Abstract
Molar apical elongation (MAE) was the leading cause for euthanasia or death in a captive breeding colony of endangered Amargosa voles (Microtus californicus scirpensis). Clinical signs included ocular discharge, abnormal mastication, dyspnea, abnormal mentation, weight loss, and death. Although the severity varied, all molars in all quadrants were affected. When severe, the overgrown molar reserve crown and apex protruded into the nasal meatuses, the orbit, the calvarial vault and through the ventral margin of the mandible. Overall prevalence in the colony was 63% (92/146 voles) and increased to 77% in aged voles (>1 year). Mean age of onset was 5.3 months (1.7–11.2 months). Progression to extreme severity occurred over 1 to 3 months. Mean survival was 10.9 months (7.1–21.7 months). Histologically, the lesion was characterized by odontogenic hyperplasia and dysplasia. MAE was also documented in museum specimens of 2 other M. californicus subspecies (M. californicus californicus, M. californicus vallicola) and 3 other Microtus species (M. montanus, M. pennsylvanicus, M. socialis). In the M. californicus californicus collection, overall prevalence was 35.1% (129/368 skulls) and increased to 77.3% in aged voles (>1 year). A probable genetic influence was identified in the museum collection of M. californicus californicus. The etiopathogenesis of MAE is likely multifactorial, due to (1) inherent continuous odontogenic proliferation, (2) inadequate occlusal attrition, and (3) possible heritable disease susceptibility. In captivity, dietary or other management of occlusal attrition to prevent or delay MAE is a fundamental concern.
The Amargosa vole (Microtus californicus scirpensis) is an endangered desert subspecies of the California vole (M. californicus). It inhabits isolated, discontinuous bulrush (Scirpus olneyi) marshes that are scattered along the Amargosa River in the Central Mojave Desert (Inyo County, CA, USA) 7,34 in the vicinity of the towns of Shoshone (extirpated) and Tecopa. Considered extinct as recently as 1976 and “rediscovered” in 1979, the species is protected under the California 7 and United States 34 Endangered Species acts. Major threats include habitat loss or degradation, increased predation pressure, competition with nonnative species, infectious disease and a small population size with low genetic diversity. 7,34 Currently, as few as 50 to 500 Amargosa voles remain in the wild. 9
To minimize threats and maximize the recovery potential of the species, the United States Fish and Wildlife Service authored a recovery plan which proposed enhancement of existing Amargosa vole populations 34 through measures such as the establishment of a captive breeding program to not only supplement wild populations but also ensure against catastrophic loss in the wild. In July 2014, a captive breeding program composed of 20 wild-caught founders was installed at the University of California, Davis as a collaborative effort between the School of Veterinary Medicine, California Department of Fish and Wildlife, United States Fish and Wildlife Service, Bureau of Land Management and other partners in the Amargosa Vole Conservation Project. In July 2015, at 1 year into the program, cases of overgrown mandibular and maxillary molar reserve crowns and apices began to occur at an alarmingly high frequency in this captive population of Amargosa voles. This condition was termed molar apical elongation (MAE) and was associated with ocular discharge, abnormal masticatory behavior, labored breathing, abnormal mentation, weight loss, and death. Currently, MAE is the leading cause for euthanasia or death in the colony.
Open-rooted (continuously growing), hypsodont (high-crowned) or elodont incisor teeth are a defining feature of rodents. Like most other myomorph rodents, the dental formula of the Amargosa vole is 1/1 incisor, 0/0 canine, 0/0 premolar, and 3/3 molar teeth. 7,11 Unlike most other myomorph rodent species, voles also have elodont molar teeth that continuously grow and erupt throughout the life of the individual. 3 The arvicoline clade, which includes voles, has evolved in periods of repeated bursts of speciation, most likely related to major climatic fluctuations. 5 Over the last 3 million years, there has been a gradual shift from radicular (rooted, finite growth period) and brachydont (low-crowned) dentition to elodont dentition. As elodont dentition primarily developed about 0.5 million years ago, 28 the adaptation likely occurred in response to expansion of open grasslands and possibly more fossorial activity, conditions that would favor continuously growing molar teeth. Specifically, full elodont dentition is postulated to have evolved to compensate for elevated attrition associated with a highly abrasive natural diet. 11,27 Changes in tooth morphology may have come about through gradual changes in gene regulation. 31
In elodonts, spontaneous dental abnormalities would be expected with changes in attrition, particularly in captive circumstances with commercially formulated diets or suboptimal roughage and nesting materials. 2,6,11 Elongation of the apices of elodont teeth, whether incisor or molar teeth, is well recognized as an acquired dental disease in rabbits and a variety of pet rodents 2 and has been previously described in other arvicoline rodents. 11,12,15,18,29,30 The proposed pathophysiologic mechanisms vary from being a heritable trait 14,15,18 to an acquired disease process 1,2,14 to a multifactorial combination of both. 11,12,29
Because of the inherent implications to management of an endangered species, a detailed investigation into MAE was pursued in this captive breeding colony of Amargosa voles and in museum specimens of other Microtus voles. The purpose of this study was to morphologically and epidemiologically characterize MAE in this captive breeding colony of Amargosa voles, to ascertain whether MAE is unique to Amargosa voles and to investigate potential causes of MAE, such as a heritable trait, that would influence colony management.
Materials and Methods
Breeding Colony of Amargosa Voles
The captive breeding colony at the University of California, Davis was started in July 2014 with a founder population of 20 wild-caught Amargosa voles (M. californicus scirpensis). At the time of this report, the colony had been maintained for 4 generations and includes 146 individuals. The majority of the colony was housed indoors, in heavy plastic cages with metal mesh lids (Ancare, Belmore, NY). The room was temperature-controlled with a 12-hour light: dark cycle. Voles were housed either individually, with same-sex siblings or with a mate. Straw substrate (at least 10 cm deep) was provided for burrowing. A small subset of the colony (1–10 voles) was housed outdoors, in landscaped pens that were exposed to natural light, weather and temperature. The outdoor pens (mesocosms) were constructed of covered structural foam planters (Rubbermaid, Winchester, VA) filled with soil and planted bulrush clones from Tecopa, California. Outdoor females may have been paired with siblings. Outdoor males were housed alone. All voles were fed ad libitum with 2018 Teklad Global 18% protein rodent chow (Envigo) and provided water in water bowls. Outdoor voles were supplemented with planted bulrush. Water bowls were changed daily.
