severe fibrosis or inflammation within the liver often carries poor prognosis and is associated with decreased regeneration and increased production of reactive oxygen species. 6,14 For example, in humans, a major risk factor for chronic liver disease is alcohol consumption, causing a syndrome characterized by hepatocellular necrosis, neutrophilic inflammation, and progressive fibrosis known as alcoholic hepatitis. The effect is thought to be related to ethanol metabolism, which results in the generation of reactive oxygen species and lipid peroxidation. 18 Very little is known about the pathogenesis of chronic liver disease in horses. Cases with moderate or severe hepatic fibrosis were found to be associated with significantly higher mortality rates, although the exact etiology is rarely confirmed. 17
New treatment modalities of chronic liver disease in humans currently focus on enhancing hepatic defense mechanisms to stimulate regeneration and decrease inflammation and fibrosis. A protein that received much attention in recent years is metallothionein (MT), which was shown to possess regenerative, antifibrotic, and anti-inflammatory activities in experimental animals. 25 MT was first identified in 1957 as a cadmium- and zinc-binding protein produced in the equine renal cortex. 27 It is a low molecular weight, highly conserved intracellular protein with a high binding affinity for divalent cations, especially copper. 49 Virtually all eukaryotic as well as some prokaryotic organisms express MT. 38 Four major isoforms of MT exist in mammals (I, II, III, IV), of which MT-I and MT-II are most abundant in mammalian cells and are considered a single MT due to their high homology. 39,50 Most mammalian cells express low levels of MT, but there is tissue specificity to expression. Liver MT is predominantly associated with copper and zinc, whereas kidney MT binds mainly copper, cadmium, and zinc. 11
MT gene expression can be induced by various hepatotoxic molecules, such as heavy metals, carbon tetrachloride, and ethanol, as well as certain cell stressors, including cellular starvation and hydric stress. 49 Studies showed that MT may play an important role as an anti-inflammatory agent. 22,23 For example, MT appears to be protective against lipopolysaccharide-induced acute lung inflammation when compared with MT-knockout mice. 46 Several proinflammatory cytokines, including interleukin 6, tumor necrosis factor α, and interferon γ, were shown to induce the expression of MT. 16 The MT gene contains several response elements that upregulate its transcription, including glucocorticoid response elements, metal response elements, and an antioxidant response element. 15
MT is normally found in the cytosol of resting, nonproliferating cells but can be translocated to the nucleus during cell proliferation and differentiation. 3 Intranuclear localization of MT is thought to be associated with gene expression during the cell cycle, as a mechanism to protect DNA from damage and cell apoptosis. 10 Elevated levels of intranuclear MT may be related to an increased demand for zinc during rapid growth, as zinc is required for transcription factors and enzyme function. 10 Furthermore, the presence of MT within the nucleus of human neoplastic cells is indicative of their mitotic activity, as MT immunostaining was most intense at the proliferative edge of malignant tumors. 9 In addition, a 4-fold increase in cellular MT levels was measured in proliferating liver cells as compared with MT levels in resting cells. 45 The role of MT in carcinogenesis has also been suggested, and in vitro studies postulated that p53 and the estrogen receptor may be involved in the induction of MT in human neoplastic epithelial cells. 11
When liver regeneration was compared between wild-type and MT-knockout mice subjected to partial hepatectomy or acute liver injury via a hepatotoxin, wild-type mice had a significantly higher proliferation index. 37 Similarly, fibrosis was reversible in wild-type mice exposed to a hepatotoxin, as compared with MT-knockout mice. 25
Very little is known about the expression of MT during chronic liver disease in domestic animals. Recent work by our group showed a positive correlation between MT expression and hepatocyte regeneration and inflammation in chronic liver disease in dogs. 44
The present study aimed to examine the relationship between MT expression and hepatocellular proliferation in horses affected by chronic liver disease. The correlation between MT expression and common liver lesions (ie, degree of hepatic inflammation, fibrosis, and bile duct proliferation) was also assessed. We hypothesized that higher MT expression would be associated with increased heptocellular proliferation and decreased fibrosis, inflammation, and bile duct proliferation.
Materials and Methods
Case Selection and Histologic Scoring
Pathology reports from the archives at Prairie Diagnostic Services between January 1, 1995, and December 31, 2014, were reviewed for cases of chronic equine liver disease by assessing for the presence of fibrosis within hematoxylin and eosin (HE)–stained histologic sections. Cases included in the study showed no autolysis and contained at least minimal fibrosis, with or without inflammation or bile duct proliferation based on evaluation of HE-stained histologic sections. All cases originated as necropsy submissions, and none were from biopsy submissions, as they did not meet the minimum requirement of 1 cm2 of available tissue. A total of 77 cases were selected for this study.
