Abstract
Feline oral squamous cell carcinoma (FOSCC) is an aggressive malignancy with invasive and metastatic behavior. It is poorly responsive to chemotherapy and radiation. Neoplastic epithelial-mesenchymal transition (EMT) portends highly malignant behavior and enhances resistance to therapy. In transitioning to a more malignant phenotype, carcinoma stem cells undergo transformation mediated by expression of proteins, endowing them with mesenchymal properties advantageous to cell survival. The goal of the current study was to identify proteins associated with EMT in FOSCC. This study documents protein expression patterns in 10 FOSCC biopsies and 3 FOSCC cell lines (SCCF1, SCCF2, SCCF3), compatible with an EMT phenotype. As markers of EMT, P-cadherin, N-cadherin, vimentin, nuclear transcription factors Twist and Snail, hypoxia inducible factor 1α (HIF-1α), programmed death ligand 1, and vascular endothelial growth factor D, as well as E-cadherin, were examined using immunohistochemistry, Western blot, and enzyme-linked immunosorbent assay. P-cadherin, Twist, HIF-1α, and programmed death ligand 1 were commonly expressed in biopsies and cell lines. N-cadherin, classically associated with EMT, was not highly expressed, and E-cadherin was coexpressed along with proteins characteristic of EMT in all specimens. Production of vascular endothelial growth factor A by cell lines, a process regulated by HIF-1α expression, was suppressed by the small-molecule inhibitor dasatinib. These data are consistent with EMT in FOSCC and shed light on cellular changes that could contribute to the aggressive behavior of FOSCC.
Keywords
Squamous cell carcinoma is an aggressive malignancy of the feline oral cavity. The prognosis for affected cats is poor due to aggressive, invasive malignant behavior. Unlike some other malignancies, death is most often due to local disease and not systemic metastasis, as feline oral squamous cell carcinoma (FOSCC) is slow to metastasize beyond draining lymph nodes. Tumors arising from gingiva are osteoinvasive early in the clinical course, and tumors arising under the tongue (the most common site) infiltrate adjacent soft tissues deeply. 96 The median survival time of 54 cats with oral squamous cell carcinoma was 44 days, and <10% of affected cats were alive at 1 year after diagnosis. 36,54 FOSCC is poorly responsive to chemotherapy and/or radiotherapy, while surgical excision, even with radical procedures such as mandibulectomy, generally fail to prevent recurrence. 7,24,54,69,113,116 Surgical removal is commonly reserved for smaller tumors that require less mutilating surgeries and without laborious postoperative care. Quality of life and the relatively short time until recurrence discourage many pet owners from choosing radical surgical procedures. Recurrence can be seen even though surgical margins are clear. 69 The outcomes have been slow to improve in more than 25 years. 20,21,40,45,77,79 By contrast, the larger size of medium to large dogs provides more options for mandibular surgery that may account for better surgical outcomes for canine oral squamous cell carcinoma. 2,12,52
Several small studies reported median survival times approaching 1 year, but this small subset of cats is unusual and may reflect low-grade tumors, more favorable clinical stages, or location within the oral cavity when diagnosed. 40,69 Toceranib was recently reported to extend survival time of cats with FOSCC (median 123 days vs 45 days without toceranib), but only 20% of treated cats survived 1 year. 113 Intralesional therapies with radioisotopes and electrochemotherapy have had positive results in preliminary reports. 93,105
Epithelial-mesenchymal transition (EMT) is a process by which epithelial cells acquire mesenchymal features that modify cell behavior. This transition alters epithelial cell morphology, adhesion, and migratory capabilities. In health, it is a normal process in wound repair, ovulation, and embryonic development. 53,67,102 In malignancy, onset of EMT is marked by altered gene expression that drives morphologic and behavioral changes in carcinoma cells, including loss of epithelial cell polarity, disaggregation of cell-to-cell adhesion, invasion into adjacent tissues, and metastasis. 12,23 Transformation to a mesenchymal phenotype is mediated by upregulation of many genes and downregulation of others. This leads to a more aggressive malignancy capable of invasion and metastasis. Tumor-initiating cells with stem cell–like properties are considered drivers of EMT, which helps to explain the plasticity of carcinomas. 47,48,57,84 This phenomenon may be partially or wholly responsible for resistance of carcinomas to chemotherapy, radiotherapy, and targeted therapy, all properties of tumor-initiating/cancer stem cells. 23,28,116,122
Transformation to a mesenchymal phenotype entails a continuum of phenotypical and behavioral changes. Changes in adhesion molecules, such as increased N- and P-cadherin and decreased E-cadherin expression (cadherin switching), increased vimentin expression, and activation of transcription factors Snail and Twist, are documented features of mesenchymal transition. Snail and Twist expression are highly specific indicators of EMT. 90 Not all cells in a tumor exhibit all of these shifts, possibly due to their location within a neoplasm (eg, parenchyma vs invasive front) or because they lack the genetic profile and plasticity needed for dedifferentiation. Heterogeneity of tumors is a contributing factor to mixed degrees of EMT. This accounts for an intermediate phase where cells express mesenchymal proteins while retaining epithelial markers. 28,39,50,84 For example, some carcinomas continue to express E-cadherin, connecting adjacent cells at adherens junctions, while at the same time acquiring migratory behavior characteristic of mesenchymal cells. 6,26,50 These observations have given rise to the concept of a “metastable phenotype” ranging from anchored sedentary growth to motile invasive growth. Shifts are mediated by malignant cells with stem cell properties that express novel, dominant adhesion molecules enabling invasive and migratory mesenchymal behavior. Upregulated P-cadherin, a marker of EMT, is also a stem cell marker and contributes to stem cell signaling. 50,84,107
Neoplastic transformation of human oral squamous epithelium is associated with upregulated P-cadherin, whereas E-cadherin expression can be either unchanged or decreased. 88 Normal epithelial cell-to-cell adhesion is maintained by E-cadherin–catenin complexes; when these are interrupted, a critical barrier to metastasis is removed. P-cadherin is a disruptor of cell-to-cell adhesion, thereby promoting cell motility and a proliferative phenotype. 50,88 In human head and neck squamous cell carcinoma (HNSCC), P-cadherin is upregulated in primary tumors and lymph node metastases. N-cadherin, another adhesion molecule considered a signature of EMT, is not seen in this malignancy. Shifts in protein expression and phenotype of EMT indicate P-cadherin, but not N-cadherin, is the major membrane protein in HNSCC. 80 P-cadherin was found to be widely expressed in high-grade malignant feline mammary tumors, and E-cadherin was coexpressed in half of these. The study concluded that P-cadherin expression was a better predictor of an aggressive phenotype than reduced expression of E-cadherin. 22
