Abstract
The chronic form of primary hepatitis occurs commonly in dogs, and the etiology is rarely found. Metallothionein (MT) is a heavy metal–binding protein found in many organs, including the liver. MT was recently shown to enhance liver regeneration and decrease hepatic fibrosis in human beings. This study examined the expression of MT in 24 cases of chronic hepatitis in dogs using immunohistochemistry. To understand the role of MT as a determinant of hepatic inflammation, fibrosis, bile duct proliferation, and regeneration, we correlated its expression with histologic lesions of chronic hepatitis, such as hepatic inflammation, fibrosis, and bile duct proliferation, as well as hepatocellular growth fraction as measured by Ki67 immunolabeling. Hepatocellular growth fraction was used as a measure of hepatic regeneration. Regression analysis revealed a significant positive correlation between MT labeling intensity and growth fraction (r2 = 0.29, P < .05). The percentage of MT-positive cells and the overall MT expression were both positively correlated with growth fraction (r2 = 0.25 and 0.26, respectively; P < .05). A negative correlation was found between the overall MT labeling and fibrosis (r2 = 0.18, P < .05). A similar trend of negative correlation was also found between the percentage of MT-positive cells and fibrosis, but the P value was not statistically significant (r2 = 0.14, P = .0684). These findings suggest a protective role of MT in dogs affected by chronic hepatitis, similar to its role in human beings. These dogs may respond to treatment modules focusing on enhancing the expression of MT.
Canine primary hepatitis comprises a group of liver diseases in which inflammation occurs in response to a disease process within the liver. In contrast, liver inflammation that occurs in response to an extrahepatic disease process is termed nonspecific reactive hepatitis. Nonspecific reactive hepatitis is also used to describe residual inflammation of a previous primary intrahepatic disease. Primary hepatitis is the most common form of liver disease in dogs. 4,17 Regularly encountered forms of this entity include acute hepatitis and chronic hepatitis (CH), with the latter being the most common. A wide variety of causes for hepatopathy in general have been identified. However, despite large efforts, the etiology of primary hepatitis in the majority of cases remains unknown. 4 Our understanding of causes of primary hepatitis in dogs is limited and thus results in limited treatment options with variable outcomes. In recent years, the defense mechanisms involved in the protection against liver injury are receiving greater attention in human beings. In fact, the focus is being shifted from identifying causes of liver injury, which is often very difficult, to understanding defense and disease mechanisms within the liver. 10,14,21 Thus, by enhancing defense mechanisms and targeting disease pathways, a better patient outcome would be achieved regardless of the primary cause.
Metallothionein (MT) is a family of proteins with a high binding capacity to heavy metals, including copper, zinc, and many others. 24 In mammals, 4 major subfamilies of MT exist: MT-I, MT-II, MT-III, and MT-IV. 24 The MT-I and MT-II isoforms are often considered a single species (referred to here as MT) due to their high homology and the inability of primary antibodies to differentiate between the 2 forms. 16 Both forms are ubiquitous in mammalian tissues and increase in the liver after heavy metal exposure, which is thought to be protective against heavy metal–induced liver injury. 2
It has been shown that COMMD1-deficient dogs, which progressively develop copper-induced CH, display a significant increase of MT expression at 12 months of age, coinciding with the massive accumulation of copper despite the absence of liver disease. 5 In recent years, the use of MT-knockout mouse models has provided evidence that MT has anti-inflammatory and antifibrotic properties and promotes hepatocellular regeneration. For example, hepatic MT is increased after bacterial infection, an effect mediated by lipopolysaccharide. 23 In addition, MT-knockout mice are more susceptible to lipopolysaccharide-induced lung inflammation as compared to wild-type mice. 23 In Doberman Pinchers affected by copper-induced hepatitis, reduced mRNA and protein expression of MT have been found in advanced stages of the disease. 22 Several proinflammatory cytokines, including IL-6, TNF-α, and IFN-γ, have been shown to induce the expression of MT. 3 Reactive oxygen species generated during inflammation also increase MT expression through multiple pathways. 19 The increased expression of MT is a defense mechanism by which the liver mediates anti-inflammatory effects and protects against heavy metal exposure. 9,11
Very little is known about hepatic defense mechanisms in dogs; therefore, this study examined the expression of MT in dogs affected by CH and correlated its expression with liver regeneration, inflammation, fibrosis, and bile duct proliferation (BDP).
