Abstract
Oligodendroglioma is a common brain tumor in dogs, particularly brachycephalic breeds. Oligodendrocyte precursor cells (OPCs) are suspected to be a possible origin of oligodendroglioma, although it has not been well elucidated. In the present study, 27 cases of canine brain oligodendrogliomas were histologically and immunohistochemically examined. The most commonly affected breed was the French Bulldog (n = 19 of 27, 70%). Seizure was the most predominant clinical sign (n = 17 of 25, 68%). The tumors were located mainly in the cerebrum, particularly in the frontal lobe (n = 10 of 27, 37%). All cases were diagnosed as anaplastic oligodendroglioma (AO) and had common histologic features characterized by the proliferation of round to polygonal cells with pronounced atypia and conspicuous mitotic activity (average, 10.7 mitoses per 10 high-power fields). Honeycomb pattern (n = 5 of 27, 19%), myxoid matrix (n = 10, 37%), cyst formation (n = 6, 22%), necrosis (n = 19, 70%), pseudopalisading (n = 5, 18.5%), glomeruloid vessels (n = 16, 59%), and microcalcification (n = 5, 19%) were other histopathologic features of the present tumors. Immunohistochemically, the tumor cells were positive for Olig2 in all cases and for other markers of OPCs in most cases, including SOX10 (n = 24 of 27, 89%), platelet-derived growth factor receptor α (n = 24, 89%), and NG2 (n = 23, 85%). The present AO also consisted of heterogeneous cell populations that were positive for nestin (n = 13 of 27, 48%), glial fibrillary acidic protein (n = 5, 19%), doublecortin (n = 22, 82%), and βIII-tubulin (n = 15, 56%). Moreover, cultured AO cells obtained from 1 case retained expression of OPC markers and exhibited multipotent characteristics in a serum culture condition. Overall, the findings suggest that transformed multipotent OPCs may be a potential origin of canine AO.
Oligodendroglioma is a common glial tumor, accounting for 20% of all intracranial neoplasms in dogs. 43 Brachycephalic breeds, including Boxer, Bulldog, and Boston Terrier, are predisposed. 24,25,42,43 Oligodendrogliomas are commonly located in the gray and white matters of the cerebral hemisphere, especially in the frontal and piriform lobes, and in the vicinity of the lateral ventricles. 43,53 Histopathologically, the tumors are composed of sheets of round to polygonal cells with a hyperchromatic nucleus and clear to lightly stained cytoplasm with a perinuclear halo. Higher-grade tumors are highly cellular and have nuclear atypia, increased mitotic activity, microvascular proliferation, and intratumoral necrosis. 23,24 Oligodendrogliomas are defined as being composed of neoplastic oligodendrocytes in the World Health Organization (WHO) histologic classification of domestic animal tumors. 23 Such definition implies that these tumors are derived from mature oligodendrocytes. Nevertheless, markers of mature oligodendrocytes, such as myelin basic protein or myelin-associated glycoprotein, are not detected in canine oligodendroglioma cells. 47 In addition, expression of neuronal markers, such as doublecortin (DCX), βIII-tubulin, and synaptophysin, have been reported in a subset of oligodendroglial tumors. 21,37 The alternative possibility is that oligodendrogliomas arise from more immature cells, which have the potential to differentiate into glial and neuronal cells. 14,19,21 However, the cellular origin and the histogenesis of canine oligodendroglioma remain poorly understood.
According to studies of humans, there is evidence to support oligodendrocyte precursor cells (OPCs) as the origin of oligodendroglioma. 1,4,10,27,34 –36,40 OPCs, the major dividing cell population in the adult brain, become myelin-forming oligodendrocytes and are widely dispersed throughout the subventricular zone, white matter, and gray matter across the major regions of the brain, including telencephalon, cerebellum, and brainstem. 12 When myelin repair is needed after a central nervous system injury, OPCs migrate into the lesion, proliferate, differentiate into mature oligodendrocytes, and remyelinate axons. 15,32,41 OPCs can be identified by their expression of transcription factors, such as Olig2 and SOX10, platelet-derived growth factor receptor α (PDGFRα), proteoglycan NG2, and ganglioside A2B5. 2,12,49,51 OPCs are normally restricted to the oligodendrocyte lineage; however, multipotential differentiation can occur under certain cultured conditions. 6,26 OPCs differentiate to neurons under serum-free conditions with growth factors (basic fibroblast growth factor), while their differentiation can be directed toward astrocyte-lineage cells under serum-rich culture conditions. 26
To investigate the role of OPCs in canine oligodendroglioma, we characterized the expression of cell lineage markers in spontaneous tumor cases and cultured cells of canine anaplastic oligodendroglioma. To examine candidate molecules that determine putative glial or neuronal lineage specificity, selected cell differentiation markers were examined (Fig. 1): Olig2, SOX10, PDGFRα, proteoglycan NG2, nestin, glial fibrillary acidic protein (GFAP), βIII-tubulin, DCX, and neurofilament (NF). Olig2 and SOX10 are transcription factors that are sufficient to drive multipotential neural stem cells toward oligodendrocyte fate and are widely expressed in oligodendrocyte lineage. 16,33,38 PDGFRα expressed in OPCs is a tyrosine-protein kinase that acts as a cell surface receptor for platelet-derived growth factor AA, which plays an essential role in their survival and proliferation. 16,33 NG2 is an integral membrane chondroitin sulphate proteoglycan expressed on OPCs in the central nervous system. 16,33 As OPCs undergo terminal differentiation into mature oligodendrocytes, they lose the expression of NG2 and PDGFRα. 16,33 GFAP is a main intermediate filament protein of mature astrocytes, whereas nestin is a main intermediate filament protein observed in many immature cell types and has been extensively used as a neuron stem cell marker. 13,21,44 The antibody for βIII-tubulin has been used to identify the early phase of neuronal differentiation in human embryonic development. 21,48 DCX plays a crucial role in neuroblast migration and is highly expressed in migrating neural precursor cells in the subventricular zone and rostral migratory stream during central nervous system development. 21,48 Neurofilament is a distinct marker for mature neurons. 21 The aim of the present study was to characterize the profile of these markers in canine oligodendroglioma, to develop new information regarding its histogenesis.