Upregulated P-cadherin correlates with shortened disease-free interval and reduced overall survival time in humans with non–small cell lung carcinoma. 42 Upregulated P-cadherin is also found in numerous human epithelial malignancies, including breast, ovarian, prostate, endometrial, skin, gastric, pancreatic, and colonic carcinoma, as well as oral squamous cell carcinoma. 88,107 Thus, P-cadherin is considered a consistent marker of EMT and is a protein of interest in this study. 88
Twist and Snail play central roles as mediators of EMT. The appearance of these transcription factors signals mesenchymal transformation and portends highly malignant behavior. Some authors indicate expression of Twist and/or Snail alone is sufficient proof of EMT. 31,41,109 More than biomarkers of EMT, expression of Twist or Snail is a negative prognostic factor in human cancers. As transcription factors, activated by hypoxia inducing factor 1α (HIF-1α), they switch on specific genes that inhibit apoptosis, promote chemotherapy resistance, and facilitate metastasis. Therefore, Twist and Snail are recognized as biomarkers of EMT because when expressed, a cell is committed to undergoing EMT. Moreover, Twist expression is a negative prognostic indicator for shortened disease-free survival and overall survival times in human patients with breast, lung, prostate, and gastric cancer. 123 The association of Twist expression and aggressive human carcinomas has stimulated efforts to target it therapeutically using small interfering RNA (siRNA). 98,124 Snail expression is an independent prognostic indicator, predicting recurrence and progression in non-muscle-invasive bladder cancer in humans and in canine mammary carcinomas. 32,41 In canine mammary carcinoma, nuclear accumulation of Snail was highest in solid and anaplastic carcinomas and was shown to be a negative prognostic factor that correlated with the degree of malignancy. 41
Vascular endothelial growth factor (VEGF)–D is a VEGF isoform associated with hypoxia and angiogenesis, especially lymphangiogenesis. It is upregulated in hypoxic microenvironments such as those conducive to EMT, where VEGF-D promotes development of new, local lymphatic networks. 1,18,27,95,114 This could be the case for FOSCC as well, but information about VEGF-D in FOSCC is lacking and therefore of interest to this study. VEGF-D binds to VEGF-3R/Flt4, the lymphatic growth factor receptor expressed on lymphatic endothelium. This initiates development and proliferation of lymphatic channels used for metastasis to lymph nodes. 27,95 When VEGF-D was reduced in the tumor microenvironment with a blocking VEGF-D antibody, lymphatic metastasis was suppressed in an animal model. 95 There is interest in development of novel targeted agents to inhibit EMT. 10,13,108 This approach could also have effects on VEGF-D. Other strategies to inhibit VEGF-D activity and lymphangiogenesis have included VEGF-D–specific blocking monoclonal antibodies, antibody blockade of VEGF-3R, and targeting proprotein convertases responsible for VEGF-D processing. 37,56,61,94
Efforts to reverse EMT (ie, redifferentiation) are also attractive as a means to mitigate aggressiveness of cancer cells. 67 For example, inhibitors of transforming growth factor–β (TGF-β), a major driver of EMT, have been pursued therapeutically in phase I trials for glioblastoma and hepatocellular carcinoma. 11,30,85,119 Targeted small-molecule inhibitors of Src and focal adhesion kinases are also of interest for their ability to reverse EMT. 39,44
EMT has been described in canine tumors, but there are few reports in cats despite the common occurrence of epithelial cancer in this species. 41,73 Although relatively brief remissions are seen, the propensity for FOSCC to recur after chemotherapy and radiation therapy suggests a population of surviving cells tolerant of lethal stress. In vitro, FOSCC cells survived long-term culture with the epidermal growth factor receptor (EGFR) inhibitor, gefitinib, by undergoing EMT. 73 Stem cells within the cultures could explain the adaptive survival responses. 73
Immunohistochemistry (IHC) has been used to enumerate protein profiles of the EMT phenotype. 11,12,91,101 The goal of the current study was to identify proteins associated with EMT in 10 archived FOSCC biopsies and 3 established FOSCC cell lines. We conducted a systematic interrogation of all samples using IHC to identify markers that could underlie EMT and account for the invasive clinical course and chemotherapeutic resistance of FOSCC.
Materials and Methods
Biopsy Selection and Preparation
Archived FOSCC formalin-fixed, paraffin-embedded (FFPE) biopsies submitted to the Oregon State University Veterinary Diagnostic Laboratory between 2006 and 2012 were the source of study material. This study employed 10 tumor biopsy samples from 10 individual, privately owned cats, submitted for routine pathological evaluation by primary care veterinary practitioners; it did not use any live animals or experimentation. All tissues were confirmed to be the conventional subtype of FOSCC by 2 board-certified veterinary pathologists (M.K., H.B.G.). 63 Cellular pleomorphism was present, and most tumors were intermediately differentiated; 1 was poorly differentiated. Serial sections (4 microns thick) were prepared from tissue blocks and placed on positively charged slides prior to IHC labeling.
Cells and Cell Culture
Three established FOSCC cell lines, SCCF1, SCCF2, and SCCF3, were kindly provided by Dr. T. Rosol (Ohio State University) for this study. 60,100 Cells were cultured in RPMI 1640 medium containing L-glutamine and sodium pyruvate; medium was supplemented with 10% fetal bovine serum (FBS), 1% penicillin-streptomycin, and 2 mM HEPES buffer solution. Cells were incubated at 37°C in a humidified 5% CO2 atmosphere, and growth was 80% to 90% confluent when cells were detached for experiments. Each cell line was tested periodically for mycoplasma expression by quantitative polymerase chain reaction (qPCR). Cell pellets were prepared by removing medium and rinsing adherent cells with warm phosphate-buffered saline. Cultures were then incubated for 5 minutes with a 10× dilution of trypsin at 37°C to detach cells, transferred to Eppendorf tubes, pelleted at 400 × g for 5 minutes, rinsed, and recentrifuged. Phosphate-buffered saline was removed and replaced with 10% formalin solution, and cells were fixed for 24 hours before being embedded in paraffin.
Immunohistochemistry
IHC was performed on serial sections from FOSCC FFPE blocks and cell pellets prepared from the SCCF1, SCCF2, and SCCF3 cell lines. Expression of adhesion proteins, hypoxia/angiogenesis proteins, structural protein, and protein transcription factors associated with EMT was examined. These included E-cadherin, P-cadherin, N-cadherin, vimentin, Twist, Snail, HIF-1α, VEGF-D, programmed death ligand 1 (PD-L1), and EGFR (Table 1). Positive control feline tissues were selected from recommendations in the manufacturers’ data sheets, published literature, and The Human Protein Atlas, which confirmed all positive control tissues highly expressed relevant messenger RNA (mRNA) or proteins. In some cases, 2 different tissue types were used as positive controls for a single antibody. The negative control for each antibody replaced the primary antibody with dilution buffer, and slides were otherwise processed identically to the study slides.
Immunohistochemistry Methods Used in This Study.
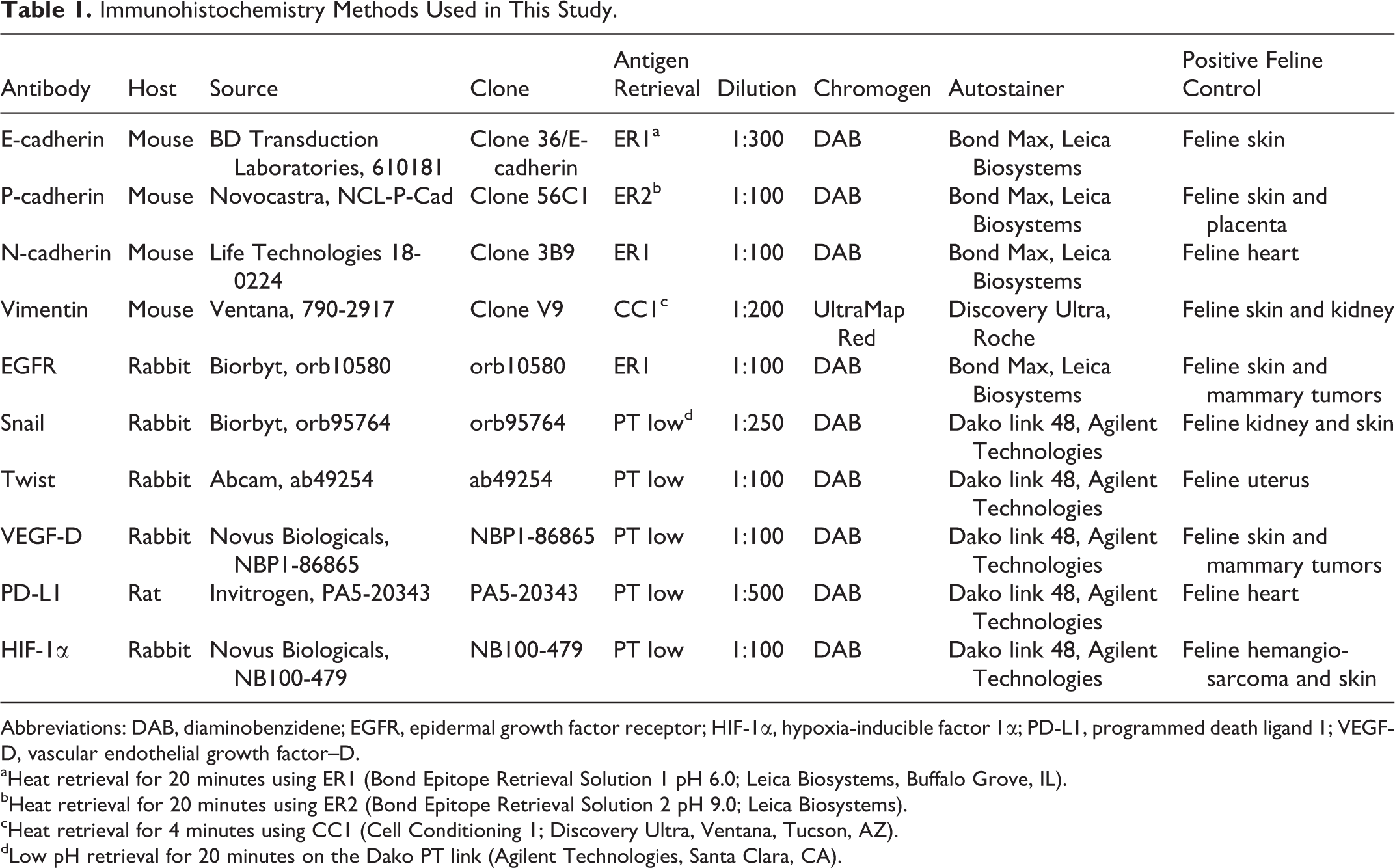
Abbreviations: DAB, diaminobenzidene; EGFR, epidermal growth factor receptor; HIF-1α, hypoxia-inducible factor 1α; PD-L1, programmed death ligand 1; VEGF-D, vascular endothelial growth factor–D.
