Abstract
Coxiella burnetii is an obligate intracellular pathogen and the cause of Q fever in many animal species and humans. Several studies have reported the association between C. burnetii and abortion, premature delivery, stillbirth, and weak offspring. However, no solid evidence indicates that C. burnetii causes endometritis, subfertility, and retained fetal membranes. For this study, histopathological and PCR evaluation were performed on 40 uterine biopsies from dairy cattle with poor fertility. Uterine swabs were concurrently tested with microbiology assays. The endometrial biopsies of 30 cows did not have any significant lesions, and no pathogens were identified by aerobic bacterial culture and PCR. Ten cows were PCR-positive for C. burnetii and negative for other pathogens by aerobic bacterial culture and PCR. These 10 cases revealed a mild to severe chronic endometritis admixed with perivascular and periglandular fibrosis. Immunohistochemical evaluation of C. burnetii PCR-positive biopsies identified, for the first time, the presence of intralesional and intracytoplasmic C. burnetii in macrophages in the endometrium of cattle.
Coxiella burnetii is a zoonotic, Gram-negative, obligate intracellular bacterium causing Q (Query) fever in humans. 6 Human and domestic animal infections may occur when contaminated dust or aerosols are inhaled. Infection apparently persists indefinitely in sheep and cattle, and bacteria are shed at parturition and via milk. 1 In cattle, Coxiella burnetii infection is generally asymptomatic, but it can also be associated with reproductive disorders. 1 The outcome of an infection of the pregnant uterus can lead to abortion, premature delivery, stillbirth, and weak offspring in addition to clinically normal progeny that may or may not be congenitally infected. 1 Several studies have addressed the possible associations between C. burnetii and a range of other reproductive conditions such as retained fetal membranes, 19 poor conception rates and calving outcomes, 18 infertility and sterility, 10 and endometritis/metritis. 18 However, there is no solid evidence of a causal relationship with these reproductive outcomes. 1 The aim of this study was to describe the presence of C. burnetii in macrophages at the site of inflammation in infertile dairy cows.
We examined 40 Holstein-Friesian cows reared in Campania region, Italy. The average age of the cows was 36 months old with a mean body condition score of 3.25 out of 5.0. Cows were submitted for veterinary examination due to repeat breeding. Clinical data are summarized in Table S1.
After the clinical assessment, uterine swabs and subsequently endometrial biopsies were collected 258 ± 58 days postpartum from each cow using a well-established method 4 to perform microbiological, histological, and immunohistochemical analysis. Endometrial biopsies were sectioned into 2 pieces and placed in a 50 ml tube for preservation in 10% neutral buffered formalin or cryopreserved in liquid nitrogen and stored at –80°C for further processing and examination. Total genomic DNA was extracted from frozen tissue samples using the QIAamp DNA mini kit (Quiagen S.r.l. Milan, Italy) according to the manufacturer’s protocol. DNA concentration was determined by UV absorption. Finally, genomic DNA was stored at –20°C until their use in PCR assays.
The infectious agents investigated for this study and the aerobic bacteriological and molecular methods used to detect them 8,9,13,14,17 are reported in Table 1. Examination for anaerobic bacteria was not performed. For histopathology, formalin-fixed and paraffin-embedded 4-μm sections were stained with hematoxylin and eosin (H&E) for morphological evaluation, Macchiavello stain for Rickettsiae, and Van Gieson to assess the grade of fibrosis. Histopathology evaluation was performed blinded to the results of microbiology assays. Endometrial lesions were classified 3 based on the quality of the biopsy, the epithelium, the inflammatory infiltrate in the stratum compactum (SC) and stratum spongiosum (SS) and the glands (criteria in Table S2). Inflammation was subjectively graded as none, mild, moderate, or severe, and the predominant cell type was assessed as mononuclear or segmented as previously described. 3
List of Infectious Agents Tested for the Study and Corresponding Detection Method.

