Abstract
Two horses that consumed well water with high fluoride content exhibited clinical signs of chronic dental and skeletal fluoride toxicosis and were later euthanized and autopsied. Both horses had degenerative disease of multiple joints and multiple dental defects. Elevated fluoride concentrations were found in bone and tooth samples of both horses, well water, and feed. Microscopically, abnormalities were noted in bone and tooth samples, and consisted mostly of foci of cement necrosis and hypercementosis. Horses exhibiting bilateral, highly symmetrical dental and/or skeletal lesions, with chronic lameness, should be evaluated for the possible presence of fluoride toxicosis.
Clinical signs and lesions associated with fluoride (F-) toxicosis (fluorosis) have been documented in a range of species, including horses, cattle, humans, cervids, water buffalo, sheep, goats, donkeys, kangaroos, and dromedary camels.3,16 Cattle are the species most susceptible to fluorosis, followed by horses and sheep. Chickens are the domestic species most tolerant to this toxicosis. 11 In a region of India with water F- concentrations of 1.4–3.3 ppm, lesions consistent with fluorosis were found in 30% of adult horses, with an incidence second only to cattle, of which 60% were affected.5–7 Although clinical fluorosis has been described in horses,9,18 and horses appear to be highly susceptible to this condition,5,7 bilateral carpal skeletal abnormalities and remodeling of navicular bones in horses with elevated F- have not been reported previously, to our knowledge. In addition, fluorine (F) has not yet been determined to be an essential dietary element in horses, 10 and information about dietary requirements and thresholds for F- toxicosis for horses is insufficicent. 14 One journal article suggested an equine dietary tolerance of 60 ppm F- on a dry matter basis. 19 However, there is no evidence-based information to establish this level. We report herein skeletal and dental abnormalities in 2 horses with fluorosis associated with consumption of well water with elevated F-.
Two Quarter Horse geldings were raised on a ranch in the area of Twentynine Palms, California. These horses were raised alongside other livestock (horses, cattle, and pigs) and ranch pets (bird, dog, and turtle). The horses and cattle were raised in fenced dry lots and were provided the same commercial hay (alfalfa and timothy) and well water ad-libitum. Data from the Twentynine Palms Water District indicated that water from wells in this region contained F- levels of 3–22 ppm. Case 1 (1997–2013) was purchased out-of-state as a yearling and remained on the ranch for 15 y. Case 2 (2004–2018) was purchased in state as a 4-y-old and remained on the ranch for 10 y. At ~ 10 y of age, these geldings began to exhibit shifting leg lameness that was accompanied by bilaterally enlarged carpi, and they eventually became unsafe to compete. Prior to the onset of enlarged carpi, flexing any limb caused pain with unusual resistance in both horses. As the disease progressed, both animals exhibited difficulty lying down and getting up. Lameness progressed quite rapidly, and after a few years, both geldings were retired and eventually euthanized at ages 16 y (case 1) and 14 y (case 2).
Both horses were autopsied. The carcasses were in good nutritional condition and weighed ~ 450 kg each. Gross lesions were similar in both horses. Externally, the dorsal aspects of both carpi were severely swollen. The swelling corresponded to severe fibrosis with focally extensive areas of soft tissue ossification and multifocal areas of yellow softening involving the peri-articular tissues on the dorsal aspect of the joint, including the joint capsule and the synovial sheath of the extensor tendons (Fig. 1). In addition, the proximal articular surface of both radial carpal bones had severe, focally extensive loss of cartilage with eburnation and pannus on the periphery of the lesion (Fig. 1). The dorsal border of the proximal articular surface of ulnar, intermediate, and radial carpal bones had multiple spikes and depressions. Similar, but less severe lesions were observed on the palmar border of the proximal articular surface of these 3 bones. The cartilage of the proximal articular surface of 2nd, 3rd, and 4th carpal bones, as well as the articular cartilage of the medial and lateral ridges of the trochlea of both femurs, had multiple ulcers, eburnation, and pannus. In case 2, ulceration, eburnation, and pannus were also noted in the articular cartilage of the first, second, and third phalanges, and the axis dens.