aHeat retrieval for 20 minutes using ER1 (Bond Epitope Retrieval Solution 1 pH 6.0; Leica Biosystems, Buffalo Grove, IL).
bHeat retrieval for 20 minutes using ER2 (Bond Epitope Retrieval Solution 2 pH 9.0; Leica Biosystems).
cHeat retrieval for 4 minutes using CC1 (Cell Conditioning 1; Discovery Ultra, Ventana, Tucson, AZ).
dLow pH retrieval for 20 minutes on the Dako PT link (Agilent Technologies, Santa Clara, CA).
IHC was done with a BOND-MAX Automated Staining System (Vision BioSystemxs; Leica, Bannockburn, IL) using the Bond Polymer Detection System (Vision BioSystems; Leica) with 3,3′-diaminobenzidine as the chromogen for E-cadherin, P-cadherin, N-cadherin, and EGFR or on the Dako link 48 Automated Staining System (Agilent Technologies, Santa Clara, CA) using the peroxidase-conjugated EnVision Polymer Detection System (Agilent Technologies) for Snail, Twist, VEGF-D, PD-L1, and HIF-1α or on the Discovery Ultra Automated Staining system (Ventana Medical Systems, Tucson, AZ) using the UltraMap alkaline phosphatase red detection system (Ventana Medical Systems) for vimentin.
Positive control tissues
Feline skin was employed as the positive control for E-cadherin, P-cadherin, and HIF-1α. The same E-cadherin clone was used in prior investigations of feline neoplasia. 34,75 Feline placenta served as another P-cadherin–positive control tissue as recommended by the manufacturer of the 56C1 antibody. 70,72 The P-cadherin antibody clone of this report has previously been shown to recognize P-cadherin in human placenta and feline mammary carcinoma. 22,87
Skin has constitutive HIF-1α expression and was used as a positive control for HIF-1α IHC. 83,117 Feline heart served as the positive control for N-cadherin, 62,81 as recommended by the manufacturer. 64
Feline skin and kidney were the positive control tissues used for vimentin IHC; The Human Protein Atlas shows high vimentin expression in skin and kidney. 78 The antibody used was previously reported to recognize vimentin in feline sarcoma and carcinoma. 75,76
Feline uterus served as the positive control tissue for Twist based on expression in normal human endometrium and in human uterine carcinoma. 4,78 Notably, the manufacturer’s Twist antibody data sheet states the clone recognizes feline Twist. 104
Feline skin and kidney were used as control tissues for Snail immunolabeling. Snail is found throughout the body in most tissues. 78 Snail is expressed in the dermis at the base of hair follicles. 25 It is constitutively expressed in the nucleus of ≥25% of cells in normal skin. 33
Feline mammary carcinoma and normal skin served as the positive control tissues for VEGF-D immunolabeling. VEGF-D is highly expressed in human breast carcinoma, 18 with gene expression reported in skin. 94 Feline heart served as the positive control tissue for PD-L1 expression based on known expression and on recommendation of the manufacturer. 106,74 Feline skin and mammary carcinoma served as the positive control tissues for expression of EGFR as previously described. 86,112 Photomicrographs of the control IHC tissues are provided as Supplemental Figures S1 to S10.
IHC scoring
Multiple (eg, ≥10) high-power fields were scanned (M.K.) and immunolabeling of individual cells was reported as positive or negative, independent of signal intensity. The percentage of positively immunolabeled cells on glass slides was estimated. Digitized images of the glass slides were reviewed independently by a second pathologist (H.B.G.) to corroborate results.
Western Blot VEGF-D
FOSCC cells were cultured under normoxic and hypoxic conditions. Lysates were extracted from cells cultured in each condition. Hypoxic cell lysates from SCCF1, SCCF2, and SCCF3 cell lines were kindly provided by Dr. E. Dickerson (St. Paul, MN). Cells in hypoxic conditions were cultured and maintained in standard tissue culture–treated flasks in Dulbecco’s modified Eagle medium/Ham’s F-12 50:50 mix supplemented with 10% FBS and 0.2% Primocin (InvivoGen, San Diego, CA). The cells were maintained under hypoxic conditions in 1% O2 at 37°C for 72 hours by adding nitrogen to the incubation. Intracellular proteins from normoxic and hypoxic cell cultures were extracted using RIPA buffer (10 mM Tris, 1% Triton X-100, 0.5% deoxycholate, 0.1% sodium dodecyl sulfate, 150 mM NaCl, 1 × Protease Inhibitor Cocktail for mammalian cells [Sigma, St. Louis, MO], 2 mM orthovanadate, and 10 mM NaF [pH 8.0]). Then, 30 to 50 µg of cell extracts was resolved on 12% NuPage Gel in MES buffer (Invitrogen, Carlsbad, CA) and transferred to polyvinylidene fluoride membranes. The membranes were blocked with Tris-buffered saline (TBS) with 0.1% Tween-20 (TBST) and 1.5% bovine albumin followed by blotting with the appropriate antibodies. The VEGF-D antibody (ab155288; Abcam, Cambridge, MA) was diluted in TBST at a 1:1000 concentration. Secondary antibody (goat anti-rat and goat anti-rabbit; Santa Cruz Biotechnology, Santa Cruz, CA) linked to horseradish peroxidase was diluted 1:20 000 in TBST. Bands were detected using Supersignal West Pico luminol (Pierce, Rockford, IL) exposed to Kodak Biomax film (Rochester, NY). Actin served as the loading control for Western blots and was detected using an actin-specific antibody (sc-47778; Santa Cruz Biotechnology).
VEGF Enzyme-Linked Immunosorbent Assay
A commercial VEGF enzyme-linked immunosorbent assay (ELISA) kit (DY293B; R&D Systems, Minneapolis, MN) that recognizes human, feline, canine, and rat VEGF-A (per product data sheet) was used according to the manufacturer’s instructions to quantitate VEGF-A production by SCCF1, SCCF2, and SCCF3 cell lines. The ELISA is specific for VEGF-A, and the data sheet indicated no cross-reactivity with isoforms VEGF-B, VEGF-C, and VEGF-D. VEGF-A was chosen for this query because a commercial VEGF-D ELISA validated for the cat could not be sourced when the experiment was performed. The kit included recombinant human VEGF-A for creating the standard curve validating the assay and served as a reference for the feline test samples. Cells were cultured in supplemented RPMI-1640 medium under normoxic conditions described above. They were grown to 80% confluence, detached, washed, and then placed in 96-well plates (100 000 cells/well) in medium lacking serum supplementation or identical medium spiked with 100 ng/ml epidermal growth factor (EGF) and incubated overnight at 37°C. At that time, some EGF-containing wells received the Src inhibitor dasatinib (10 nM) and remained in culture for 1 additional hour to assess effect on VEGF production. For each cell line, all conditions were performed in triplicate. The supernatants were collected from wells and the VEGF concentration in each sample determined using the VEGF ELISA.