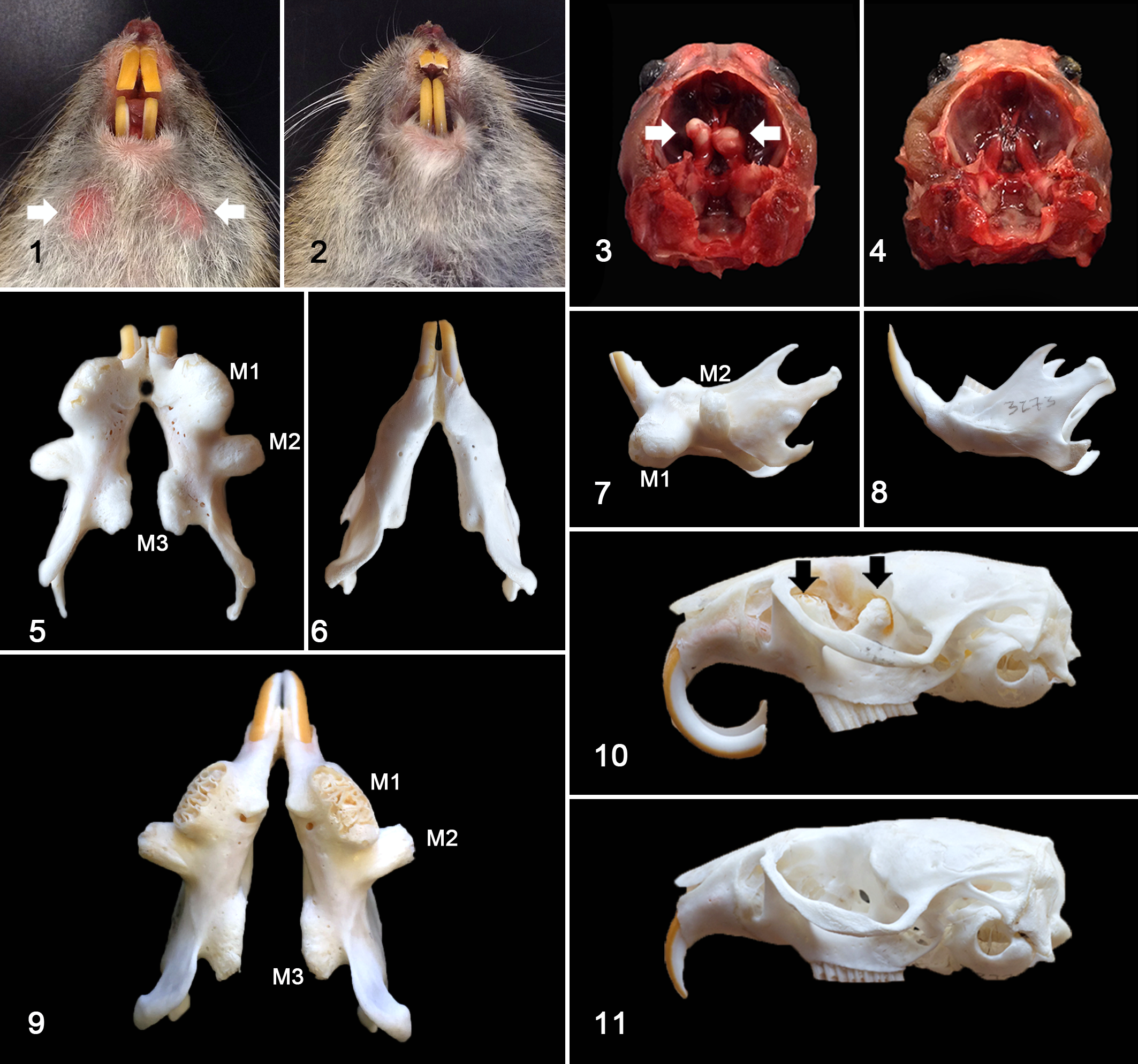
The presence and severity of hard protrusions (Fig. 1) from the ventral mandible was recorded for each individual Amargosa vole at monthly colony health assessments, over a time period of 21.7 months. Evaluations were performed by visual examination and manual palpation and categorized as follows: normal (N), palpable but not visible (P), visible (V) and visible extreme (VE).
Museum Collections of Microtus sp
Archived skulls of Microtus spp. (2 M. californicus subspecies and 3 other Microtus species) from the Museum of Vertebrate Zoology (MVZ) at the University of California, Berkeley were evaluated for dental abnormalities (Supplemental Table S1). The California vole (M. californicus) specimens originated from the San Francisco Bay area and were derived from two different breeding experiments (“Age Set” and “Buffy Set”) with complete age, sex, lineage and captive status data sets. Given the similarity in their origins and methods of rearing, the collections were combined for some analyses. The “Age Set” was originally used to create an algorithm for age prediction. 21 The “Buffy Set” was originally used to study vole coat colors, particularly a coat color referred to as buffy. 20 The meadow vole (M. pennsylvanicus) specimens came from a laboratory colony in the Psychology Department at the University of California, Berkeley. Age and sex data were not available for most specimens but all were assumed to be adults. The social vole (M. socialis) specimens were captive-reared from a laboratory colony in Iran (Supplemental Table S2). Age data were unavailable for this specific collection. The montane vole (M. montanus) and Owens valley vole (M. californicus vallicola) specimens originated from the same laboratory at University of California, Davis as the Amargosa voles and were housed under similar husbandry conditions.
Skulls from the California vole collection were visually examined for abnormalities in mandibular molars apical regions, maxillary molar apical regions, molar occlusal surfaces, and incisor teeth. Presence or absence of MAE was ranked 1 or 0, respectively. Abnormalities in the molar apical regions were ranked as follows: 0, no visible molar apical protrusions; 1, <0.2 cm long protrusions; 2, 0.2 to 0.3 cm long protrusions; 3, >0.3 cm long protrusions. As skulls were intact, apical regions of the maxillary molar teeth could not be evaluated in detail; therefore, abnormalities were ranked as follows: 0, no abnormalities; 1, abnormalities present. Molar occlusal surfaces and incisor teeth were ranked similarly.
Microcomputed Tomography (microCT)
Microcomputed tomography (CT) scans were performed at the Center for Molecular and Genomic Imaging (UC Davis) on an InveonCT scanner (Siemens Preclinical Solutions; Knoxville, TN). The standard resolution scans were acquired with the following parameters: 80 kVp, 420 uA, 925 ms per frame exposure, and 50 calibration images (light and dark fields). Four hundred and forty projections were acquired over 220 degree rotation. Scan time was approximately 23 minutes.
Reconstructions were performed with Cone Beam Reconstruction Algorithm (COBRA) using a FeldKamp algorithm with beam hardening correction. Resulting images had a corresponding voxel size of 0.048 mm × 0.048 mm × 0.048 mm. 3D renderings were generated using Drishti 2.4 (Vizlab, Australian National University).