In each section, hepatic inflammation, fibrosis, and bile duct proliferation were scored independently by 2 pathologists (A.N.A. and A.L.A.) with a grading scheme modified after Sridharan et al. 44 Hepatic inflammation was subjectively scored as 0, 1, 2, or 3 based on the assessment of the entire HE-stained section. A score of 0 indicated the absence of inflammatory cells; 1, the presence of inflammatory cells in <10% of the section; 2, 10% to 50%; and 3, >50%. Similarly, fibrosis was scored 0, 1, 2, or 3 based on the assessment of the same sections stained with Masson trichrome stain. A score of 0 indicated the absence of fibrosis; 1, the presence of periportal fibrosis not extending into the adjacent parenchyma; 2, fibrosis extending away from portal tracts into the parenchyma; and 3, extensive bridging fibrosis between portal areas, centrilobular areas, or portal and centrilobular areas. Bile duct proliferation was evaluated by counting the number of bile duct profiles in 5 random fields that contained at least 1 bile duct (magnification, 400×; field of view, 500-µm diameter). The number of bile ducts counted per pathologist was then averaged, and the cases were subsequently divided into 2 groups based on the presence or absence of bile duct proliferation, to improve statistical power. Cases with >4 bile ducts per field were considered to have bile duct proliferation, whereas cases with 0 to 4 bile ducts per field were regarded as not proliferated. The cutoff value of 4 bile ducts per field was determined by utilizing the bile duct counts of 6 normal liver sections (10 random fields per section) measured by both pathologists. In the histologically normal livers, ≤4 bile ducts per field were found in 95% of the 60 counted fields.
Immunohistochemistry for MT and Ki-67
Immunohistochemistry for MT and Ki-67 was performed with consecutive 5-μm sections from formalin-fixed, paraffin-embedded tissue blocks mounted onto charged slides (ProbeOn Plus or SuperFrost Plus; Fisher) and oven baked at 60°C for 60 minutes. Tissue sections were deparaffinized in xylene for 5 minutes, followed by rehydration in successive graded ethanol (5 minutes each: 100%, 95%, 70%) and then rinsed in distilled water. The tissue sections were then placed in 3% hydrogen peroxide solution (in methanol) for 10 minutes, to block any nonspecific endogenous peroxidase activity, and then rinsed twice in PBST (phosphate-buffered saline with Tween 20) for 5 minutes. Heat-induced antigen retrieval was performed in a water bath (Dako PT Link) at 97°C in Tris/EDTA buffer (pH 9) for 20 minutes and allowed to cool in immunohistochemical buffer.
The MT primary antibody (clone E9, mouse anti-horse, monoclonal IgG1; Dako) was diluted 1:1000 in antibody diluent (Dako) and incubated on the slides for 30 minutes at room temperature. The Ki-67 primary antibody (clone MIB-1, mouse anti-human, monoclonal IgG1; Dako) was diluted 1:50 in antibody diluent and applied overnight at 4°C. Slides were rinsed 3 times for 5 minutes with PBST and incubated with the Dako Envision+ System. Color development was performed with diaminobenzidine substrate buffer and chromagen per manufacturer’s instructions. Slides were counterstained with hematoxylin at room temperature for 4 minutes, rinsed with water, and mounted with coverslips.
Positive controls for MT staining included equine skin and kidney as previously described. 28,42 Positive controls for Ki-67 staining included equine duodenum with Peyer patches. Negative controls were processed similarly, with omission of the primary antibody as well as substitution with an irrelevant isotype-matched monoclonal antibody (clone 15.c.5, mouse anti-bovine viral diarrhea virus, monoclonal IgG1; IDEXX). Six histologically normal liver samples obtained during necropsy were immunostained for MT and Ki-67 to examine the expression within normal liver.
Scoring of Hepatic MT and Ki-67 Expression
MT expression in hepatocytes was evaluated by counting the number of positive cells via an intraocular grid at 400× measuring 25 µm2 (J.N.C.V.). Ten fields were counted in each section in a diagonal square zig-zag pattern, and an average per field was calculated. On average, 104.5 hepatocytes were counted within the 25-µm2 grid.