Immunohistochemical analysis was performed with anti–C. burnetii Cb10B10 mouse monoclonal antibody, identified by Western immunoblot as an IgG1 that reacts with a 25–26-kDa band. 6 Immunohistochemistry was performed using horseradish peroxidase (HRP) method described elsewhere. 12 Antigen retrieval pretreatments were performed using a HIER citrate buffer pH 6.0 (Bio-Optica, Milan, Italy) for 20 minutes at 98°C. Primary antibody was diluted (1/1000) in PBS (0.01 M PBS, pH 7.2) and applied at 4°C overnight. Antigen-antibody binding was detected with a polymer detection kit (MACH1, Biocare Medical LLC, Concord, CA, USA) and labeled with 3, 3’-diaminobenzidine tetrahydrochloride (DAB) chromogen with hematoxylin counterstain. For each tissue section, 2 controls were performed incubating 1 section with PBS alone and the other with Negative Control Mouse IgG1 (NC490 AA, H, Biocare Medical LLC, Concord, CA, USA). For double color immunofluorescence staining, we used a procedure described elsewhere. 11 Slides were incubated overnight at 4°C with primary anti–C. burnetii Cb10B10 mouse monoclonal antibody diluted (1/1000) in PBS and a goat polyclonal anti-CD68 antibody (M-20: sc-7084, Santa Cruz Biotechnology, Inc, Dallas, TX, USA) diluted (1/50) in PBS for macrophage detection. Secondary antibodies for C. burnetii and CD68 comprised, respectively, a fluorescein isothiocyanate (FITC)-conjugated pure goat anti-mouse secondary antibody (Alexa Fluor® 488 conjugated, A-11029 Thermo Fisher PO Box 117 Rockford, IL, USA) diluted (1/100) in PBS and rhodamine (TRITC) conjugated donkey anti-goat IgG-TR (sc-2783 Santa Cruz Biotechnology, Inc, Dallas, TX, USA) diluted (1/50) in PBS.
Using the standard aerobic bacteriological culture, none of the 40 samples had growth. Coxiella burnetii was detected by PCR in 10 out of the 40 uterine biopsy tissues (25%). Interestingly, none of the other pathogens tested were identified. Moreover, no significant histological changes were observed in C. burnetii-negative endometrial biopsies. C. burnetii-positive uterine biopsies revealed the presence of mild to severe endometritis that was classified as class 1 to 3 according to the severity of the lesions (Figs. S1–S6).
In biopsies showing class 1 endometritis (2 cases), the luminal epithelium had 1 layer of columnar cells. The stratum compactum exhibited a chronic, mild, diffuse, interstitial infiltrate consisting mostly of macrophages, lymphocytes, and plasma cells. Few inflammatory cells were also found in the stratum spongiosum, mostly around the glands. Moderate numbers of small, round to tortuous glands were separated by mild stromal edema with small, regularly distributed blood vessels. The epithelium of the glands had a slight pseudostratification with occasional mitotic figures and a few clear cells. The glandular lumen was occasionally filled with a pale eosinophilic and amorphous material interpreted as uterine milk. Neither stromal (periglandular) nor perivascular fibrosis were observed. In biopsies with a class 2 endometritis (6 cases), the luminal epithelium was normal with a slight pseudostratification of columnar cells. A mild infiltration with lymphocytes and granular, pale pink fibrin deposits were also present on the epithelial surface. Both stratum compactum and stratum spongiosum had a moderate, diffuse, interstitial, periglandular and perivascular inflammatory infiltrate consisting of lymphocytes, plasma cells and macrophages. Endometrial biopsies also contained numerous round to tortuous, occasionally ectatic glands with a hyperplastic epithelium, containing thickened dark pink fluid. Furthermore, glandular nesting with a moderate stromal fibrosis was observed. In biopsies with a class 3 endometritis (2 cases), the luminal epithelium was multifocally necrotic and hyperplastic with numerous lymphocytes transmigrating the superficial epithelial surface. A severe, diffuse, and mixed inflammatory infiltrate consisting of lymphocytes, plasma cells, rare neutrophils, and macrophages was observed within the stratum compactum and the stratum spongiosum. Glands were round to tortuous, occasionally ectatic, and filled with amorphous and eosinophilic material. The epithelium was pseudostratified and infiltrated with mononuclear cells. Arteries and small arterioles had moderate to severe perivasculitis and mild vasculitis with transmural lymphoplasmacytic inflammation. Glandular nesting and a moderate to severe periglandular and perivascular fibrosis were also seen.