Necropsy and Histopathology
A complete gross necropsy evaluation was performed on 29 of the 146 Amargosa voles in the colony. Skulls from 12 (Supplemental Table S3) of the 29 necropsied voles, in addition to routine sets of tissue, were collected for histopathology. Tissues were submersion-fixed in 10% neutral-buffered formalin for 72 hours. Skulls were decalcified in 15% formic acid for 48 to 96 hours, as needed. Skulls were sectioned along oblique parasagittal planes to capture the molar apices. Formalin-fixed and decalcified tissues were processed routinely, embedded in paraffin, sectioned 4–5 μm thick and stained with hematoxylin and eosin.
Mandibular and maxillary molar apices were evaluated and graded for the following features: hyperplasia, dysplasia and atrophy. Hyperplasia was defined by elongation of the apices with collapse of intercuspal loops, bony remodeling and irregular expansion of dentin deposits. Hyperplasia was graded as follows: 0, none; 1, minimal < 0.1 cm apical elongation with mild intercuspal loop collapse and minimal bony remodeling; 2, mild to moderate 0.1, 0.3 cm apical elongation with intercuspal loop collapse and bony remodeling; 3, moderate to marked > 0.3 cm apical elongation with intercuspal loop collapse, bony remodeling and broadening of dentin. Dysplasia was defined by the disorganization of cervical and intercuspal loop components (absence of normal architectural orientation) with abnormal matrigenesis and was graded based on percentage of apices involved: 0, none; 1, <25%; 2, 25–50%; 3, >50%. Atrophy was defined by the loss of germinal inner and outer enamel epithelium with replacement of pulp by reparative dentin. Atrophy was graded based on percentage of apical region involved: 0, none; 1, <25%; 2, 25–50%; 3, >50%.
Incisor apical abnormalities were not histopathologically evaluated as sections that targeted molar apices were not ideal for evaluating incisor apices. Observations of incisor abnormalities were limited to the changes grossly evident in the apices (bony protrusion or perforation) and clinical crown (elongation, recurvature, malocclusion).
Skull Preparation
A subset of 6 skulls (Supplemental Table S4) from 29 necropsied Amargosa voles was frozen and subsequently cleaned by a combination of dermestid beetle, maceration and chemical methods. Defrosted, partially desiccated skulls were stripped of soft tissues by dermestid beetles. Stripped skulls were cleaned in enzymatic detergent (Hard Rock laundry detergent, Rockin’ Green, Charlotte, NC) and bleached in 30% hydrogen peroxide.
Onset and Survival Analysis in Amargosa Vole Colony
Data from individual Amargosa voles were used for either or both onset and survival analyses. Criteria for inclusion in the onset analysis included (1) presence of the MAE, (2) monthly assessment for MAE, or (3) absence of MAE at first assessment. Criteria for inclusion in the survival analysis included (1) presence of MAE, (2) monthly assessment for MAE, (3) presence in the colony, or (4) spontaneous (noniatrogenic) cause of death or euthanasia. Number of months surviving was calculated as ([date of death – date of birth]/30). Survival curves for the Amargosa voles were generated in R, using the packages “survival” and “survminer.” Both Kaplan-Meier and Nelson-Aalen nonparametric estimators were used to analyze survival. Plots were generated with the R package “ggplot2.”
Pedigree analysis was not performed for Amargosa voles in the breeding colony because of the limited number of lineages (10 founder pairs), extensive intermixing between lineages and low numbers of voles per lineage.
Pedigree Analysis in California Vole Collection
Pedigree analysis for the California vole collection was performed using a previously validated mathematical modeling program, SOLAR (Sequential Oligogenic Linkage Analysis Routines) 10 and R, using the package “kinship2.” 32 SOLAR uses maximum likelihood estimation and tests for significance between a model with genetic variance of MAE set to zero and a model with genetic variance estimated. The trait, MAE, was tested against lineage.
Onset and survival analyses were not performed for the California vole collections as these data were not available (age of onset was not recorded) or was predetermined (voles were euthanized at designated time points based on experimental protocol).
Statistical Analysis
Statistical analyses were performed using GraphPad Prism 6.07 (La Jolla, CA). Ordinal data were evaluated by Fisher’s exact (contingency) or Mann-Whitney tests. Strength of association was analyzed by nonparametric Spearman correlation. P values < .05 were considered significant.
Results
Affected individuals exhibited bilaterally symmetric, hard protuberances that extended from the ventral aspect of the mandible (Fig. 1, an unaffected vole is provided for comparison in Figs. 2, 4, 6, 8 and 11). Similar bilateral hard protuberances extended from the floor of the calvarium, adjacent to the sella turcica, and into the vault (Fig. 3). Evaluation of prepared skulls established that the ventral mandibular protuberances corresponded to the elongated reserve crowns and apices of the first mandibular molar teeth (Figs. 5, 7, 9), with either deformity of (Fig. 5) or perforation through (Fig. 9) bone. The second and third mandibular molar teeth protruded from or through the lateral and medial aspects of the mandible, at an acute to right angle to the crown (Figs. 5, 7, 9). The first and second maxillary molar teeth extended into the nasal cavity and orbit (Fig. 10), respectively. Protrusion of the third maxillary molar teeth into the calvarium resulted in compression of the ventral aspect of the brain (Fig. 12), corresponding to the caudate/putamen and anterior hypothalamic regions. Incisor teeth abnormalities, such as recurved and splayed incisor teeth (Figs. 1, 10), fracture and malocclusion, were often but not always observed. No bony protrusion or perforation that would correlate with incisor apical elongation was observed. Other common necropsy findings included gastric trichobezoars. All voles evaluated were in adequate to good body condition. No history of inappropriate behavior (cage-chewing) or other cause for oral trauma was reported.