Ki-67 expression was evaluated by noting the presence or absence of nuclear staining within hepatocytes, Kupffer cells, biliary epithelium, and any lymphocytic infiltrates within the entire liver section(s) (J.N.C.V.).
Copper Staining
Ten slides with elevated MT expression and 6 normal liver samples were stained with rhodanine stain and assessed for copper content as previously described (J.N.C.V.). 41
Statistical Analysis
Statistical analysis was performed with Stata 14.2 (StataCorp). Interrater agreement between the pathologists was determined by calculating a kappa score for inflammation, fibrosis, and bile duct proliferation. The kappa score was interpreted according to Landis and Koch. 29 A Spearman rank test was used to determine correlations among inflammation, fibrosis, and bile duct hyperplasia. The data on MT expression were not normally distributed; therefore, nonparametric statistics were used. A Dunn test with post hoc Sidak (to account for multiple comparisons) was used for comparing group differences in hepatocyte MT expression by inflammation score (0–3) and fibrosis score (0–3) as determined by each pathologist. A Mann-Whitney U test was used to assess potential differences in hepatocyte MT expression by nuclear Ki-67 expression (presence/absence) within hepatocytes, bile duct epithelium, intrasinusoidal Kupffer cells, and lymphocytic infiltrates. Additionally, a Mann-Whitney U test was used to assess potential differences in hepatocyte MT expression by the presence or absence of bile duct proliferation as determined by each pathologist. In all analyses, the results were considered statistically significant when P < .05.
Results
A significant moderate agreement was found between the pathologists, who independently examined all cases for each evaluated parameter (Table 1).
Interobserver Agreement for 2 Pathologists’ Histologic Scores of All 77 Diseased Liver Samples for Hepatic Inflammation, Fibrosis, and Bile Duct Proliferation Based on the Kappa Statistic.a
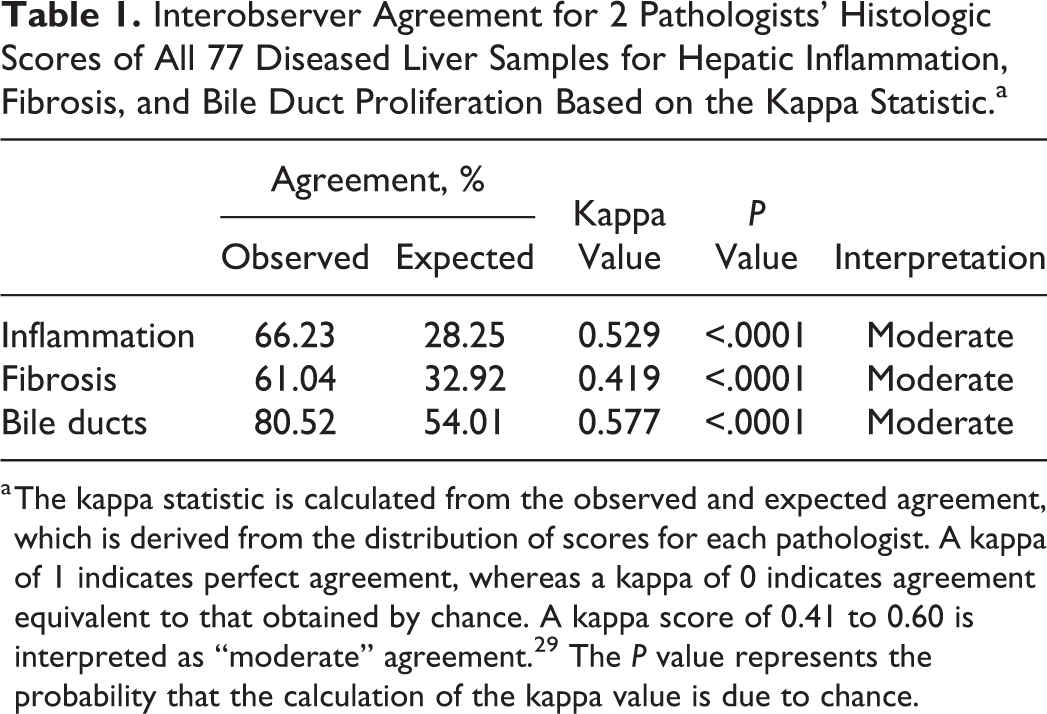
a The kappa statistic is calculated from the observed and expected agreement, which is derived from the distribution of scores for each pathologist. A kappa of 1 indicates perfect agreement, whereas a kappa of 0 indicates agreement equivalent to that obtained by chance. A kappa score of 0.41 to 0.60 is interpreted as “moderate” agreement. 29 The P value represents the probability that the calculation of the kappa value is due to chance.