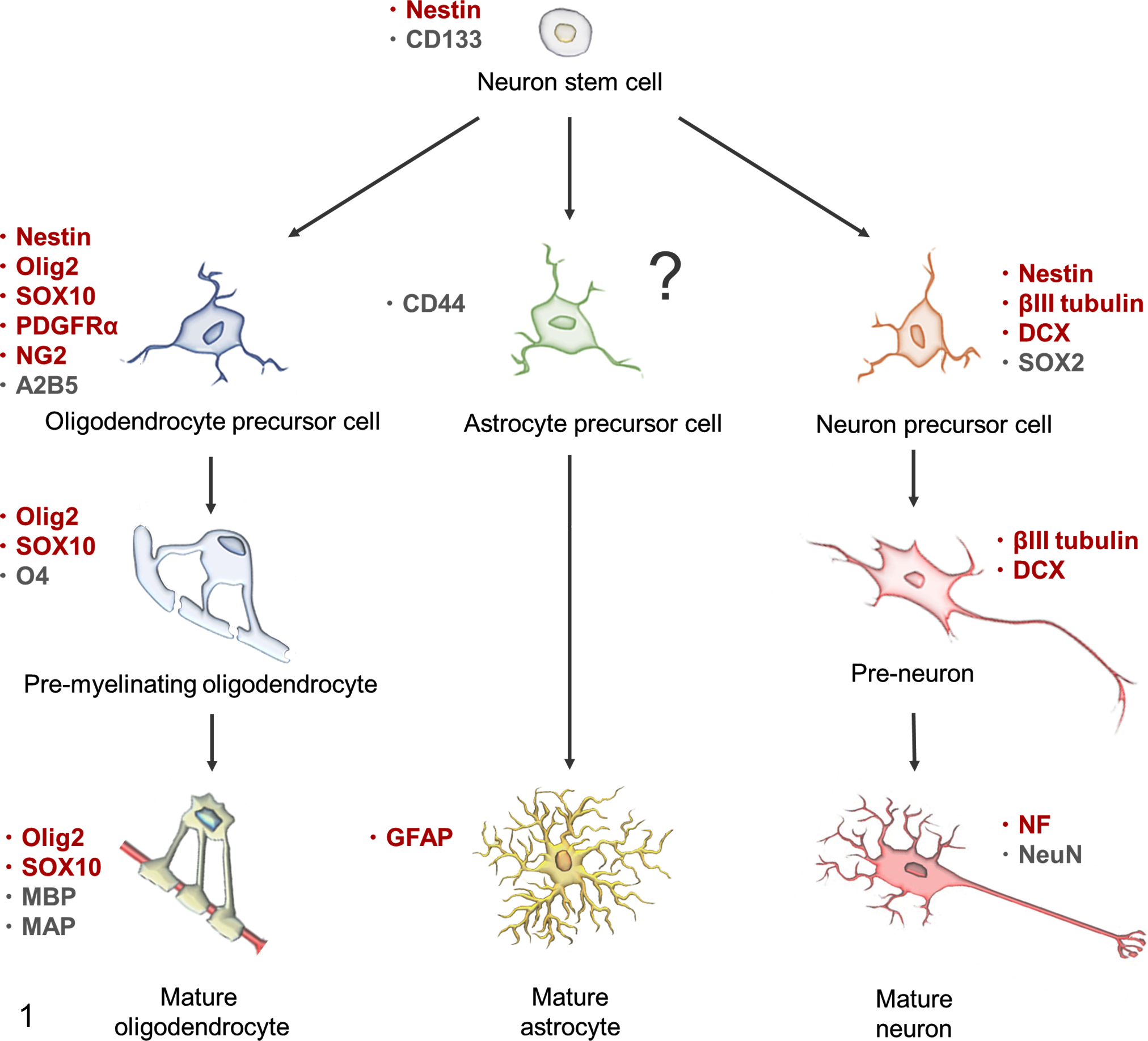
Markers expressed during neural and glial lineage development. Markers of the early astrocytic lineage (astrocyte precursor cell and preastrocyte) remain elusive. 5,13,16,33,48 The markers used in the present study are shown in red. DCX, doublecortin; GFAP, glial fibrillary acidic protein; MAP, myelin-associated protein; MBP, myelin basic protein; NF, neurofilament; PDGFRα, platelet-derived growth factor receptor α.
Materials and Methods
Samples and Histology
Twenty-seven canine brain oligodendrogliomas (7 necropsy and 20 biopsy cases) were reexamined from the archive of the Department of Veterinary Pathology, Graduate School of Agricultural and Life Sciences, University of Tokyo. This represented all cases of brain oligodendroglioma within the period examined (2013–2017). Specimens were routinely fixed in a 10% phosphate-buffered formalin solution, embedded in paraffin, sectioned at 4 μm, and stained with hematoxylin and eosin. Histologic classification of the brain tumors was determined according to the criteria defined by the animal and human classification. 23,29 The grading of the tumor was based on prominent cellular atypia, high cellularity, and brisk mitoses. Other features, including prominent microvascular proliferation and necrosis, were used as aids in diagnosis.
Immunohistochemistry
Immunohistochemistry was performed as summarized in Table 1. The positive control used for these markers was nontumorous normal canine brain tissue, including subventricular zone and gray and white matter. In all experiments, negative controls were performed by omitting the primary antibody. Deparaffinized tissue sections were treated with 10% hydrogen peroxide in methanol at room temperature for 5 minutes and incubated in 8% skim milk–Tris-buffered saline at 37°C for 30 minutes to prevent nonspecific reactions. The sections were incubated at 4°C overnight with primary antibodies. Envision horseradish peroxidase–labeled polymer anti-mouse or anti-rabbit IgG (Dako) was then applied at 37°C for 40 minutes. For the detection of DCX, tissue sections were reacted with an anti-goat immunoglobulin G (1:400; KPL) at 37°C for 40 minutes and with horseradish peroxidase-conjugated streptavidin (1:300; Dako) at room temperature for 40 minutes. Finally, the reactions were visualized with 0.05% 3-3′-diaminobenzidine and 0.03% hydrogen peroxide in Tris–hydrochloric acid buffer, followed by a counterstain with Mayer’s hematoxylin. Immunopositive tumor cells were counted in 10 randomly selected areas (high-power field, 400×). Semiquantitative scores included 4 categories: −, no positive cells; +, 1%–30% positive cells; ++, 31%–60% positive cells; +++, >60% positive cells.
Primary Antibodies Used for Immunohistochemistry in 27 Canine Oligodendrogliomas.