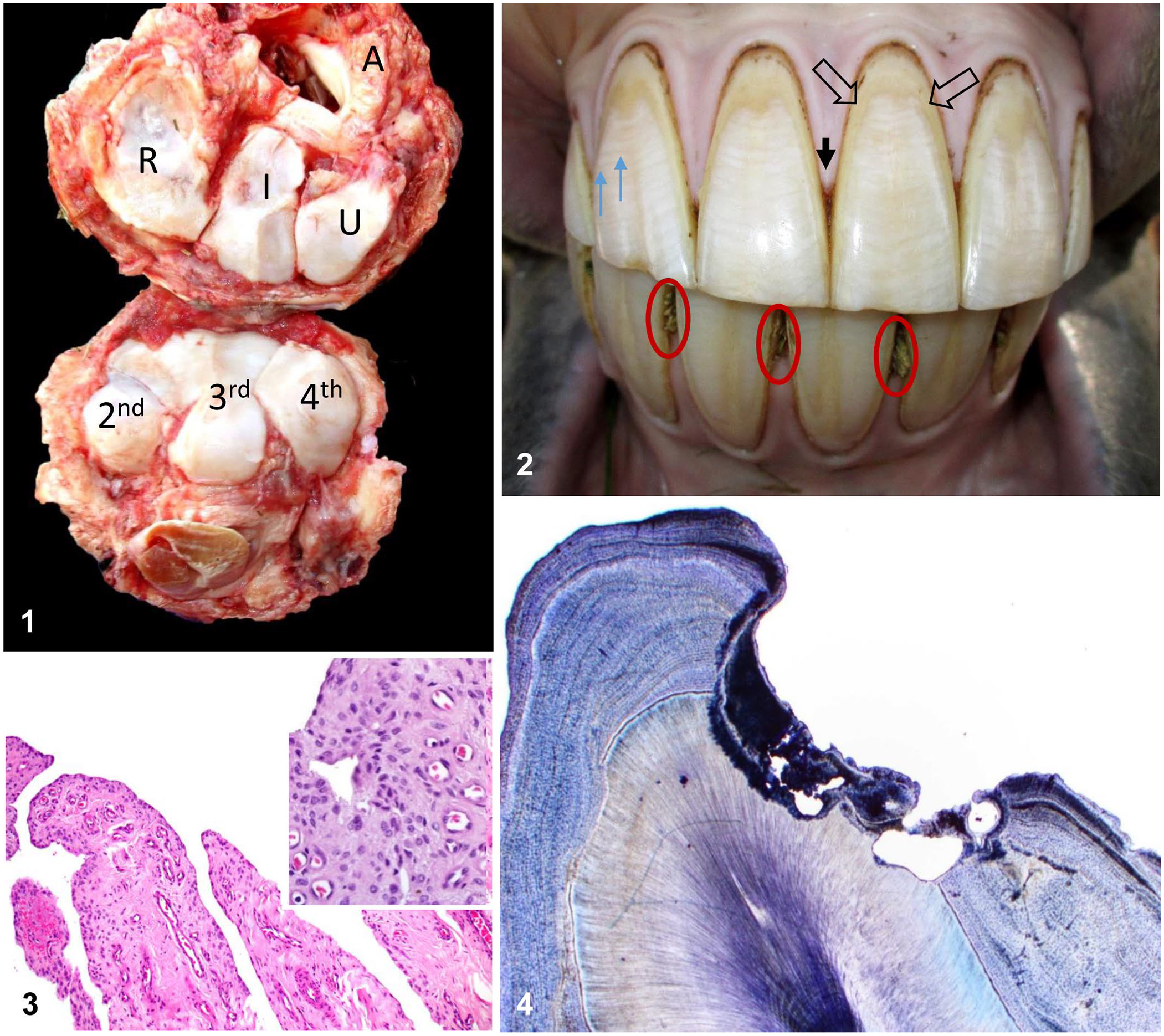
Dental and skeletal lesions in 2 horses with fluorosis.