Inflammation was present in 81.8% of cases, and bile duct proliferation was present in 35.7% of cases. A significant moderate correlation was observed between inflammation and fibrosis scores (Spearman rho = 0.4046, P = .003). 12 No significant correlation was found between bile duct hyperplasia with inflammation or fibrosis.
The expression of MT and Ki-67 in normal liver samples was low to absent in comparison with diseased livers (Figs. 1, 2). MT expression was observed within hepatocytes in 73 of 77 (94.8%) cases of equine liver disease, with a median of 42.0 positive cells and a range of 0 to 166.3 positive cells per 25 µm2. Both cytoplasmic and nuclear staining were observed. Minimal expression was also occasionally noted within bile duct epithelium and lymphocytes.
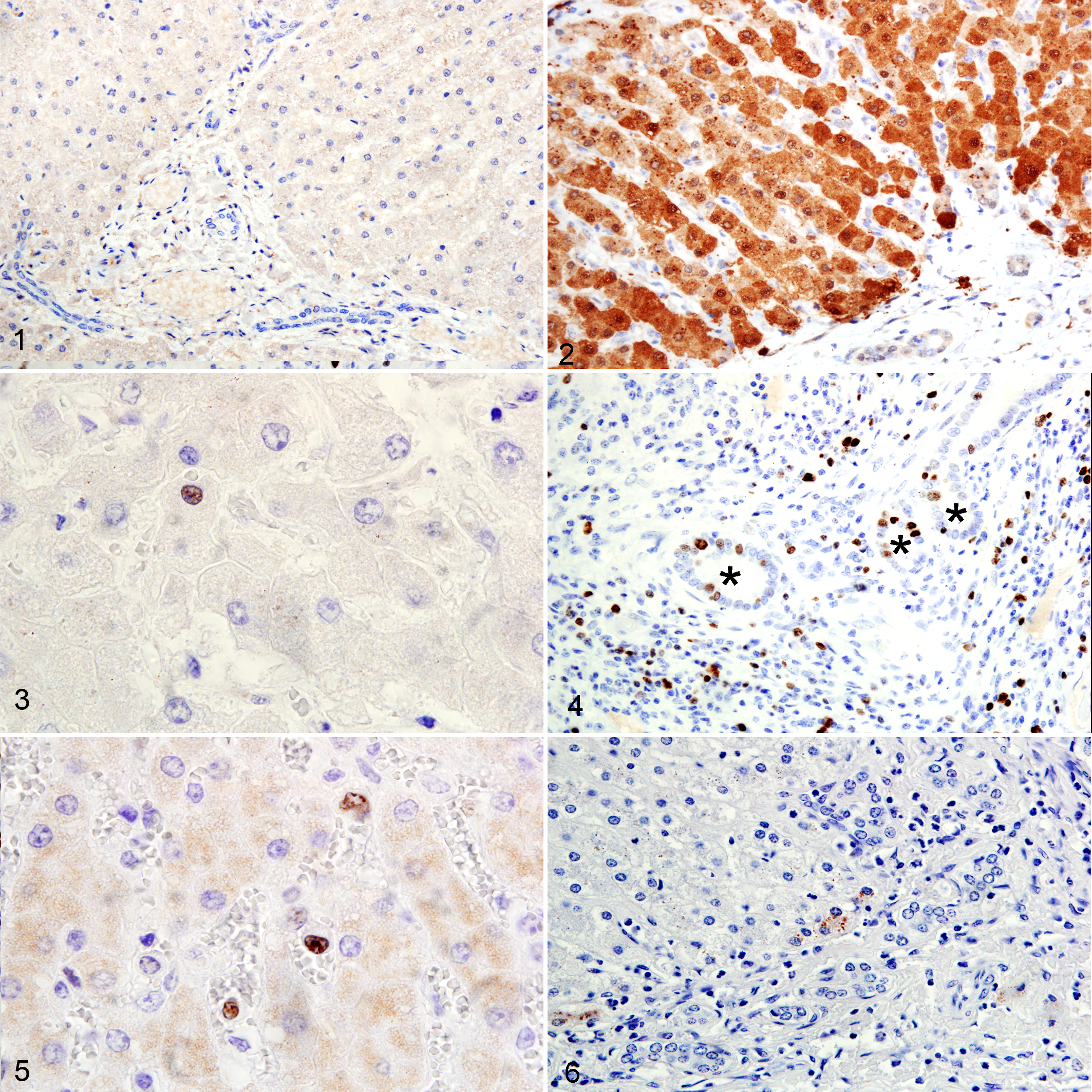
Of 77 cases, 8 (10.4%) had evidence of Ki-67 expression within hepatocyte nuclei (Fig. 3); 10 (13%) had nuclear expression within bile duct epithelium (Fig. 4); and 42 (54.6%) displayed nuclear expression within Kupffer cells (Fig. 5). Staining was more prominent within bile duct epithelial cells than hepatocytes, where the pattern of expression was sparse and limited to individual random hepatocytes. Strong Ki-67 expression was found in all lymphocytic inflammatory foci (Fig. 4).