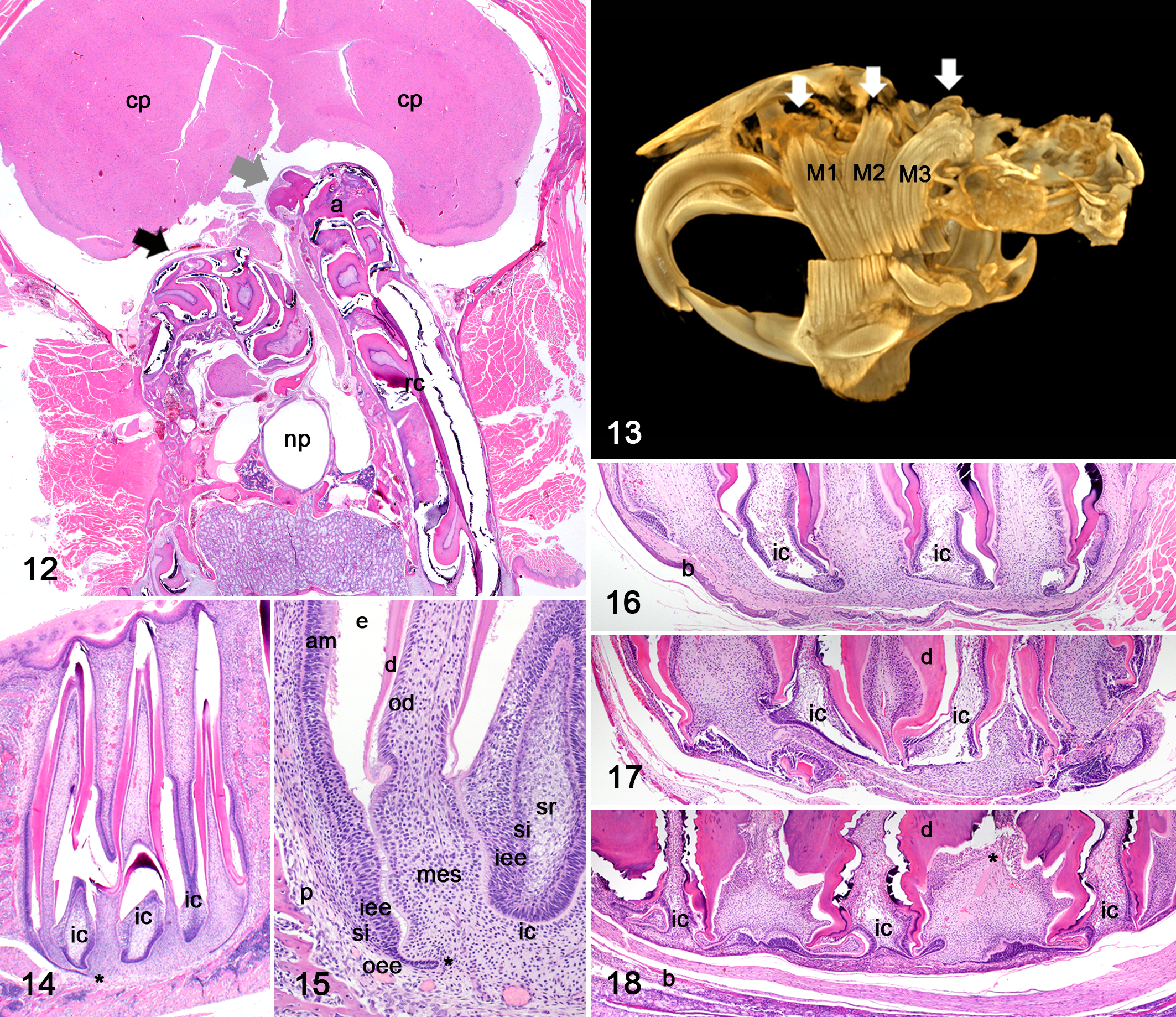
MicroCT analysis of an affected vole confirmed that the intracranial protuberances were elongated reserve crowns and apices of the third maxillary molar teeth (Fig. 13). The protuberances into the nasal cavity and orbit were confirmed to be the elongated reserve crowns and apices of the second and third maxillary molar teeth, respectively (Fig. 13). This imaging modality revealed that even individuals deemed unaffected on clinical and gross evaluation exhibited mild elongation of molar reserve crowns and apices (data not shown), in the absence of deformity of or perforation through bone.
The expected histologic appearance of the apices of an elodont tooth includes complex folds of apical odontogenic epithelium that form the cervical and intercuspal loops or apical buds 26 (Figs. 14, 15). The cervical and intercuspal loops begin as linear, hook to paddle-shaped projections that extend linearly downward into the dental mesenchyme. 33 In voles with mild disease, cervical and intercuspal loops were easily identifiable, though the reserve crowns and apices were elongated and the surrounding alveolar or cortical bone was remodeled to accommodate the apical growth (Fig. 16). In voles with marked disease, cervical and intercuspal loops were progressively compressed and distorted (Figs. 17, 18) with collapse and buckling of the inner enamel epithelium, irregular expansion and folding of dentin and shortening of the pulp cavity with more proximal or lower capping by reparative dentin (Fig. 18).
Dysplasia and atrophy were prominent features in moderate to advanced MAE. Dysplasia occurred in both mandibular and maxillary apices, tended to be associated with bony perforation or microfracture (P = .16), and was characterized by disorganization of cervical and/or intercuspal loop components with continued proliferation (Figs. 19, 20, 21). In 2 of 12 voles, there were cauliflower-like masses of well-differentiated but disorganized odontogenic epithelium intermingled with matrical conglomerates of dentin, reparative dentin, cementum, and enamel (Figs. 20, 21). Occasionally, cementum and dentin were directly opposed. These masses were consistent with elodontomas (previously known as and sometimes still referred to as pseudo-odontomas) and when present, were only observed in the apices of maxillary third molar teeth (Fig. 20). Atrophy was characterized by loss of odontogenic epithelium and extensive replacement of the dental mesenchyme with an admixture of dentin and reparative dentin (Fig. 22).