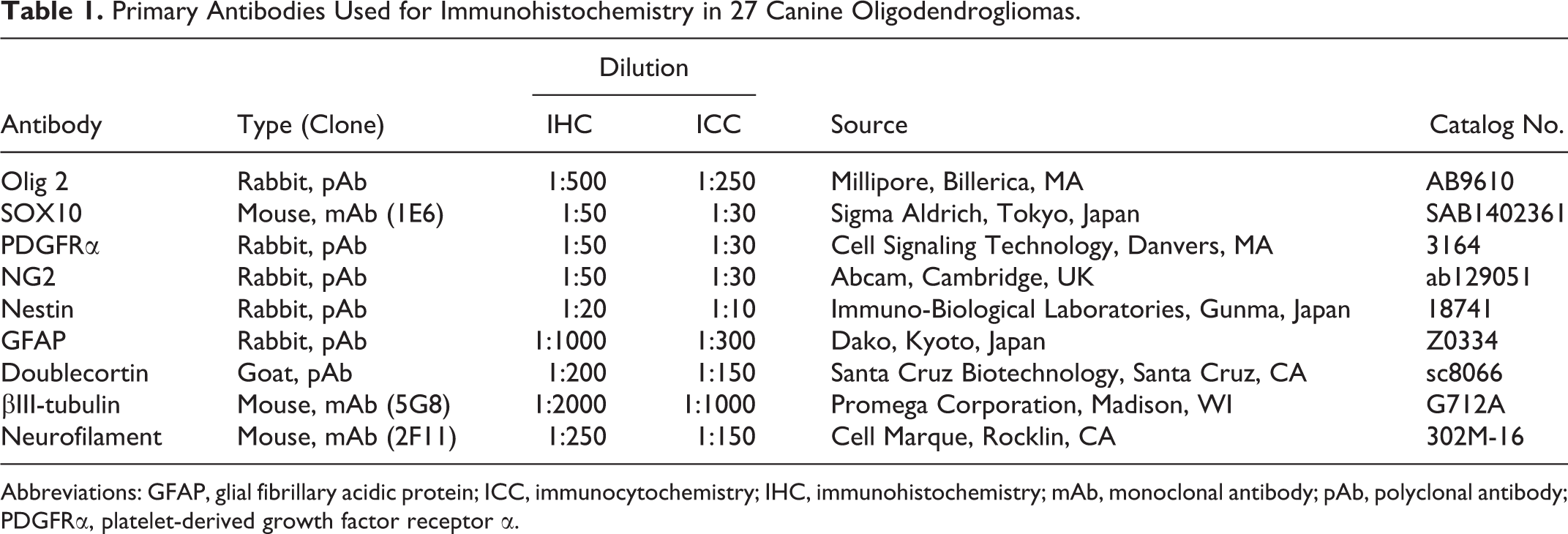
Abbreviations: GFAP, glial fibrillary acidic protein; ICC, immunocytochemistry; IHC, immunohistochemistry; mAb, monoclonal antibody; pAb, polyclonal antibody; PDGFRα, platelet-derived growth factor receptor α.
Tissue Culture of Anaplastic Oligodendroglioma
The primary tumor cells were isolated from a 12-year-old neutered male French Bulldog (case No. 10). By the owner’s approval, the dog was euthanatized and necropsy immediately performed. At necropsy, asymmetric enlargement of the left piriform lobe was observed. Frontal slices through the brain at necropsy showed an ill-defined mass occupying the left brain base. The tumor included large cystic, hemorrhagic, and necrotic areas in the center of the mass and a shift of the midline structure toward the right hemisphere. Histologic examinations confirmed the diagnosis of anaplastic oligodendroglioma. For cell culture, the fresh tumor samples were cut into small pieces and washed twice with sterile phosphate-buffered saline (PBS; pH 7.4) containing 1% penicillin-streptomycin. The samples were then minced in Dulbecco’s modified Eagle medium (DMEM; Gibco, Invitrogen) containing 0.25% crude trypsin (BD Difco) and incubated at 4°C for 18 hours. The cell suspension was then collected by centrifugation, washed with culture medium consisting DMEM supplemented with 10% heat-inactivated fetal bovine serum (FBS; GE Healthcare Life Sciences) and 1% antibiotics (penicillin streptomycin; Wako Pure Chemical), and filtered through a cell strainer (70-μm nylon; Falcon). The suspended cells were seeded in cultured medium and subsequently incubated at 37°C in a humidified incubator at 5% CO2. Three months after the initial cultivation, tumor cells were cloned by a serial dilution method. After cells had reached 70% to 80% confluency, cloned cells were removed and seeded into 60-mm petri dishes with 10% FBS in DMEM. The culture medium was then changed twice a week and cell passages performed every 3 to 5 days. The cells were routinely frozen at various passage levels in DMEM containing 10% FBS and 10% dimethylsulfoxide for later use.
For a morphologic study, the cells were grown on 2-well chamber slides (Nunc Lab-Tek II CC2 Chamber Slide System; Thermo Scientific) and incubated in a humidified atmosphere of 5% CO2 and 95% air at 37°C. At about 80% confluence, the cells were washed with PBS, fixed in methanol, and then stained with Giemsa solution (Merck Millipore).
Cell Growth and Cytogenetic Analysis (Karyotypic Analysis)
The anaplastic oligodendroglioma (AO) cells were plated at 1 × 104 cells in 24-well multiplates and maintained in the growth medium at 37°C for 14 days. The number of cells in each well was counted at 1, 2, 4, 6, 8, 10, 12, and 14 days after the initiation of cell culture. The growth curve was made, and the cell population doubling time was calculated with the following equation: doubling time (hours) = T × log(2) / log(Ne/Nb), where T is duration (hours) of culture, Nb is the cell number at the beginning of the incubation time, and Ne is the cell number at the end of the incubation time.
Briefly, for cytogenetic analysis, confluent monolayer cells were treated with 0.02 µg/ml of colcemid (KaryoMax; Invitrogen) for 2 hours at 37°C and then dispersed with 0.5% trypsin and 0.02% EDTA. After having been washed and treated with a prewarmed hypotonic solution of 0.075M potassium chloride (Wako), the cells were then fixed in a methanol/acetic acid (1:1) solution. The cell suspension was dropped on a glass slide and subsequently stained with Giemsa solution. Two hundred metaphases were counted with an oil immersion objective lens (1000×) and light microscope to estimate the modal chromosome numbers.
Fluorescence Immunocytochemistry
The primary cultured cells were seeded and grown on chamber slides 24 hours prior to the experiments. After fixation with 4% paraformaldehyde, the cell membrane was permeabilized by incubating the cells with 0.25% Triton X-100 for the detection of intracellular markers. After being rinsed with PBS, the cells were treated with 1% bovine serum albumin in PBS–Tween 20 to block nonspecific reactions. Primary antibodies (Table 1) were applied at 4°C overnight. Cells were further incubated with appropriate fluorochrome-labeled secondary antibodies, including fluorescein horse anti-mouse immunoglobulin G (IgG), fluorescein goat anti-rabbit IgG, or fluorescein streptavidin (Vector Laboratory), at room temperature for 1 hour and with DAPI (Vectashield; Vector Laboratory) for nuclear labeling. Negative controls were performed by omitting the primary antibodies. Immunofluorescence was visualized under a Zeiss LSM 700 laser scanning confocal microscope (Carl Zeiss Meditec).