Incisor, canine, and cheek teeth of both horses had multiple foci of cement necrosis and hypercementosis. Gum recession and diastemas with associated alveolar bone loss and/or loss of peripheral tooth cementum was also observed (Fig. 2). Enamel points on cheek teeth were exacerbated by the loss of supporting peripheral cementum. Hypomineralized enamel resulted in peripheral fractures in incisor and cheek teeth. Dental lesions were bilateral and exhibited a high degree of symmetry. No other significant gross abnormalities were observed in the remainder of either carcass.
The following tissues from both horses were collected and fixed in 10% neutral-buffered formalin, pH 7.2, for a minimum of 48 h, then processed routinely for the production of H&E sections: kidney, liver, spleen, lung, adrenal gland, thyroid gland, aorta, stomach, small intestine, colon, heart, cranial celiac mesenteric ganglion, sciatic nerves, and skeletal muscle. In addition, brain (cortex, corpus striatum, thalamus, midbrain at the level of superior colliculi, pons, cerebellar peduncles, cerebellum, pineal gland, and medulla at the level of obex) was collected from case 1, and the carpal joint capsule and coccygeal vertebrae from case 2. Bone was demineralized in 8% formic acid before being processed for the production of H&E sections. Non-demineralized, ~ 100-µm thick, cross-sections of the right corner mandibular incisor teeth were embedded (Sample-Kwick; Beuhler) and sectioned (IsoMet saw; Beuhler), followed by hand polishing.
Microscopically, the carpal joint capsule of case 2 had fibrosis, nodular synovial hyperplasia, and occasional hemosiderin-laden macrophages and lymphocytes in the synovial villi (Fig. 3). The coccygeal vertebrae of this horse had mild irregularity of the cortical surface with enthesophyte extension into sites of ligamentous attachment. The incisor teeth from case 1 had single, irregular, large resorption cavities that extended into the dentin (Fig. 4). Minimal amounts of irregular cementum deposition were present within and surrounding this area. There were a few other small areas of irregular cementum deposition associated with small resorption cavities. The incisor teeth of case 2 had minimal irregularity of the outer, and to a lesser extent, inner cementum surface, but significant resorption cavities were not observed. No other microscopic abnormalities were observed in other tissues from either case.
Upper and lower dental arcades including the canine and incisor teeth of case 1, and the skull, atlas, axis, first rib, and all 4 distal limbs of case 2, were collected and cleaned by composting in damp pine shavings for several months until all soft tissues were removed. Cleaned specimens of case 1 revealed severe bilateral mandibular canine and incisor teeth caries and hypercementosis; the severity of these lesions diminished toward the central incisors (Fig. 5). The arcades of both horses contained multiple diastemas, with loss of adjacent peripheral bone. Peripheral tooth cementum was abnormal and irregular. Cleaned skull, rib, and limb bones from case 2 had severe enthesophytes, which were most notable in areas of attachment of tendons and ligaments, in peri-articular regions. Mineralized ligaments and exostoses fused the distal intertarsal hock joint (Fig. 6). The dens of the axis had several joint osteophytes. All 4 distal sesamoid (navicular) bones had roughened irregular surfaces with exostoses at the attachment of ligaments and tendons (Fig. 7). The lingual surface of the right mandibular cheek teeth had peripheral hypercementosis and horizontal cementum necrosis; fully developed enamel remained unaffected (Fig. 8). All skeletal and dental lesions in cases 1 and 2 were bilateral and were highly symmetrical.