Rhodanine staining for intracellular copper within hepatocytes was seen in 3 of 10 cases of diseased liver. Staining was random, sparse, and observed only within individual or small aggregates of cells, predominantly in periportal areas (Fig. 6). Staining within hepatocytes of normal liver samples was very weak to absent.
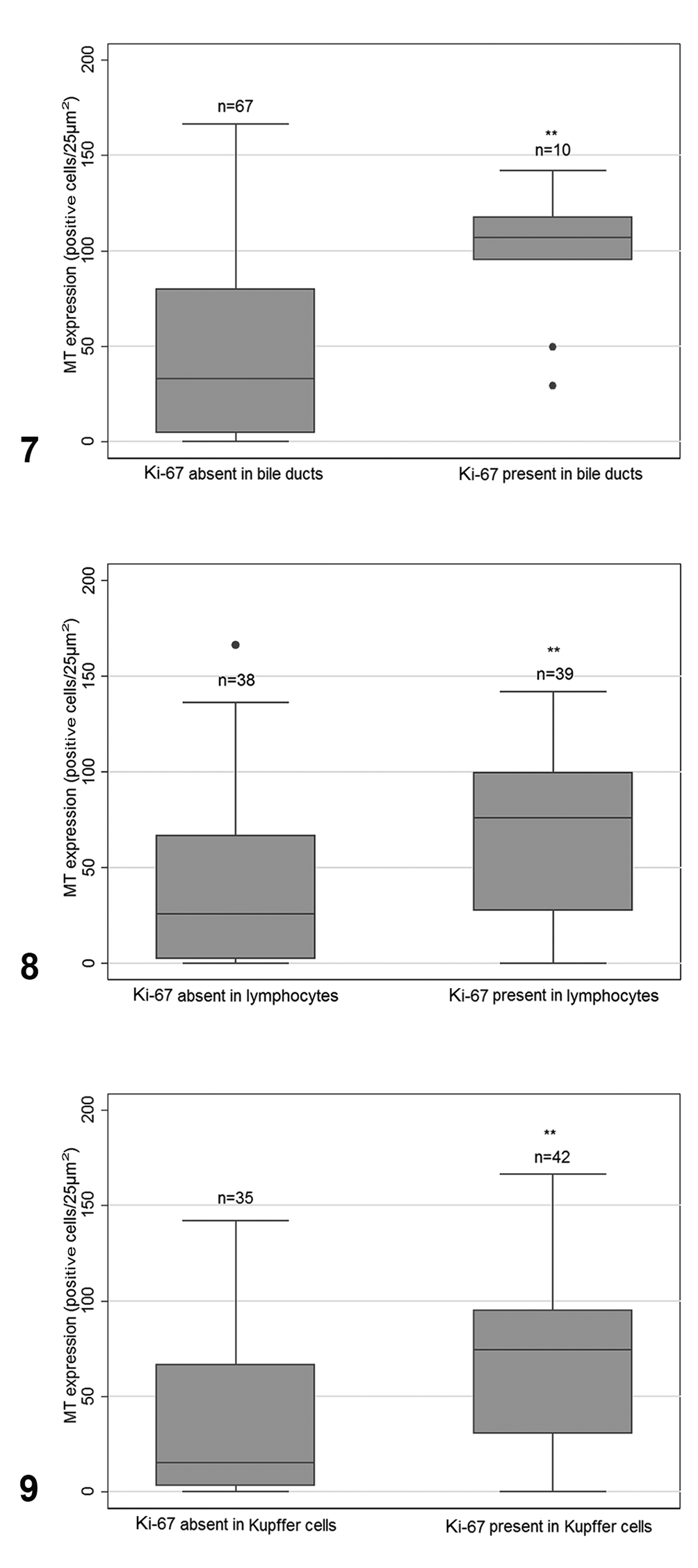
MT expression was significantly associated with Ki-67 staining in bile duct epithelium and Kupffer cells (Table 2). Specifically, median MT expression within hepatocytes was significantly increased when Ki-67 expression was observed within bile duct epithelium, in contrast to when it was absent (median: 106.8 vs 33.0 cells/25 µm2, n = 10 and 67, respectively, P = .0004; Fig. 7). In addition, when MT expression was compared in samples with and without Ki-67 expression in sinusoidal Kupffer cells (Fig. 8), there was a significant increase in MT expression in the Ki-67-positive group (median: 74.6 vs 15.2 cells/25 µm2, n = 42 and 35, respectively, P = .0045). Median MT expression was also higher in cases containing lymphocytic infiltrates as compared with cases with no lymphocytic infiltrate (median: 76.1 vs 25.8 cells/25 µm2, n = 39 and 38, respectively, P = .0017; Fig. 9). Strong Ki-67 expression was found in all lymphocytic inflammatory foci.