Results
Occurrence of Oligodendrogliomas
Twenty-seven dogs were examined in this study. The signalment, major neurologic signs, and tumor locations of the cases are listed in Table 2. Of the 27 cases, oligodendroglioma occurred in 14 males (neutered = 57.1%, intact = 42.9%) and 13 females (neutered = 53.8%, intact = 46.2%) with a mean age of 7.8 years (range, 3–12 years). Breeds were French Bulldogs (n = 19, 70.4%), Miniature Dachshunds (n = 2, 7.4%), Boston Terriers (n = 2, 7.4%), Bulldog (n = 1, 3.7%), Yorkshire Terrier (n = 1, 3.7%), German Shepherd (n = 1, 3.7%), and Labrador Retriever (n = 1, 3.7%). Various neurologic signs were recorded in 25 cases, which included seizure (n = 17, 68.0%), gait abnormality (n = 9, 33%), altered level of consciousness (n = 6, 24%), circling (n = 5, 20.0%), vision abnormality (n = 4, 16.0%), tremor (n = 3, 12.0%), torticollis (n = 2, 8.0%), hypersalivation (n = 2, 8.0%), behavioral change (n = 1, 4.0%), olfactory disorder (n = 1, 4.0%), tachypnea (n = 1, 4.0%), and paralysis (n = 1, 4.0%). The frontal lobe was the most affected area (n = 10 of 27, 37.0%), followed by the temporal lobe (n = 8, 29.6%), lateral ventricle wall (n = 3, 11.1%), and piriform lobe (n = 3, 11.1%). The exact locations of the tumors were not recorded in 3 cases (11.1%). Complete postmortem examination was performed in 7 cases, and distant metastases were not detected in any cases.
Signalment and Clinical Features of 27 Canine Intracranial Oligodendrogliomas.
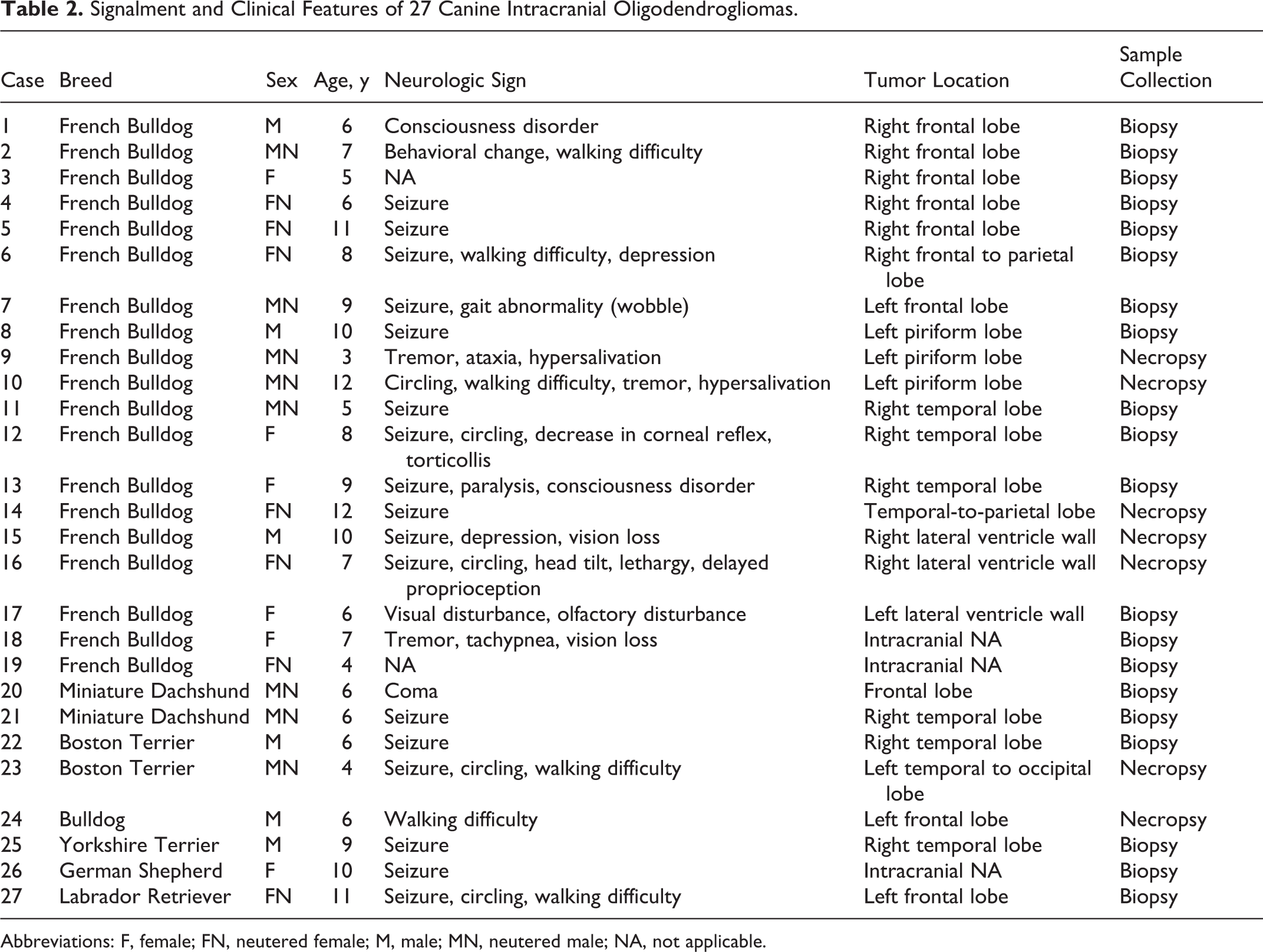
Abbreviations: F, female; FN, neutered female; M, male; MN, neutered male; NA, not applicable.
Histologic Characteristics
All the cases were histologically diagnosed as AO based on the presence of a monomorphous population of round cells with hyperchromatic nuclei, high cellularity, increased cellular atypia, and brisk mitotic activity. Tumors were characterized by sheets of round to polygonal cells with a hyperchromatic nucleus and inapparent to moderate amounts of cytoplasm. The nuclei were round to ovoid, and the nucleoli were obscure. Pronounced anisocytosis and anisokaryosis were noted with an average 10.7 mitoses per 10 high-power fields (range, 4–37; Fig. 2). A honeycomb pattern was present in 5 (18.5%) cases (Fig. 3). An extracellular mucinous matrix was present in 10 (37.0%) cases and microcystic degenerative changes in 6 (22.2%) tumors (Fig. 4). Intratumoral necrotic areas were present in 19 (70.4%) cases and pseudopalisading of tumor cells surrounding necrotic foci in 5 (18.5%) cases (Fig. 5). Prominent vascular endothelial proliferation, namely glomeruloid microvascular proliferation, was present in 16 (59.3%) cases (Fig. 6). Focal calcification was present in 5 (18.5%) cases (Fig. 7).
