Abstract
Spontaneous choriocarcinomas are rare, highly vascular, malignant trophoblastic tumors that occur in humans and animals. This report describes the unusual spontaneous presentation of 4 choriocarcinomas within the subcutaneous tissues of 4, multiparous but nongravid, Amargosa voles (Microtus californicus scirpensis) from a captive breeding colony. Two subcutaneous neoplasms were composed of multifocal discohesive and infiltrative aggregates of medium to large trophoblasts and cytotrophoblasts within a fibrovascular stroma. Neoplastic cells were associated with variably sized thrombi and cavitary areas of hemorrhage and necrosis. Two subcutaneous tumors were predominantly composed of expansile, blood-filled, cystic spaces lined by neoplastic cytotrophoblasts and occasionally contained medium to large trophoblasts. Trophoblasts and cytotrophoblasts were positive for pancytokeratin and cytokeratin 8/18, negative for alpha-fetoprotein, and contained intracytoplasmic Periodic acid–Schiff (PAS)-positive glycogen in all 4 tumors. In species with hemochorial placentation, migration of trophoblasts into maternal circulation with embolization to distant nonreproductive tissues occurs and may explain the unusual subcutaneous distribution of these 4 tumors. The 2 multiloculated paucicellular tumors may represent an early stage of neoplastic transformation. To the authors’ knowledge, this is the first report characterizing choriocarcinomas in extrareproductive sites in rodents.
The Amargosa vole (Microtus californicus scirpensis) is one of the most endangered mammals in North America and is currently protected under the California and the US Endangered Species Acts.4,25 It is a desert subspecies of the California vole (M. californicus) and inhabits highly localized and isolated three-square bulrush (Schoenoplectus americanus) marshes and wetlands scattered along the Amargosa River in the Mojave Desert in Inyo County, California.4,25 Major stressors proposed in the decline of the Amargosa vole population include habitat loss or degradation caused by drought and climate change, increased predation pressure, competition with nonnative species, low genetic diversity, and infectious diseases.4,8,25 Currently, it is estimated that less than 500 Amargosa voles remain in the wild.4,22
As part of recovery efforts, a captive breeding colony composed of 20 wild-caught Amargosa voles was initiated at the University of California, Davis in 2014, as a collaborative effort between the School of Veterinary Medicine, California Department of Fish and Wildlife, US Fish and Wildlife Service, Bureau of Land Management, US Geological Survey, and other partners in the Amargosa Vole Conservation Project. 7 Through the University of California, Davis Comparative Pathology Laboratory, a health surveillance program, including infectious disease screening and complete postmortem examinations, monitors for diseases in the colony. A recent survey documenting common pathological conditions seen in this Amargosa vole colony showed that neoplasia represented 15% (6/40) of the histopathological diagnoses, with choriocarcinomas representing 50% (3/6) of neoplastic cases. 8
Gestational trophoblastic disease is a group of benign and malignant gestational tumors in which abnormal trophoblast cells grow inside the uterus after conception. 23 Choriocarcinoma is a rare subtype of gestational trophoblastic disease and can be of gestational or nongestational origin.13,23 Gestational choriocarcinoma is derived from placental trophoblastic lineage cells, while nongestational choriocarcinoma arises from totipotential germ cells in the ovaries, testes, or midline tissues in the mediastinum or abdomen. 23 In humans, gestational choriocarcinoma often occurs following a molar pregnancy presenting as an abnormal overgrowth of placental chorionic tissue in the uterus (also known as hydatidiform mole), which can invade the myometrium and spread hematogenously to extrauterine sites. Nongestational choriocarcinomas are germ cell-derived trophoblastic tumors that rarely metastasize via the lymphatics to the lung, liver, brain, and vulvovaginal region.13,23
While uncommon, choriocarcinomas have been reported in domestic and laboratory animals and in wildlife, including spontaneous or experimentally induced gestational choriocarcinomas in the rhesus macaque, armadillo (Dasypus novemcinctus mexicanus), and 2 rabbits.10,12,17 Spontaneous nongestational choriocarcinomas have been described in the ovaries in 2 rhesus macaques (Macaca mulatta), a cynomolgus macaque (M. fascicularis),6,16,24 and laboratory mouse strains, such as B6C3F1 and Crl:CD1 (ICR), and once in the testes of a Swiss albino mouse.1,9,18 Spontaneous ovarian choriocarcinomas have also been described in genetically engineered mice carrying a targeted null mutation of both Hic1 and Trp53 on a C57BL/6 × 129S4/SvJae background or a conditional deletion of Trp53 alleles under the control of Tie2 promoter in a Crl:CD1(ICR) × 129X1/SvJ genetic background.2,3 Reports of choriocarcinomas in nonreproductive tissues are exceedingly rare, represented by a primary gastric choriocarcinoma in a dog, and none have been reported in rodents. 21 Here, we describe a cohort of spontaneous extrareproductive choriocarcinomas in the subcutis of 4 female Amargosa voles maintained in a captive breeding colony.