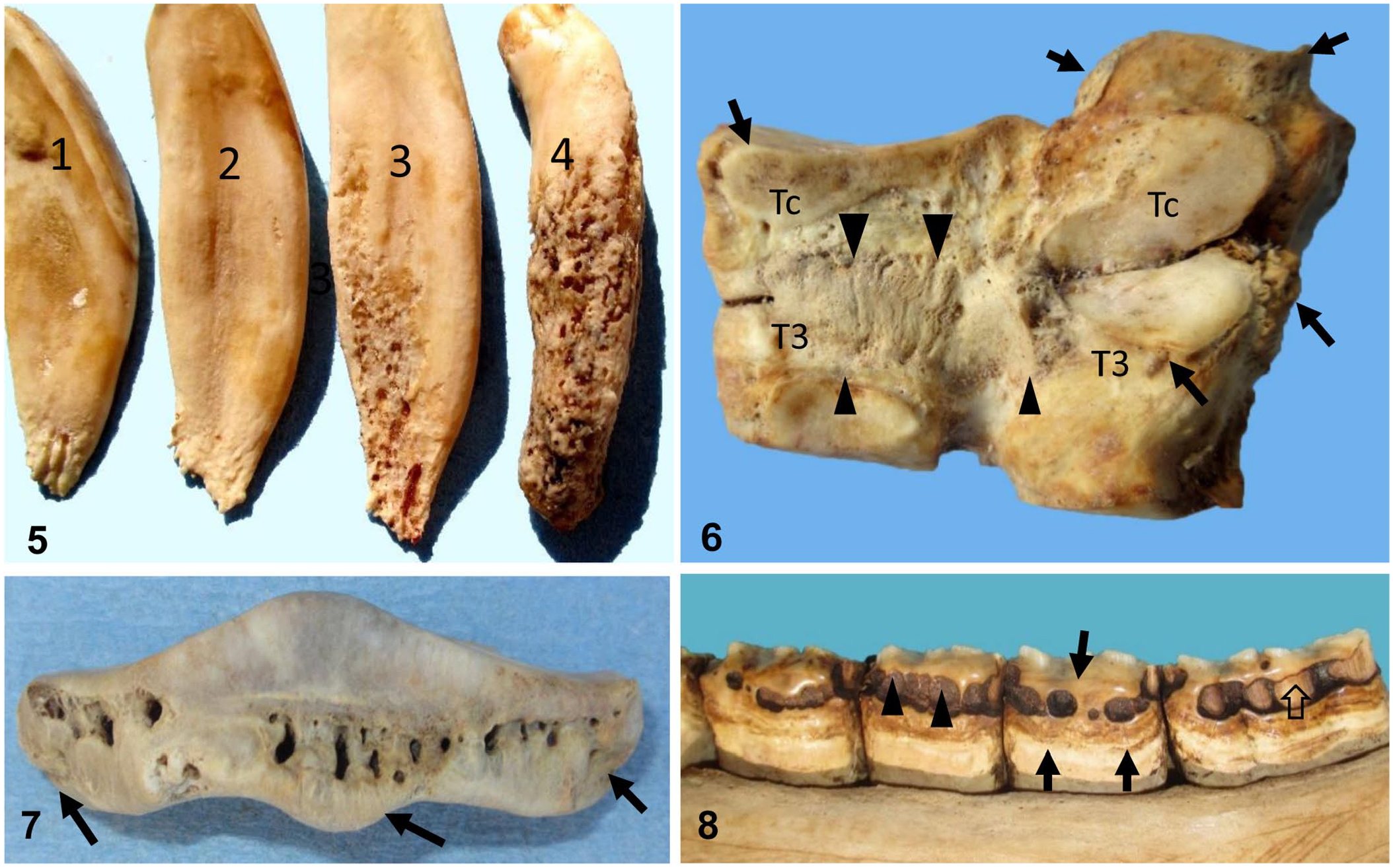
Dental and skeletal lesions in 2 horses with fluorosis.
Fresh samples of feces of case 2, and the proximal right mandibular corner incisor tooth, mid-section of humerus and first rib, the last 2 coccygeal vertebrae, and urine and composted mandibular incisor alveolar bone of both cases, along with composted samples of the distal tip of the third phalanx, midsagittal section of navicular bone of case 2, and multiple samples of the hay consumed by these horses were submitted for determination of F- levels. Hay had been sourced from the same commercial hay ranch in Brawley, CA (~ 190 km [120 mi] from the horse ranch), for the past 25 y. In addition, a ranch well water sample was collected (after allowing water to flow for 15 min) in a clean container for F- testing.