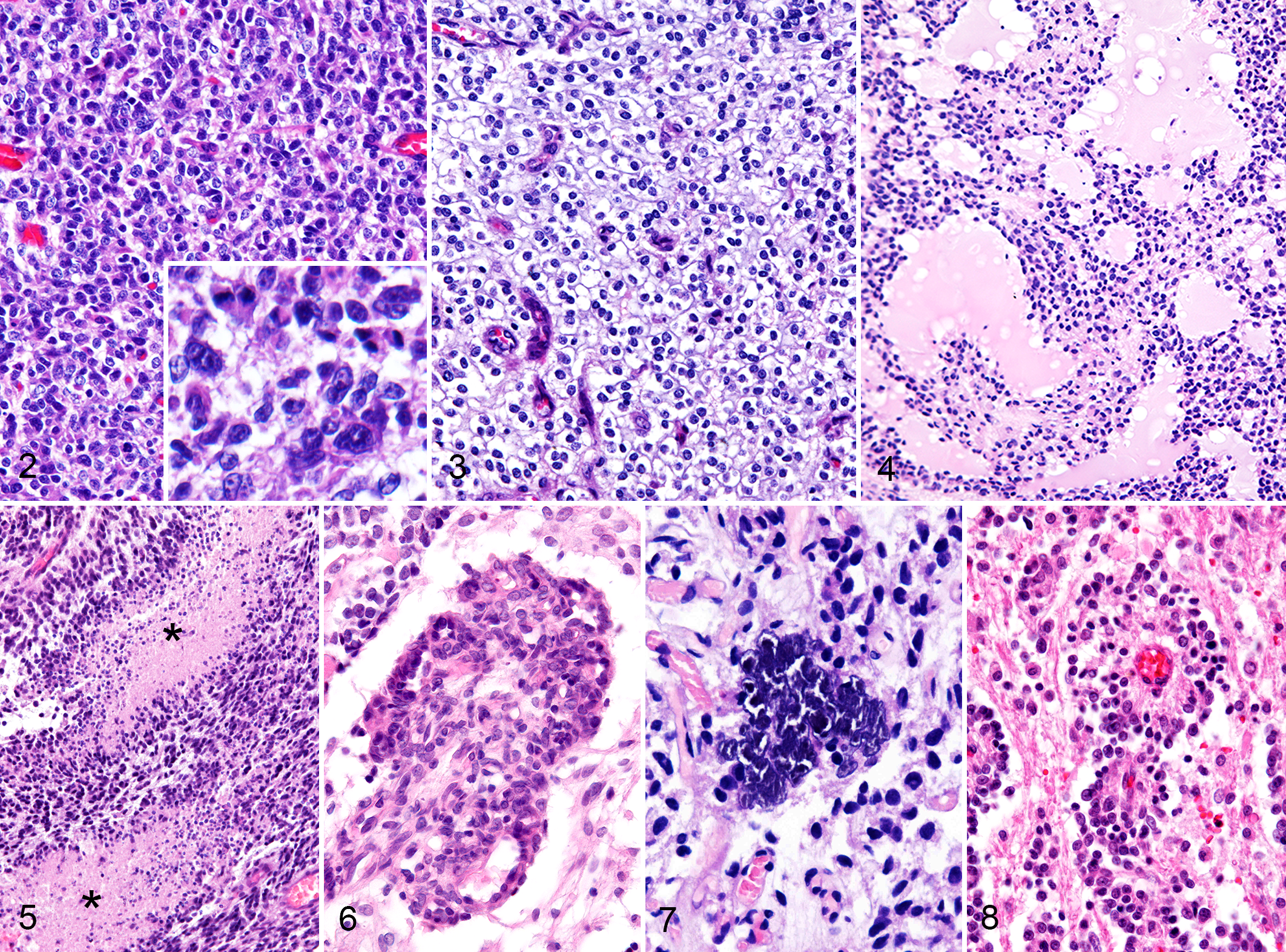
Anaplastic oligodendroglioma, cerebrum; dog. Histologic characteristics of canine anaplastic oligodendroglioma.
We evaluated the histopathologic patterns of tumor cell infiltration in 14 cases, whereas other cases could not be assessed, since no brain parenchyma was included in the samples. Perineuronal satellitosis was observed in 4 (28.6%) cases. Arrangement of tumor cells surrounding blood vessels (perivascular aggregate) was present in 5 (35.7%) cases (Fig. 8). Subpial and/or leptomeningeal spread of tumor cells was present in 3 (21.4%) cases (Supplemental Table 1).
Immunohistochemical Characteristics
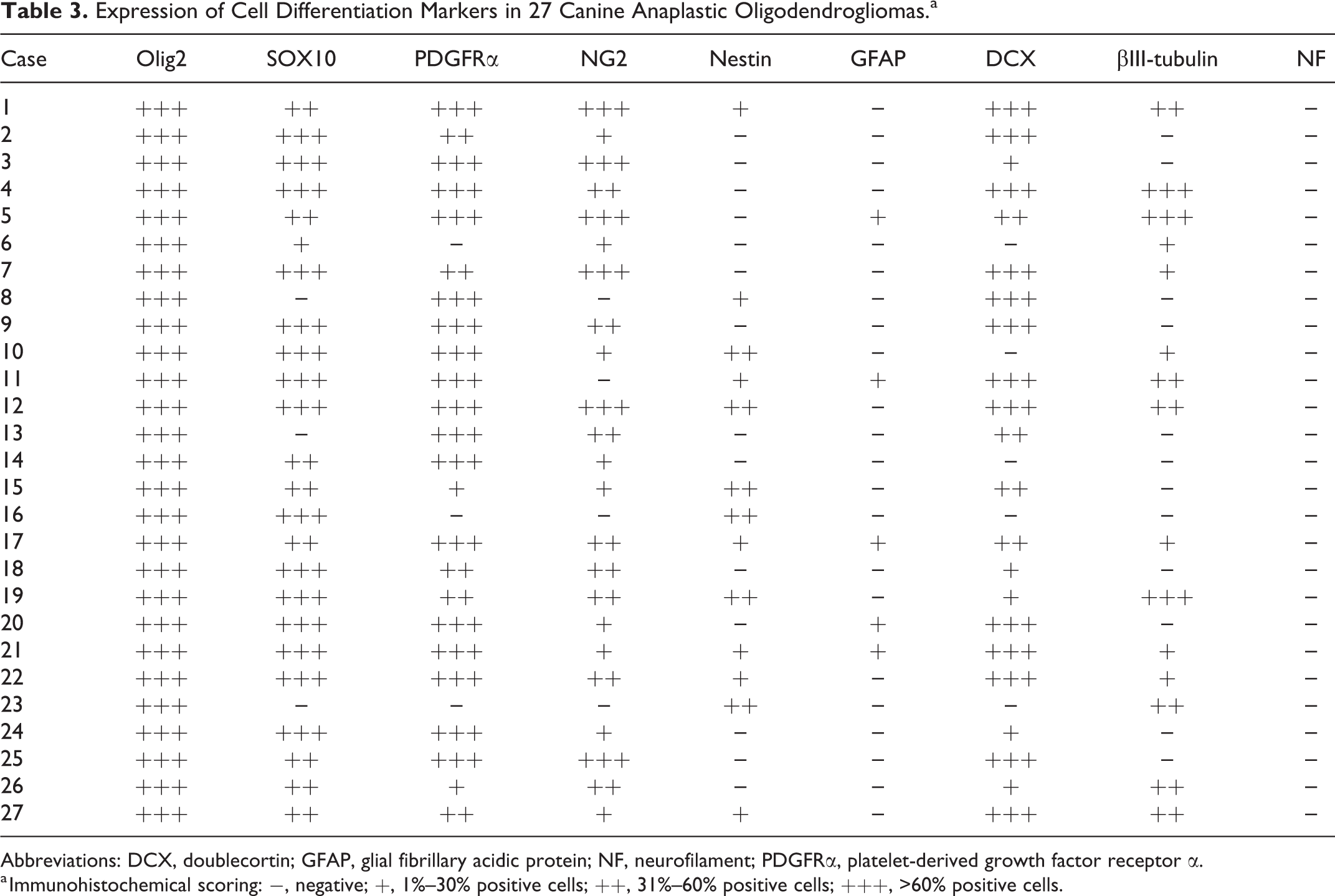
Immunohistochemical characteristics of canine AO are summarized in Table 3. Intense nuclear staining for Olig2 was observed in all 27 (100.0%) tumors (Fig. 9a). Nuclei of tumor cells were weakly but diffusely positive for SOX10 in 24 (88.9%) cases (Fig. 9b). Strong diffuse cytoplasmic staining for PDGFRα was detected in 24 (88.9%) cases (Fig. 9c). Tumor cells were strongly positive for NG2 in 6 (22.2%) cases while weakly to moderately positive in 17 (63.0%) cases (Fig. 9d). Focal to diffuse cytoplasmic staining for nestin was present in 13 (48.1%) cases (Fig. 9e). A few neoplastic cells were positive for GFAP in 5 (18.5%) cases (Fig. 9f). Tumor cells were positive for DCX, with a strong cytoplasmic pattern in 22 (81.5%) cases (Fig. 9g). Various amounts of tumor cells were positive for βIII-tubulin in 15 (55.6%) cases (Fig. 9h). Tumor cells in all the cases were negative for neurofilament, while non-neoplastic neurons were positive.