A complete gross necropsy evaluation was performed on 4 Amargosa voles (M. californicus scirpensis). Tissues were fixed in 10% neutral-buffered formalin for at least 72 hours. Formalin-fixed tissues were processed routinely, embedded in paraffin, sectioned at thicknesses of 5 µm, and stained with hematoxylin and eosin. The hematoxylin and eosin-stained slides from these cases were reviewed by at least 2 board-certified veterinary pathologists (SEC, ALJ, KMC, DMI). Immunohistochemistry for pancytokeratin AE1/AE3 (Biocare Medical, Pacheco, California), cytokeratin 8/18 (Fitzgerald, North Acton, Massachusetts), and alpha-fetoprotein (Proteintech, Rosemont, Illinois) and periodic acid-Schiff reactions were performed on 5-µm sections of paraffin-embedded tissues from all control tissues and cases. Sections from normal murine placenta were included in the immunohistochemical study as comparison. Immunohistochemistry panel details are reported in Table 1. All work in the study was performed according to the guidelines for humane use of animals in research, approved by the UCD Institutional Animal Care and Use Committee (protocols 17996 and 19741), and authorized by US Fish and Wildlife Service Endangered Species Recovery Permit TE54614A-1.
Antibodies and methods used for immunohistochemistry.
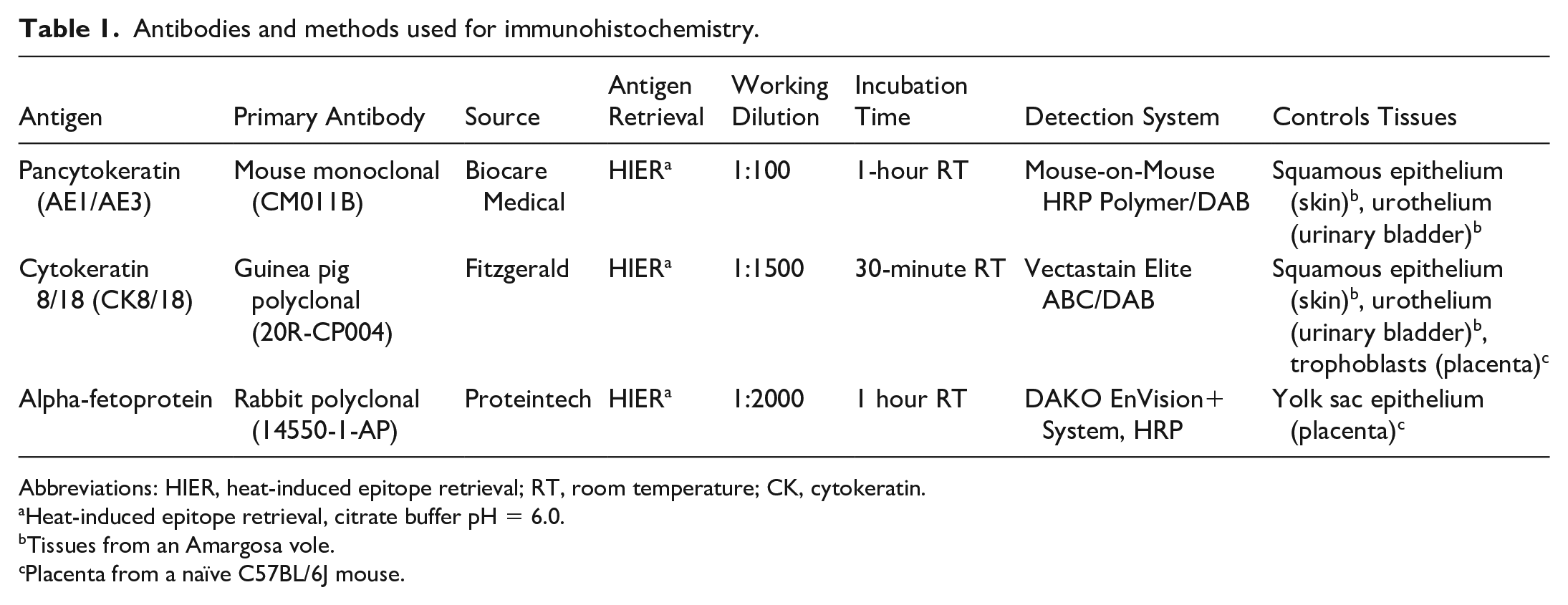
Abbreviations: HIER, heat-induced epitope retrieval; RT, room temperature; CK, cytokeratin.