Materials and Methods
Tissue Selection and Histologic Scoring
The pathology archives at Prairie Diagnostic Services Inc, Saskatoon, Saskatchewan, Canada, were searched for histologic liver samples diagnosed with canine CH of unknown etiology in which toxicologic analysis of hepatic metals was performed. Twenty-four cases were selected for which at least 1 cm2 of tissue was available for evaluation. Cases were included when the histologic findings were consistent with a diagnosis of CH according to criteria developed by the World Small Animal Veterinary Association Liver Standardization Group. Hepatic analysis of metal levels was performed using inductively coupled plasma spectrometer as previously described. 27 Normal liver copper level is <400 parts per million, and normal liver iron level is 400–1200 parts per million (dry weight basis). 7,18 Cases with elevated hepatic metals, such as copper and iron, were excluded. Also, dog breeds known to accumulate copper for genetic or familial reasons such as Bedlington Terriers, Dalmatians, Doberman Pinschers, Labrador Retrievers, Skye Terriers, and West Highland White Terriers were excluded from this study. 6,8,10,12,25,26 In each section, hepatic inflammation, fibrosis, and BDP were evaluated and scored. Hepatic inflammation was subjectively scored from 0 to 3 based on the percentage area of liver affected on hematoxylin and eosin sections. A score of 0 indicated the absence of inflammation; 1, the presence of mild inflammation (<10% of hepatic parenchyma affected); 2, the presence of moderate inflammation (10%–50% of hepatic parenchyma affected); and 3, severe inflammation (>50% of hepatic parenchyma affected). Hepatic fibrosis was highlighted utilizing Masson’s trichrome–stained sections and was scored from 0 to 3. A score of 0 indicated the absence of fibrosis; 1, the presence of fibrosis around portal areas only; 2, the presence of nonbridging fibrosis extending beyond portal areas to the hepatic parenchyma or other portal or central areas without any 2 areas being connected; and 3, the presence of bridging fibrosis. BDP was assessed by averaging the number of bile ducts in 5 random high-power fields (400×). Each liver section was scanned randomly at high power to locate a bile duct. Once found, the bile duct was placed at the center of the field, and all bile ducts in that field were counted. Each of the 3 histologic scores was assessed by 2 pathologists independently (A.N.A. and A.L.A.) and a mean calculated. In addition, 3 liver biopsies that were diagnosed as histologically normal were selected per the availability of tissue and the absence of histologic lesions of inflammation, fibrosis, and BDP.
Immunohistochemistry for MT and Ki-67
Immnuohistochemistry for MT and Ki-67 was performed as described by Shimada et al 20 and Al-Dissi et al, 1 respectively, with some modifications. Consecutive 5-µm sections from formalin-fixed, paraffin-embedded liver tissue blocks were cut, mounted, and dried on glass slides. Tissue sections were deparaffinized through a series of xylene baths, rehydrated in graded alcohol (100%, 95%, and 70% ethanol), and rinsed in distilled water. Heat-induced antigen retrieval was performed by microwaving tissue sections using 0.01M sodium citrate buffer (pH of 6.8) for 23 minutes, followed by cooling for 40 to 50 minutes at room temperature. This was followed by washing in 0.05% Tween-20 solution diluted in phosphate-buffered solution (PBST). The sections were then subjected to 3% hydrogen peroxide in absolute methanol for 10 minutes to block the endogenous peroxidase activity. Nonspecific protein binding was saturated using 2.5% normal horse serum in phosphate-buffered saline for 20 minutes (Vector Laboratories). The sections were incubated overnight at 4°C with either mouse monoclonal anti-canine MT IgG1 (1:500 in 0.05% PBST, no. ab12228, Abcam) or monoclonal mouse anti-human Ki-67 antigen (ready to use, Dako). After primary antibody incubation, the sections were washed 3 times (5 minutes each) in 0.05% PBST and then incubated with ImmPRESS reagent micropolymer containing anti-mouse Ig (Vector Laboratories) for 30 minutes. Color development was performed utilizing diaminobenzidine (DAB)–substrate buffer and DAB–chromogen per the manufacturer’s instructions (ImmPACT DAB Peroxidase Substrate, Vector Laboratories). To stop the DAB activity, the slides were rinsed with deionized water. Finally, the slides were counterstained using Mayer’s hematoxylin for 1 minute, rinsed with tap water, and mounted with coverslips using Permount mounting medium. Exclusion of primary antibodies in a few known positive sections was carried out to assess the specificity of the primary antibodies and revealed no staining. Three sections from normal canine livers were also stained for MT and Ki-67.