All F- analyses (Tables 1, 2) were performed by ion-selective electrode (Orion combination F- electrode; Fisher Scientific). Bone and hay samples were ashed at 600°C for 12 h. Ashed samples were then ground with a mortar and pestle, 0.01–0.02 g dissolved in hydrochloric acid, and subsequently neutralized with sodium hydroxide solution. Samples were next brought to a 5-mL volume with 18 mega-ohm water. One-half mL of total ionic strength adjustment buffer III (TISAB III) was mixed with 0.5 mL of each sample and analyzed by F- electrode. F- was quantified by comparison with external calibration solutions. Quality control materials analyzed with the samples included a method blank, a laboratory control spike, and, if the sample amount permitted, a matrix over spike. After a minimum of every 10 samples, a drift check was analyzed to ensure the stability of the electrode. The detection limit of F- in bone and water was 0.1 ppm.
Fluoride levels in tissues from 2 horses with fluorosis as determined by ion-selective electrode.
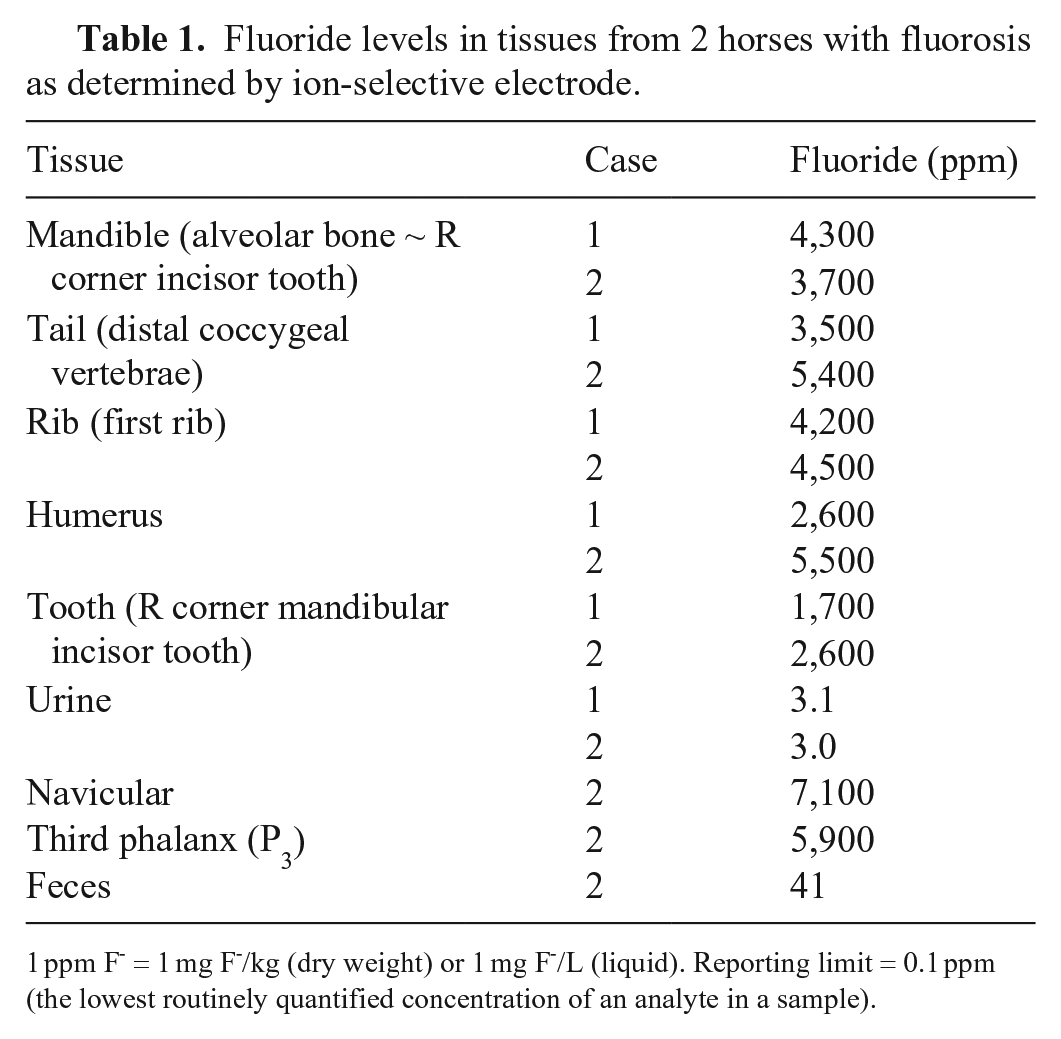
1 ppm F- = 1 mg F-/kg (dry weight) or 1 mg F-/L (liquid). Reporting limit = 0.1 ppm (the lowest routinely quantified concentration of an analyte in a sample).