Heat-induced epitope retrieval, citrate buffer pH = 6.0.
Tissues from an Amargosa vole.
Placenta from a naïve C57BL/6J mouse.
All 4 cases were nongravid, female, adult Amargosa voles euthanized due to approximately 1.0- to 3.0-cm-diameter, well-demarcated, variably ulcerated subcutaneous masses in the medial aspect of the right pelvic limb (case 1; Fig. 1a), right inguinal region (case 2), or left inguinal region (case 4), or were found dead with a 0.5-cm raised, red, subcutaneous perineal nodule (case 3). On cut section, the masses were composed of pale-tan, soft connective tissue separated by irregular blood-filled cavitations and hemorrhage (Fig. 1b). Breeding records from these voles indicated that females were pregnant approximately 2–6 months prior to the necropsy dates. Case 1 had 1 successful litter yielding 3 pups followed by 3 unsuccessful litters. Case 2 had 1 successful litter yielding 3 pups. Case 3 had 1 unsuccessful litter followed by 1 successful litter yielding 2 pups. Case 4 had 2 successful litters yielding 1 pup and 2 pups, respectively. Additional secondary pathological findings are described in the Supplemental Material.
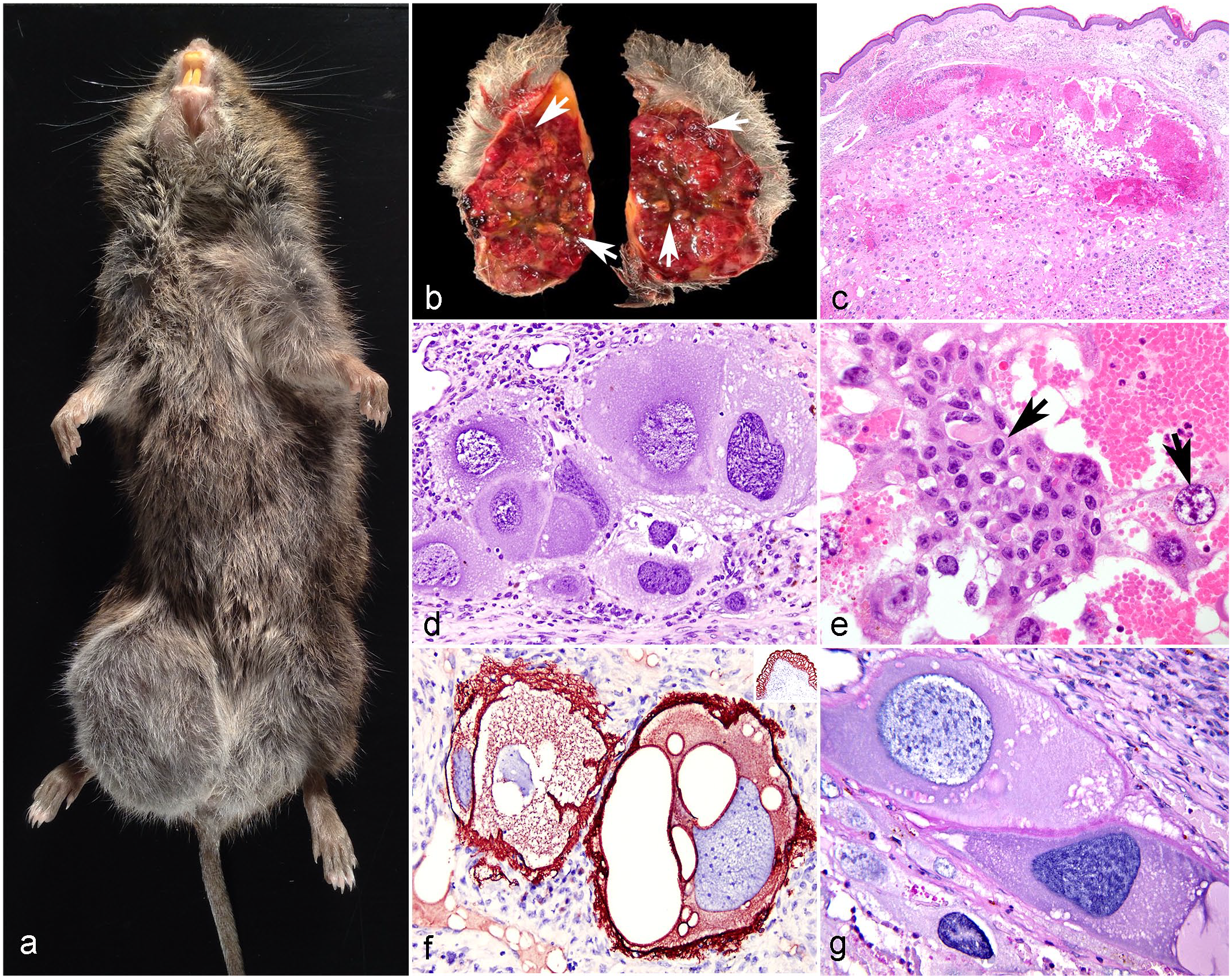
Subcutaneous choriocarcinoma, Amargosa voles. (a) The inguinal region near the right hind limb is expanded by a well-demarcated subcutaneous mass. Case 1. (b) On cut section, the mass is composed of pale-tan, soft connective tissue separated by irregular blood-filled cavitations (arrows). Case 2. (c) The subcutis and underlying fibrovascular stroma are expanded by a moderately cellular neoplasm composed of pleomorphic, atypical medium to large trophoblasts admixed with zones of necrosis and hemorrhage. Case 2. Hematoxylin and eosin (HE). (d) Neoplastic trophoblasts have 50–100 μm diameter, pleomorphic, round to oval, sometimes elongated reniform nuclei. Case 1. HE. (e) Scattered within the neoplasm, there are discohesive small aggregates of round to polygonal, 20- to 50-µm-diameter cytotrophoblasts (arrows) admixed with blood-filled spaces. Case 2. HE. (f) Neoplastic trophoblasts have strong and diffuse cytoplasmic immunolabeling for pancytokeratin AE1/AE3. Inset: vole’s urothelium expresses pancytokeratin. Case 1. Pancytokeratin AE1/AE3 immunohistochemistry. (g) Trophoblasts have moderate cytoplasmic and membranous reactivity with periodic acid-Schiff. Case 1.