Scoring of Hepatocellular MT Expression and Cell Proliferation
In each liver tissue section, the percentage of MT-positive cells and the intensity of brown labeling were assessed by a single evaluator (S.S.). The percentage of MT-positive cells was calculated in 5 random high-power fields (400×) by counting the number of positive hepatocytes against the total number of cells in each field; a mean of the 5 fields was then calculated. Labeling intensity was subjectively scored from 0 to 3 in 5 random high-power fields and a mean calculated. A score of 0 indicated the absence of labeling; 1, the presence of low labeling intensity; 2, the presence of moderate labeling intensity; and 3, the presence of high labeling intensity. An overall MT score was calculated by adding the percentage of positive cells and the MT labeling intensity.
Hepatocellular growth fraction (GF) was assessed (by S.S.) in Ki-67 immunolabeled sections in 12 random high-power fields (400×). In each field, the percentage of cells with positive nuclear labeling was calculated by counting the number of positive cells against the total number of cells. A mean of the 12 fields was then calculated. Tissue from a normal canine lymph node was used a positive control for Ki-67 (Figs. 1–6).
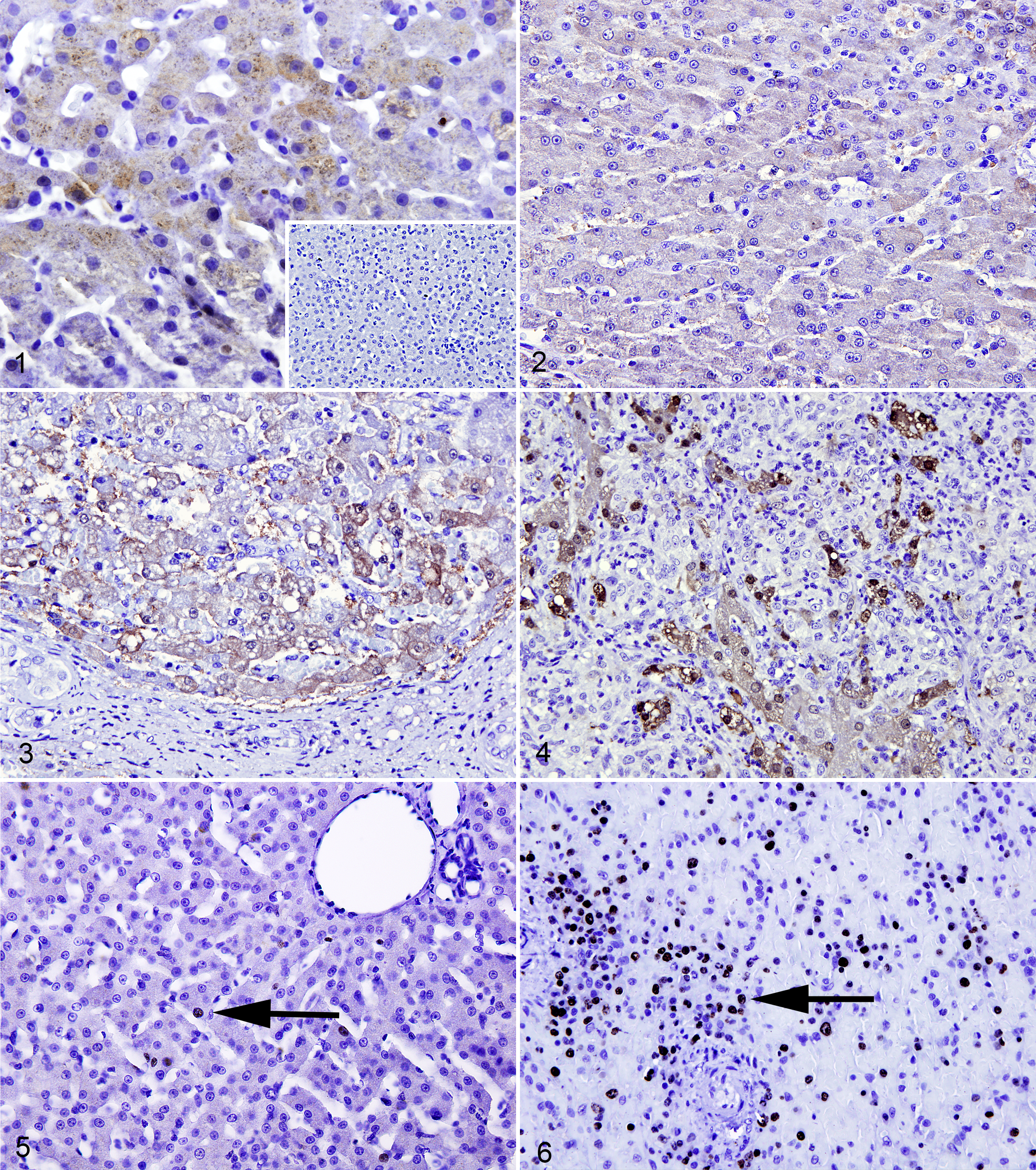