Fluoride levels in hay 2012–2014, rock phosphate fertilizer, and well water as determined by ion-selective electrode.
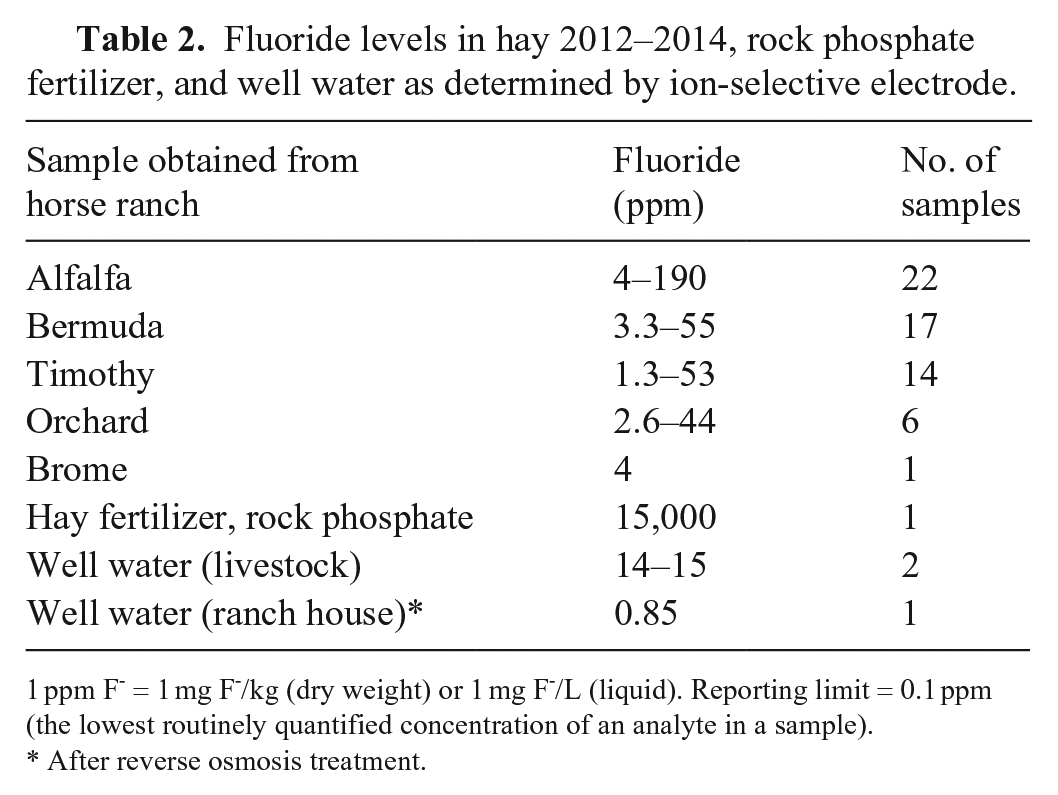
1 ppm F- = 1 mg F-/kg (dry weight) or 1 mg F-/L (liquid). Reporting limit = 0.1 ppm (the lowest routinely quantified concentration of an analyte in a sample).