Metallothionein Expression in 77 Diseased Equine Livers for Each Dichotomized Outcome Variable.
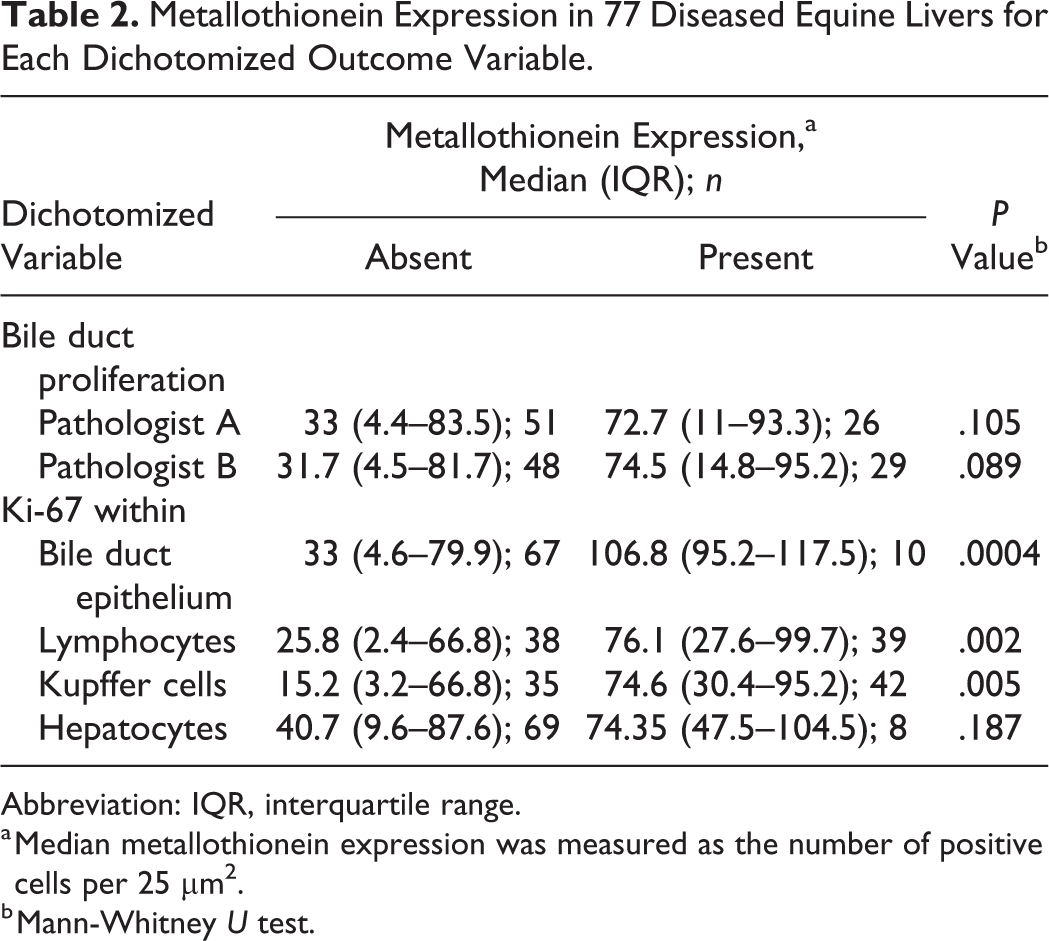
Abbreviation: IQR, interquartile range.
a Median metallothionein expression was measured as the number of positive cells per 25 µm2.
b Mann-Whitney U test.