Liver, dog. Hepatitis is scored from 0 to 3. Immunohistochemistry for metallothionein (MT).
Statistical Analysis
Statistical analysis was performed using the SAS 9.3 (SAS Institutes). Regression analysis was performed after square root or logarithmic transformation of data, as appropriate, to evaluate the role of MT expression as a determinant of hepatocellular GF, fibrosis, inflammation, and BDP scores. P < .05 was considered significant.
Results
Interobserver variation was assessed for the scores of the 2 independent pathologists using Spearman rank correlation coefficient, which was statistically significant for each evaluated parameter (r = 0.46, 0.61, and 0.70 for inflammation, fibrosis, and BDP, respectively). Of the 24 cases of CH, 18 (75%) displayed lymphoplasmacytic inflammation, while 6 (25%) displayed neutrophilic inflammation. Hepatic copper and iron concentrations were variable but within the established reference interval. 7,18 The expression of MT within hepatocytes was mainly cytoplasmic and rarely nuclear and was found in all cases of CH. The expression was randomly distributed in all hepatic zones without an apparent zonal predilection. The percentage of MT-positive cells ranged from 1.7 to 44.6, and the intensity of MT labeling ranged from 0.6 to 2.8. Hepatocellular GF ranged from 0.4% to 25.2%. The results for the parameters evaluated are shown in Table 1.