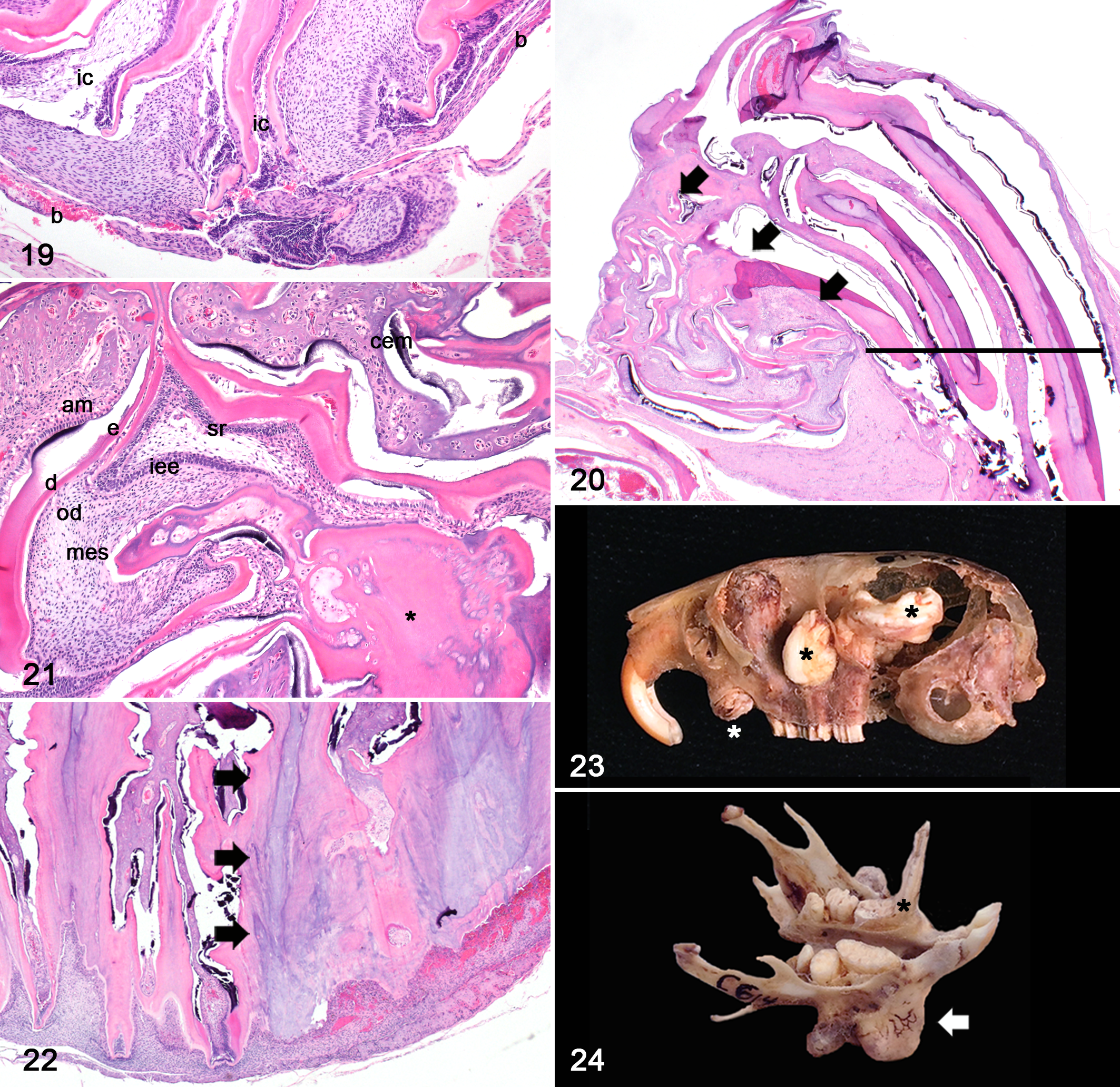
The histopathologic scoring system used to evaluate hyperplasia, dysplasia and atrophy directly and positively correlated (r = .89, P = .02) with the clinical scoring system developed for use during monthly health assessments (Fig. 25). No significance was found between the mandibular and maxillary molar apices in terms of the degree of apical odontogenic hyperplasia (P = .86), dysplasia (P = .85) or formation of elodontoma (P = .48) (Fig. 26). Atrophy tended to be more severe in overgrown mandibular molar apices than in maxillary molar apices (P = .10) (Fig. 26). The progressive elongation and impaction of molar teeth was associated with adjacent osteolysis and proliferative woven bone (secondary bony remodeling), obstruction of nasal passages and compression of soft tissues (including cranial nerves and the ventral aspect of the brain) (Fig. 12). Despite the marked compression of the ventral aspect of the brain, no reactive parenchymal changes (edema, neuronal necrosis, hemorrhage, gliosis, etc) were observed. Molar apical alveolar or periodontal inflammation (periodontitis) was only identified in one vole, affecting one mandibular molar tooth.
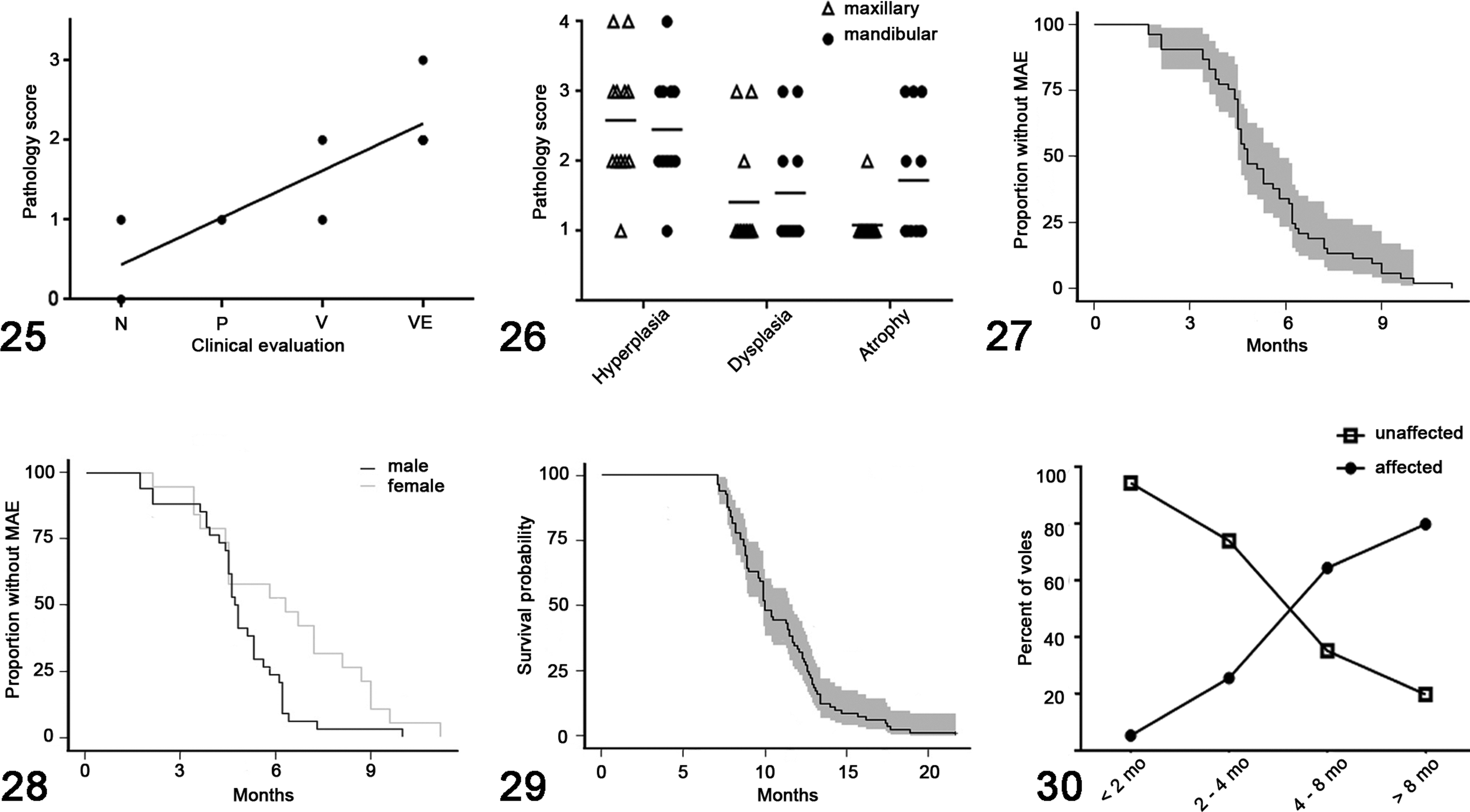
MAE was identified in 92 out of 146 voles (63.0%) during health assessments or postmortem examination. In voles greater than 1 year of age, 45 out of 58 voles (77.6%) were affected. Excluding capture and transport-related casualties, death or euthanasia due to clinical implications of MAE accounted for 49 out of 65 mortalities (75.4%) in the colony. Rate of progression varied. Some individuals progressed from mild (palpable but not visible) to moderate (visible) grades within 1 month, while others remained in the mild stage for up to 3 months before the condition progressed. In one individual vole, temporary regression was observed where severe (visible extreme) MAE regressed to undetectable (normal) disease over the span of 5 months. Following 8 months of undetectable disease, MAE became palpable again but the individual has not progressed to developing clinical signs and is currently alive (>21.7 months old).