No significant differences were observed in MT expression between samples with and without Ki-67 expression in hepatocytes, and no association was found between MT expression and inflammation, fibrosis, and bile duct proliferation (Table 3).
Metallothionein Expression in 77 Diseased Equine Livers for Each Categorized Outcome Variable.
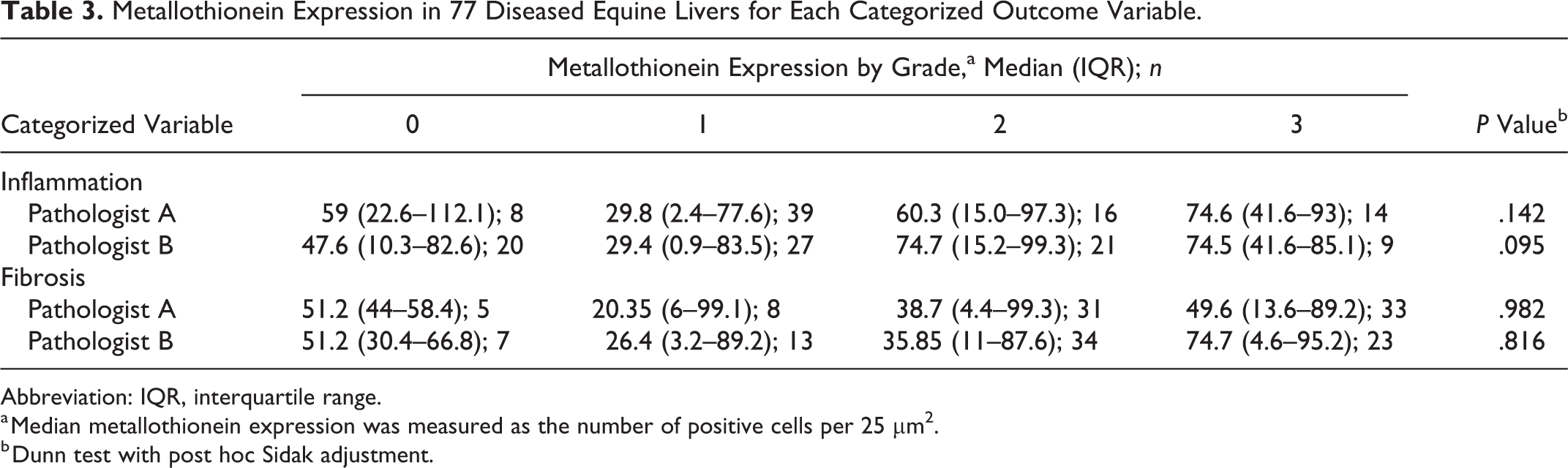
Abbreviation: IQR, interquartile range.
a Median metallothionein expression was measured as the number of positive cells per 25 µm2.
b Dunn test with post hoc Sidak adjustment.
Discussion
The objective of the current study was to examine the relationship between hepatic MT expression and common pathologic findings in the diseased liver, as well as cellular proliferation in different cell types within the liver. We report for the first time the presence of a significant relationship between MT expression within hepatocytes and the presence of lymphocytic inflammation or Ki-67 immunostaining within bile duct epithelium and Kupffer cells.