Results
Immunohistochemistry
Immunohistochemical results are summarized in Table 2. There was near-total agreement (ie, within 5%–10%) between pathologists’ scores for E-cadherin, N-cadherin, Snail, Twist (nuclear), and PD-L1–labeled specimens; 11 of 13 VEGF-D-labeled specimens; 10 of 13 vimentin-labeled specimens; 9 of 13 HIF-1α–labeled specimens; and 8 of 13 P-cadherin–labeled and EGFR-labeled specimens. Differences were attributed to variability of areas examined on the glass slides vs the digitized images. For biopsies where scores differed by >10% between pathologists, the lower score was reported. Where there were differences, scoring of digitized images was consistently higher than scores from evaluation of glass slides. Given high concordance between pathologists’ scores for nearly 86% of the IHCs, the results of scores from the glass slides are reported in Table 2.
Immunolabeling Patterns in Cell Lines and Biopsies of Feline Oral Squamous Cell Carcinoma.a
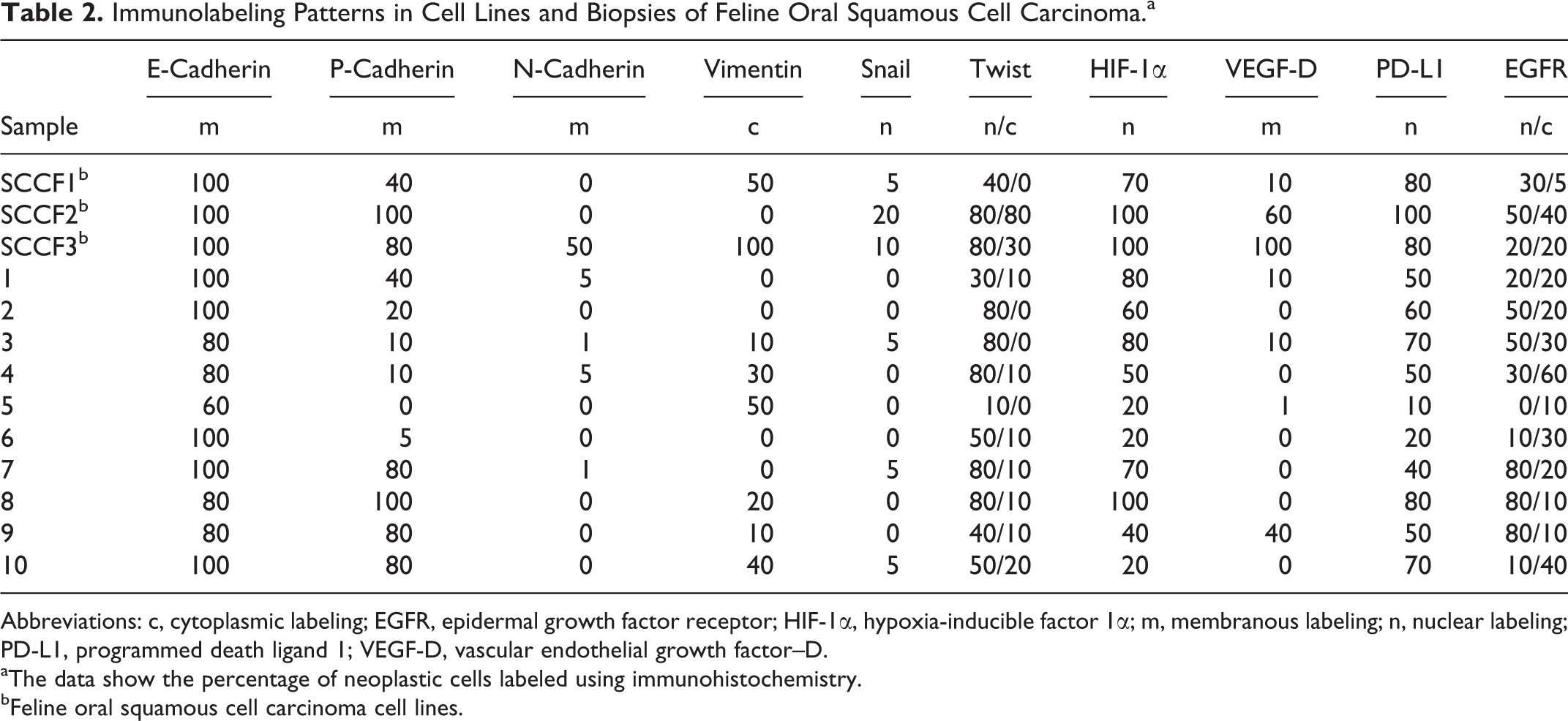
Abbreviations: c, cytoplasmic labeling; EGFR, epidermal growth factor receptor; HIF-1α, hypoxia-inducible factor 1α; m, membranous labeling; n, nuclear labeling; PD-L1, programmed death ligand 1; VEGF-D, vascular endothelial growth factor–D.
aThe data show the percentage of neoplastic cells labeled using immunohistochemistry.
bFeline oral squamous cell carcinoma cell lines.
E-, P-, and N-cadherins
All 10 biopsies expressed membranous E-cadherin in 80% to 100% of the malignant cells, with the exception of biopsy No. 5 (60%) (Fig. 1). The 3 FOSCC cell lines each showed 100% membranous labeling for E-cadherin. Membranous P-cadherin expression (Fig. 2) was present in 9 of 10 tumors ranging from 5% (biopsy No. 6) to 100% (biopsy No. 8), with ≥40% labeling in half the biopsies. P-cadherin was undetectable in only 1 tumor (biopsy No. 5). Membranous P-cadherin was expressed by each of the 3 FOSCC cell lines with 40% to 100% cells immunolabeling. In contrast, N-cadherin expression was low. The highest expression of N-cadherin was seen in the SCCF3 cell line, with 50% of cells immunolabeling (Fig. 3). The other 2 cell lines were negative for N-cadherin. N-cadherin was uncommon in the FOSCC biopsies, with ≤5% membranous expression in 4 of 10 (40%) tumors.