Liver Biopsies of Dogs Affected With Chronic Hepatitis: Inflammation, Fibrosis, Bile Duct Proliferation, Metallothionein Scores, and Growth Fraction (Ki-67).
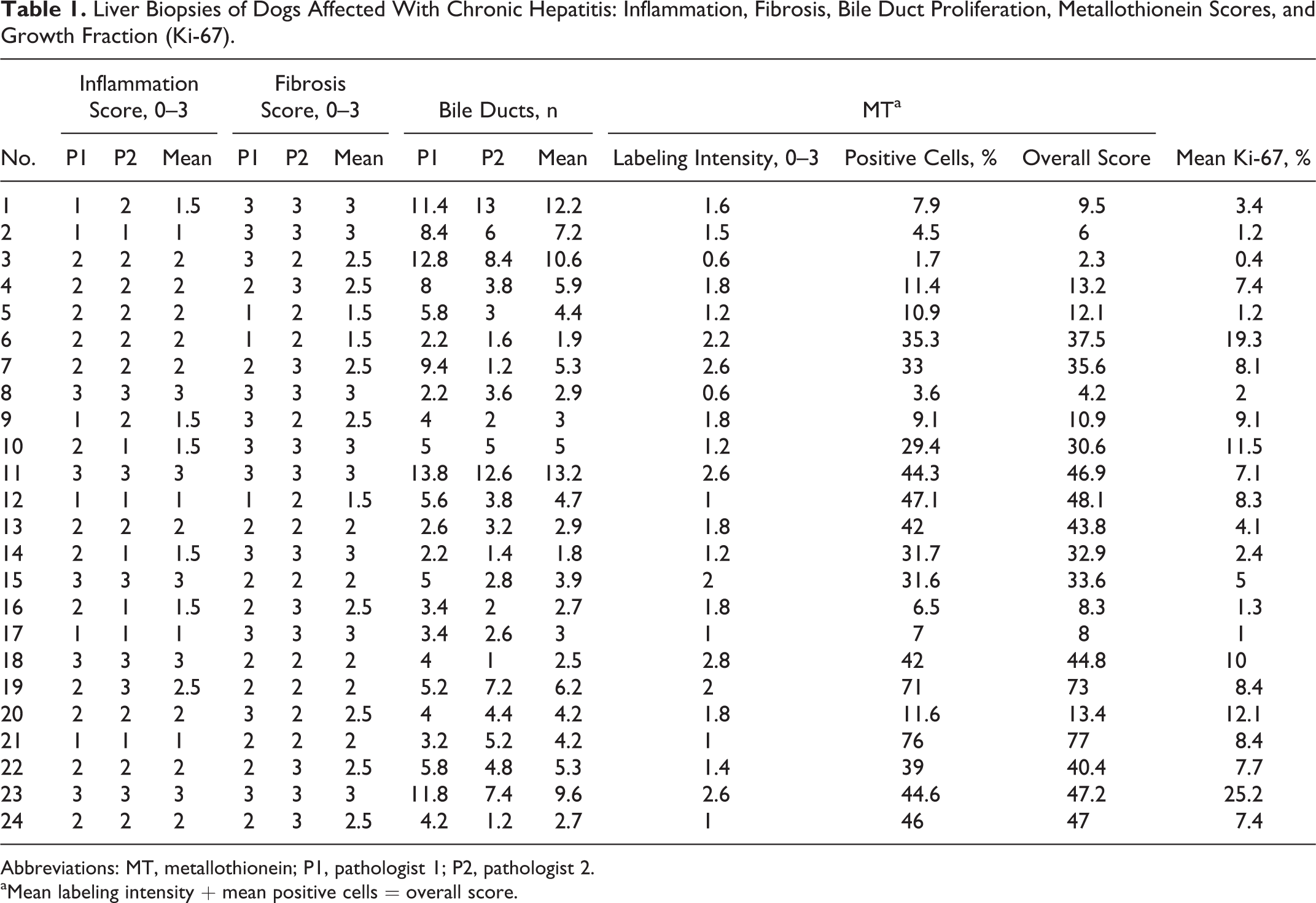
Abbreviations: MT, metallothionein; P1, pathologist 1; P2, pathologist 2.
aMean labeling intensity + mean positive cells = overall score.
Regression analysis revealed that MT labeling intensity was positively correlated with both GF and inflammation (r 2 = 0.29 and 0.25, respectively; P < .05). The percentage of MT-positive cells and the overall MT expression were both positively correlated with GF (r 2 = 0.25 and 0.26, respectively; P < .05). A negative correlation was found between the overall MT labelling and fibrosis (r 2 = 0.18, P < .05). A similar trend of negative correlation was also found between the percentage of MT-positive cells and fibrosis, but the P value was not statistically significant (r 2 = 0.14, P = .0684). No correlation was found between MT expression and BDP. For detailed results, including r 2 values, see Table 2.
Regression Analysis Results for the Role of MT Expression as a Determinant of Histology Scores and Growth Fraction.