In our study, hepatic MT expression was increased when Ki-67 was present within lymphocytes. Note that the directionality of this relationship cannot be determined by our methods nor whether a third factor (eg, hepatocellular damage) may be driving MT expression and lymphocytic Ki-67 expression. MT is known to play a role in mediating inflammation, 23 and its gene expression can be induced by cytokines, glucocorticoids, and oxidative stress. 34 Although primarily localized intracellularly within many cell types, MT has been found extracellularly within biological fluids, including serum, urine, and milk. 4,32,36,47,51 Interestingly, extracellular MT was shown to be involved in leukocyte chemotaxis, proliferation, and activation. For example, Yin et al reported that inflammatory cells migrate toward an increasing MT gradient, a response that can be specifically blocked by antibodies targeting MT. 55 Additionally, MT was shown to induce the proliferation of B cells and enhance their capacity to differentiate into plasma cells. 8,31
A significant positive relationship was found between the presence of Ki-67 immunoreactivity within sinusoidal Kupffer cells and increased MT within hepatocytes; however, the directionality of this relationship could not be determined by our methods. The expression of Ki-67 within Kupffer cells suggests that these resident macrophages are actively engaged in the cell cycle. Intrasinusoidal Kupffer cells play a major role in the first line of defense against toxic and infectious agents within the liver, acute and chronic, and activation of these cells causes a release of cytokines and reactive oxygen species. 1,54 Kupffer cells may contribute to hepatocellular injury 24,30 or may be protective against it, 26 as in the case of acetaminophen toxicity. Additionally, acetaminophen alone had only a minimal effect on Kupffer cell chemotaxis and phagocytosis, suggesting that factors released from hepatocytes themselves may modulate Kupffer cell activation. 30 In a thioacetamide-induced liver injury study, pretreatment of rat liver with gadolinium chloride prior to thioacetamide selectively inactivated Kupffer cells, enhanced hepatic MT expression, and decreased the overall hepatotoxic effect of thioacetamide in rats. 3
This study also showed increased MT expression within hepatocytes when Ki-67 immunostaining was present within bile duct epithelium. Although bile duct hyperplasia was not significantly associated with MT expression, an increasing trend was observed (data not shown), and nonsignificance may reflect limited statistical power. MT may play a role in cellular regeneration by regulating the pool of available zinc, which is essential for cell growth and division. 10
Liver regeneration occurs by the proliferation of mature hepatocytes, by ductular reaction, or both. Ductular reaction originates from bipotent hepatic progenitor cells within hepatic portal areas and represents an array of reaction patterns, depending on the types and severity of hepatic injury. 19 Unexpectedly, very little Ki-67 immunostaining was observed in hepatocytes. The immunostaining was repeated 3 times, all with positive staining within our control tissue (equine Peyer patches). In our previous study, where MT was assessed in the diseased canine liver, Ki-67 expression within hepatocytes ranged from 0.4% to 25.2%. 44 It is possible that Ki-67 was absent because these cells reached the resting phase of the cell cycle and were no longer dividing. However, MT staining was observed within the nuclei of hepatocytes, suggesting that hepatocytes were regenerating, as MT immunostaining within the nucleus is associated with hepatocyte proliferation. 10,51,52 Additional studies are needed to examine cellular proliferation in general and the expression of Ki-67 in the equine liver after chronic injury. Note that hepatocyte regeneration may be accomplished in a less conventional way. For example, in a 70% partial hepatectomy (mouse) model, liver regeneration was achieved mainly by cell hypertrophy prior to hyperplasia. Cells entered S phase of the cell cycle but did not always progress to M phase. 33 Kupffer cells may also influence differentiation, but not the proliferation, of hepatic progenitor cells during liver injury in mice. 53
Rhodanine staining for copper was absent in normal liver samples and in 7 of 10 cases of diseased liver, while the remaining cases displayed only sporadic staining. It is therefore possible that copper does not play a role in chronic hepatic disease in horses. There is only a limited amount of information on hepatic copper in horses, although they seem to be relatively resistant to toxicosis. 43 Rare reports do exist of copper causing acute hepatic disease. 5 Although MT gene transcription can be induced by binding of heavy metals, 20 copper does not appear to be driving MT expression in equine liver. Additional studies are needed to elucidate the role of copper in the development and progression of liver disease in horses.
Agreement between pathologists was moderate, with the degree of fibrosis being the most difficult lesion to agree on, despite being provided a rigorous scoring guideline prior to liver section evaluation. Cohen kappa statistic is often used to determine the amount of interrater agreement (0 = no agreement, 1 = perfect agreement). In general, kappa values in this study were consistent with other studies involving scoring systems for chronic liver disease. 21
In conclusion, elevated MT expression within hepatocytes was associated with Ki-67 expression within lymphocytes, Kupffer cells, and bile duct epithelia. Additional studies are needed to further evaluate the role of MT during chronic hepatic disease in horse. These findings are the first known report of MT expression within chronic equine hepatic disease.
Footnotes
Acknowledgements
We kindly acknowledge the invaluable expertise from Melissa Koehnlein and Dale Godson (Prairie Diagnostic Services, Saskatoon) for their assistance with immunohistochemistry protocol development, as well as Larhonda Sobchisin and Ian Shirley for their imaging expertise.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding was kindly provided by the Townsend Equine Health Research Fund and the Western College of Veterinary Medicine Interprovincial Graduate Fellowship (J.N.C.V.).