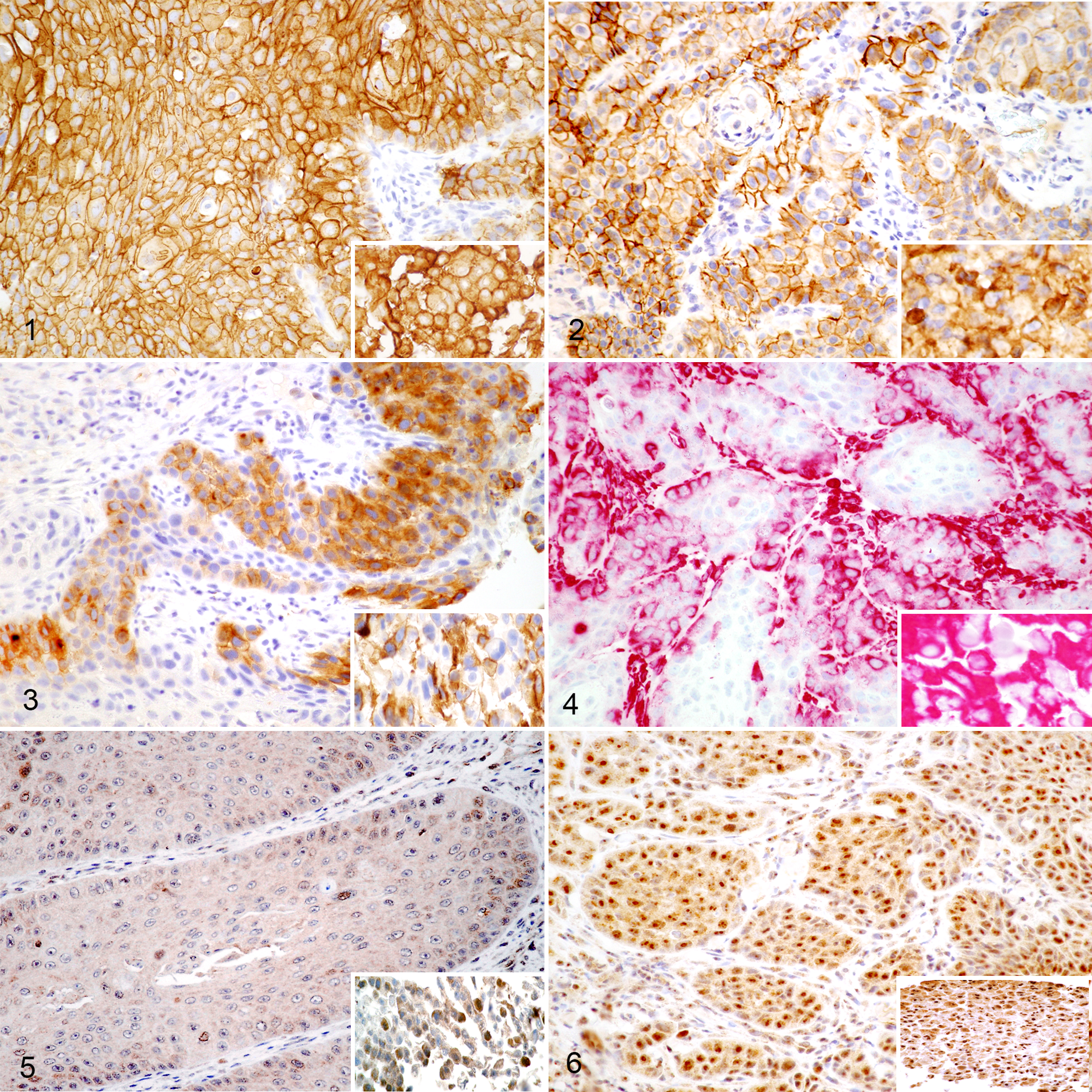
Vimentin
Cell lines SCCF1 and SCCF3 showed positive vimentin immunolabeling, with 50% and 100% positive labeling, respectively. Six of 10 biopsies were positive, ranging from 10% to 50% cell labeling (2 at 10% and 1 each 20%, 30%, 40%, and 50%). In every sample, immunolabeling was cytoplasmic (Fig. 4).
Transcription factors Twist, Snail
Snail immunolabeling was nuclear, with the highest percentage of positive cells in the FOSCC cell lines (Fig. 5). The SCCF2 cell line exhibited the greatest percentage of positive cells (20%), while 3 biopsies (Nos. 3, 7, and 10) expressed nuclear Snail in 5% of the malignant cells. Twist was more highly expressed in all specimens (Fig. 6). The greatest concentrations were in the nucleus (normal for transcription factors), although cytoplasmic Twist labeling was detected in up to 20% of cells in all biopsies and in cell lines SCCF2 and SCCF3 (80% and 30% positive cells, respectively). Seven of 10 biopsies expressed nuclear Twist in ≥50% of cells, and the remaining biopsies had 40%, 30%, and 10% positive cells (Fig. 6). The SCCF2 cell line had high Twist expression (80%) in both the nucleus and cytoplasm of positive cells.
VEGF-D, HIF-1α
The 3 cell lines and 3 of 10 biopsies expressed VEGF-D on cell membranes (Fig. 8).
The SCCF2 and SCCF3 cell lines had the highest expression, with 60% and 100% of cells immunolabeling, respectively. HIF-1α was detected in all 13 specimens (Fig. 7). There was high nuclear expression in the SCCF1, SCCF2, and SCCF3 cell lines (70%, 100%, and 100%, respectively). The biopsies expressed HIF-1α, with 6 of 10 exhibiting ≥50% positive nuclear immunolabeling. Biopsy No. 5, despite being one of the higher vimentin expressers, had the lowest expression of nuclear Twist (10%), HIF-1α (20%), and P-cadherin (0%, the only negative). Biopsy Nos. 5 and 6 had the fewest HIF-1α–positive cells (20% each) compared to the other samples.
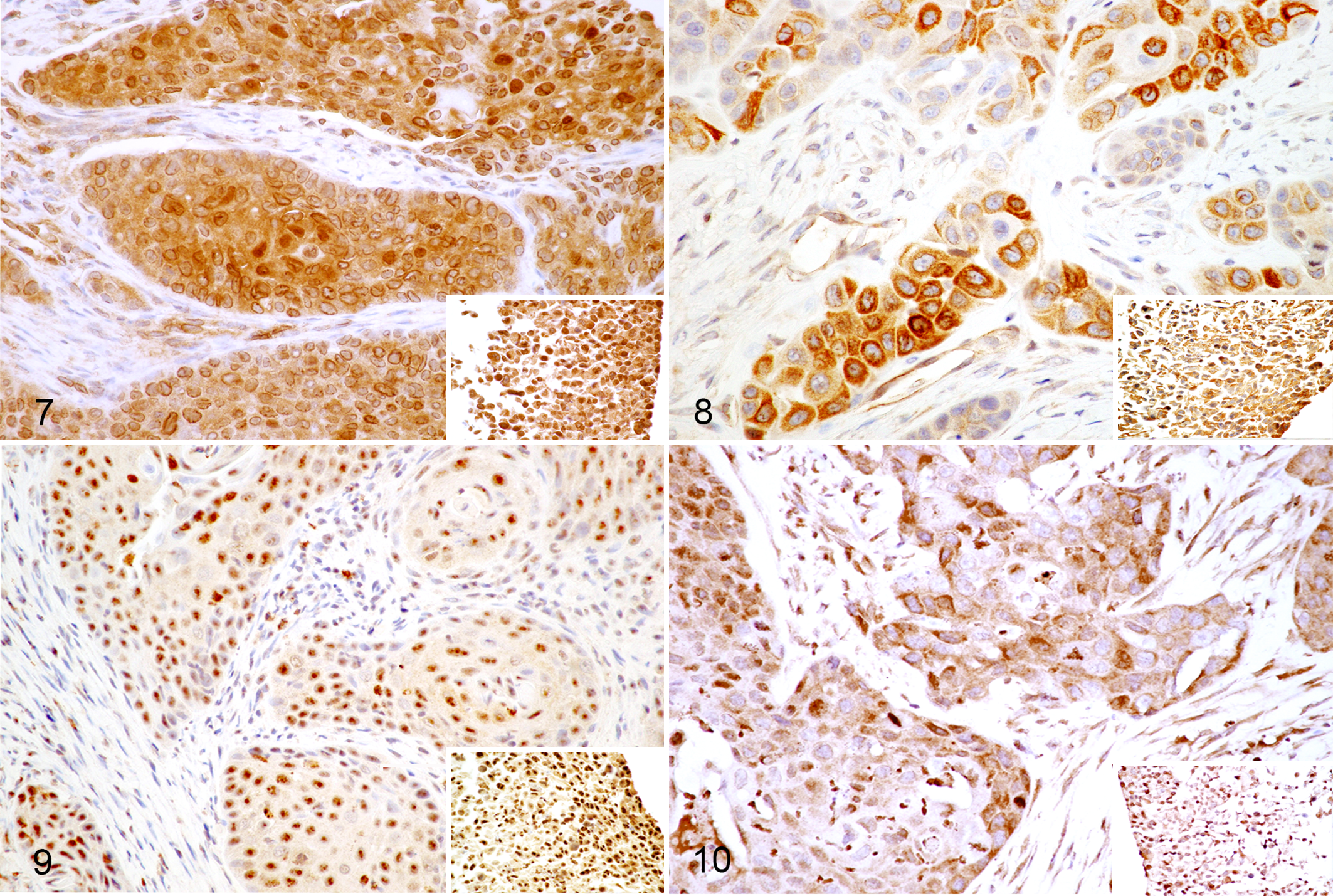
PD-L1, EGFR
PD-L1 was detected in nuclei of all 10 biopsies and 3 cell lines (Fig. 9). The cell lines had the greatest percentage of immunolabeled cells, ranging from 80% to 100%. Membranous expression was not detected in cell lines or biopsies; occasional cytoplasmic labeling was present. EGFR labeling was present in all 13 samples (Fig. 10). Perinuclear labeling was seen in 10% to 60% of cells in 4 biopsies, and nuclear labeling was found in cells from 5 biopsies, 50% to 80% positive. There was equal cytoplasmic and nuclear labeling in biopsy No. 1 as well as 2 cell lines (SCCF2, SCCF3), and EGFR labeling was primarily nuclear in 30% of SCCF1 cells. Membrane expression was not detected. EGFR was found in all specimens in the cytoplasm (perinuclear), nucleus, or both.