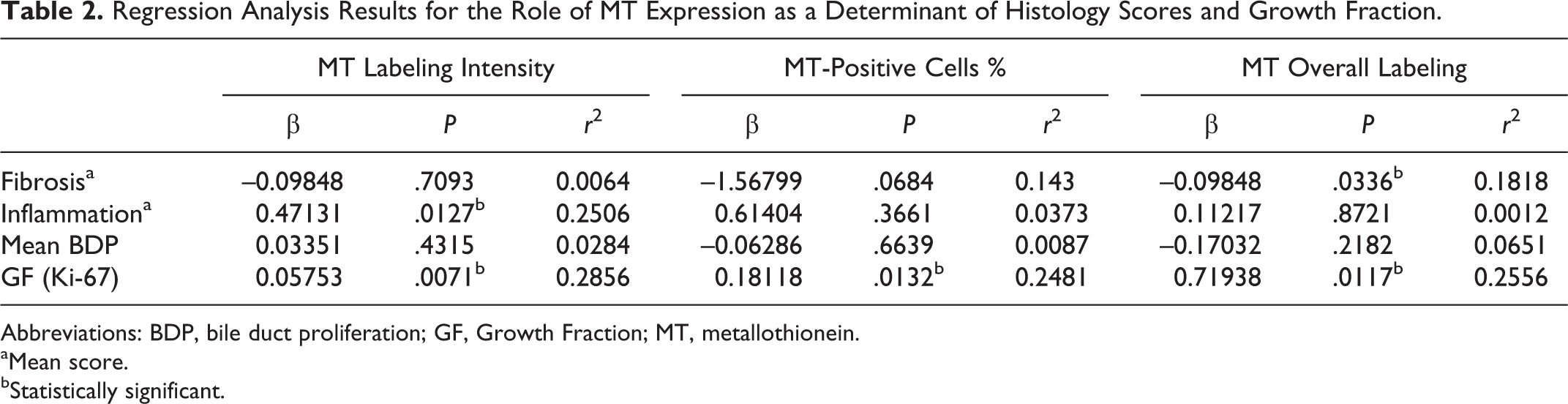
Abbreviations: BDP, bile duct proliferation; GF, Growth Fraction; MT, metallothionein.
aMean score.
bStatistically significant.
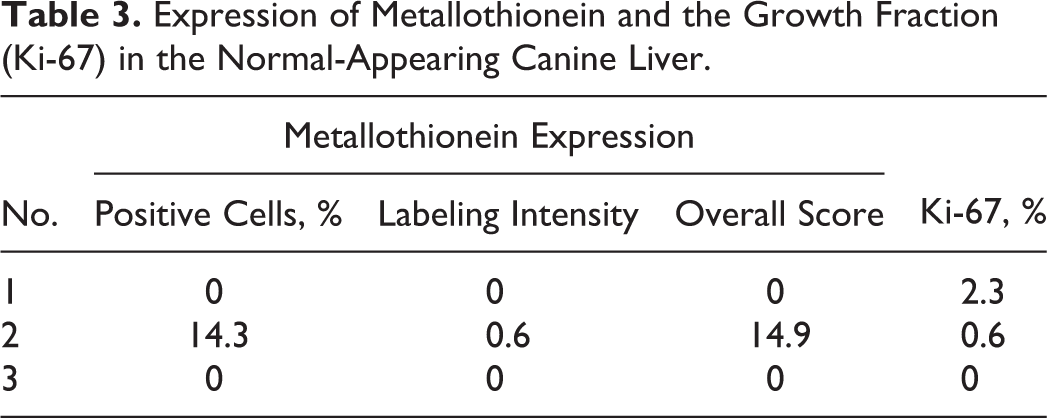
In normal liver sections (n = 3), the expression of MT was absent in 2 of 3 sections. The expression of MT in the remaining section was cytoplasmic with weak labeling intensity. The details of MT expression and GF in normal liver samples are shown in Table 3.
Expression of Metallothionein and the Growth Fraction (Ki-67) in the Normal-Appearing Canine Liver.
Discussion
This study examined the expression of MT in 24 cases of CH in dogs using immunohistochemistry. Immunohistochemical localization of MT in physiologically normal dogs in various organs was studied by Shimida et al 20 and showed tissue specific variations in MT expression. However, the expression pattern of MT in liver during CH is not known. This is the first study that examined the expression of MT in canine CH and the role of MT as a determinant of histologic lesions of CH, such as hepatic inflammation, fibrosis, BDP, and regeneration. The lesions of hepatic fibrosis and BDP are often present in CH but can be found in other types of liver diseases.
CH is the most common form of primary hepatitis in dogs, representing 66% of all forms in 1 study. 4 The final outcome of CH depends on our ability to control the deterioration of liver function and is often guarded, as the exact etiology of CH is rarely identified. A new approach to therapy for chronic liver diseases in humans focuses on enhancing hepatic defense mechanisms.