Expression of Cell Differentiation Markers in 27 Canine Anaplastic Oligodendrogliomas.a
Abbreviations: DCX, doublecortin; GFAP, glial fibrillary acidic protein; NF, neurofilament; PDGFRα, platelet-derived growth factor receptor α.
a Immunohistochemical scoring: −, negative; +, 1%–30% positive cells; ++, 31%–60% positive cells; +++, >60% positive cells.
Anaplastic oligodendroglioma, cerebrum (left ventricle wall); dog, case 17. Immunohistochemical characteristics of canine anaplastic oligodendroglioma. (a) Strong nuclear labeling with Olig2. (b) The nuclei of the tumor cells weakly label with SOX10. (c) The plasma membrane and cytoplasm of the tumor cells are strongly immunoreactive for platelet-derived growth factor receptor α. (d) The plasma membrane and cytoplasm of the tumor cells diffusely label with NG2. (e) The cytoplasm of the tumor cells focally expresses nestin. (f) Cytoplasmic labeling with glial fibrillary acidic protein is scant. (g) The cytoplasm of the tumor cells diffusely labels with doublecortin. (h) Scant cytoplasmic labeling with βIII-tubulin.
Characterization of Canine AO-Cultured Cells
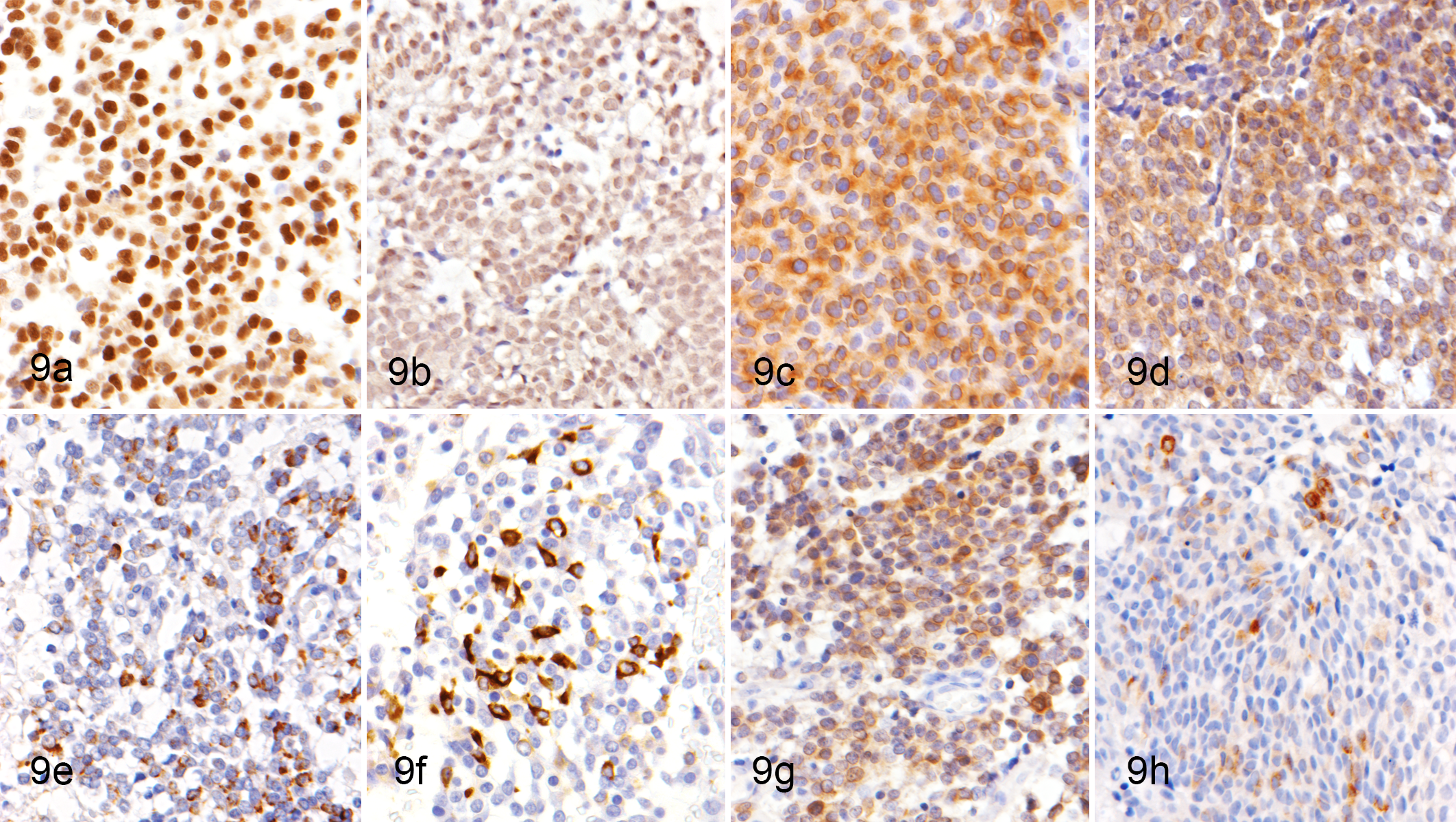
The cultured tumor cells grew continuously as far as the 80th passage during the collection of data described herein. The cells grew as adherent monolayers and were spindle to polygonal in shape with abundant cytoplasm. Their nuclei were round to oval with prominent single or multiple nucleoli. Multinucleated giant cells were sparsely present (Fig. 10, arrowhead). The growth curve of the cultured AO cells at the 75th passage is shown in Figure 11. The cells showed an initial growth lag on the first day, then an exponential phase of growth between days 1 and 8 after plating. The population doubling time was 41.3 hours at the 75th passage. Abnormal chromosome numbers were observed in the cultured AO cells. The cultured tumor cells of the sixth passage showed a mean chromosome number of 90.9 with hyperdiploidy.