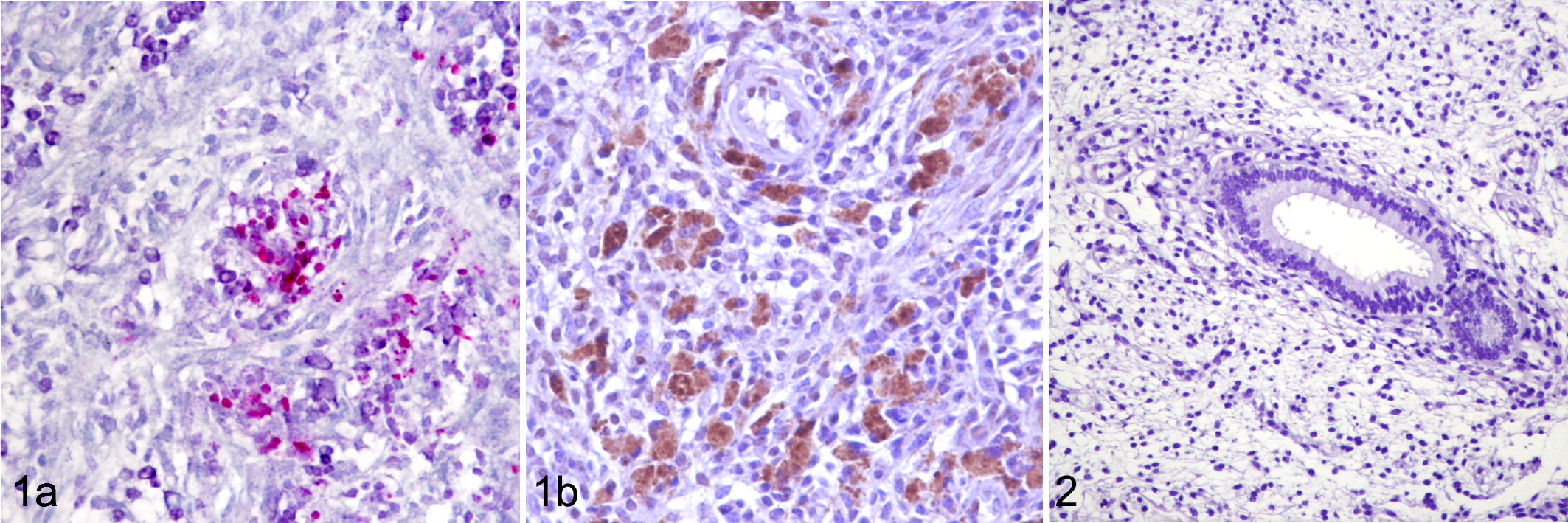
PCR C. burnetii-positive endometrial biopsies had bacteria positive both to Macchiavello stain and anti-C. burnetii antibody, within the cytoplasm of macrophages (Fig. 1a and 1b). None of the C. burnetii-negative endometrial biopsies had immunoreaction for C. burnetii antibody (Fig. 2). Negative controls had no immunoreactivity, confirming the specificity of the antibody (Supplemental Figure 7).
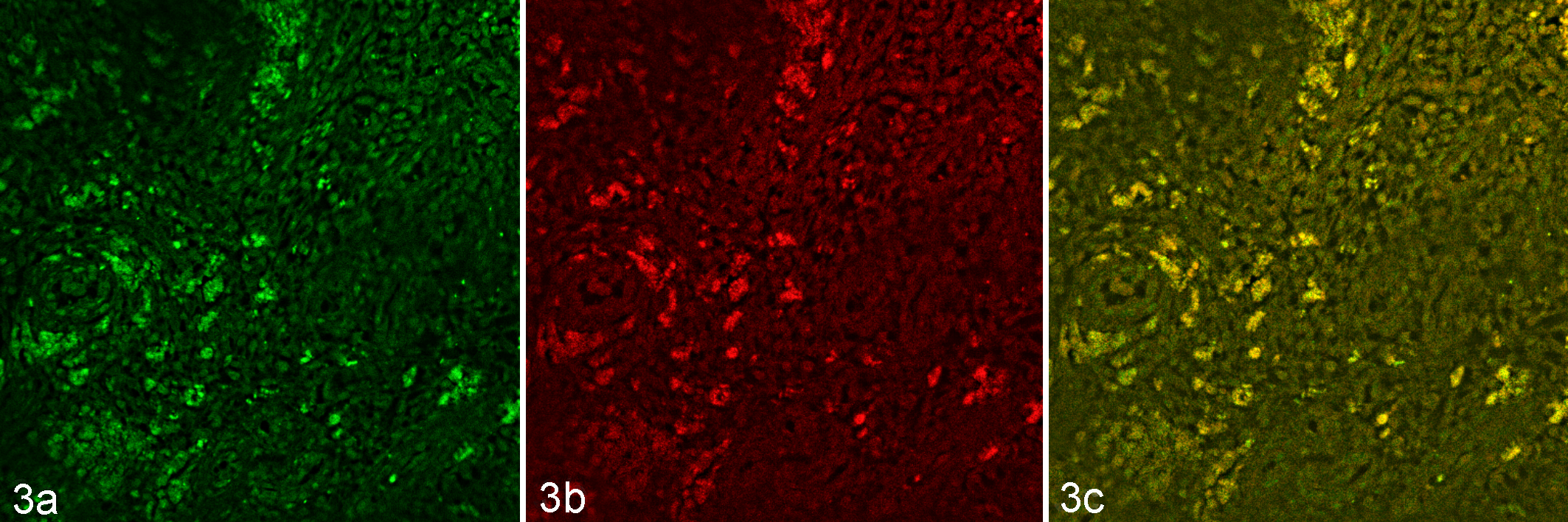
Doublecolor immunofluorescence revealed colocalization labeling with FITC-conjugated C. burnetii and TRITC-conjugated CD68 antibodies, evident as yellow fluorescence (Fig. 3a-c).