MT is a protein with a high binding capacity to heavy metals and is thought to be protective against liver injury. Two studies explored the potential role of MT in liver regeneration in wild-type and MT-knockout mice utilizing 2 models: (1) surgical resection of 35% of the liver and (2) acute liver injury induced by the intraperitoneal injection of the hepatotoxin (ie, thioacetamide). 14,15 In wild-type mice, both models resulted in increased expression of MT synthesis in the liver within 12 hours. Furthermore, hepatocyte proliferation index was 40% to 55% higher than sham-operated controls after partial hepatectomy or after treatment with thioacetamide. In MT-knockout mice, hepatic proliferation index was low (approximately 18% to 25%) after partial hepatectomy and chemical injury compared to control mice. These 2 studies demonstrate that in the absence of MT, hepatic cell proliferation is low, thereby suggesting an essential role for MT in cell proliferation and liver regeneration. In our study, the percentage of MT-positive cells, MT labeling intensity, and overall MT expression were all positively correlated with GF (also known as proliferation index), which indicates that MT expression enhances liver regeneration in dogs affected by CH.
Studies with MT gene therapy provide strong evidence for the protective role of MT against hepatic fibrosis. 10 Wild-type mice treated with carbon tetrachloride for 4 weeks developed a reversible liver fibrosis associated with increased expression of MT, whereas MT-knockout mice developed irreversible fibrosis after similar treatment. A similar but longer treatment (8 weeks) of wild-type mice with carbon tetrachloride resulted in irreversible fibrosis and low hepatic levels of MT. Interestingly, the fibrosis was reversed when these mice were treated intravenously with adenoviral delivery of the human MT gene. The hepatic MT elevation in these mice was associated with an increase in activities of collagenases in the liver. Consistent with the above findings, we found that the percentage of MT-positive cells and the overall MT expression were both negatively correlated with fibrosis, which supports a protective role of MT against fibrosis in dogs affected by CH. It would be interesting to examine the correlation between MT expression and collagenases within the liver in dogs with CH in future studies.
In people, the presence of severe hepatic fibrosis is often associated with a guarded prognosis. 2 Given the negative correlation between MT and fibrosis in this study, it would be reasonable to think that MT may have prognostic value in dogs with CH. In support of this, it has been shown that high hepatic MT expression in people with hepatitis C virus contributes to their improved viral clearance and favorable prognosis. 13
The expression of MT in the normal canine liver was very weak, patchy, and lower than its level in CH, although the expression in both tissues was not compared statistically. Although no cases of acute hepatitis were included in this study, it is still plausible that MT expression would be elevated. This is supported by the findings of elevated MT expression shortly after partial hepatectomy or the intraperitoneal injection of thioacetamide in mice. 14,15 In our study, MT labeling intensity, but not the percentage of MT-positive cells, was found to be positively correlated with inflammation. It is thus possible that the increased intensity of MT expression is related to the presence of active inflammation close to MT-positive areas.
No correlation was found between MT expression and BDP, which suggests that BDP is not related to MT expression. The antibody used to detect MT in this study cross-reacts with MT-I and MT-II, both of which are induced by metal ions, glucocorticoids, inflammatory cytokines, and oxidative stress. Our finding of a significant correlation between MT expression and inflammation supports previous studies implicating inflammation as an inducer of MT expression. It would be interesting to examine the role of these 2 proteins separately during liver inflammation in dogs, which would shed some light on their differential expression and function. The absence of primary antibodies that differentiate the 2 isoforms currently precludes this examination.
Although significant, the level of correlation between MT and both regeneration and fibrosis in this study was relatively weak (r 2 < 0.3). This could be due to the small sample size or to the fact that other proteins are important in controlling regeneration and fibrosis. The absence of correlation between MT and BDP suggests that MT has no effect on BDP.
In summary, this study found positive correlation between hepatic MT expression and hepatocellular GF and negative correlation between MT expression and hepatic fibrosis. Therefore, dogs affected by CH may respond to therapies focusing on enhancing the expression of MT.
Footnotes
Acknowledgement
We thank Dr C. Feng for statistical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by a grant from the Western College of Veterinary Medicine Companion Animal Health Fund, Saskatoon, Saskatchewan, Canada. Dr Sridharan was funded by an Interprovincial Graduate Fellowship from the Western College of Veterinary Medicine.