VEGF-D Western Blot
Cell lysates from the 3 cell lines incubated in normoxic (21% O2) and hypoxic (1% O2) culture conditions were probed for expression of VEGF-D using Western blot (Fig. 11). All 3 cell lines expressed VEGF-D under normoxic incubation, but in hypoxic culture conditions, there was a marked increase in VEGF-D expression by SCCF1 cells (Fig. 11). Under normoxic culture conditions, the SCCF1 cell line had the lowest percentage of cells with membrane expression of VEGF-D by IHC (10%) yet exhibited more than a 2-fold increase of VEGF-D expression when stressed in a hypoxic environment (Fig. 12). Conversely, IHC revealed SCCF2 and SCCF3 cells from normoxic cultures exhibited high percentages (60%, 100%) with membrane VEGF-D but only slightly increased VEGF-D expression in hypoxic conditions (Fig. 12).
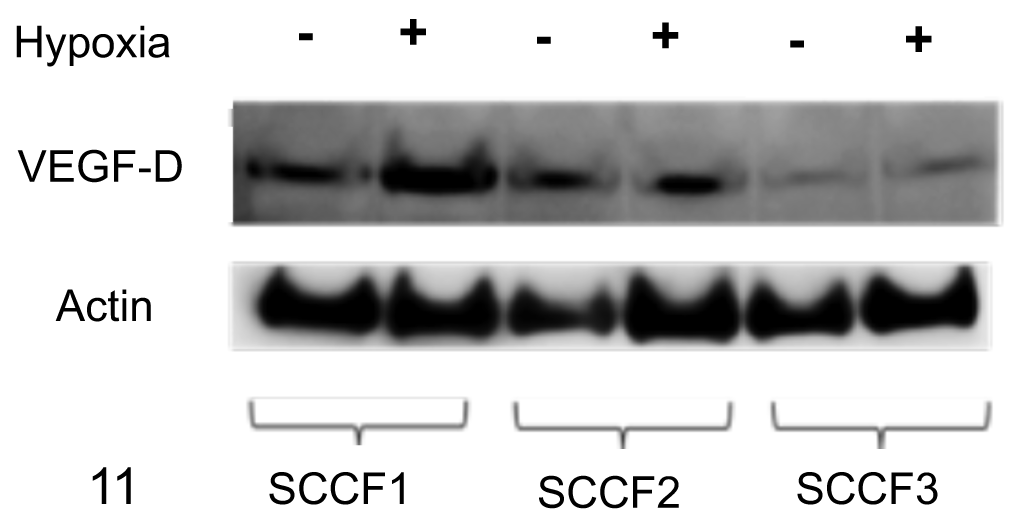
Vascular endothelial growth factor (VEGF)–D western blot in feline oral squamous cell carcinoma cell lines. Hypoxic culture conditions upregulate expression of VEGF-D in the feline oral squamous cell carcinoma cell line SCCF1 with minor increases in the cell lines SCCF2 and SCCF3. Lysates from hypoxic and normoxic cell cultures were probed with anti–VEGF-D antibody. Actin served as the loading control.
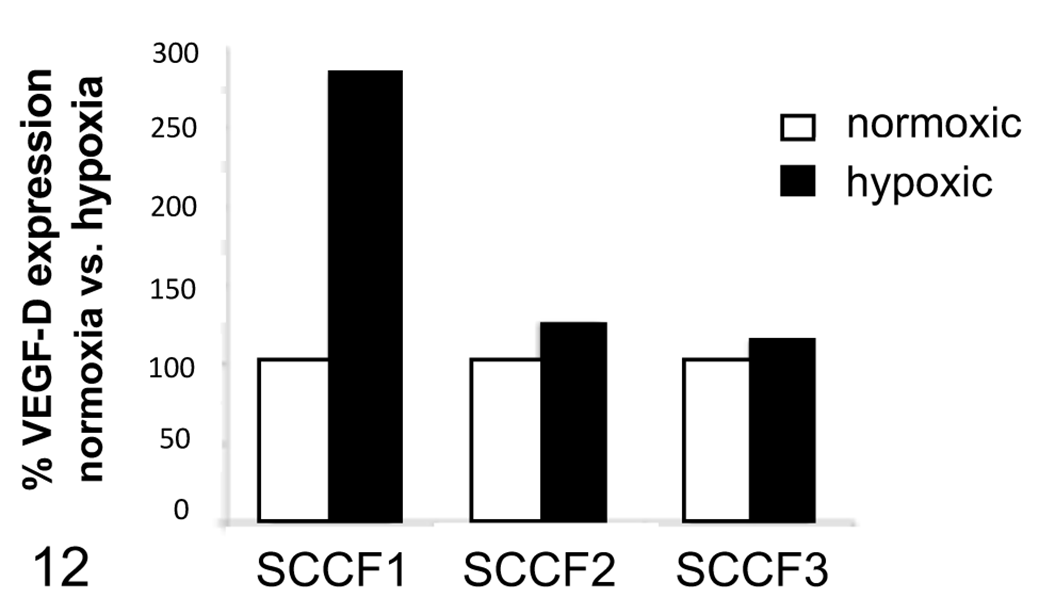
Densitometry of bands in western blot (Fig. 11). Basal (untreated) vascular endothelial growth factor (VEGF)–D production by cells from normoxic cultures normalized to 100% (white bars) and percent increase by hypoxic cells shown as percent of basal level (black bars). There was a 2.5-fold increase of VEGF-D expression by hypoxic SCCF1 cells, with slight increases by SCCF2 and SCCF3 cells.
VEGF ELISA
VEGF-A production by the 3 FOSCC cell lines was determined using ELISA (Fig. 13). SCCF1 and SCCF2 cell lines exhibited low levels of VEGF-A production when cultured aerobically in serum-starved medium (R0, Fig. 13). The SCCF3 cell line, on the other hand, had high basal VEGF production. When EGF was included in the culture medium, SCCF1 and SCCF2 lines had marked increases in VEGF production (Fig. 13). The SCCF3 cell line had a modest increase in VEGF production secondary to EGF stimulation. When dasatinib, a targeted small molecule that inhibits signaling by Src kinase was added, VEGF production was markedly suppressed in SCCF1 and SCCF2 cell lines and, to a lesser degree, in SCCF3, despite concurrent stimulation by EGF (Fig. 13).
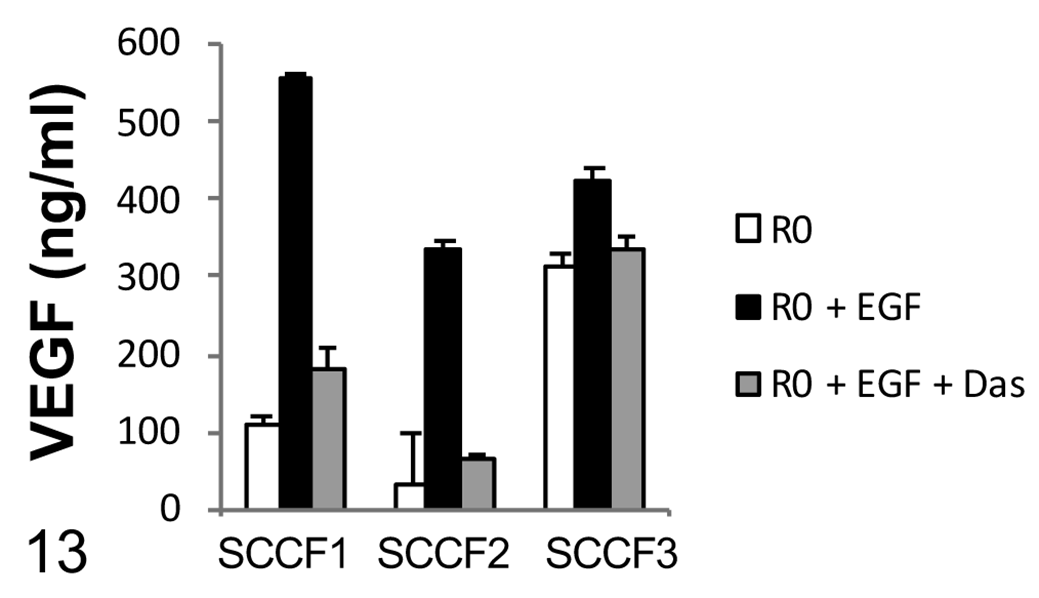
Vascular endothelial growth factor (VEGF) production in feline oral squamous cell carcinoma cell lines. Cells from each cell line were seeded into microwells in serum-free medium (R0, white bars); epidermal growth factor (EGF, 10 nM, black bars) was added to some wells. After overnight incubation, dasatinib (Das, gray bars) was added to some of the EGF-treated wells and incubated for an additional hour before quantitating VEGF in supernatants. There were marked increases in VEGF production by the SCCF1 and SCCF2 cell cultures stimulated by EGF. Dasatinib had a marked inhibitory effect on EGF-stimulated VEGF production with concentrations returning to near-basal levels in all cell lines. Bars represent the mean ± SEM of triplicate cultures.