Onset analysis was based on 53 of 146 Amargosa voles (19 females, 34 males) that fit inclusion criteria. Onset of MAE occurred at 5.3 months of age on average, ranging from 1.7 to 11.2 months (Fig. 27). On average, MAE occurred 1.5 months earlier (P = .024) in males (4.8 months) than in females (6.3 months) (Fig. 28). More than 50% of voles exhibited evidence of MAE by 5 months of age and by 8 months of age, 90% had MAE. Survival analysis was based on 81 of 146 Amargosa voles (29 females, 54 males) that fit inclusion criteria. Survival (or life expectancy) for voles with MAE was 10.9 months on average, ranging from 7.1 to 21.7 months. Mortality among Amargosa voles with MAE began to increase between 7 and 8 months of age (Fig. 29). Fewer than 10% of voles that developed MAE survived beyond 14 months. The longest-lived vole with MAE was 21.7 months but this animal experienced an unusual period of regression. The longest-lived vole with MAE that had not experienced a period of regression was 18.9 months. There was no significant difference in survival between males and females with MAE (P = .75).
In the museum specimens of captive laboratory-reared M. californicus, evidence of MAE was observed in 129 of 368 skulls (35.1%). In museum specimens of voles greater than 1 year of age, 17 of 22 skulls (77.3%) exhibited evidence of MAE. Incisor abnormalities (overgrowth, recurvature, fracture, malocclusion) were observed in 27 of 368 skulls (7.3%). In one individual (Fig. 23), incisor apical elongation with perforation through maxillary bone was observed. The average age of museum specimens of M. californicus with MAE was 7.3 months, ranging from 1.4 to 35.3 months. There was no significant difference in prevalence of MAE between male and female (P = .75) museum specimens of M. californicus. Age was the only significant risk factor; museum specimens of M. californicus older than 3 months were significantly more likely to exhibit MAE (P < .001) (Fig. 30). Museum specimens of M. californicus older than 4 months were significantly more likely to exhibit the severe form of MAE (P < .01) (Fig. 23). A genetic influence was present as heritability of MAE was significantly different from 0 in the Buffy (P < .001), age (P < .001) and combined sets (P < .001) (S1).
MAE was also identified in museum specimens from captive laboratory-reared collections of M. californicus vallicola, M. montanus, M. pennsylvanicus and M. socialis (Table 1). In the M. pennsylvanicus collection, MAE was identified in 29 out of 157 voles (18.5%). Mandibular molar apical abnormalities were more prevalent than maxillary apical abnormalities. With the exception of 2 individuals, maxillary MAE occurred only in voles where MAE was advanced in the mandible. MAE was observed in several M. socialis, M. montanus, and M. californicus vallicola specimens but evaluations were not completed for these collections.
Molar and Incisor Apical Elongation in Arvicoline Rodents.
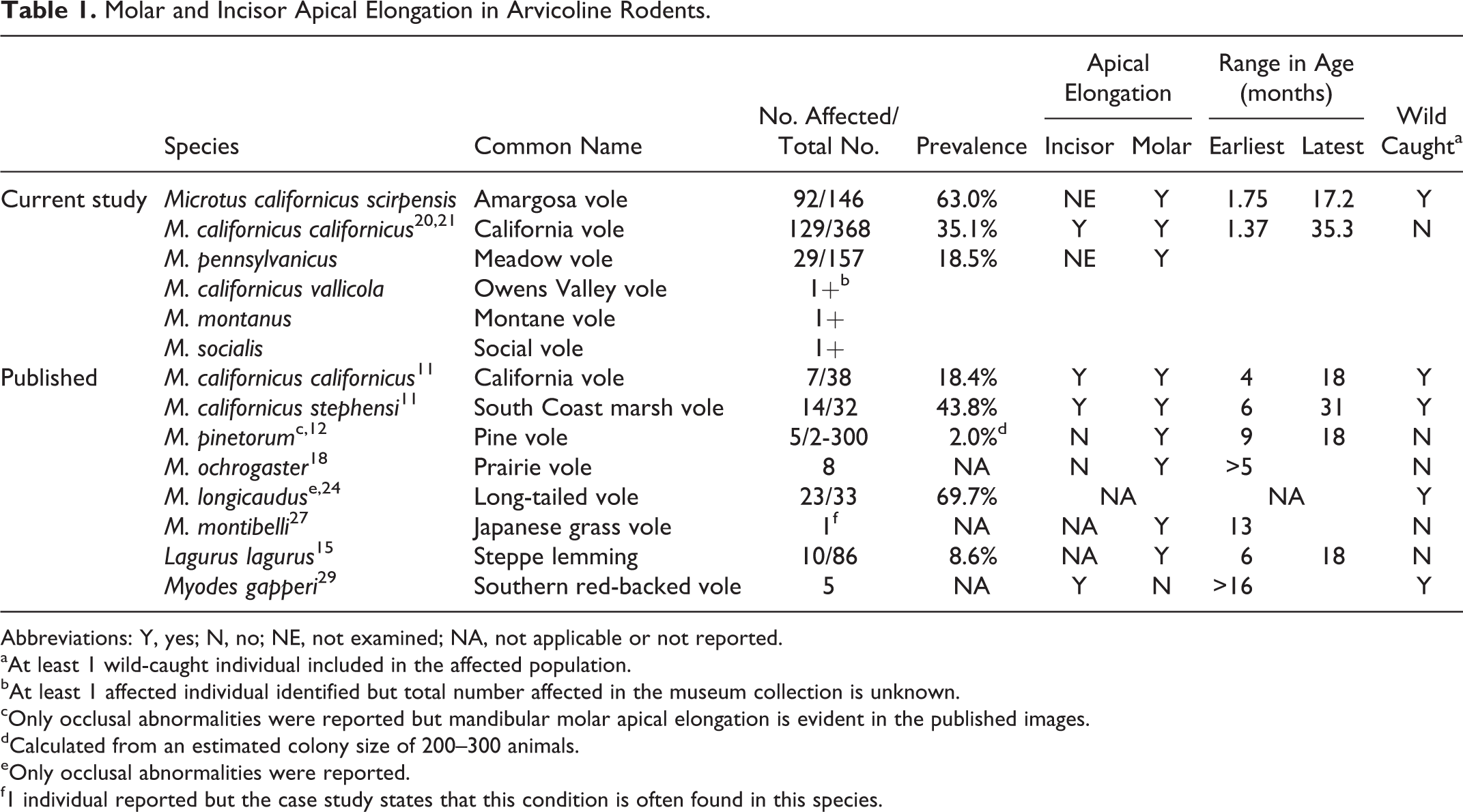
Abbreviations: Y, yes; N, no; NE, not examined; NA, not applicable or not reported.