After reverse osmosis treatment.
All samples, except urine from both horses, had elevated F- concentrations (Table 1). Fluoride in hay fed to ranch animals contained 4–15 ppm F-. The well water had F- concentrations of 14 and 15 ppm. An additional sample of water collected after a reverse osmosis water purification system was installed in the ranch home had 0.85 ppm of F- (Table 2). Assuming an intake of ~ 9 kg hay/horse/d, with 4 (alfalfa) or 15 (timothy) mg F-/kg, total daily F- from hay was 36–135 mg. Assuming daily water intakes of ~ 57 L containing 15 mg/L of F-, total daily F- intake from water was ~ 855 mg F-. Based on the results of the feed and well water analysis, each 450-kg gelding was estimated to have consumed 891 (36 + 855) to 990 (135 + 855) mg of F-/d from well water and hay, with 90% of the F- originating from well water.
The results of F- analysis, coupled with the gross and microscopic findings, confirmed a diagnosis of fluorosis. Additional elevated bone F- levels in livestock and ranch pets (data not shown) supported this diagnosis. Fluoride is naturally found in soil and water, and it has an affinity for positive ions. Absorbed fluoride can replace weaker halogen ions (Cl-, Br-, I-, and At−), and is capable of inhibiting enzymes that require divalent cations, metalloenzymes, and enzymes with hydroxyl groups. 11 Fluoride is readily absorbed transplacentally, 16 through ingestion, inhalation, or when applied topically. 4 Once absorbed, F- has an affinity for the calcium hydroxyapatite of the pineal gland, 12 teeth, and bone tissues. 11 Calcium hydroxyapatite is converted to calcium fluoride apatite when F- replaces OH- in the hydroxyapatite crystal by an exchange and adsorption reaction. The dental and skeletal lesions associated with fluorosis are a result of this replacement given that the osteoblasts, osteoclasts, and ameloblasts are affected.8,11,17 Fluoride is not irreversibly bound to bone and can be released back into the plasma when plasma concentrations fall. 4
Lesions associated with chronic fluorosis typically occur in teeth and bones.8,18 Dental lesions that occur during tooth development can affect both enamel and dentin. Enamel F- concentrations, unlike dentin concentrations, do not increase once the tooth erupts, because mature erupted enamel does not undergo remodeling. 1 Dentin, however, does undergo remodeling after eruption and is likely to have F- levels closer to those of compact bone. 11 Corner mandibular incisor tooth F- levels were ~ 50% less than those in cancellous bone (Table 1). 11 The permanent buds of the first molar teeth of the horse develop in utero, 1 potentially making them susceptible to transplacental fluorosis. Characteristic dental lesions include discoloration, hypomineralization, hypoplasia, and excessive abrasion.5,13 Pathognomonic enamel discoloration and pitting usually noted in brachydont teeth of humans and cattle with fluorosis have been previously reported in deciduous hypsodont teeth of foals, 16 and was likewise observed in several foals of the ranch described in our report (not shown).
Additional oral lesions associated with fluorosis include hypercementosis, cementum necrosis, hypoplasia with diastemas, necrosis of alveolar bone with recession of bone and gingiva, delayed eruption of permanent incisors, and oblique eruption of permanent teeth. Lesions in tissues adjacent to hypsodont teeth as a result of fluorosis can occur after dental eruption.8,13 Clinically, the incisor teeth in case 2 exhibited horizontal mottling, gum recession, diastemas, and minimal peripheral cementum. In case 1, antemortem radiographs would have been required to appreciate the hypercementosis and caries present in canine and incisor teeth (Fig. 3).