Discussion
FOSCC is an aggressive malignancy that resists long-term control regardless of treatment modality. Carcinomas that invade adjacent tissues and resist chemo- and radiotherapy have morphed through EMT to express features characteristic of aggressive mesenchymal neoplasia. These features are well described in human HNSCC. 80 This study investigated EMT in FOSCC to begin to determine expression of cell proteins characteristic of resistant transformed cells in this malignancy. Phenotypic protein footprints of EMT can be detected in cells by various methods. The study used IHC to probe FOSCC biopsies and cell cultures for membrane and intracellular proteins linked to EMT in transformed human cancer cells, particularly human HNSCC. 15,38 These included vimentin, E-cadherin, N-cadherin, P-cadherin, and transcription factors Twist and Snail. In addition, we also examined HIF-1α and PD-L1 expression as both are recognized contributors to the EMT phenotype.
We were unable to examine the invasive front of the biopsies since completely excised tumors were not generally submitted by the primary care clinicians. This is relevant because mesenchymal changes in EMT transdifferentiation are more pronounced at the tumor’s invasive front, but results from this study suggest EMT changes can also be detected in other locations within FOSCC tumors. 15,16,46,65 Next steps for follow-up studies should include larger tissue samples to enable examination of the invasive front and metastatic tissue to determine if EMT changes at the primary FOSCC tumor site are carried forward to metastatic sites. This would be particularly interesting in light of a report that showed metastatic canine mammary carcinoma cells had lower Snail (and Slug) expression than the primary tumor. 82
E-, P-, and N-Cadherin
Cadherin switching is a hallmark of EMT. Cadherin expression varies during embryological development to allow cells to move and shift locations, changing expression of cadherin isoforms within adherens junctions affecting cell-to-cell contact. Changes in cadherin isoform expression are termed cadherin switching. 111 In cancer as part of EMT, malignant cells use the same mechanisms to invade surrounding tissue by undergoing cadherin switching. In this study, only the SCCF3 cell line expressed N-cadherin, with 50% of cells immunolabeling. Its expression was low in the biopsies and absent in the other cell lines.
P-cadherin, on the other hand, was expressed by the 3 FOSCC cell lines and cells from 9 biopsies. Only 1 biopsy was negative for membranous P-cadherin. These findings show membrane expression of P-cadherin was common in the FOSCC cells examined. P-cadherin expression is associated with the invasive, metastatic phenotype in numerous human carcinomas and is considered a reliable indicator of EMT. 88 E-cadherin expression, without appreciable N-cadherin expression in all biopsies and cell lines, does not rule out EMT. These cells could be a metastable phenotype known to express E- and P-cadherin concurrently. In fact, E-cadherin expression was shown to be present in high-grade canine mammary malignancies with histologic and IHC features of EMT. 82 Similarly, E-cadherin expression in feline cutaneous squamous cell carcinoma predicts a poor prognosis. 82,86 High P-cadherin expression with stable E-cadherin and low N-cadherin expression, as seen in FOSCC cells in this study, is a common phenotype in human oral squamous cell carcinoma. 88 While the significance of P-cadherin expression by FOSCC is unknown, P-cadherin has been recognized as a predictor of poor prognosis in human and feline mammary malignancies. 22,92
The data reported herein suggest that P-cadherin expression may be common in FOSCC. The finding in this study that cadherins identified in FOSCC mimic those of human HNSCC may suggest shared biological perturbances for this malignancy, further supporting FOSCC as a model for human HNSCC. 60 P-cadherin’s interactions with subcellular signaling partners is another area to be explored. Targeting vulnerabilities in subcellular pathways is an area of interest for development of new and more effective therapies for FOSCC. Suppression of VEGF production from FOSCC cell lines by the Src inhibitor dasatinib, shown in this report, is such an example. Targeting STAT3 with a molecular inhibitor blocked intracellular signaling and growth of identical FOSCC cell lines reported here. 8
HIF-1α, Twist, and Snail
The major driver of tumor angiogenesis is HIF-1α expressed by hypoxic cells. 3,9,120 HIF-1α switches on VEGF production and activates multiple transcription factors, including Snail and Twist. They are highly active in the control of genes that promote mesenchymal transformation. 43,109
FOSCC biopsies and cell lines expressed nuclear HIF-1α, suggesting a hypoxic phenotype, in vivo and in vitro. Most biopsies and all cell lines expressed Twist in the nucleus. Hypoxia induces HIF-1α, which has a stimulatory effect on Twist expression. 121 In this report, HIF-1α expression correlated with expression of Twist in most FOSCC specimens. Twist’s roles as a trigger of EMT, inhibitor of apoptosis, and contributory effect toward cell stemness and drug resistance have generated enthusiasm for developing it as a therapeutic target in oncology. 49,68,124 Based on results of this study, FOSCC offers a unique opportunity to investigate novel therapies to suppress Twist.
Low Snail expression was detected in the nucleus in the minority of biopsies (3/10) compared to relatively high expression (10%–20%) in SCCF2 and SCCF3 cell lines.
What constitutes positive Snail labeling differs between investigators. For example, Snail immunolabeling was considered high when ≥10% of cells were positive in 2 separate reports. 66,115 Different antibody clones were used in each report. The anti-Snail antibody employed in this study was the same as that used in one of the prior studies. 115 In another investigation, gastric carcinoma cells were considered positive for Snail expression if >1% of cells labeled with the anti-Snail antibody (ie, any immunolabeling). 99 Based on these studies, the 3 FOSCC cell lines and 3 biopsies showed high expression of Snail. The low thresholds for a positive Snail result imply Snail is not highly expressed, even in tissues that have undergone EMT. Alternatively, differences in antibody binding affinities might account for the discrepancy, or the biology of Snail and Twist differs so that Twist expression is more abundant than Snail in certain instances.
However, this result (high Twist and low Snail expression) matches that of human HNSCC. 31 Interestingly, the high Twist, low Snail, high P-cadherin, low N-cadherin, and high E-cadherin expression seen in the FOSCC of this report is also seen in human HNSCC. 80 This overlap may represent a unique signature of oral squamous cell carcinoma in EMT transformation.
VEGF
Hypoxia induces angio- and lymphangiogenesis, which is largely mediated by VEGF. VEGF’s role in cancer growth and metastasis is indisputable. We were interested in determining VEGF-D expression in FOSCC because it is an isoform of VEGF that is both angiogenic and lymphangiogenic. 95,114 When metastasis occurs, FOSCC spreads via lymphatic channels, and we therefore hypothesized a role for VEGF-D.