aAt least 1 wild-caught individual included in the affected population.
bAt least 1 affected individual identified but total number affected in the museum collection is unknown.
cOnly occlusal abnormalities were reported but mandibular molar apical elongation is evident in the published images.
dCalculated from an estimated colony size of 200–300 animals.
eOnly occlusal abnormalities were reported.
f1 individual reported but the case study states that this condition is often found in this species.
Discussion
Molar apical elongation is a highly prevalent dental disease of arvicoline rodents, described here in a captive breeding colony of Amargosa vole (M. californicus scirpensis) and the museum specimens of California vole (M. californicus californicus). The disease was defined by elongation of the molar reserve crown and apex in the apical direction with secondary bony remodeling and soft tissue compression. Histopathologically, there was apical odontogenic hyperplasia and dysplasia with atrophy and occasional elodontoma formation. Compression of the caudate/putamen and anterior hypothalamus and obstruction of nasal passages were considered the direct causes of associated debilitation and mortality. Antemortem palpation and visual examination for mandibular MAE correlated well with histologic assessment of mandibular and maxillary MAE severity. In the California vole collection, age was the sole risk factor identified and though a heritable trait was suspected, the mode of inheritance could not be determined. MAE was also observed in museum specimens of Owens valley vole (M. californicus vallicola), montane vole (M. montanus), meadow vole (M. pennsylvanicus), and social vole (M. socialis).
Histologically, MAE spans a continuum of apical odontogenic hyperplasia and dysplasia. As MAE evolves, there is progressive apical odontogenic hyperplasia with reactive bony remodeling. Advanced stages involve collapse and deformation of the cervical and intercuspal loops, widening and plication of dentin and shortening of the pulp cavity as the apex is crowded against the continuously remodeling alveolar or cortical bone, features that are consistent with inhibited coronal eruption and subsequent apical compression or impaction against bone. 2 Dysplastic changes in this study tended to be associated with fracture or perforation through bone. Although dysplasia was present in both the mandibular and maxillary molar apices, the elodontomas occurred only in the third maxillary molars, perhaps due to a higher likelihood of fracture in maxillary versus mandibular bone. Atrophy tended to be more prominent in the mandibular molar apical regions than the maxillary regions and may reflect ischemic damage due to compression against the less compliant mandibular ventral cortex. 23 The lack of statistical significance in the association between (1) the presence of dysplasia and bony fracture/perforation, (2) the presence of elodontoma formation in maxillary versus mandibular molar apices, and (3) the presence of atrophy in mandibular versus maxillary molar apices was likely a limitation of the small sample size (N = 12).
Elodontoma was proposed as a term to represent locally proliferative masses of well-differentiated but haphazardly organized odontogenic epithelium and closely apposed dental matrix that develop at the apical bud of elodont teeth. 1 This term was considered the most appropriate for the histopathologic changes (Figs. 20, 21) observed in 2 of the Amargosa voles evaluated. Elodontoma was meant to replace the term odontoma to reflect a distinct difference in pathogenesis as elodontomas were considered nonneoplastic and may be traumatically induced. 1 The definition of elodontomas as hamartomas is contentious as the term hamartoma implies a developmental anomaly that has completely benign behavior. 25 Because the elodont apical bud is a site of continuous cellular proliferation (from the inner enamel epithelium of the cervical or intercuspal loop) as well as an adult stem cell niche (from the stellate reticulum), 26,33 an abnormal apical proliferation could be considered to occur “during development” as development never ceases at that site. However, a major point of contention is the proposed acquired, traumatic pathogenesis 1 as trauma is not a recognized pathway to hamartoma development. 25 Another point of contention would be the supposed benign clinical behavior as an expansile mass of fragmentable, dysplastic, and continuously proliferating germinal tissue can be invasive and secondarily destructive. Pseudo-odontoma was considered appropriate for some of the dysplastic changes, particularly the abnormal matrigenesis, observed at the molar apices but not for the masses of markedly disrupted, haphazardly organized and proliferative odontogenic epithelium. Despite these challenges, the gross and histopathologic features here are very similar to those described in rat-like and squirrel-like rodents, 1,2,13,22 rabbits, 17 and other voles 29 though less proliferative and destructive than those described in guinea pigs and degus. 2,4,16,19,23
Observations in CSF-1-deficient, 14 Snx10-deficient and bisphosphonate-treated (personal observations, D. Imai) osteopetrotic rodents have confirmed that microtraumatic disruption and fragmentation of odontogenic epithelium by bone can produce isolated and disorganized islands of proliferative odontogenic epithelium that can subsequently proliferate into odontoma-like masses. 14 This microtraumatic disruption of the odontogenic epithelium is one possible transitional step between apical elongation and elodontoma formation. Rather than requiring direct trauma (such as falls from height or chewing on metal bars), 1,3,29 microtraumatic disruption may only require crowding pressure of continually expansile, hyperplastic odontogenic epithelium against less compliant and sometimes fractured bone. Thus, not only could elodontoma formation be dependent on the rate of coronal attrition in proportion to apical growth, but elodontoma formation could also be dependent on the rate of reactive accommodation by bony remodeling. 6,17 Instead of separate entities, these disease processes could be part of a pathologic continuum of possible ways in which elodont teeth inherently grow and respond to injury. Unfortunately, elodontoma occurrence was rare in this study and insufficient to test this hypothesis.
The pathogenesis of apical elongation in elodonts is thought to be inadequate coronal attrition resulting in clinical and reserve coronal elongation, abnormal occlusal pressure, inhibited coronal eruption, apical elongation and compression, and remodeling or perforation of bone. 3,17,23 The occlusal pressure is thought to directly inhibit continuous coronal eruption into the buccal cavity as well as transmit compressive forces to the apex, induce bony remodeling and allow for apical elongation. In the absence of direct occlusal contact and vertical pressure, coronal eruption may not be inhibited as quickly and apical elongation may be delayed. This difference in occlusal apposition and masticatory force between molar and incisor teeth and the absence of any repetitive incisor trauma 1,2,22 could explain why apical elongation (with one exception in the California vole collection) was not observed in the incisors of the Amargosa voles. Histopathologic incisor apical abnormalities may still be present in the Amargosa voles, but were not evaluated in this study.