The neoplasms from cases 1 and 2 had histologically similar features. The subcutis was infiltrated by multifocal clusters of neoplastic cells (Fig. 1c) and expanded by large foci of hemorrhage and necrosis with variably sized venous thrombi. Neoplastic cells were irregularly oval to polygonal, 100- to 500-µm-diameter trophoblasts (Fig. 1d) that formed loosely cohesive sheets interspersed in blood-filled spaces. These neoplastic cells had distinct borders, abundant pale-basophilic, finely granular to vacuolated cytoplasm, and 50- to 150-µm-diameter, round, oval to reniform nuclei (Fig. 1d) with 1–6 variably distinct nucleoli. There was moderate to marked anisocytosis and anisokaryosis, mild binucleation, and variable mitoses. Mitotic figures averaged 3–4 in ten 400× fields (2.37 mm2). Scattered throughout the tumors, trophoblasts were interspersed with small clusters of round to polygonal, 20- to 50-µm-diameter cytotrophoblasts (Fig. 1e) exhibiting distinct cell borders; high nuclear to cytoplasmic ratios; scant, homogenous, pale eosinophilic cytoplasm; and single round to oval nuclei with finely stippled chromatin and 1–2 magenta nucleoli. Cytotrophoblasts exhibited less pleomorphism and nuclear atypia compared to the trophoblasts. Within the tumors, medium to large veins were distended; segmentally occluded by thrombi; and surrounded by hemorrhage, erythrophagocytosis, low to moderate numbers of neutrophils and macrophages, and individual clusters of neoplastic trophoblasts. The vascular walls of thrombosed veins (Fig. 2d) were segmentally and variably effaced by fibroplasia and necrosis mixed with clusters of necrotic neutrophils and hemosiderin (recanalized thrombi). Neoplastic cell aggregates were variably separated by fibrous connective tissue and granulation tissue and infiltrated by mixed macrophages, plasma cells, and lymphocytes.