IHC detected membranous expression of VEGF-D in the 3 cell lines and in 4 biopsies. In a preliminary unpublished study of 32 FOSCC biopsies, we found 20 tumors (62%) expressed VEGF-D when examined by IHC with the same anti–VEGF-D antibody used in this report. Here, 40% of the biopsies expressed VEGF-D. Taken together, VEGF-D expression appears to be a common feature in FOSCC. Furthermore, we found that subjecting FOSCC cell lines to hypoxic stress increased their expression of VEGF-D (Fig. 11). VEGF-D is likely not the only VEGF isoform induced by hypoxia, but growing interest in developing it as a therapeutic target in cancer stimulated our interest, especially since there is no prior published information about feline VEGF-D to our knowledge.
VEGF-D’s enhancement of lymphatic metastasis has prompted development of therapies targeting the VEGF-D/VEGF-3R signaling axis. These include VEGF-D–specific blocking monoclonal antibodies, analogous to bevacizumab developed to block VEGF-A–induced angiogenesis, antibody blockade of VEGF-3R, and targeting proprotein convertases responsible for VEGF-D processing. 37,56,61,94
An exploratory experiment was conducted looking at VEGF-A production by FOSCC cell lines. A commercial ELISA kit with specificity for VEGF-A, but no other VEGF isoforms, was used to quantitate secreted VEGF-A by FOSCC cell cultures to begin to answer questions about production, stimulation, and suppression of VEGF-A. The kit was validated for recognition of feline VEGF-A by the manufacturer. VEGF-A is a major driver of angiogenesis and binds the VEGF-2 receptor but not VEGFR-3/Flt4. These 2 features differentiate it from VEGF-D, but both isoforms play important roles in malignant neovasculature formation. Basal VEGF-A secretion by all cell lines was identified to varying degrees. Activation of EGFR by its ligand, EGF, induces VEGF production. As predicted, production of VEGF-A was augmented when EGF was added to cultures; however, unexpectedly, it had dramatic effects in 2 cell lines, inducing 5 to 11 times more VEGF than unstimulated cells. IHC demonstrated that the FOSCC cell lines expressed EGFR, consistent with previous reports. 55,86 Binding of EGF to EGFR in FOSCC cells during culture activated multiple internal pathways upregulating VEGF expression. 118 These findings, although preliminary, suggest EGF could play a role in facilitating FOSCC survival in the hostile tumor microenvironment. On the other hand, the results present a rationale to target EGFR therapeutically in FOSCC.
Particularly striking was the suppressive effect of dasatinib on cellular production of VEGF that could override the stimulatory effect of EGF in the cell cultures (Fig. 13). Dasatinib is a tyrosine kinase inhibitor that prevents activation of intracellular Src, blocking phosphorylation of binding sites used for Src signaling. Full activation of EGFR requires crosstalk with activated Src. 51,110 VEGFR is not a known target of dasatinib. Its potent inhibitory effect on upregulated VEGF secretion may be attributed to downstream ripple effects of the Src blockade that interfere with VEGF production or failure of EGFR to become fully activated by Src. Dasatinib could also have had a suppressive effect on TGF-β, a cytokine stimulatory to EMT; it has been shown to reverse EMT. 5
These findings bear follow up because dasatinib is now being used therapeutically in veterinary oncology and may have a role in the treatment of FOSCC similar to interest in it for human HNSCC. 17,58,59 These data suggest dasatinib could be an inhibitor of feline Src as it is in other species, but further investigation is needed to clarify its effect in FOSCC. Additional actions of dasatinib against other Src-family kinases continue to be discovered that may contribute to inhibiting EMT. For example, dasatinib has been shown to promote formation of adherens junctions, increase cell-to-cell adhesion, and demonstrate activity against murine cutaneous squamous cell carcinoma through inhibition of Fyn kinase, a Src-family member. 5,19
PD-L1
PD-L1 was expressed in the nucleus of all samples, but it was not found in any other cell compartment. Nuclear PD-L1 expression is well described in numerous human malignancies in EMT-transformed cells, including HNSCC, colon cancer, and prostate cancer. In each malignancy, nuclear PD-L1 was a negative prognostic factor associated with shortened survival times. 71,89 Cells with nuclear PD-L1 may be associated with loss of cell-cell contact. 89 Surface expression of PD-L1 is downregulated while nuclear expression increases in human breast cancer cells treated with doxorubicin, endowing cells with chemoresistance. Nuclear PD-L1 expression, in association with markers of EMT, represents a highly chemoresistant phenotype. 29
PD-L1 expression has been associated with EMT gene expression signatures in HNSCC and esophageal carcinoma. Patient outcomes were worse when cells expressed PD-L1, especially at the invasive front, where cells exhibited phenotypical features of EMT. 35,71,103 Our sample size was relatively small, and it was not possible to say with certainty that nuclear localization of PD-L1 in FOSCC cells is a uniform feature of EMT in FOSCC without interrogating a larger sample size.
In contrast, PD-L1 has received much attention as a checkpoint target for immunotherapy when expressed on cell membranes of malignant cells. Surface PD-L1 can engage the PD-1 receptor on the membranes of cytotoxic lymphocytes, triggering their death by apoptosis. In this way, membrane PD-L1 helps the cancer cell to avoid immune destruction. 71
Monoclonal antibodies (eg, pembrolizumab and nivolumab) that block interaction between PD-L1 on the surface of the tumor cell with the PD-1 receptor on immune cells have revolutionized human cancer immunotherapy. These checkpoint inhibitors have shown promise in human HNSCC, significantly extending the life span of some patients with recurrent HNSCC, but only when ≥50% of tumor cells expressed PD-L1 on the membrane. Patients with surface PD-L1 expression <50% did significantly worse. 14 These results indicate membrane expression of PD-L1 in human HNSCC is heterogeneous. A study of nearly 100 human HNSCCs showed only 25% were positive for membrane expression of PD-L1. 97 Examination of a larger FOSCC sample size would help to clarify if the same is true for FOSCC and whether PD-L1 inhibition could be a relevant treatment strategy for FOSCC.
This study has shown that EMT is a common feature of FOSCC. This conclusion is based on expression of specific membrane, nuclear, and cytoplasmic changes reflecting increased expression of proteins recognized as markers of EMT in human malignancies. While classical upregulation of N-cadherin and vimentin has served as a canonical criterion for EMT, relying exclusively on their expression may be overly restrictive given the role of other proteins that are unambiguously associated with the EMT phenotype. Restricting the diagnosis of EMT to high expression of N-cadherin and vimentin overlooks the variability of their expression within a given tumor as was seen here as well as in human HNSCC. This could result in underestimating predicted malignant behavior if other EMT markers, such as P-cadherin, PD-L1, Twist, and Snail, are not considered.
Although preliminary, the effect of dasatinib on blocking VEGF production by FOSCC cells with an EMT phenotype was unambiguous and robust. Extending this line of research could help to develop dasatinib as a treatment for FOSCC.
This study employed a panel of antibodies for IHC to investigate EMT protein expression in FOSCC biopsies and cell lines. The proteins evaluated have been associated with EMT in other biological systems and species. The consistency of protein expression between samples examined could suggest an “EMT phenotype” in FOSCC. Prospective interrogation of clinical FOSCC biopsies for EMT markers described in this report, correlated with clinical outcomes of treated cats, would provide a more complete understanding of how EMT transformation relates to FOSCC prognosis. This information could also facilitate exploration of novel therapies such as targeted small molecules that would be selected for specificity with knowledge of candidate proteins to target. The behavioral changes that EMT confers on carcinoma cells help to explain the aggressive clinical course of FOSCC. This study begins to shed light on proteins that could mediate the process.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985819859873 - Immunohistochemical Features of Epithelial-Mesenchymal Transition in Feline Oral Squamous Cell Carcinoma
Supplemental Material, DS1_VET_10.1177_0300985819859873 for Immunohistochemical Features of Epithelial-Mesenchymal Transition in Feline Oral Squamous Cell Carcinoma by Krystal Harris, Howard B. Gelberg, Matti Kiupel and Stuart C. Helfand in Veterinary Pathology
Footnotes
Acknowledgements
We thank Dr. Erin Dickerson for providing lysates from cells cultured in hypoxia and Dr. Thomas Rosol for providing feline oral squamous cell carcinoma cell lines. Kevin Marley contributed technical support.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We are grateful to the Morris Animal Foundation for support of this research by grant D14FE-015 (SCH).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