The rates of coronal attrition, apical growth and reactive bony remodeling are likely determined by multiple genetic and environmental factors which could account for the species and individual variation in MAE prevalence, age of onset and clinical severity. In this study, the pedigree analysis of the museum collection of California voles was supportive of a heritable trait associated with MAE, although the magnitude and mode of inheritance was not confirmed. Inheritance could not be evaluated in the captive breeding colony of Amargosa voles due to the small sample size in each lineage. Other investigators consider this condition to be heritable with a complex, multifactorial to semicontinuous and variable expression pattern. 11,15,18 In captivity, one of the major environmental factors contributing to decreased coronal attrition is an inappropriate diet as laboratory rodent chow is a poor substitute for the natural Amargosa vole diet of highly abrasive bulrush. Additional proposed causes for MAE in rabbits and laboratory rodents include inflammation, aging changes, calcium/phosphorus imbalance, hypovitaminosis A, hypomagnesemia, hypophysectomy, and toxin exposure. 2,17,22,23 In this study, many of these proposed causes were not directly evaluated.
True prevalence in the entire Amargosa vole breeding colony was likely higher than 63.0% as many individuals reported as “not affected” were either (1) transferred out of the colony after only a few assessments, (2) too mildly affected to be palpable, or (3) too young to have developed the condition. Similarly, in the entire California vole museum collection, the 35.1% prevalence of MAE was likely an underestimate based on the young age of most specimens. The prevalence of MAE in both the Amargosa and California voles that were greater than 1 year of age was extremely high (77.6% and 77.3%, respectively) and equivalent across species. Age of onset (1.7–11.2 months) and rate of progression (1–3 months to progress from mild disease) varied, with at least one instance of temporary regression, which could explain the lack of correlation between age and severity of MAE. The one instance of temporary regression may be similar to what occurs in equine vertical impaction, in which mandibular bony deformities that correspond to mandibular MAE can resolve with removal of the inhibitory eruption cap. 8 Though males generally experienced MAE onset at an earlier age, the difference in average onset was only 1.5 months and there was no difference in progression of or survival with MAE between the sexes.
Although a high prevalence of MAE is a concern in this breeding colony of endangered Amargosa voles, it may be of limited relevance to individuals in the wild. Due to major threats, including habitat loss, increased predation, nonnative species competition, and infectious disease, 7,34 the average life expectancy of a wild Amargosa vole is only 2–3 months which is earlier than the average age of onset of MAE (5.3 months) in the captive breeding colony. Because of this disparity, very few wild individuals are likely to be affected. In a captive breeding colony, MAE could have a significant impact. Management to delay the onset or reduce the prevalence of this condition could increase the number of litters produced by a given female and increase numbers available for repopulation. Delay of even a month could generate 4–5 pups extra as females are capable of producing litters every 3–4 weeks. 7,34 Currently, colony management changes aim to increase the rate of occlusal attrition to prevent impaired eruption and apical elongation. The colony diet has been changed to Laboratory Rabbit Diet HF (high fiber) 5326 (LabDiet, Land O’Lakes Inc., St Louis, MO) that is specially milled to a larger pellet size and bulrush continues to be offered. The change has been too recent to assess efficacy but it is the same diet used to successfully manage the campus prairie vole (M. ochrogaster) colony. Exclusion of Amargosa voles that display early onset or rapid progression of MAE from breeding schemes is possible but not ideal as genetic diversity is already limited and other adverse traits could be selected.
The clinical, computed tomographic, pathologic and histopathologic features of MAE in voles are similar, if not identical, to those described for acquired dental disease of cheek teeth in rabbits, chinchillas, degus, and guinea pigs, odontogenic dysplasia of aging rodents and lagomorphs and even, vertical impaction of cheek teeth in horses. 2,8,18,20,22 With the observations from this study (Table 1), MAE has now been identified in 6 Microtus species, 12,18,30 4 subspecies of M. californicus, 11 and another arvicoline rodent, Lagurus lagurus. 15 In the pine voles, only molar occlusal abnormalities were reported but MAE with protrusion of mandibular molar apices through cortical bone was evident in the gross images. 12 Similarly, in long-tailed voles (M. longicaudus), molar occlusal abnormalities have been reported but apical elongation was likely present as well. 24 In another arvicoline rodent, Myodes gapperi, 29 apical elongation with elodontoma formation has been reported but affected the incisors only, as Myodes sp. have radicular, brachydont molar teeth. 5,27
In summary, MAE is not unique to Amargosa voles, or to Microtus spp., or even to arvicoline rodents. Rather, all evidence suggests that MAE is a spontaneous acquired condition inherent to species with continuously growing teeth (elodonts) that is due to impaired occlusal attrition and impaired eruption and influenced by heritable and external factors. Though not a disease exclusively of captivity, the limitations and provisions of the captive environment could directly influence the prevalence, severity and relevance of the disease. Because MAE can be a limiting factor to the health and lifespan of an individual, the condition should be managed in captivity. Palpation and visual examination of the ventral mandible for mandibular molar apical overgrowth is an effective method of identifying and staging disease. Increased access to roughage for dietary or behavioral enrichment can increase coronal attrition and has decreased the incidence of MAE in at least one captive colony. 12
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985818758469 - Apical Elongation of Molar Teeth in Captive Microtus Voles
Supplemental Material, DS1_VET_10.1177_0300985818758469 for Apical Elongation of Molar Teeth in Captive Microtus Voles by Denise M. Imai, Risa Pesapane, Christopher J. Conroy, Christina N. Alarcón, Nora Allan, Russell A. Okino, Jennifer Fung, Brian G. Murphy, Frank J. M. Verstraete, and Janet E. Foley in Veterinary Pathology
Footnotes
Acknowledgements
The authors would like to recognize the technical contributions of Melea Hunrath from Necropsy service at the Comparative Pathology Laboratory and Christopher Kwong, Kurt Takahashi, Amber Villareal, Felicia DonRussello, and Richard Wolfe at the Histopathology Laboratory in the Veterinary Medical Teaching Hospital, University of California, Davis. We are grateful to Dr William Lidiker for the California vole collection and data sets. Mijoo Kim assisted with data collection and analysis of museum specimens at the MVZ. Charles Peterson from SOLAR provided technical assistance with the pedigree analysis of MVZ voles. Mai Ueda assisted with Japanese to English language translation of published literature.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Funds for vole colony management and disease assessment were provided by the California Department of Fish and Wildlife, the Bureau of Land Management, and the US Fish and Wildlife Service Palm Springs Field Office and Cooperative Endangered Species Conservation Fund Award No. F14AP01006.
Supplementary material for this article is available online
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