Skeletal fluorosis is associated with osteosclerosis, osteoporosis, periosteal hyperostosis, osteomalacia, or osteophytosis, 14 including enthesophytes and degenerative joint disease, as also observed in our cases. Fluoride not only affects osteoblasts, resulting in bone deformation, subperiosteal enthesophytes, spurs and bridging around joints, but is also responsible for the mineralization of tendons and ligaments, which was also observed in our cases.11,15,18 Skeletal changes occur after long-term exposure to fluoride, and are dose dependent. 8 Initially, fluoride toxicosis increases bone density, which is followed by osteoporosis and osteomalacia. At higher concentrations, spontaneous fractures, especially of the third phalanx, may occur. 11 The third phalanx may also be thickened and have a rough texture 18 ; similar changes were observed in all 4 distal phalanxes of case 2. In addition, all 4 navicular bones of case 2 had roughened irregular surfaces indicative of bone remodeling, as well as enthesophytes at areas of attachment of ligaments and tendons. All of these skeletal changes were most likely responsible for the progressive shifting leg lameness and pain that was observed in our 2 cases.
The rate of F- turnover in alveolar bone is higher 2 than in other bones; therefore, bone loss in cases of fluorosis is higher in alveolar than in other skull bones, and decreases progressively in ribs, vertebrae, and long bones. In alveolar bone, the first indication of chronic fluorosis is recession of alveolar bone and gingiva.5,9 Clinically, the recession of bone and gingiva in our cases resulted in the formation of diastemas between adjacent incisors where hay became trapped.
Well water was the primary source of F- exposure in our 2 cases. It is possible that the water was contaminated naturally and not from anthropogenic activities, such as fertilizer use or industrial emissions, but this remains undetermined. After diagnosis of fluorosis in our 2 cases and in other livestock and pets from the same ranch (data not shown), a reverse osmosis (RO) system was installed in the ranch to treat well water for use in the ranch house, which reduced the F- concentration in the water from 15 ppm F- to 0.85 ppm.
Fluorosis may be exacerbated by altitude, tropical climate, high annual mean temperatures, humidity, the presence of phosphates, sulfates, molybdenum, arsenic, lead, acidic pH levels, and impaired renal function.4,15,16 Some of these factors may have contributed to fluorosis in our cases. Twentynine Palms is 606 m above sea level, and average daily maximum temperature ranges from 15.5°C in the winter to 39.4°C in the summer (http://www.usclimatedata.com/climate/twentynine-palms/california/united-states/usca1173).
Previous reports of fluorosis in horses identified F- sources as mineral supplements and industrial contamination. Although F- exposure in the United States has been reduced by government regulations over the years, animals remain at risk in areas with naturally high F- levels, and in areas contaminated by F- from other sources such as super phosphate fertilizers (Table 2). 11
Normal bone F- concentrations in livestock have been reported at 300–600 ppm. In horses, > 500 ppm F- in bone is considered abnormal. 3 Fluoride bone concentrations were 2,600–4,300 ppm in case 1 and 3,700–7,100 ppm in case 2 (Table 1).
The gold standard for the diagnosis of chronic fluorosis in humans is determination of F- in urine, because 50% of ingested F- is excreted in the urine.4,15 Our cases had F- concentrations in urine of 3 ppm. However, because normal F- values in urine have not been determined for horses to date, urine F- testing is of little value in the diagnosis of fluorosis in horses. Fecal F- concentration in case 2 was 41 ppm, suggesting that fecal F- concentration, rather than urine concentration, may be more reflective of ongoing F- exposure in horses.
Several authors have reported periosteal enthesophyte involving the skull, ribs, metacarpus and metatarsus, and hoof deformities in horses with fluorosis.5,9,11,18 Similar lesions were also observed in our cases. In addition, both horses had bilateral carpal lesions, and remodeling changes of all 4 navicular bones were observed in case 2, which, to our knowledge, has not been described previously in horses with fluorosis. Horses exhibiting bilateral and highly symmetrical dental and/or skeletal lesions associated with chronic lameness should be evaluated for F- toxicosis using methods described above.
Footnotes
Acknowledgements
We thank Donna K. Fernandez for writing assistance, and Kelli J. Maddock for editorial assistance.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