Characteristics of cultured canine anaplastic oligodendroglioma (AO) cells; case No. 10.
Immunocytochemical Characteristics of Canine AO-Cultured Cells
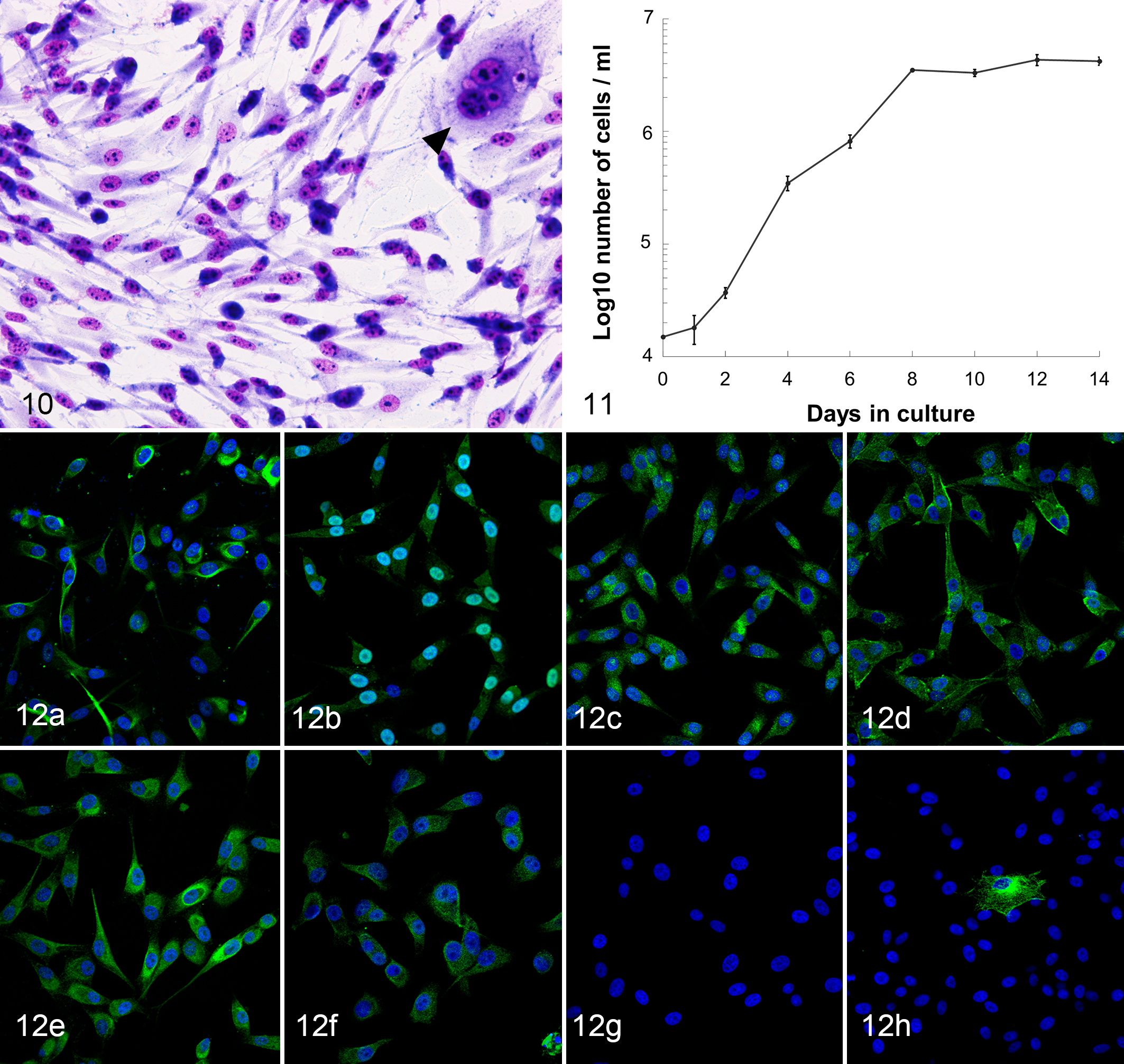
Immunocytochemical characterization of the primary AO cell culture for OPCs and additional differentiation markers was performed with fluorescence immunocytochemistry. The AO cells were consistently positive for Olig2, SOX10, PDGFRα, and NG2 (Fig. 12a–d). The cells predominantly exhibited nuclear localization of the SOX10 protein, but weak cytoplasmic localization was also observed (Fig. 12b). Expression of PDGFRα and NG2 was identified in the cytoplasmic membrane and/or cytoplasm (Fig. 12c, d). Contrary to the nuclear localization observed by immunohistochemistry, expression of Olig2 was exclusively identified in the cytoplasm (Fig. 12a). The majority of tumor cells showed cytoplasmic expression of nestin and GFAP, while few cells expressed βIII-tubulin in the cytoplasm (Fig. 12e, f, h). Tumor cells were negative for DCX (Fig. 12g) and neurofilament.
Discussion
In the present study, canine cerebral oligodendroglioma was most frequent in the frontal lobe, followed by the piriform lobe and the temporal lobe, which is consistent with previous studies. 43,53 Middle-aged dogs (mean age, 7.8 years) were often affected. No sex predilection was recorded. Seizure was the most common neurologic sign, while other clinical signs were sporadically observed. These properties were similar to those of humans. 29 The various clinical manifestations of AO might be associated with the tumor size, rate of growth, and areas in the brain affected.
A prior study 43 reported that glial neoplasms occurred more frequently in large breed versus small breed dogs. Additionally, Boxers, Boston Terriers, and other brachycephalic breeds were reported to be prone to oligodendrogliomas. 24,42,43 In the present study, oligodendrogliomas in the brain frequently occurred in brachycephalic breeds, which is consistent with the results of previous studies. Moreover, oligodendrogliomas had a high incidence among French Bulldogs. 3 Since the tumor incidence depends on the population of breeds, the data obtained in this study might be unique to Japan and differ from data in other countries. Furthermore, a previous study suggested that genetic predisposition is one of the theories for an increased prevalence of gliomas in brachycephalic dog breeds. 43 In humans, the most common genetic aberration in oligodendrogliomas is the 1p/19q codeletion 17,22,50 or mutations of the isocitrate dehydrogenase 1. 50,52 However, the underlying genetic mutations are yet to be elucidated for dogs. The fact that brachycephalic breeds are predisposed to developing oligodendrogliomas may offer a unique opportunity to study and understand the genetic mechanisms of this tumor.