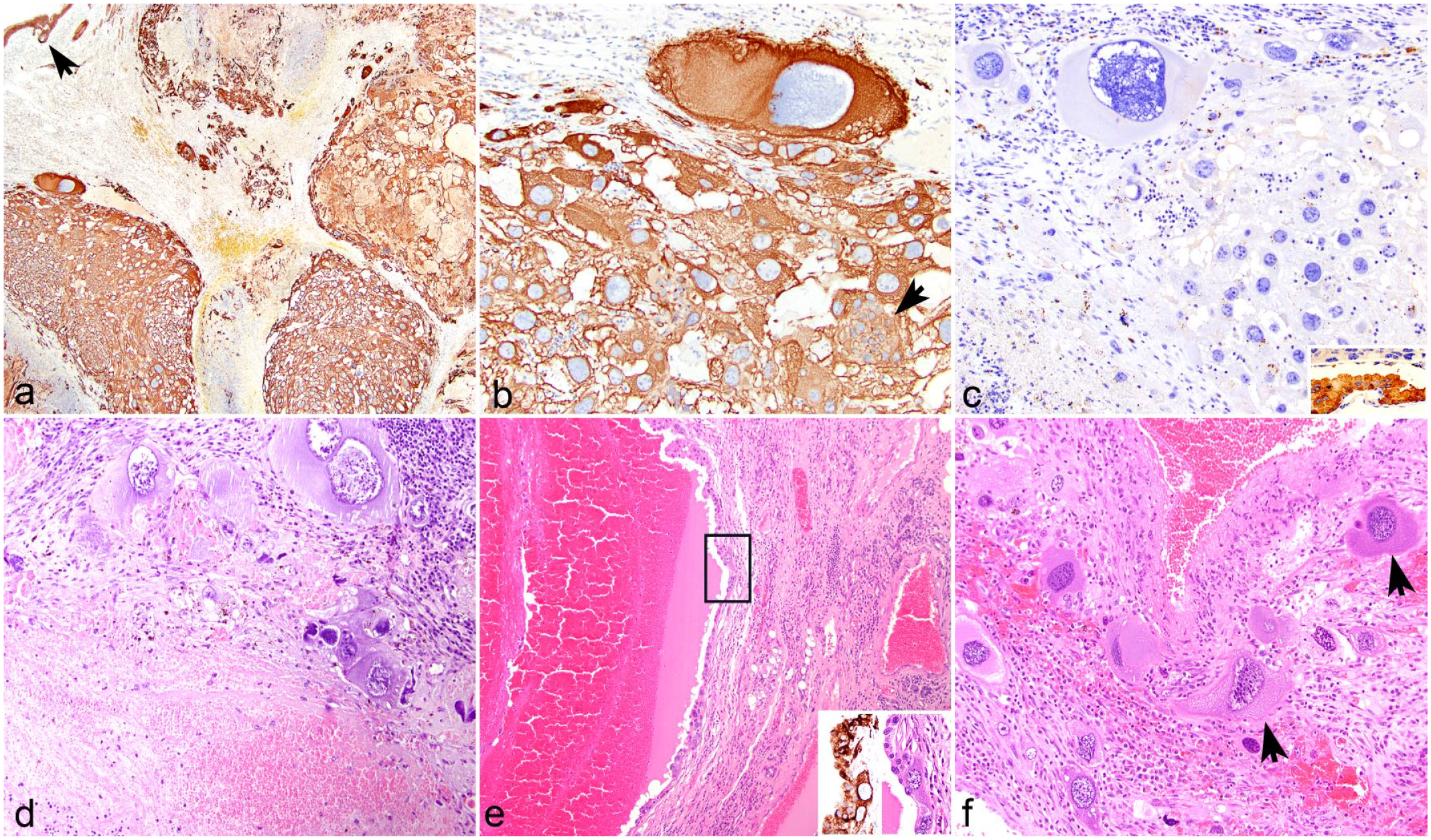
Choriocarcinomas, skin. Amargosa voles. (a) Multifocally, islands of trophoblasts and cytotrophoblasts in the right inguinal neoplasm are diffusely immunolabeled for cytokeratin (CK) 8/18. The arrow shows the immunolabeled epidermis. Immunohistochemistry (IHC) for CK 8/18. Case 2. (b) Neoplastic trophoblasts and cytotrophoblasts (arrow) exhibit strong and diffuse cytoplasmic CK 8/18 immunolabeling. Case 2. IHC for CK 8/18. (c) Neoplastic trophoblasts and cytotrophoblasts are negative for alpha-fetoprotein (AFP). Case 2. Inset: AFP immunolabeled a mouse’s yolk sac. IHC for AFP. (d) In the subcutis, a large-sized vein is distended and occluded by a thrombus that is surrounded by a fibrovascular stroma infiltrated by individual medium to large neoplastic trophoblasts, lymphocytes, and plasma cells. Case 1. Hematoxylin and eosin (HE). (e) The inguinal mass is composed of blood-filled cystic spaces lined by cytotrophoblasts and separated by fibrovascular stroma. Case 4. HE. Insets (corresponding to black box): neoplastic cytotrophoblasts exhibit strong cytoplasmic immunolabeling for CK 8/18. Case 4. CK 8/18 IHC and HE. (f) Scattered within the tumor, the fibrovascular stroma is infiltrated by individual medium and large trophoblasts (arrows) and low numbers of plasma cells and histiocytes. Case 4. HE.
The subcutaneous tumors from cases 3 and 4 exhibited similar histological features compared to tumors from cases 1 and 2, but these tumors were predominantly expanded by blood-filled, cystic spaces (Fig. 2e) lined by 40- to 60-µm-diameter cytotrophoblasts (Fig. 2e). These neoplastic cells formed a delicate, continuous 1- to 2-cell thick layer and had distinct cell borders with uniform round nuclei and rare mitoses (Fig. 2e). The cavitations were separated by thin bands of fibrous connective tissue infiltrated by mixed leukocytes and occasional medium to large neoplastic trophoblasts (Fig. 2f).