In cattle, natural infections with C. burnetii are generally not associated with significant clinical signs. Several studies have evaluated the association between infection with C. burnetii and a range of reproductive disorders other than abortion, but there was no evidence that C. burnetii alone could be the cause of disorders such as endometritis and subsequent poor fertility. 1 In this report we describe the identification of C. burnetii antigen within the macrophages in the areas of endometritis in infertile dairy cattle. This is the first report describing the presence of C. burnetii as associated with endometritis and uterine vasculitis and fibrosis.
In vertebrates, C. burnetii infects cells of the macrophage lineage 15 and has the ability to induce chronic persistent infections in humans and animals. 2,16 The strategies that C. burnetii uses to control the fate of the host cell have still to be completely understood. Several studies seem to indicate that C. burnetii infection inhibits apoptosis of macrophages, allowing bacterial survival and multiplication. 7,20 One of the proposed mechanisms for this anti-apoptotic effect is the inhibition of the release of cytocrome c that prevents the activation of executioner caspases. 7 The downregulation of macrophage responses to lymphokines and induction of suppressor factors as well as defective phagosome maturation and impairment of the antimicrobial activities may also allow C. burnetii to survive within these cells promoting the chronic inflammation. 5 Activated macrophages normally produce reactive oxygen and nitrogen intermediates and mediators of inflammation that play a pivotal role in the host response to bacterial infections. The persistence of bacteria-laden macrophages may contribute to chronic inflammation and to progressive damage and functional impairment of the infected tissue. In light of these considerations, the presence of C. burnetii within the uterine tissue may explain the lesions we observed and may also be related to a progressive reproductive disorder such as infertility in affected dairy cows. Our results seem to indicate that endometrial lesions can be observed in C. burnetii infection in the apparent absence of other pathogens, although it should be noted that the association of C. burnetii and endometritis does not prove causality. Furthermore, we show that endometrial biopsy may be a significant source of information regarding the endometrial status 4 and may help in the study of reproductive disorders; and that morphologic, immunohistochemical, and qPCR evaluation of endometrial biopsies seems to be a very promising tool for the diagnosis of bovine coxiellosis. Additional investigations may be able to elucidate how C. burnetii modulates the immune response in vivo in determining endometrial lesions.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985818760376 - Coxiella burnetii in Infertile Dairy Cattle With Chronic Endometritis
Supplemental Material, DS1_VET_10.1177_0300985818760376 for Coxiella burnetii in Infertile Dairy Cattle With Chronic Endometritis by Davide De Biase, Alessandro Costagliola, Fabio Del Piero, Rossella Di Palo, Domenico Coronati, Giorgio Galiero, Barbara Degli Uberti, Maria Gabriella Lucibelli, Annalisa Fabbiano, Bernard Davoust, Didier Raoult, and Orlando Paciello in Veterinary Pathology
Footnotes
Acknowledgements
The authors thank Mr Ilsami Raffaele for his valuable technical support and Prof Didier Raoult (Unité des Rickettsies, Faculté de Medecine, Université de la Méditerranée) who provided C. burnetii primary antibody.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was in part funded by “Progetto di Ricerca Corrente 06/2013 IZSM Portici.”
Supplementary material for this article is available online
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