Based on histologic similarities to human oligodendrogliomas, the human WHO classification system of central nervous system tumors has been tentatively applied to the taxonomy and grading for canine AO used in this study. 23,29 AOs are classified as anaplastic (high grade) when having anaplasia and high cellularity, prominent microvascular proliferation, nuclear pleomorphism, increased mitotic activity, necrosis, and/or leptomeningeal infiltration. 23,29 In addition, the latest human WHO classification adds the palisading of neoplastic cells around areas of necrosis. 29 However, there are no firm diagnostic criteria to differentiate high grade (anaplastic) from low grade beyond the recognition of the predominance of the features of anaplasia. Additionally, there is a possibility that these features (microvascular proliferation, necrosis, and pseudopalisading) may not be observed at the sampling place or method. Hence, in this study, increased cellular atypia, high cellularity, and increased mitotic activity were consistently observed and emphasized to differentiate high grade from low grade. Other histologic features, including prominent microvascular proliferation, necrosis, and palisading, were used to support the diagnosis. However, honeycomb pattern, extracellular myxoid matrix, cystic formation, and microcalcification, which are classical features observed in low-grade oligodendroglioma, help in the preliminary diagnosis of this tumor. Similar to human AO, canine AO in the present study showed hypercellularity, pleomorphism with anisokaryosis, conspicuous mitotic figures, and intratumoral microvascular proliferation. Necrosis was frequently observed, and some cases showed pseudopalisading, resembling the characteristics of glioblastoma. Occasionally, the classic histopathologic characteristics of lower-grade oligodendroglioma—namely, the honeycomb pattern, extracellular myxoid matrix, cystic formation, and microcalcification—could also observed. 29 Additional microscopic features included perineuronal and perivascular satellitosis as well as subpial and/or leptomeningeal neoplastic spread in some cases. These patterns of glioma cell invasion are referred to as “the secondary structures of Scherer.” These structures are not necessary for the diagnosis of AO; however, they do describe the appearance and behavior of glioma cells migrating away from the main tumor mass through the brain parenchyma. 11,39
In humans, accumulating evidence supports the existence of OPCs as the origin of oligodendroglioma. 1,4,10,27,34 –36,40 For example, tumor cells express a set of genes that are specific to OPCs (ie, Olig2, SOX10, PDGFRα, NG2, and A2B5). 1,34,36,40 Additionally, several studies 27,35 showed that some OPCs underwent malignant transformation and formed glioma with histologic features of oligodendrogliomas. Furthermore, glioma-initiating cells (GICs) in oligodendroglioma exhibit similar properties as OPCs, which implies that the cell of origin may be a transformed OPC. 35 With the heterogeneous components in canine oligodendroglial tumors, the cellular origin of the neoplastic cells was narrowed down to immature cells residing in the brain. 21,37 Since GICs were identified from canine glioblastoma, they have since been discovered in canine oligodendrogliomas in recent years. 14,19,45 The identification of GICs in canine oligodendrogliomas indicates immature cells as the origin of the glioma. 14,19 These studies also suggested that neuron stem cells or neuron precursor cells have the capability to be the origin of the canine oligodendroglioma. Moreover, recent findings demonstrated that canine AO highly expressed PDGFRα, which is the marker of OPCs. 20 However, unlike human studies, there is currently no information indicating that OPCs could be the origin of canine oligodendroglial tumor cells.
As described in human oligodendrogliomas, most of the present canine AO cases showed an enriched expression of OPC markers, including nuclear transcription factors (eg, Olig2 and SOX10) and cell surface markers (eg, PDGFRα and NG2). Expression of these lineage-specific markers may suggest that canine AO arises from OPCs. Immunohistochemical analysis has demonstrated nestin expression in the cytoplasm of AO cells. While nestin has been widely used as a neuron stem cell marker, its expression was well documented in human OPCs. 28 However, some tumor cells also expressed an astrocytic marker and some neuronal markers. This suggests that canine AO may be derived from immature multipotent cells with the ability to differentiate into the oligodendroglial, astrocytic, and neuronal lineages. Although OPCs have generally been regarded as oligodendrocyte lineage-restricted precursor cells, several studies 6,26 demonstrated that the OPCs may possess a multipotent differentiation ability. Gliomas that do not express NG2, PDGFRα, and SOX10 may have arisen from different precursor cells, including multipotent neuron stem cells. As another possibility, these markers may have been downregulated due to acquired genetic changes or environmental influences. To better elucidate the origin of canine AO, we performed immunocytochemistry on a primary explant culture of canine AO. The results demonstrated that explanted canine AO cells cultured in DMEM supplemented with FBS retained a typical precursor morphology and the expression of OPC markers, which clearly indicated their OPC state. As we could expect from immature cells with multipotential competence, in vitro tumor cells were characterized by aberrant expression of several cell lineage markers. The primary serum-cultured AO cells not only contained neuronal components but also exhibited astrocytic phenotypes. The serum-supplemented culture medium may trigger the differentiation of AO cells, as observed in the in vitro studies of cultured neural stem cells and other precursor cells. 9,18,30 Olig2, a basic helix–loop–helix transcription factor, has been recognized as a key regulator of oligodendrocyte development. 31,46,54 In the present study, nuclear localization of Olig2 was observed in histologic sections of an original AO, while cultured AO cells exhibited cytoplasmic Olig2 expression. Expression of Olig2 has been normally detected in the nucleus of oligodendrocyte lineage cells, while the cytoplasmic translocation of Olig2 was often observed during astrocytic differentiation of precursor cells. 7,8
Spontaneous canine oligodendroglioma showed epidemiologic, histologic, and immunohistochemical characteristics similar to those of human oligodendroglioma. The present histologic and in vitro studies indicated that canine AO consisted of an enriched OPC population. Moreover, in the culture conditions, AO cells were uncommitted to terminal differentiation, as the tumor cells predominantly expressed OPC markers and exhibited multipotent characteristics. Taken together, as with human oligodendrogliomas, our results suggest that multipotent OPCs may be a potential glioma-initiating population in canine AO and that dogs may be a suitable animal model for a comparative study of human oligodendroglioma. Additionally, the tumor cell line established in the present study is useful for understanding the cancer development. Therefore, further in vitro studies with our cultured AO cells may provide a useful framework for investigating the origin of oligodendroglial tumors.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985818777794 - Expression of Oligodendrocyte Precursor Cell Markers in Canine Oligodendrogliomas
Supplemental Material, DS1_VET_10.1177_0300985818777794 for Expression of Oligodendrocyte Precursor Cell Markers in Canine Oligodendrogliomas by Takuya E. Kishimoto, Kazuyuki Uchida, Atigan Thongtharb, Tokuhiro Shibato, James K. Chambers, Kazumi Nibe, Yumiko Kagawa, and Hiroyuki Nakayama in Veterinary Pathology
Footnotes
Acknowledgements
We thank S. Kato for her technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