In all cases, the proliferating trophoblasts and cytotrophoblasts exhibited strong intracytoplasmic immunoreactivity for pancytokeratin AE1/AE3 (Fig. 1f) and had abundant amounts of periodic acid-Schiff-positive of intracytoplasmic glycogen (Fig. 1g). Trophoblasts and cytotrophoblasts from all subcutaneous masses exhibited strong intracytoplasmic immunoreactivity for cytokeratin 8/18 (Fig. 2a, b, e). Neoplastic cells from all neoplasms were negative for alpha-fetoprotein (Fig. 2c). In the control mice, yolk sac epithelium from placenta (Fig. 2c) and fetal hepatocytes demonstrated strong immunoreactivity for alpha-fetoprotein.
In humans, the microscopic diagnosis of choriocarcinomas requires the presence of at least 2 cell populations such as trophoblasts, cytotrophoblasts, and/or syncytiotrophoblasts. 23 Microscopically, gestational choriocarcinomas grow in an infiltrative pattern and are typically composed by clusters of cytotrophoblasts and trophoblasts with mild to moderate nuclear pleomorphism and multinucleated syncytiotrophoblasts surrounding areas of hemorrhage and necrosis. 23 The immunophenotype depends on the evaluated trophoblastic cell population as all trophoblastic cell subtypes express AE1/AE3 (pancytokeratin) and cytokeratin 7. 19 3β-Hydroxysteroid dehydrogenase, human chorionic gonadotropin, melanoma cell adhesion molecule, human leukocyte antigen G, and cytokeratin 8 are additional immunohistochemical markers used to characterize choriocarcinomas in humans.15,23 The diagnosis of choriocarcinoma in these voles was based on the presence of cytotrophoblasts and medium to large trophoblasts with supportive immunophenotyping within the expansile and peripherally invasive tumors. Cytokeratin 18 is an epithelial cytoskeletal protein expressed in trophoblasts (including intermediate trophoblasts, cytotrophoblasts, and syncytiotrophoblasts) in the mouse placenta 5 and was expressed in the neoplastic cells of all subcutaneous masses presented in this report. In addition, our immunohistochemistry results demonstrated that alpha-fetoprotein was not expressed by trophoblasts and cytotrophoblasts (Fig. 2c), which allowed us to rule out a neoplasm of germ cell of origin.
The anatomical location of these choriocarcinomas is unique in these cases. While previous studies have shown choriocarcinomas in rodents arising from the ovary and testis,1,2 none have described the development of choriocarcinomas in the subcutis. In humans, cutaneous choriocarcinomas are extremely rare and reported as metastatic foci from a primary neoplasm. 20 In these Amargosa voles, no other neoplasm in the reproductive tract or other tissue was present that could be considered primary. None of the voles exhibited concurrent neoplasms of germ cell origin in the ovaries or uterus, nor high-grade epithelial neoplasms in other organs. Occasionally, choriocarcinomas have been reported to coexist with ovarian teratomas and embryonal carcinoma in nonhuman primates and testicular yolk sac carcinoma, embryonal carcinoma, and teratoma in a Swiss albino mouse.6,9,24 One of the 4 cases (case 3) exhibited a uterine leiomyoma and ovarian cystadenomas, while the rest of the cases had normal reproductive organs. Thus, it seems unlikely that these tumors originated from germ cells in the uterus or ovaries from these voles.
In these cases, the subcutaneous choriocarcinomas most likely represent gestational disease due to malignant transformation of trophoblasts that embolized to the subcutis during past pregnancies. Spontaneous trophoblastic embolism has been reported in animals with hemochorial placentation and as incidental findings associated with normal pregnancies in hamsters, chinchillas, gerbils, cotton rats, porcupines, and a snowshoe hare.11,14 Embolism typically occurs in lung, adrenal gland, spleen, or liver,11,14 but has not yet been described in the subcutis. Gestational choriocarcinoma in humans is usually preceded by an invasive hydatidiform mole which forms a mass in the uterus, a lesion that has not been reported in rodents but could nevertheless occur. It is possible that the trophoblastic cell population that embolized to small blood vessels in the maternal subcutis was inherently abnormal, ultimately led to the proliferation and neoplastic transformation into a trophoblastic tumor. The presence of paternal deoxyribonucleic acid by genotyping and short-tandem repeat analysis in these tumors are techniques used to distinguish gestational from nongestational choriocarcinomas in humans. 23 Gestational choriocarcinomas in humans are also correlated with significantly elevated levels in serum beta-gonadotropin hormone, which if available for the vole could support the proposed pathogenesis. 23
In summary, this case series documents an unusual presentation of subcutaneous choriocarcinomas in 4 Amargosa voles maintained in a breeding colony and proposes a pathogenesis of gestational disease, involving subcutaneous trophoblastic embolization and malignant transformation.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858231203647 – Supplemental material for Subcutaneous choriocarcinomas in captive Amargosa voles (Microtus californicus scirpensis)
Supplemental material, sj-pdf-1-vet-10.1177_03009858231203647 for Subcutaneous choriocarcinomas in captive Amargosa voles (Microtus californicus scirpensis) by Sebastian E. Carrasco, Amanda L. Johnson, Kerriann M. Casey, Nora Allan, Mia Reed, Janet E. Foley and Denise M. Imai in Veterinary Pathology
Footnotes
Acknowledgements
The authors would like to recognize the technical contributions of Melea Hunrath from Necropsy service at the UCD Comparative Pathology Laboratory, Caroline Atkinson from Histology service at the MIT Comparative Pathology Laboratory and Maria Jiao from the Histology service at the MSKCC / WCM Laboratory of Comparative Pathology. We thank Dr. Isabel Casanova for her useful suggestions and comments on the manuscript and Dr. Krista La Perle for immunohistochemistry services.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funds for vole colony management and disease assessment were provided by the California Department of Fish and Wildlife, the Bureau of Land Management, and the US Fish and Wildlife Service Palm Springs Field Office and Cooperative Endangered Species Conservation Fund (Award No. F14AP01006). Sebastian E. Carrasco and the Laboratory of Comparative Pathology are also supported in part by the NIH Core Grant P30CA008748.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
